Abstract
Background
Preclinical and clinical evidence suggests a role for the dysregulation of the endocannabinoid system in migraine pain, particularly in subjects with chronic migraine.
Methods
The gene expression of endocannabinoid system components was assayed in peripheral blood mononuclear cells of 25 subjects with episodic migraine, 26 subjects with chronic migraine with medication overuse (CM-MO) and 24 age-matched healthy controls. We also evaluated the protein expression of cannabinoid receptors 1 and 2 as well as DNA methylation changes in genes involved in endocannabinoid system components.
Results
Both episodic migraine and CM-MO subjects showed higher cannabinoid receptor 1 and cannabinoid receptor 2 gene and protein expression compared to controls. Fatty acid amide hydrolase gene expression, involved in anandamide degradation, was lower in migraine groups compared to healthy control subjects. N-arachidonoyl phosphatidylethanolamine phospholipase D gene expression was significantly higher in all migraineurs, as were monoacylglycerol lipase and diacylglycerol lipase gene expressions. The above markers significantly correlated with the number of migraine days and with the days of acute drug intake.
Conclusion
The findings point to transcriptional changes in endocannabinoid system components occurring in migraineurs. These changes were detected peripherally, which make them amenable for a wider adoption to further investigate their role and applicability in the clinical field.
clinicaltrials.gov NTC04324710.
Introduction
Migraine is a neurovascular disease whose pathophysiology is far from being completely clarified. This is mainly due to the complex mechanisms that underlie migraine attack as well as its recurrence. The endocannabinoid system (ES) is a complex signalling system involved in different biological processes (e.g. neuronal activity, pain sensation and immune functions) and it plays a crucial role in the maintenance of body homeostasis (1,2). The ES components include the endogenous lipids, the most studied ones being N-arachidonoylethanolamide (AEA) and 2-arachidonoylglycerol (2-AG), their metabolic enzymes and at least two cannabinoid receptors (CB1 and CB2). The biosynthesis of AEA mainly occurs by N-acylphosphatidylethanolamide-phospholipase D (NAPE-PLD), whereas 2-AG is produced through the action of diacylglycerol lipase (DAGL). AEA is metabolized by fatty acid amide hydrolase (FAAH) and 2-AG mainly by monoacylglycerol lipase (MAGL).
Alterations in gene expression of ES components may involve various cell types, multiple catabolic pathways and the generation of active metabolites via epigenetic mechanisms, under both physiological and pathological conditions (3). CB genes, for instance, may interact with different transcriptional factors, many of which are related to DNA methylation and histone post‐translational modifications (4). A dysfunctional ES has been associated to numerous disorders (5,6) including migraine (7). The ES, indeed, modulates multiple activities and neuromodulators/neurotransmitters that play a crucial role in migraine pathogenesis (8,9). ES is also implicated in the descending modulation of the trigeminovascular nociceptive transmission from the brainstem afferents (10). Previous studies from our laboratory, using the migraine-specific animal model based on nitroglycerin administration in rat, demonstrated the existence of interactions between the ES and pain mediation (7). In particular, we showed a key role for AEA and for FAAH-regulated AEA activity in the processing of trigeminal nociceptive signals (11–13). AEA inhibits neurogenic dural vasodilatation, as well as calcitonin gene-related peptide-induced and nitric oxide-induced dural vessel dilation, an activity that is reversed by CB1 antagonism (14). The interaction between the ES and migraine pain is also suggested by clinical observations (see (7) and (15) for review). FAAH activity was higher in platelets of women with episodic migraine (EM) (16), to suggest a more marked degradation of AEA. Subjects with chronic migraine (CM) and medication overuse (MO) showed an altered endocannabinoid metabolism not only in platelets, but also in the cerebrospinal fluid (CSF) (17–19). Although the above clinical findings are scattered and replicated, their re-consideration in the light of more recent data from the increasing pre-clinical evidence prompt the need to investigate in more depth the role of ES in migraine pathophysiology.
Changes in ES components detected in peripheral blood mononuclear cells (PBMCs) seem to correlate with central dysfunction of the ES in different neurological diseases (20,21). For instance, in patients with Parkinson’s disease or multiple sclerosis, increased CSF levels of AEA were associated with a reduction in the activity and protein content of FAAH in PBMCs, which is indicative of an increased AEA tone. AEA levels were elevated in the CSF and in the blood of schizophrenic subjects (22,23), with a significant drop in AEA blood levels and in mRNA transcripts of CB2 and FAAH in PBMCs following clinical remission (23).
The aim of the present study was the identification of a specific expression pattern of ES components at the peripheral level in subjects with migraine. To this end, we performed a thorough evaluation of multiple peripheral components of the ES (gene expression, protein expression and DNA methylation) in PBMCs of representative samples of subjects with EM without aura, CM-MO and in healthy controls (CT).
Materials and methods
Subjects
Twenty-five subjects with EM, 26 with CM-MO and 24 CT were enrolled in the headache center of the IRCCS Mondino Foundation of Pavia (Italy). The study was approved by the local Ethics Committee (CE Pavia, Aut. Number 20170023686) and all subjects enrolled signed a written informed consent, in accordance with the Declaration of Helsinki and existing national ethics regulation. The study was registered on clinicaltrials.gov (NTC04324710).
Inclusion criteria for EM subjects were: i) satisfaction of the diagnostic criteria for migraine without aura according to the International Headache Classification 3rd edition (ICHD-3) (24); and ii) an episodic pattern of migraine for at least 10 years without any period of chronification. Inclusion criteria for patient with CM-MO patients were: i) satisfaction of the ICHD-3 diagnostic criteria for chronic migraine and for one of the subtypes of medication overuse headache; and ii) a history of stable chronification for at least 5 years.
Sex- and age-matched CT without any history of migraine or other primary headaches were recruited. Infrequent tension-type headache episodes were, however, allowed. They were normal in physical and neurological examinations and did not have any systemic diseases, psychiatric disorders or any other clinically significant conditions. The subjects were instructed to avoid any type of painkiller in the 24 hours prior to the blood sampling.
Isolation of peripheral blood mononuclear cells (PBMCs)
Blood samples (20 ml) were collected within ethylenediamine tetra-acetic acid (EDTA) containing tubes from participants. Blood samples were diluted in 1:1 ratio with phosphate buffer saline 1X (PBS 1X) (Sigma). Diluted blood samples were slowly loaded onto Ficoll separating solution (15 ml) (Sigma) and centrifuged at 800 g without brake for 30 min at room temperature. PBMCs accumulated as the middle white monolayer, were washed twice in sterile PBS 1X and centrifuged at 300 g for 15 min. For each sample, a batch of PBMCs was used for RNA or DNA extraction and another one for flow-cytometer analysis.
In the PBMCs isolated from the subjects in the three study groups we assayed:
CB1 and CB2 protein expression Expression of the following genes: CB receptors, FAAH, NAPE-PLD, MAGL and DAGLα DNA methylation of ES components
CB1 and CB2 protein expression
CB1 and CB2 protein expression was evaluated in CD45+ PBMCs by flow cytometry with a FACS Canto flow cytometer (Becton-Dickinson). CD45 Isolation Kit and MidiMACS™ Separator (Miltenyi Biotec), an indirect magnetic labelling system, were used to obtain highly purified CD45+ cells from freshly isolated PBMCs. After a preincubation of 10 min with human FcR Blocking Reagent (Miltenyi Biotec) to prevent non-specific binding to the Fc receptor, cells (100,000 per reaction) were incubated for 30 min at room temperature with the following antibodies: CD45 APC h7 (BD Biosciences, 1:50), CB1 APC (R&D system, 1:50, (25)) and CB2 FITC (Cayman Chemical, 1:30, (26)), diluted according to the manufacturer instructions. Unstained cells; that is, incubated in the absence of fluorescently labelled antibodies, were used to assess background fluorescence to set voltages and negative gates appropriately. Single stained samples were used to check for fluorescence spread, to compensate for spectral overlap and to determine positive gating.
Cell population was identified on the basis of its forward and side scatter characteristics, excluding signals from cellular debris or clumps. For each sample, a total of 10,000 cell-gate events were acquired and data were expressed as percentage of total events co-expressing CD45 with CB1 or CB2.
Gene expression (rtPCR)
Total RNA from PBMCs was isolated using standard procedure (Zymo Research) and RNA quality was assessed using a NanoDrop spectrophotometer (NanoDrop Technologies, Thermofisher); cDNA was generated using the iScript cDNA Synthesis kit (Bio-Rad) following the supplier’s instructions. Gene expression of CB receptors, FAAH, NAPE-PLD, MAGL and DAGLα was analyzed using the Fast Eva Green supermix (Bio-Rad). Primer sequences obtained from the AutoPrime software (http://www.autoprime.de/AutoPrimeWeb) are reported in Table 1. Ubiquitin C (UBC), whose expression remained constant in all experimental groups, was used as a housekeeping gene. The amplification was performed with a light Cycler 480 Instrument rt-PCR Detection System (Roche) following the supplier’s instructions. All samples were assayed in triplicate and gene expression levels were calculated according to 2−ΔΔCt = 2−(ΔCt gene − ΔCt housekeeping gene) formula by using Ct (cycle threshold) values.
Primers sequences used.

DNA methylation
Since PBMCs contain the full complement of epigenetic enzymes found in most tissues, including neurons and peripheral nucleated cells, we evaluated the role of DNA methylation in the regulation of ES gene transcription in all the enrolled subjects.
DNA was extracted from whole blood using QIAmp DNA Blood Mini Kit (Qiagen) and its concentration was determined by NanoDrop quantification (NanoDrop Technologies, Thermofisher). Arrays preparation and data analysis were performed by Genomix4Life srl (Baronissi, Italy). High-quality DNA (500 ng) was bisulfite converted using EZ DNA methylation kit (Zymo Research, Irvine, CA, USA). Bisulfite converted DNA (200 ng) was used for analysis of whole-genome methylation, using the HumanMethylation 450 K BeadChip (Illumina, San Diego, CA, USA), which contains 485 577 probes covering 21 231 (99%) RefSeq genes. In brief, bisulfite converted DNA was whole-genome amplified for 20 h followed by end-point fragmentation. Fragmented DNA was precipitated, denatured and hybridised to the BeadChips for 20 h at 48°C. The BeadChips were washed and the hybridised primers were extended and labelled before scanning the BeadChips using the Illumina iScan system. GenomeStudio software (version 2011.1; Illumina Inc.) was used for the extraction of DNA methylation signals from scanned arrays. The methylation level for each cytosine was expressed as a beta value calculated as the fluorescence intensity ratio of the methylated to unmethylated versions of the probes: Beta values ranged between 0 (unmethylated) and 1 (methylated). The annotation relating to CGIs uses the following categorisation: “shore”, each of the 2-kb sequences flanking a CGI; “shelf”, each of the 2-kb sequences next to a shore; “open sea”, DNA not included in any of the previous sequences or in CGIs (4). TSS200 and TSS1500 indicate the region between position −200 bp and −1500 bp from the TSS, respectively. The significant methylation difference between two given loci is indicated by a delta-beta value and determined with GenomeStudio Methylation Module using Illumina custom algorithm for calculating DiffScores (DiffScore ≤−30.0 (≈pval < 0.001) = hypo-methylation; DiffScore ≥30.0 (≈p-value < 0.001) = hyper-methylation).
Statistical analysis
In a preliminary study, we evaluated FAAH expression in the PBMCs of a group of CT subjects (n = 12, Mean = 1.70 ± 0.6) and EM subjects (n = 12, Mean = 1.12 ± 0.7) reporting an effect size of 0.88. To compare FAAH expression in the PBMCs of CM-MO patients with those of CT and EM subjects, we hypothesized an effect size of 0.45. Thus, to obtain a statistical power of 0.80 and alpha = 0.05, it was estimated that the number of subjects needed in each group was at least 17.
The statistical analysis was performed with SPSS (Statistical Package for Social Sciences), version 21.0 for Windows.
Continuous data are expressed as mean ± standard error of the mean (SEM). Apart from age, all data showed a not-normal distribution at the Shapiro-Wilk test, therefore variables were compared using non-parametric tests. At baseline, differences between EM and CM-MO patients were evaluated with a Mann-Whitney test. Differences in proteins and genes expression between EM, CM-MO, and CT groups were assessed with a Kruskal-Wallis test. The level of significance of this test was adjusted for total number of Kruskal-Wallis tests performed (n = 6) with a Bonferroni’s correction. Only when the main Kruskal-Wallis tests proved to be significant were further analyses conducted. In particular, inter-group differences were assessed with a post-hoc analysis performed with Dunn's Multiple Comparison test. In EM and CM-MO groups, a bivariate correlation analysis between clinical-demographic data and levels of NAPE, FAAH, DAGL, MAGL, as well as of CB1 and CB2 mRNA levels was performed by means of the Spearman’s test. According to the results of the correlations, a linear regression was also performed to control for age and sex. As the main variable of interest we used the number of migraine days per month, while the number of days of acute drug intake was not used in the model because of collinearity. For the linear regression analysis, each set of variables was used two times (correlation and regression analysis), and for this reason we applied an appropriate Bonferroni’s correction in the regression model. Differences between categorical variables were analyzed using the chi-square test. A probability level of less than 5% was regarded as significant, always corrected for multiple comparison as described above.
Results
Clinical and demographic characteristics of the study population
Patients with EM (n = 25, 24 females, age 42 ± 2.06 years) presented with 6.6 ± 0.5 headache days per month, 6.2 ± 0.4 migraine days per month, and 6.28 ± 0.5 days of acute drug intake per month. Patients with CM-MO (n = 26, 22 females, age 47 ± 2.2 years) reported 25.9 ± 1.0 headache days per month, 24.0 ± 1.1 migraine days per month, and 23.7 ± 1.1 days with acute drug intake per month. CT subjects (n = 24, 18 females, age 41.46 ± 2.5 years) were comparable for age and sex to the migraine patients. CT subjects did not report any acute medication intake in the month prior to enrolment. EM and CM-MO groups were comparable for demographic features as well as age at migraine onset, presence of migraine with aura, and intake of preventive medication at baseline. Clinical and demographic data are summarized in Table 2.
Clinical and demographic characteristics of the study population.
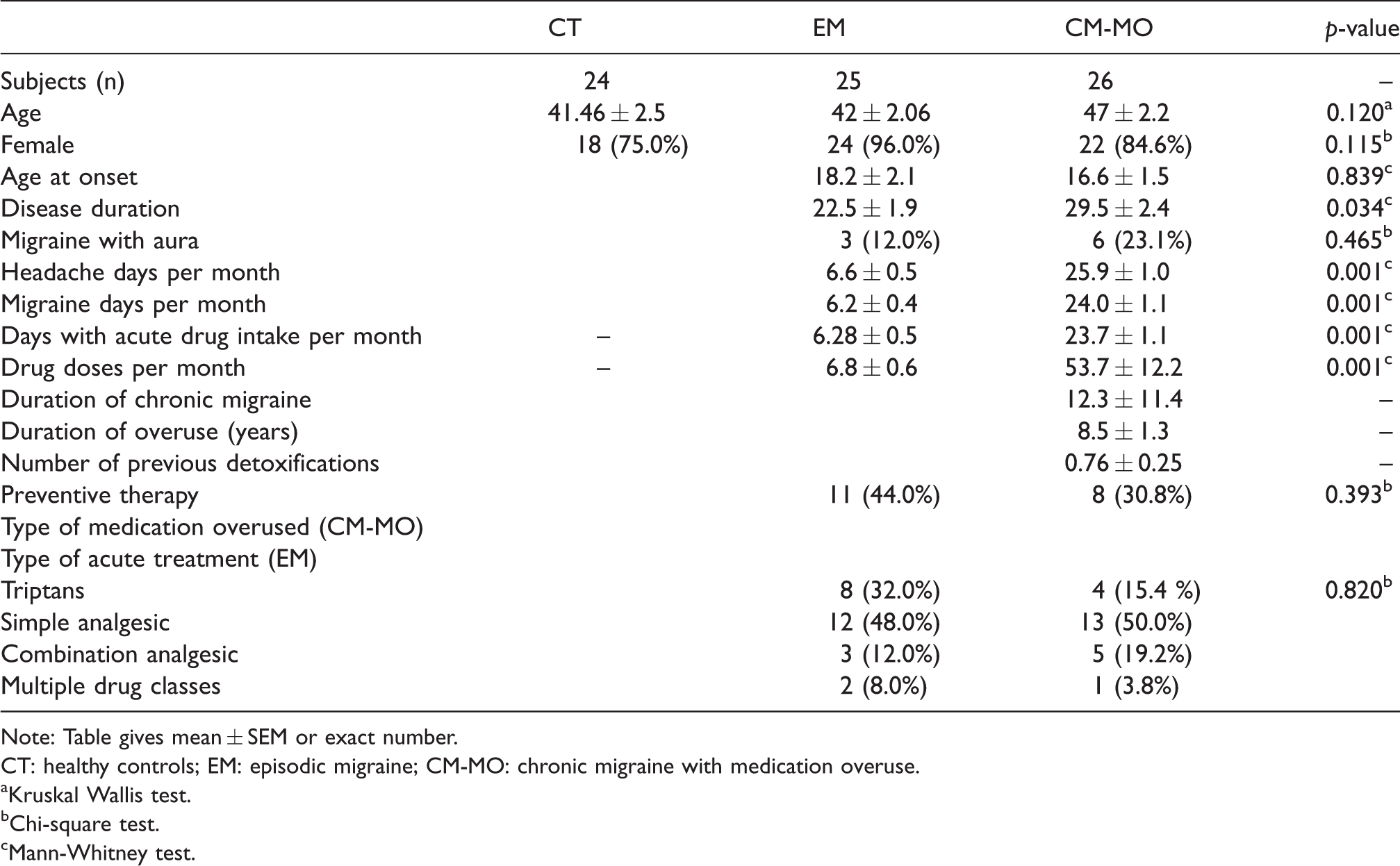
Note: Table gives mean ± SEM or exact number.
CT: healthy controls; EM: episodic migraine; CM-MO: chronic migraine with medication overuse.
aKruskal Wallis test.
bChi-square test.
cMann-Whitney test.
CB1 and CB2 expression levels
Both EM and CM-MO groups showed higher levels of CB1 and CB2 protein expression, as illustrated by the significantly higher percentage of CB1 and CB2 events compared to CT group (Figure 1). The levels of CB1 and CB2 mRNAs were significantly higher in both patient groups when compared to CT (Figure 2), with the CM-MO group showing a significantly higher expression of CB2 mRNA when compared to the EM group (Figure 2, Table 3).

Protein expression of CB1 and CB2. Upper panel: Representative histograms of unstained (upper row) and single-labelled (lower row) cells analyzed by flow cytometry for surface expression of CD45 and CB1 or CB2. Lower panel: Bar plots representing the percentage of CB1+ and CB2+/CD45+ events in the three population groups: Healthy controls (CT), episodic migraine (EM) and chronic migraine with medication overuse (CM-MO). The rectangles represent the scatter plots of flow cytometric measurements showing the percentage of CB1+ and CB2+/CD45+ events as blue dots in migraine groups (EM or CM-MO) and healthy controls (CT). Data are represented as means ± SEM; Kruskal-Wallis test followed by Dunn’s Multiple Comparison Test. p < 0.01 and *p < 0.001 vs. CT.

CB1 and CB2 gene expression in PBMCs of healthy controls (CT), subjects with episodic migraine (EM) and subjects with chronic migraine and medication overuse (CM-MO). Expression was normalized to ubiquitin and data are represented as mean ± SEM. Kruskal-Wallis test followed by Dunn’s Multiple Comparison Test, ***p < 0.001 vs. CT; °°p < 0.01 vs EM.
mRNA levels of the endocannabinoid components assayed in the study.

CT: healthy controls; EM: episodic migraine; CM-MO: chronic migraine with medication overuse; NAPE: N-acylphosphatidylethanolamide-phospholipase D; FAAH: fatty acid amide hydrolase; MAGL: monoacylglycerol lipase; DAGL: diacylglycerol lipase; CB1: cannabinoid receptor 1; CB2: cannabinoid receptor 2.
Note: The p-values presented are adjusted for multiple comparisons according to Bonferroni’s correction. Data are expressed as mean of the relative mRNA expression (RQ) ± SEM.
Gene expression of ES enzymes
FAAH gene expression was significantly lower in both EM and CM-MO patients compared to CT (Figure 3(a), Table 3); moreover, FAAH gene expression was significantly lower in CM-MO patients when compared to EM group. At variance, the levels of NAPE-PLD, MAGL and DAGL mRNAs were significantly higher in both patients’ groups compared to CT. NAPE-PLD, MAGL and DAGL mRNAs levels were also significantly higher in CM-MO compared to EM subjects (Figure 3(b)–(d), Table 3).

Gene expression of ES enzymes in PBMCs of healthy controls (CT), subjects with episodic migraine (EM) and subjects with chronic migraine and medication overuse (CM-MO). mRNA levels of fatty acid amide hydrolase before (FAAH) (a), N-acylphosphatidylethanolamide-phospholipase D before (NAPE-PLD) (b), monoacylglycerol lipase before (MAGL) (c) and diacylglycerol lipase before (DAGL) (d). Expression was normalized to ubiquitin and data are represented as mean ± SEM. Kruskal-Wallis test followed by Dunn’s Multiple Comparison Test, *p < 0.05 and ***p < 0.001 vs CT; °p < 0.05 and °°°p < 0.001 vs EM.
Associations and correlations of ES gene components with clinical and demographic data
The mRNA levels of the genes coding for NAPE, FAAH, MAGL, DAGL, CB1 and CB2 were not associated to gender, type of drug used for the acute treatment of migraine, or use of preventive treatment.
In migraine patients, we found significant and positive correlations between “headache days per month”, “migraine days per month”, “days of acute drug intake per month”, and “doses of acute drug intake per month” and NAPE, MAGL, DAGL, CB1, and CB2 mRNA expression levels (Table 4). A negative correlation was instead detected between FAAH mRNA expression levels and “headache days per month”, “migraine days per month”, “days of acute drug intake per month”, and “doses of acute drug intake per month”. Moreover, CB2 levels were also significantly positively correlated with age and disease duration (Table 4). Headache days and migraine days per month significantly and positively correlated with the days of intake (Spearman’s Rho 0.952, p = 0.001, and Spearman’s Rho 0.998, p = 0.001, respectively) and the doses (Spearman’s Rho 0.948, p = 0.001, and Spearman’s Rho 0.977, p = 0.001, respectively) of acute drug per month.
Matrix of the correlation analysis output.
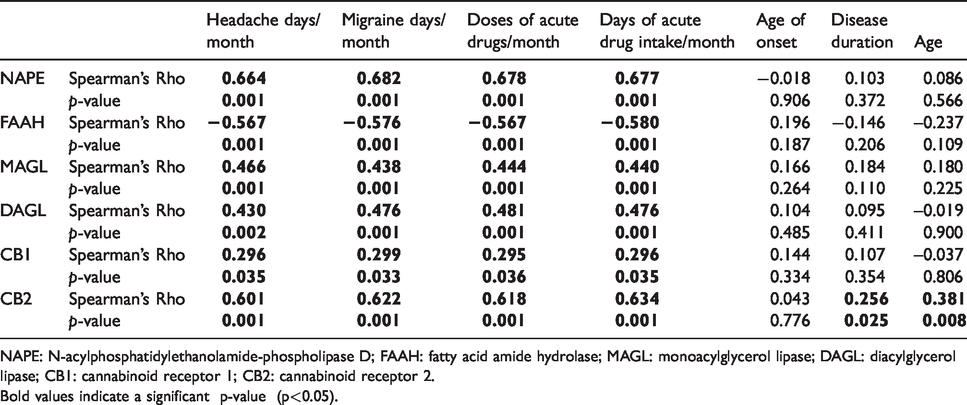
NAPE: N-acylphosphatidylethanolamide-phospholipase D; FAAH: fatty acid amide hydrolase; MAGL: monoacylglycerol lipase; DAGL: diacylglycerol lipase; CB1: cannabinoid receptor 1; CB2: cannabinoid receptor 2.Bold values indicate a significant p-value (p<0.05).
It is worth noting that after correction for age and gender in a linear regression model, the number of migraine days per month was still significantly associated with NAPE (Beta 1.188, p = 0.001) and FAAH (Beta −0.042, p = 0.001) levels, with MAGL (Beta 0.328, p = 0.004) and DAGL (Beta 0.272, p = 0.001) mRNA levels, and CB2 (Beta 0.187, p = 0.001) levels. After correction for age and sex, the number of migraine days per month was no longer associated to CB1 levels (p = 0.064). In CM-MO patients we described a bivariate correlation between “duration of chronic migraine” and CB1 levels, but the correlation did not survive after correction for age and sex.
DNA methylation of ES gene components
DNA methylation analysis did not show any significant differences between patients and CT as regards the detected CpG sites at the promoter region levels of all the evaluated genes (Table 5).
DNA methylation analysis.

Chromosome: chromosome where the probe sequence maps; Position (bp): genomic coordinates indicating the start and end positions of the probe according the design of array; Probe: probe sequence; SE: standard error computed considering the β values between migraine subjects and controls; p-value: detected p-value of the CpG probe averaged between cases and controls; β1 value: mean probe beta value in all migraineurs samples; β2 value: mean probe beta value in control samples; Δβ value: difference of mean probe beta value between migraineurs and controls; Feature: position of each probe relative to CpG Islands-genomic regions in which the frequency of CpG sites is higher than other regions; Shore: 0–2 kb from island; Shelf: 2–4 kb from island; N: upstream (5’) of CpG island; S: downstream (3’) of CpG island; Gene: reference gene; Position (bp): the location of the CpG relative to the CpG island.
Discussion
Blood-based biomarkers have been the object of several investigations, given the accessibility and minimal invasiveness and cost of phlebotomy, and following the idea that pathophysiological events that occur in several neurological disorders may not only originate in central nervous system (CNS) signaling, but may also reflect events taking place at the peripheral level. In this frame, various blood components may represent an ideal “mirror” into the CNS regarding the pathogenesis of neurological disorders (27–29). Endocannabinoids are highly lipophilic and can equilibrate from organs and tissues into the circulation, suggesting that their circulating levels may be an indirect biomarker of the endocannabinoid tone in different tissues. Indeed, circulating endocannabinoids are altered in several conditions including inflammation and pain (30). PBMCs share much of the non-synaptic neuronal biochemical environment and contain the epigenetic enzymes found in most tissues, including neurons (31).
In the present study, we detected transcriptional changes in the ES components in PBMCs of migraine subjects, in some cases with a gradient of severity from EM to CM-MO. Both EM and CM-MO patients showed an increase in CB2 and CB1 protein and gene expression compared to CT. Interestingly, the CB2 gene expression was higher in CM-MO compared to EM patients, showing a generalized adaptive response of ES also in the immune system, as previously found at central level (19). Accordingly, an increased binding of CB1 receptors during the interictal phase was found with PET in EM subjects, especially in brain regions involved in pain modulation (32). The mechanisms related to inflammation may contribute to migraine pathophysiology (33), and thus pro-inflammatory mediators could modulate gene expression of CB receptors by activating transcription factors, such as nuclear factor kappa B (34).
In our study, FAAH gene expression was lower in the PBMCs of CM-MO and EM patients compared with CT subjects. We speculate that this finding reflects a dynamic compensatory mechanism aimed at maintaining higher AEA levels in a challenged system. In this frame, it is worth noting that the alteration of FAAH gene expression was more pronounced in CM-MO patients. This interpretation is indirectly supported by the demonstration by Cupini et al. of lower AEA levels in subjects with CM (16). At partial variance from our present findings, Cupini et al. (17) failed to detect any differences in FAAH activity in platelets of subjects with EM as compared to healthy controls. It must be noted, however, that they used a different methodology (for example high performance liquid chromatography) and did not provide clinical details of their EM population, which prevents comparability of the two studies.
In our study, mRNA levels of NAPE-PLD, one of the enzymes responsible for AEA synthesis, were significantly higher in both patients’ groups compared to CT, with a more marked increase in CM-MO compared to EM subjects. This finding speaks again in favor of a compensatory mechanism aimed at increasing AEA synthesis, possibly to counteract a decreased endocannabinoid tone. In accordance, the expression of the DAGL gene was increased in both migraine groups, though more markedly in CM-MO subjects, which points toward an activation of the synthetic pathway of 2-AG. The apparently surprising increase in MAGL mRNA levels observed in both EM and CM-MO groups may be explained as a more active turnover of 2-AG. In general, the entity and direction of the changes observed in our present study is consistent with an increased turnover of 2-AG and AEA, with a consequent rapid degradation.
The results of the correlation analysis, which show that all peripheral changes observed in gene expression of components of ES are significantly associated with the number of migraine days, further support the idea that an alteration of AEA and 2-AG metabolism is present in migraine subjects, with a gradient of severity that parallels the severity of disease.
In this frame, caution is needed in the interpretation of our findings, since gene expression of the components of ES significantly correlated also with the days of intake of acute drugs for migraine and with the doses of these drugs. It is therefore possible that the changes observed in our patient groups are influenced by acute drugs, rather than being the pure manifestation of a migraine-associated derangement of the ES. Indeed, several of the drugs used for the acute treatment of migraine, namely some non-steroidal anti-inflammatory drugs, may interfere with the ES when considering that 2-AG and AEA are oxygenated by the second isoform of the cyclooxygenase enzyme, COX-2. Nimesulide for instance, which is a preferential inhibitor of COX-2, showed an effect on CB1 receptors activity, though not on AEA or 2-AG levels (35). Ibuprofen is an inhibitor of anandamide metabolism (36), whereas studies on the interaction of paracetamol with the ES have yielded contradictory results (37). The effects of triptans, the migraine-specific acute medications, on the ES have not been studied, to the best of our knowledge, but an interaction between the serotonergic and ES in the processing of somatosensory nociceptive information has been demonstrated in a validated animal model of migraine (10). There is also very scarce information available about the modulation of the ES by chronic exposure to medications for the acute treatment of migraine. Cupini et al. (16) reported that the platelet activity of FAAH was comparable in CM-MO patients and CM patients without MO, thus suggesting that AEA levels may be related to an adaptive response induced by chronic headache itself more than to overused drugs. Interestingly, no significant association was reported among the type of symptomatic drug of overuse and FAAH platelet activity. By contrast, we previously demonstrated that in patients with MO headache, withdrawal from overused medications was associated to a significant reduction in FAAH activity in the platelets (38).
The parallel changes in migraine days and days of acute drug intake observed in our patients do not allow separation of the respective roles of these two variables on the expression of ES components, thus specifically targeted studies are necessary to better define the role of acute medications in the dysregulation of the ES observed in EM and CM-MO patients in the present study.
Our findings show a dysregulation in the ES – both in terms of enzymes and receptors binding – in patients with migraine, thus confirming and expanding on previous studies (18,19,32,38). They also raise the question of the possible role of acute drugs in the observed differences. The answer to this question will be very important, as it will provide a neurobiological basis for the negative impact of acute medication overuse on migraine course and will hopefully pave the way for specific approaches to CM-MO.
Interestingly, the findings obtained in the clinical setting with peripheral biomarkers are largely consistent with the data obtained by our group in the CNS using the animal model of migraine based on nitroglycerin administration. Indeed, we reported that nitroglycerin-induced hyperalgesia was associated with an increased activity of MAGL in the mesencephalon and an increased density of CB binding sites in the mesencephalon and in the hypothalamus (11). It is worth noting that in the same study we found an increase in FAAH activity in the medulla, mesencephalon and hypothalamus, which is apparently in contrast with the changes we detected in the PBMC of our migraine patients. However, since the ES is highly complex, region-specific and cell type-specific, this discrepancy may be only apparent, and may actually reflect different engagement mechanisms and metabolic activation of the ES in different areas of the CNS and in peripheral tissues (39). Our data do not allow further speculation on this issue and additional research will be necessary to unravel this fascinating topic. Taken together, the findings suggest that gene expression of ES components in PBMCs may be a potential biomarker of migraine, although we cannot derive any definite information about the level (central, peripheral or both) of the dysfunction and, at present, we cannot rule out a role of acute medications on the observed peripheral changes in ES components.
To date, there are very few studies about the epigenetic regulation of the ES components in a clinical context. An upregulation of CB1 gene expression has been observed in the blood of patients with different eating disorders, but these alterations were not found to be associated with DNA methylation of the CB1 promoter (40). Moreover, FAAH gene expression in PBMCs of subjects with Alzheimer’s disease was related to a reduced DNA methylation at its promoter compared with healthy subjects (41).
Here, we did not find difference in the methylation level at the CpG unit in ES components of migraineurs compared to CT. It is possible that other epigenetic mechanisms are implicated in gene expression regulation of the ES in migraine. For instance, DNA hypomethylation and histone hyperacetylation can regulate CB1 and CB2 gene expression in different cells of the nervous and immune systems (42).
Gene expression is also regulated by the control of mRNA degradation, since the steady-state concentration of mRNA is determined both by its rates of synthesis and decay. Additionally, the methylation patterns can change over an individual’s lifetime in response to genetic and environmental factors, and thus it cannot be necessarily attributed only to migraine pathogenesis.
Limitations of the study
First, in this study we did not assay the circulating levels of endocannabinoids. Peripheral concentrations of AEA and 2-AG are influenced by several physiological factors (physical activity, stress, depression, anxiety disorders, etc.) and their levels are highly unstable in biological samples. These considerations suggest that circulating levels of endocannabinoids may not represent reliable biomarkers of a trait condition. Second, as discussed above, we cannot exclude a role of MO in the more marked alteration observed in our CM-MO group. The hypothesis seems indeed likely, since we previously demonstrated that withdrawal from overused medications was associated to changes in FAAH activity in the platelets of migraineurs (38). To this end, we are presently running a study aimed at addressing the impact of MO in peripheral changes of markers of ES activity.
Conclusions
Here, we describe a peripheral alteration in gene expression of ES components in both EM and CM-MO migraine subjects. The observed changes were more marked in CM-MO subjects, as compared to EM. Further studies are needed to extrapolate and quantify the possible role of acute medications in the observed changes. Nonetheless, the present findings show for the first time that gene expression profiling of peripheral ES components may represent an interesting potential biomarker of migraine.
Clinical implications
Peripheral gene and protein expression of CB1 and CB2 receptors was increased in all migraineurs Peripheral gene expression of enzymes involved in the AEA and 2-AG metabolism was altered in migraineurs, but more markedly in the chronic subtype Gene expression profiling of peripheral ES components is a potential biomarker of migraine
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: In the last 3 years CT has received honoraria for the participation in in advisory boards for Allergan, ElectroCore, Eli Lilly, Novartis and Teva; she is on the speaker bureau for Allergan, Eli Lilly, Novartis and TEVA; she is PI or collaborator in clinical trials sponsored by Alder, Amgen, Eli-Lilly and Teva. She has received grants from the European Commission, the Italian Ministry of Health and the Italian Ministry of University.
The other authors declare no conflict of interest.
Ethical approval and conduct
The study was approved by the local Ethics Committee (CE Pavia).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Italian Ministry of Health to IRCCS Mondino Foundation, Pavia, Italy (RC19015D).
Patient information and consent
Informed written consent was obtained from each subject before conducting any procedures. Additional information on the study was provided verbally by the study investigator or in a written format.
