Abstract
Introduction
Wound healing disturbances as possible side effects of calcitonin gene-related peptide (CGRP) antibody treatment have been discussed previously but not yet described in humans. Basic research suggests that calcitonin gene-related peptide plays an important role in keratinocyte migration, vascularization and immune response and lack of calcitonin gene-related peptide may lead to impaired wound healing.
Case
A 51-year-old female migraine patient was treated with the CGRP receptor antibody erenumab for 6 months, which led to a relevant reduction of migraine days. During the treatment, two periods of severely impaired wound healing occurred after a trivial skin injury without spatial relation to the injection site. Skin biopsy confirmed a deep perivascular and interstitial lymphohistiocytic infiltrate with admixed eosinophils, ulceration of the epithelium, a heavy edema of the papillary dermis and focally thrombosed vessels.
Conclusion
Impaired wound healing might be relevant side effects of CGRP antibody therapy and anamnesis within the course of treatment should also include possible observation of impaired wound healing or planned surgery.
Abbreviations
CGRP: calcitonin gene-related peptide; TNF: tumor necrosis factor-α; VEGF: vascular endothelial growth factor.
Introduction
Calcitonin gene-related peptide (CGRP) antibodies are a new and promising therapeutic option in prophylactic migraine treatment and are thought to have fewer side effects compared to conventional migraine prophylactics.
CGRP and CGRP receptors are located in trigeminal neurons, including the trigeminovascular system (1). During migraine attacks CGRP is released there. In addition, it may also trigger migraine attacks (2). CGRP itself modulates nociception and maintains neurogenic inflammation, which leads to pain sensitization. With regard to the above-mentioned features of CGRP, CGRP antagonistic antibodies have become an encouraging novel option to treat episodic and chronic migraine.
Despite its involvement in migraine pathophysiology, CGRP is critical for physiological processes as well. It has strong vasodilatative properties, which might mediate protective effects in the cardiovascular system and in the gastrointestinal system especially during inflammatory states.
In wound healing processes, CGRP is critical based on several aspects. It promotes revascularization by upregulating vascular endothelial growth factor (VEGF) expression, reduces the expression of inflammatory mediators such as tumor necrosis factor-α (TNF-α) and macrophage infiltration, and it enhances proliferation of keratinocytes (3–5).
Several studies proved good tolerability of these monoclonal antibodies (6). Reported side effects are injection-site pain, constipation or upper respiratory tract infections. Although clinical data are promising regarding CGRP inhibitors in migraine patients, the ubiquitous distribution of CGRP and its receptors underline the need to continuously screen for potential side effects.
We present a migraine patient who developed a possible CGRP receptor antibody-associated skin wound healing disturbance twice within the course of drug application. To our knowledge, this is the first case of possible CGRP receptor antibody-related skin wound healing impairment as a systemic side effect of CGRP antibody therapy.
Case presentation
In November 2018, a 51-year-old, otherwise healthy Caucasian patient presented at our headache outpatient clinic with severe migraine refractory to common treatment. She had been treated in our center for almost 10 years because of a migraine that had started during childhood. Several prophylactic medications had been tested but did not substantially reduce migraine frequency, were effective only for a short time, or were associated with intolerable side effects.
At the time of consultation, she suffered from 13 migraine days per month. Recurrent headache and an attack duration of 2 days each were also reported. Zolmitriptan was used to treat attacks on 10 days per month. Opipramol 50 mg was taken for treatment of sleep disturbances. No other disorders or medication intake were reported by the patient.
Following informed consent, prophylaxis with erenemab 70 mg subcutaneously was started every 4 weeks. Follow-up after 3 months showed a consistent reduction of migraine headache attacks from 13 to 5 days per month (for details see Figure 1). Initially, no side effects were reported by the patient.

Migraine frequency and preventive medication from 2009 to 2019.
In the middle of April 2019, the patient noticed a singular erythematous papular skin lesion with a diameter of one centimeter at the inner surface of the left forearm. Erenumab had been injected in the thigh 13 days before the occurrence of the lesion. The patient presumed the skin lesion to be the consequence of an insect bite, as no other trauma could be remembered. In that context, the patient remembered having had an annular necrosis on the left lower leg after a similar lesion at the beginning of this year, which healed after approximately 8 weeks with residual post-inflammatory hyperpigmentation (see Supplemental Figure S1).
The patient had never taken glucocorticoid steroids. She is a non-smoker and does not suffer from a peripheral vascular disease. She had no febrile infection, no tick bite, no trauma and no burns. In the past, she had not noticed any relevant reactions to insect bites or any problems with wound healing.
In the following 6 days, the lesion increased rapidly in diameter and showed signs of inflammation (e.g. flush), however without fester. Necroses formed around the efflorescence. The main symptoms were itching and less pronounced burning pain. A dermatologist was consulted in late April 2019, who collected a smear from the wound that did not reveal any pathogens. Topical treatment with gentamycin and bethamethasone twice daily was initiated, but the lesion did not improve. Therefore, topical treatment with triamcinolone and clioquinol was started once to twice daily in the middle of May. During this period a picture was taken by the patient (see Supplemental Figure S2).
At the end of May, the patient presented at our headache outpatient clinic for follow-up and reported further improvement of the migraine (see Figure 1).
Due to the skin lesion, the patient was referred to the specialized wound ambulance at our Department of Dermatology. The lesion presented as three crescent-shaped, almost annular necroses with slightly erythematous skin in the center (Figure 2). The necroses had a length of approximately 5 cm and a width of approximately 2 cm. The wound margins and surroundings were unsuspicious, there was no evidence of acute inflammation.
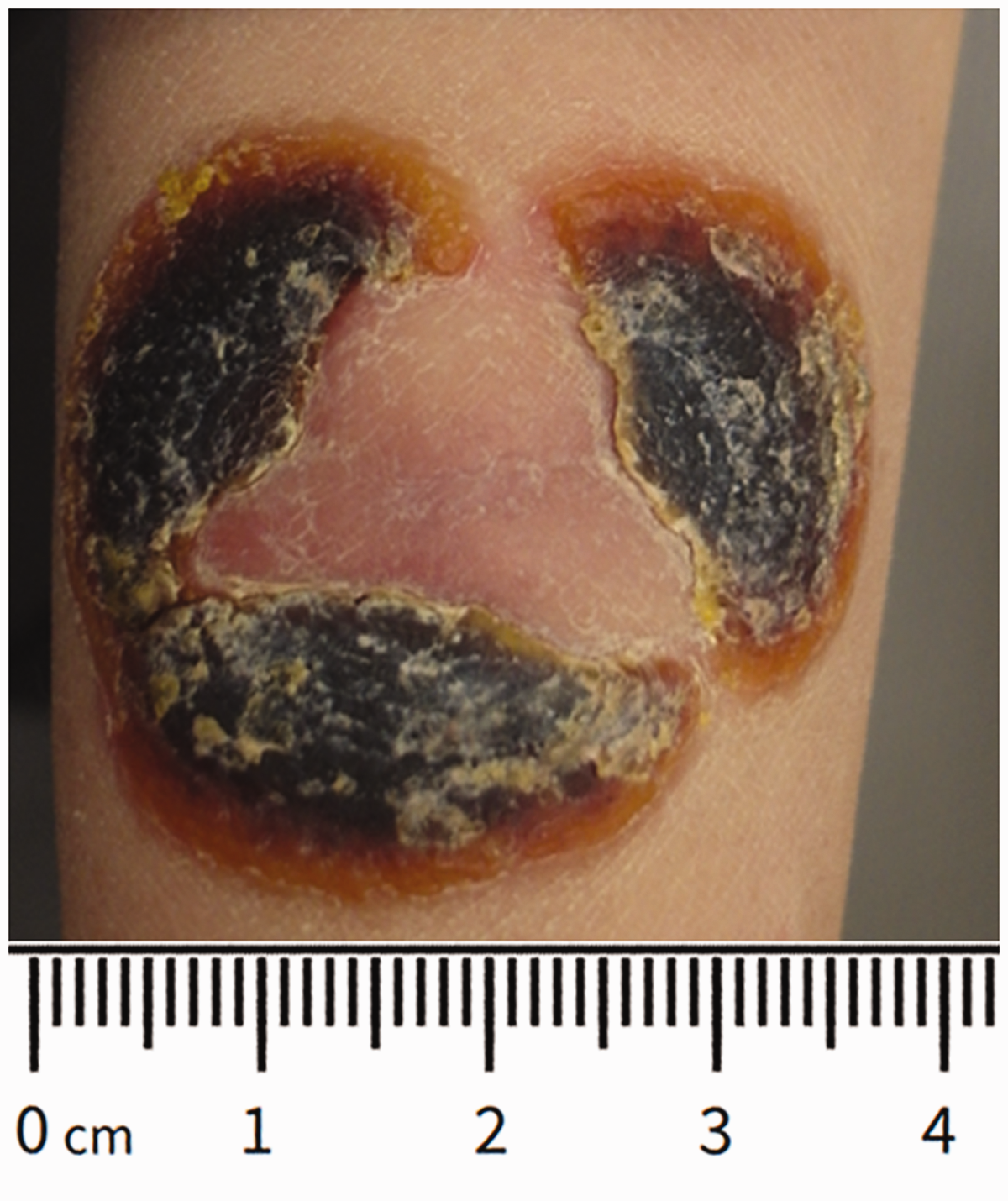
Detail view of the second efflorescence. Annular dry necroses with light erythematous skin in the center (29 May 2019).
Skin biopsy revealed a pseudocarcinomatous hyperplasia of the epithelium ulcerated to one side of the biopsy. The dermis showed a pronounced papillary edema. In the upper and middle corium, lymphohistiocytic inflammatory infiltrates with plasma cells and eosinophilic granulocytes were found, which were also abundant in the deep corium. The vessels of the upper plexus were thrombosed in the area of the corium and distinct erythrocyte extravasations were found in the surrounding area (Figure 3).

Histological findings of the skin biopsy. There is a heavy dermal inflammation, the epithelium reveals pseudocarcinomatous hyperplasia (a). In addition there is a strong papillary edema with extravasated erythrocytes (b). In the deeper dermis there are perivascular and interstitial lymphohistiocytic infiltratations with numerous admixed eosinophilc granulocytes (c). The vessels in the upper dermis show eosinophilic material in their lumina with numerous surrounding extravasated erythrocytes and a lymphohistiocytic infiltrate with neutrophils (d). Hematoxylin and eosin stain x20 (a), x200 (b), x200 (c), x200 (d).
The clinical examination found no evidence for the presence of a systemic dermatosis, an underlying rheumatic or systemic autoimmune disease such as vasculitis or autoimmune collagen vascular diseases.
The laboratory data including routine blood test results were all within the normal range. In addition, risk factors for skin wound healing impairment such as diabetes or uremia were unremarkable. Workup for autoimmune disorders (e.g. vasculitis, antiphospholipid syndrome, autoimmune connective tissue disorders) revealed completely unremarkable titers of antibodies and rheumatic markers were within the normal values (see Supplemental Table 1). Nerve conduction studies provided no evidence of neuropathy.
Since we judged the wound healing disturbances as probably related to erenumab, therapy was stopped. Eight months after stopping erenumab, both wounds still a have a residual light hyperpigmentation, but the patient reported no further wound healing disturbances.
Discussion
We present to our knowledge the first case of impaired wound healing with probable relationship to the treatment with a CGRP receptor antibody (erenumab), which developed without temporal and spatial relation to the injection in a migraine patient. The patient did not report any other side effects under therapy. Before treatment with erenumab, the patient had neither impaired wound healing nor hyperergic reactions regarding insect bites.
In addition to the absence of specific clinical symptoms and serological findings suggestive of autoimmune diseases including vasculitis, antiphospholipid syndrome and rheumatic disorders, there was no evidence of an underlying dermatosis or several systemic diseases (e.g. diabetes mellitus, polyneuropathy) as causal explanation for the impaired wound healing or as a causal predisposition to the spontaneous development of wounds.
Potential clinical differential diagnoses include vasculitis, pyoderma gangrenosum or antiphospholipid syndrome. Histologically, in the present case, there were perivascular infiltrates, edema and erythrocyte extravasates but no changes in the vessel walls or neutrophilic infiltration with leucocytoclasia as required for the diagnosis of true vasculitis. Thus, in this case, a vasculitis can be excluded by histological and clinical facts.
Both primary and secondary antiphospholipid syndrome typically show livedo racemosa and ulceration of skin, in rare cases subcutaneous nodules or ulceration. These clinical symptoms are usually long-lasting, and do not disappear without therapy or heal without scarring. Neither these symptoms nor associated antibodies could be detected in this case.
Pyoderma gangrenosum is clinically characterized by a fast-growing ulcer with violaceus borders, it heals with leaving scars. Histologically, pyoderma gangrenosum reveals a primarily neutrophilic inflammation in the papillary dermis. The clinical presentation of the current case did not match these clinical and histological criteria.
In conclusion, neither the above-mentioned nor other potential clinical differential diagnoses were fulfilled in the present case.
CGRP has been shown to have different effects of the various cell types relevant for skin inflammation: It triggers keratinocyte proliferation and conveys a rather Th17-prone inflammatory environment, thus supporting psoriasis plaque development and maintenance (7). However, regarding the effects on the endothelium, CGRP has been shown to rather downregulate proinflammatory cytokines (7).
Therefore, we speculate that the inhibition of CGRP might have led to unrestricted release of inflammatory cytokines from the endothelium – maybe after minimal trauma or an insect bite – finally causing a deep perivascular and lymphohistiocytic infiltration. As a consequence of endothelium-driven inflammation after inhibition of CGRP by the receptor antibody, the disturbed inflammatory circuit might have led to the thrombi seen in the vessel lumina. All changes present in the upper part of the dermis; for example, edema of the papillary dermis and the pseudocarcinomatous hyperplasia of the epithelium, are known consequences of focally occluded vessels.
Although a causal relationship between the impaired wound healing in this case and the therapy with CGRP receptor antibody remains unproven, it is most likely based on pathophysiological aspects and the function of this peptide with regard to wound healing and the time of occurrence. The present case seems to represent a CGRP receptor antibody-induced altered course of wound healing. There may be several reasons why this was not reported as a side effect before. One reason might be that wounds resulting from a trivial injury and possibly delayed wound healing were not seen in relation to the substance and therefore were not interpreted as worth reporting. This may especially account for the phase 2 and phase 3 trials, where patients possibly did not report little wounds as adverse events maybe as these may not have been bothersome, or even in fear of being excluded from the trial. Furthermore, this may indeed be a very rare side effect and it cannot be ruled out that the patient in our case report might have had a predisposition for wound healing disturbances that has not been identified yet and that has now been unmasked under erenumab treatment.
Clinicians should be aware and vigilant in diagnosing this complication. Therefore patients undergoing CGRP blocking therapy should be more intensively monitored for impaired wound healing, as many patients would not report these side effects by themselves when they occur for the first time.
Clinical implications
We present a migraine patient who developed a possible CGRP receptor antibody (erenumab) associated skin wound healing disturbance twice within the course of therapy. This is the first case of possibly CGRP receptor antibody-related skin wound healing impairment as a systemic side effect of CGRP antibody therapy. Impaired wound healing might be relevant side effects of CGRP antibody therapy and anamnesis within the course of treatment should also include possible observation of impaired wound healing or planned surgery.
Supplemental Material
sj-pdf-1-cep-10.1177_0333102420933571 - Supplemental material for Impaired wound healing in a migraine patient as a possible side effect of calcitonin gene-related peptide receptor antibody treatment: A case report
Supplemental material, sj-pdf-1-cep-10.1177_0333102420933571 for Impaired wound healing in a migraine patient as a possible side effect of calcitonin gene-related peptide receptor antibody treatment: A case report by Sebastian Wurthmann, Steffen Nägel, Eva Hadaschik, Swantje Schlott, Armin Scheffler, Christoph Kleinschnitz and Dagny Holle in Cephalalgia
Supplemental Material
sj-pdf-2-cep-10.1177_0333102420933571 - Supplemental material for Impaired wound healing in a migraine patient as a possible side effect of calcitonin gene-related peptide receptor antibody treatment: A case report
Supplemental material, sj-pdf-2-cep-10.1177_0333102420933571 for Impaired wound healing in a migraine patient as a possible side effect of calcitonin gene-related peptide receptor antibody treatment: A case report by Sebastian Wurthmann, Steffen Nägel, Eva Hadaschik, Swantje Schlott, Armin Scheffler, Christoph Kleinschnitz and Dagny Holle in Cephalalgia
Footnotes
Author contributions
SW initiated the case report after observing the wound healing disturbance and reviewing the literature. He summarized and analysed the data and was a major contributor to writing the manuscript. SN and AS reviewed the case report and were major contributor to the manuscript. EH interpreted the skin biopsy and gave final approval of the version to be published. CK reviewed the manuscript and was a major contributor in the discussion of the case report. SS did the biopsy and reviewed the manuscript. DH analysed the data, revised the case report critically for important intellectual content and was a major contributor to the manuscript.
Consent
Informed consent was obtained from the patient to utilize her medical records and case for the purpose of this report. All information has been appropriately de-identified.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SW has received honoraria for contribution to advisory boards or lectures from Allergan and Teva. He received fees for contribution to advisory boards from Novartis, which manufactures the drug that is mentioned in this case report.
SN has received honoraria for contribution to advisory boards or lectures from Teva. He received fees for contribution to advisory boards from Novartis, which manufactures the drug that is mentioned in this case report.
EH declares no conflict of interest.
SS declares no conflict of interest.
AS declares no conflict of interest.
CK declares no conflict of interest.
DH has received research grants from Gruenenthal and Allergan, and the German Ministry for Education and Research (BMBF). She received honoraria for contribution to advisory boards from Lilly, Teva and Allergan. She received fees for contribution to advisory boards from Novartis, which manufactures the drug that is mentioned in this case report.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
