Abstract
Background
Immunogenicity of erenumab, a human anti-calcitonin gene-related peptide receptor monoclonal antibody developed for migraine prevention, has been evaluated throughout clinical development.
Methods
Integrated post hoc analysis assessing immunogenicity of erenumab across six clinical trials in patients with episodic and chronic migraine (N = 2985). Anti-erenumab antibody incidence and potential impact on pharmacokinetics, efficacy, and safety were evaluated in short-term (double-blind treatment phase 12–24 weeks) and long-term (double-blind treatment phase plus extensions of up to 5 years) analyses.
Results
Anti-erenumab binding antibody incidence was low with few patients developing neutralizing antibodies. Antibody responses were mostly transient with low magnitude. Binding antibodies developed as early as 2–4 weeks after the first dose; the majority developed within the first 6 months and very few after the first year. Serum concentrations of erenumab in antibody-positive patients were generally lower than, but within the range of, antibody-negative patients. There was no impact of anti-erenumab antibodies on erenumab efficacy or safety with no differences between antibody-positive and antibody-negative patients in change in monthly migraine days or adverse event rates.
Conclusions
This pooled analysis showed that immunogenicity had no meaningful clinical impact on efficacy or safety of erenumab in patients with migraine.
Introduction
Immunogenicity is the propensity of the therapeutic protein product to generate immune responses to itself and to related proteins or to induce immunologically related adverse clinical events (1). Numerous factors may contribute to the immunogenicity of protein therapeutics including both product-related (e.g., drug’s amino acid sequence, mechanism of action, and attribute properties) and patient-related factors (e.g., disease indication, dosing regimens, prior and concomitant medications). Immunogenicity is usually monitored by anti-drug antibody (ADA) formation during clinical development, and the consequence of immunogenicity can range from no apparent clinical effect to altered pharmacokinetics, reduced drug efficacy, and/or increased risk of immune-mediated adverse events (2–5). Therefore, immunogenicity is recognized as a major potential risk for drug development.
Erenumab (in the US, erenumab-aooe) is a fully human anti-calcitonin gene-related peptide (CGRP) receptor monoclonal immunoglobulin G2 (IgG2) antibody developed for migraine prevention (6). Development of anti-erenumab antibodies would be expected to be less frequent when compared to therapeutic antibodies containing non-human coding sequences. Following regulatory guidance (1,7–9), immunogenicity was assessed longitudinally throughout clinical studies with reliable and robust methods to define the incidence of anti-erenumab antibody development and identify any potential clinical consequences.
In this post hoc analysis, we evaluated the immunogenicity profile of erenumab across six Phase 2 and 3 clinical trials with long-term extensions to understand the potential clinical consequences in terms of pharmacokinetics, efficacy, and safety.
Methods
Patients and clinical studies
This was a post hoc analysis of data from the initial erenumab global clinical development program and from the clinical development program in Japan. Adults (aged 18–65 years) with episodic migraine (EM; ≥ 4 to < 15 migraine days per month and < 15 headache days per month) or chronic migraine (CM; ≥ 15 headache days per month of which ≥ 8 were migraine days) were enrolled in four multicenter studies (three in EM and one in CM) conducted in North America, Europe, Russia, and Turkey and two multicenter studies (one in EM and one in EM and CM) conducted in Japan. Detailed inclusion and exclusion criteria for each study has been reported (10–16).
This analysis included six double-blind, randomized trials and extension phases with varying erenumab dosage regimens and treatment durations (Figure 1). All studies included a screening visit, 4-week baseline phase, 12- or 24-week placebo-controlled, double-blind treatment phase (DBTP), an extension phase comprising an open-label treatment phase (OLTP) or dose-blinded active treatment phase (ATP) ranging from 28 weeks to 5 years. Safety follow-up visits were required 12–16 weeks after the last dose of erenumab (
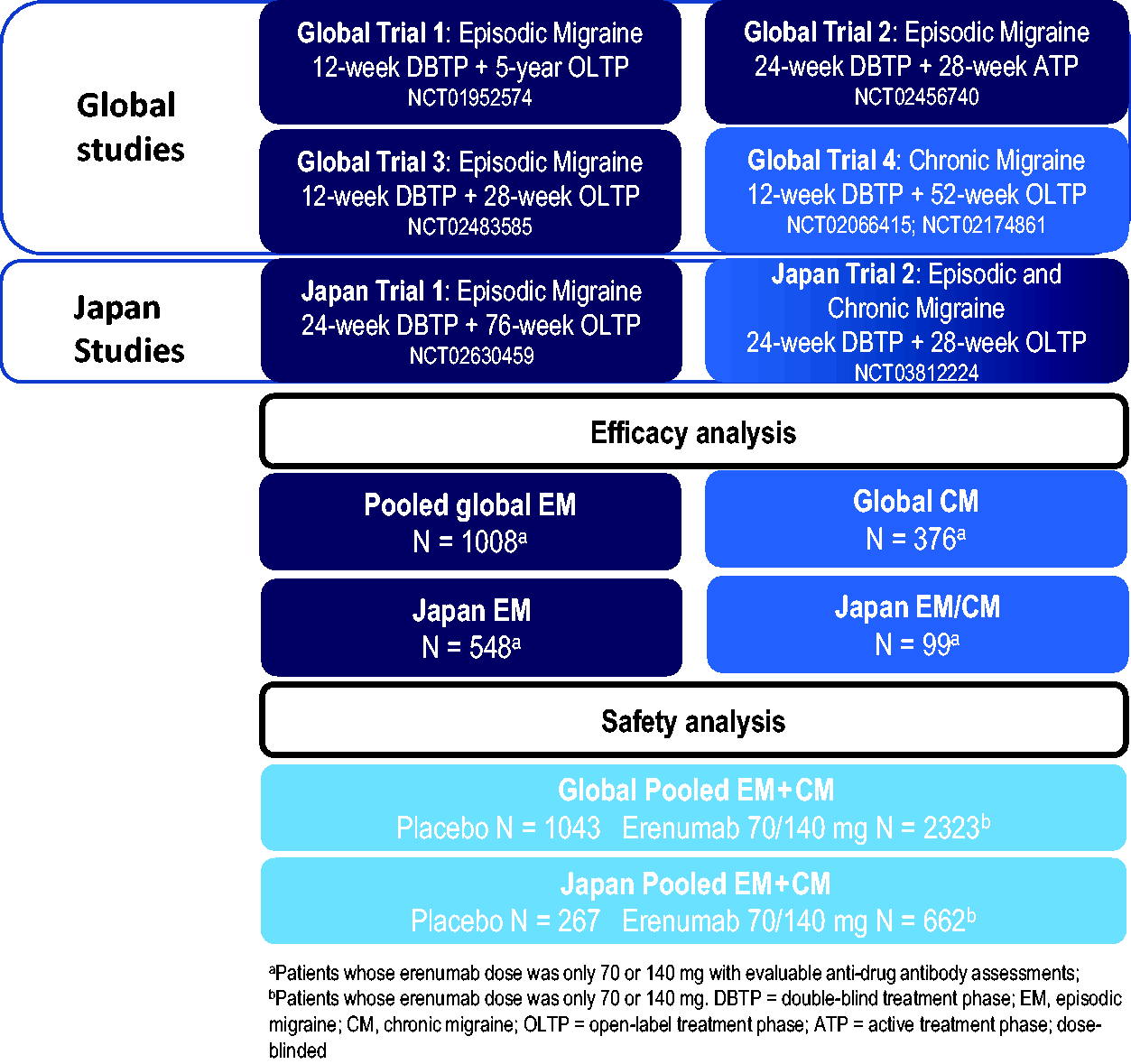
Study design.
Studies were conducted in compliance with the Declaration of Helsinki, International Council for Harmonisation Guidelines for Good Clinical Practice, and local country regulations. The study protocols were approved by the Institutional Review Board or Independent Ethics Committee at each center. Patients provided written informed consent before study initiation. Site investigators collected the data and Amgen conducted the data analyses.
Anti-erenumab antibody assessment
The immunogenicity testing strategy and methods for erenumab generally followed the recommendations of white papers and draft regulatory guidance effective at the time of method validation (1,7–9). In all six studies, serum samples for immunogenicity analysis were collected at baseline (before the first drug administration) and at regular intervals ranging from every 2 weeks to every 6 months over the study period (
Anti-erenumab binding antibodies were detected using a validated electrochemiluminescence (ECL)-based bridging immunoassay on the Meso Scale Discovery (MSD) platform and followed a two-tiered assay approach (screening and specificity) with an assay sensitivity of 24.73 ng/mL (specificity assay) and drug tolerance (specificity assay) of 100 μg/mL or 10 μg/mL erenumab at an anti-erenumab antibody concentration of 500 ng/mL or 100 ng/mL, respectively. The assay met criteria for the use of signal-to-noise (S/N) values as an alternative to titer for characterization of the magnitude of the ADA response (17). The screening assay was performed on serum samples to detect the presence of binding antibodies to erenumab, using biotinylated and ruthenylated erenumab as capture and detection reagents, respectively. Specificity was confirmed in samples with S/N greater than the screening assay cut-point using the same assay in the presence of excess unlabeled erenumab. An S/N reduction greater than the depletion cut-point was reported as positive for the presence of anti-erenumab binding antibodies.
Anti-erenumab neutralizing antibodies were detected using a validated cell-based assay and followed a two-tiered assay approach (screening and confirmatory) with a sensitivity of 0.553 μg/mL and drug tolerance of 0.1 μg/mL erenumab at an anti-erenumab antibody concentration of 1.14 μg/mL. The screening assay used an SK-N-MC, a human neuroblastoma cell line expressing α-CGRP receptor, in which erenumab inhibits intracellular cAMP production induced by α-CGRP binding to α-CGRP receptor. Increased intracellular cAMPin the presence of erenumab and α-CGRP indicated that the inhibitory activity of erenumab was neutralized. This neutralizing activity was further confirmed, by the reversal of erenumab neutralization following protein G/L depletion, indicating the presence of anti-erenumab neutralizing antibodies. Intracellular cAMP producation was measured using an MSD assay kit that is based on competitive binding with ruthenium-labeled cAMP.
Assessment of serum erenumab concentrations
Pharmacokinetics (PK) sampling times for the six clinical studies are shown in
ADA impact analysis
To assess the potential impact of anti-erenumab antibodies on drug exposure, PK data were evaluated from patients whose erenumab dose was 70 mg or 140 mg throughout the four global double-blind, placebo-controlled erenumab studies and their extensions, and PK data from the two Japan studies were evaluated separately. For the global studies, the short-term analyses were assessed at week 12 within the DBTP, and the long-term analyses were assessed at week 36, 52, and 64 during the entire studies. For the Japan studies, the short-term analyses were assessed at week 12 and 24 within DBTP, and the long-term analyses were assessed at week 36 and 52 during the entire studies. The serum concentrations of erenumab by ADA status and levels (measured by S/N values) were assessed over time within each dose group (70 mg or 140 mg). To qualify the assessment, the ADA-negative groups only include pure ADA-negative patients with an ADA-negative status by end of DBTP (short-term) or by the last on-study assessment (long-term). The ADA-negative patients at each assessment timepoint were from this pure ADA-negative group with observed PK data at the corresponding visit. The ADA-positive patients at each assessment timepoint were those who had observed PK data and ADA-positive status at the corresponding visit. For the long-term analysis, the patient had to be receiving the indicated dose (70 or 140mg) for at least 3 months before the assessment timepoint.
To assess ADA impact on efficacy, the change from baseline in monthly migraine days (MMD) from patents whose erenumab dose was only 70 mg or 140 mg throughout the six double-blind, placebo-controlled erenumab studies and their extensions was assessed over time by ADA status and S/N values. In the global studies, efficacy data from the CM study are presented separately from the pooled data from the EM studies. Data from the global studies are presented separately from the Japan studies. To qualify the assessment, the ADA-negative groups only include pure ADA-negative patients who never developed anti-erenumab antibody throughout the assessment period (short-term analysis by end of DBTP and long-term analysis by the last on-study assessment). The ADA-negative patients at each assessment timepoint were from this pure ADA-negative group with observed efficacy data at the corresponding visit. The ADA-positive patients at each assessment timepoint were those who were tested ADA-positive and with observed efficacy data at the corresponding visit. Patients with pre-existing antibodies were excluded from the efficacy analysis.
To assess the potential impact of anti-erenumab antibodies on clinical safety, injection site reaction, hypersensitivity, and immune system disorder adverse events (AE) were identified based on injection site reaction Amgen Medical Query (AMQ), hypersensitivity standardized MedDRA (version 22.1 [global] or 23.1 [Japan]) query (SMQ, narrow and broad), and immune system disorders System Organ Class (SOC). The safety data were assessed for the placebo-treatment group and the erenumab-treatment group separated by ADA status. These AEs were selected based on the biologic plausibility of a potential association with immunogenicity. Exposure-adjusted patient incidence rates (per 100 patient-years) of AEs were calculated as total number of patients reporting at least one event divided by total time at risk for reporting the AEs (in years, summed across all patients) multiplied by 100. Time at risk only included time to first occurrence of the AE. If a patient did not have the corresponding AE to report, then time at risk included time to safety follow-up visit. Multiple occurrences of the same event for an individual are counted as single events within a treatment group.
Data availability
Qualified researchers may request data from Amgen clinical studies. Complete details are available at the following: https://wwwext.amgen.com/science/clinical-trials/clinical-data-transparencypractices/
Results
ADA incidence
Short-term analysis of the double-blind treatment phase (DBTP)
During the DBTP of the six migraine studies, the incidence of anti-erenumab binding antibody development was low, with only a few patients developing neutralizing antibodies (Table 1). For the global studies, 5.0% (69/1389) of patients developed anti-erenumab binding antibodies and 0.2% (3/1389) developed anti-erenumab neutralizing antibodies. For the two Japan studies, 2.7% (11/401) of patients developed anti-erenumab binding antibodies with no neutralizing antibody development.
Anti-erenumab antibody incidence during short-term (double-blind treatment phases) and long-term (entire studies) analysis

Data reported as number n (% = n/N * 100) unless otherwise indicated; N = Number of patients whose erenumab dose was only 70 mg or 140 mg during the indicated treatment period and had a result at indicated timepoint.
aLong-term analysis included overall data from entire studies (double-blind treatment phase and extension phases).
bNegative or no result at baseline.
cNegative result at last on-study assessment time point during the indicated treatment period; percentage was calculated using the number of patients who developed binding or neutralizing antibodies, respectively, as denominator; Of the two patients who remained neutralizing antibody positive by end of the studies, one refused to participate in antibody follow-up tests and one withdrew from the study earlier.
The incidence of developing anti-erenumab antibody was lower in the 140-mg group compared with the 70-mg group. In the global studies, the incidence of anti-erenumab antibody development during the DBTPs was 6.3% (56/885) among patients receiving the 70 mg dose of erenumab (three of whom had in vitro neutralizing activity) and 2.6% (13/504) among patients receiving the 140 mg dose of erenumab (none of whom had in vitro neutralizing activity). Comparable results were obtained from the two Japan studies. In the Japan studies, the incidence of anti-erenumab antibody development during the DBTPs was 4.2% (11/264) among patients receiving the 70 mg dose of erenumab and 0% (0/137) among patients receiving the 140 mg dose of erenumab. None of the binding ADA-positive patients developed neutralizing antibodies.
Long-term analysis (DBTP + extension phase)
In the long-term analysis, the incidence of anti-erenumab binding antibody development remained low (8.0% [185/2303] in the global studies and 3.9% [26/661] in the Japan studies) among patients whose erenumab dose was only 70 mg or 140 mg throughout the entire studies (Table 1). Among the 185 patients who developed anti-erenumab binding antibodies in the global studies, 8 developed neutralizing antibodies. Among the 26 patients who developed anti-erenumab binding antibodies in the Japan studies, no patient developed neutralizing antibodies. In addition, 3 patients (0.1%) in the global studies and 1 patient (0.2%) in the Japan studies had pre-existing antibodies (non-neutralizing) prior to the first erenumab dose.
ADA kinetics
ADA onset
Anti-erenumab binding antibodies developed as early as 2–4 weeks after the first dose of erenumab, with the majority developing within the first 6 months post erenumab exposure (65.4% [121/185] in the global studies and 57.7% [15/26] in the Japan studies), and only a few with onset after the first year (7.6% [14/185] in the global studies and 11.5% [3/26] in the Japan studies) (Table 2).
ADA onset in developing anti-erenumab antibody-positive patients during the long-term analysis (entire studies).

Data reported as number n (% = n/N1*100). N1 = Number of patients who developed anti-erenumab binding antibody and whose erenumab dose was only 70 mg or 140 mg during the entire studies.
aTime from erenumab exposure to the first occurrence of developing binding antibody-positive readout.
ADA duration
Most developing anti-erenumab antibody responses were “transient” with a high reversion rate; 51.9% (96/185) of patients in the global studies and 65.4% (17/26) of patients in the Japan studies who developed binding ADAs reverted to a binding ADA-negative status by the last on-study assessment (Table 1 and Table 3). In addition, 39.5% (73/185) of patients in the global studies and 46.2% (12/26) of patients in the Japan studies developing ADAs had a single ADA-positive timepoint during the entire study (Table 3).
S/N values over time in developing anti-erenumab antibody-positive patients during the long-term analysis (entire studies).a

Data are shown as number n (% = n/N * 100) [% = n/N1 * 100]; N = Number of patients with a post-baseline ADA result and whose erenumab dose was only 70 mg or 140 mg during the entire studies. N1 = Number of patients who developed anti-erenumab binding antibody from N.
aBy the last on-study ADA assessment timepoint.
bExclude developing ADA-positive exactly once.
Of the 8 patients from the global studies who developed neutralizing antibodies, 75.0% (6/8) reverted to a neutralizing antibody-negative status by the last on-study assessment. One of the two patients who remained neutralizing antibody positive refused to participate in antibody follow-up tests and one withdrew from the study earlier.
ADA magnitude
The magnitude of the ADA response, characterized using the S/N value from binding ADA-positive samples as an alternative to titer (17), was low in the majority of anti-erenumab binding antibody-positive patients (Figure 2). Of the 188 anti-erenumab binding antibody-positive patients (185 developing and 3 pre-existing) from the global studies, 80.3% (151/188, including the 3 pre-existing antibody-positive patients) had a maximum S/N ratio (S/Nmax) ≤ 5 and 93.6% (176/188) ≤ 20 (Figure 2). Of the 27 anti-erenumab binding antibody-positive patients (26 developing and 1 pre-existing) from the Japan studies, 26 (96.3%, including the patient with pre-existing) had S/Nmax ≤ 5, and one had S/Nmax of 18.0 (data not shown). Moreover, for the ADA-positive patients with more than one ADA-positive timepoints, majority had S/Nmax before the last ADA-positive timepoint (Table 3).

S/Nmax in anti-erenumab binding antibody-positive patients from the global studies.
The kinetics of ADAs had similar pattern regardless of study or dosage (data not shown).
Impact of ADA on pharmacokinetics
Potential impact of anti-erenumab antibodies on erenumab pharmacokinetics was assessed by comparing serum trough concentrations of erenumab over time by anti-erenumab antibody status and levels within each dose group (Figure 3). In the global studies, the serum concentrations of erenumab in the anti-erenumab antibody-positive patients were 30–70% lower according to the mean trough concentrations, but within the range of the anti-erenumab antibody-negative patients, when compared to the same time point within the same dose group. While limited numbers, the patients who developed anti-erenumab neutralizing antibodies or binding ADAs with higher S/N values were more likely to have lower drug exposure than those ADA-negative patients. Similar ADA impact on PK results were observed in the Japan studies (Figure 3).
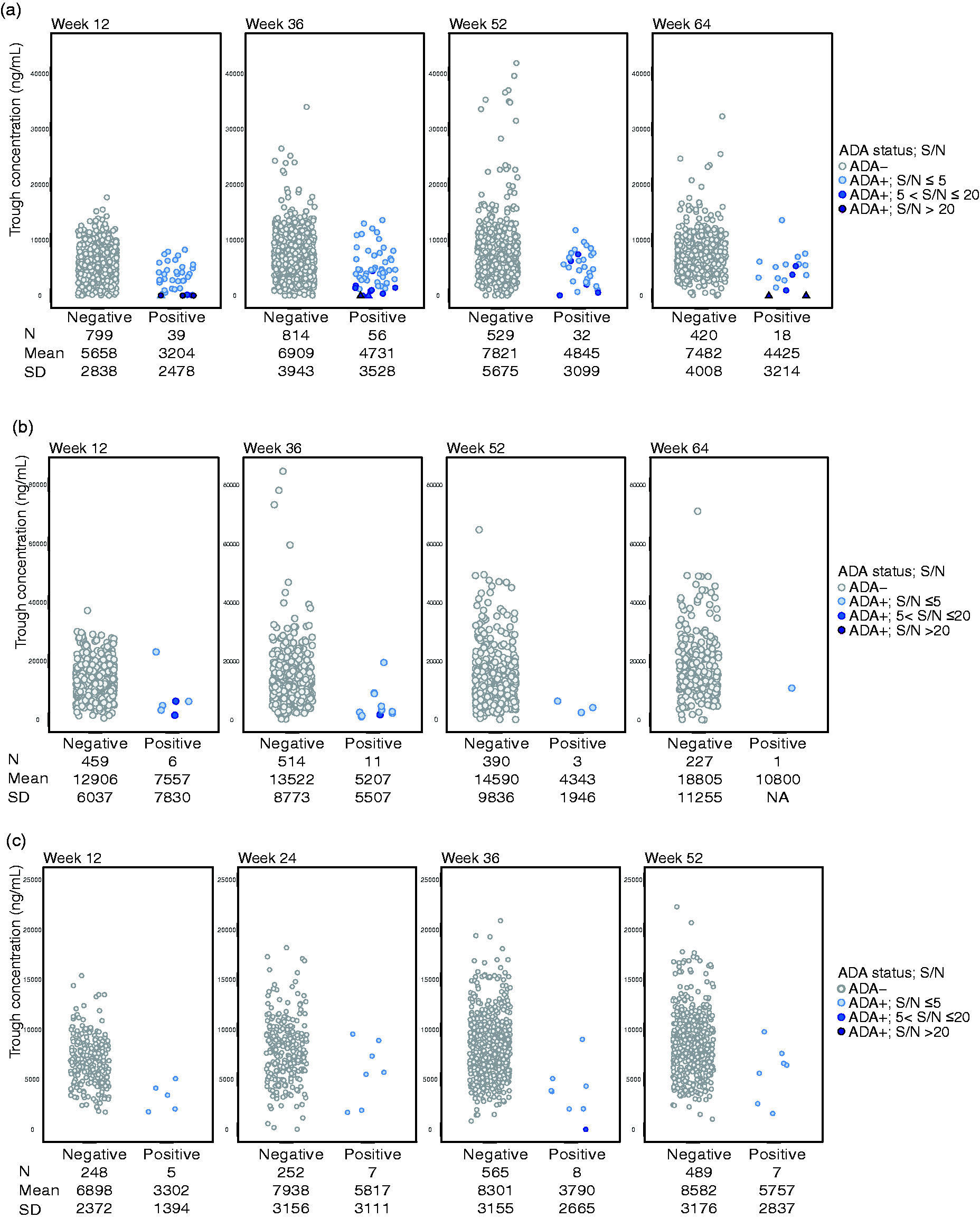
Erenumab serum concentrations by anti-erenumab antibody status and levels. Concentration of erenumab following a dose of 70 mg SC from the global studies (a), 140 mg SC from the global studies (b), and 70 mg SC from the Japan studies (c) are shown separately. There is only one ADA-positive patient from the 140 mg dose group of the Japan studies, who qualified the comparation analysis, therefore, no diagram was generated for the 140 mg SC from the Japan studies. The ADA status and levels (measured by S/N ratios) from individual patient at indicated time points were labeled by different colors. The neutralizing antibody-positive patients were marked with triangle labels.
Impact of ADA on efficacy
Efficacy of erenumab does not appear to be impacted by the ADA status or S/N level (Figure 4). The change from baseline in monthly migraine days for developing anti-erenumab antibody-positive patients is consistent with that for ADA-negative patients over time. For the global studies, the range of MMD changes observed in developing ADA-positive individuals (regardless of S/N value) was within the range observed for ADA-negative individuals in both the short-term (week 12; Figure 4A) and long-term ADA/efficacy analysis (through week 64; Figure 4B). The mean change in MMD (SE) from baseline to week 64 was −3.0 (1.5) days for patients developing anti-erenumab binding antibodies and −4.9 (0.4) days for patients who remained ADA-negative in patients with EM, and in patients with CM, the mean change in MMD from baseline to week 64 was −11.2 (2.9) and −9.1 (0.4), respectively (Figure 4B). While the number of patients who developed anti-erenumab antibodies were limited from the two Japan studies, comparable results were observed (Table 4).

Change from baseline in MMD by developing anti-erenumab antibody status and levels from the global studies. The efficacy data from the EM and CM studies are shown separately. (a) the efficacy data were plotted by developing ADA status and levels (measured by S/N ratios) at week 12 during the DBTP for short-term analysis; (b) the efficacy data were plotted by developing ADA status over time for long-term analysis. The ADA status and levels from individual patients were labeled by different colored circles. ADA, anti-drug antibody; CM, chronic migraine; DBTP, double-blind treatment period; EM, episodic migraine; MMD, monthly migraine days.
Change from baseline in MMD by developing anti-erenumab antibody status in the Japan studies.

n = Number of patients with observed efficacy data by antibody status at the corresponding visit, whose erenumab dose was only 70 mg or 140 mg during the entire studies. One pre-existing antibody-positive patient was excluded from the analysis. No patients were neutralizing antibody-positive, so no results are presented based on neutralizing antibody status.
MMD, monthly migraine days.
Impact of ADA on safety
The safety profiles of patients who tested positive for anti-erenumab binding antibodies were evaluated and found to be consistent with the safety profiles of ADA-negative patients, including for AEs related to hypersensitivity, injection site reaction, or immune system disorder (Table 5).
Exposure adjusted incidence rate of selected adverse events by anti-erenumab antibody status in long-term analysis.

Data reported as number n [r]; n = number of patients reporting at least one occurrence of an adverse event; r = exposure-adjusted patient incidence rate per 100 pt-yr (n/e * 100); e = sum across all patients, the total time at risk in years; Long-term analysis included overall data from entire studies (double-blind treatment phases and extension phases); Only includes preferred terms from the following search categories: Injection site reactions AMQ; Hypersensitivity SMQ and Immune system disorders SOC. Anti-erenumab antibodies were not tested in the placebo group.
aIncludes patients with pre-existing antibodies, three in global studies and one in Japan studies.
bNeutralizing antibody status reported as a subset of binding antibody-positive patients; there were no neutralizing antibodies observed in the Japan studies.
Discussion
In this integrated post hoc analysis, the immunogenicity profile of erenumab was assessed across six clinical trials with long-term extensions from patients with CM and EM whose erenumab dose was only 70 mg or 140 mg (N = 2985). The incidence of developing anti-erenumab antibodies was low, and with lower immunogenicity in the 140-mg group compared with the 70-mg group. The majority of patients who developed anti-erenumab antibodies had low magnitude of response with transient ADA-positivity, were often ADA-positive at only a single timepoint, and most reverted to an ADA-negative status by the last on-study assessment.
The lower ADA incidence observed in the 140 mg group compared with the 70 mg group is unlikely to be due to the drug tolerance of the binding ADA method. The assay used to detect anti-erenumab binding antibodies in clinical studies was validated to be able to detect 500 ng/mL anti-erenumab antibody in the presence of 100 μg/mL erenumab (specificity assay).
Although the circulating drug can interfere with detection of antibodies present at lower magnitude, drug concentrations in antibody samples from clinical patients administered either the 70 mg or the 140 mg dose were below the 100 μg/mL drug tolerance level and therefore unlikely to have interfered with the detection of anti-erenumab antibodies most likely to be clinically meaningful. Moreover, there was no apparent increase of ADA incidence during safety follow-up when drug concentrations were below the limit of detection. In summary, drug interference does not appear to have played a role in the lower ADA incidence observed in the 140 mg dose group. It is possible that the circulating drug may have interfered with detection of anti-erenumab neutralizing antibodies in some patients since the assay could only tolerate 0.1 μg/mL erenumab at an anti-erenumab neutralizing antibody concentration of 1.14 μg/mL. However, there was no apparent increase in the incidence of neutralizing antibodies during safety follow-up when drug concentrations were below the limit of detection. Moreover, the overall analyses of anti-erenumab antibodies including neutralizing antibodies did not reveal any meaningful impact on clinical efficacy or safety of erenumab.
Although fully human or humanized monoclonal antibody drugs are expected to be less immunogenic in humans compared to monoclonal antibody drugs of non-human origins (e.g., murine/human chimeric), they have the potential to elicit unwanted immunogenicity (18–21). Erenumab, a fully human immunoglobulin IgG2 antibody, was assessed for the potential clinical impact of anti-erenumab antibody on the pharmacokinetics, clinical efficacy, and clinical safety of erenumab.
Both binding and neutralizing ADAs have the potential to alter the clearance of therapeutic proteins (22). Despite a modest (30-70%) reduction in mean trough concentrations of erenumab among ADA-positive patients, the levels measured were within the range of the ADA-negative patients when compared to the same timepoint within the same dose group. Further, the changes from baseline in MMD observed in ADA-positive individuals were within the range observed for ADA-negative individuals regardless of the magnitude of the ADA response (measured by S/N values), supporting a lack of clinically meaningful impact of the observed immunogenicity on erenumab efficacy. Immunogenicity was not shown to be a mediator of hypersensitivity events or AEs related to injection site reactions or immune system disorders. Although these data do not demonstrate an impact of anti-erenumab antibody development on the drug efficacy or safety in these patients, the small number of ADA-positive patients limited our ability to fully evaluate the clinical impact according to ADA status.
Although ADA incidence information is typically included in the prescribing information and addressed in some of the trials of other CGRP-monoclonal antibodies (23,24), comparison of incidences of ADA across products may be misleading, due to many factors including the heterogeneity in ADA detection methodology, sample handling, timing of sample collection, concomitant medications, and underlying disease. The goal of presenting this immunogenicity data is not only to provide patients, physicians and regulators with ADA incidence information, but more importantly to provide meaningful, clinically relevant information including, specifically, the clinical consequences of immunogenicity (25).
Conclusions
Together, these findings indicate that erenumab has a low immunogenicity rate, with the majority of ADA responses of low magnitude and transient in nature. While the number of anti-erenumab antibody-positive patients is limited, results of these analyses suggest that efficacy does not appear to be impacted by the presence of anti-erenumab antibody regardless of ADA magnitude. Furthermore, development of antibodies to erenumab was not associated with safety findings potentially attributable to ADA. The low incidence of ADA in patients treated with erenumab imparts no change to the overall erenumab benefit-risk profile.
Clinical Implications
Erenumab (erenumab-aooe in the US) is a fully human anti-calcitonin gene-related peptide (CGRP) receptor monoclonal antibody approved for migraine prevention. In this integrated immunogenicity analysis, the incidence of anti-erenumab antibodies was low, with majority of the ADA responses of low magnitude and transient in nature; immunogenicity had no meaningful clinical impact on efficacy or safety of erenumab in patients with migraine. The low incidence of ADA in patients treated with erenumab imparts no change to the overall erenumab benefit-risk profile.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024221075621 - Supplemental material for Immunogenicity of erenumab: A pooled analysis of six placebo-controlled trials with long-term extensions
Supplemental material, sj-pdf-1-cep-10.1177_03331024221075621 for Immunogenicity of erenumab: A pooled analysis of six placebo-controlled trials with long-term extensions by Yanchen Zhou, Feng Zhang, Marta Starcevic Manning, Zheng Hu, Cheng-Pang Hsu, Po-Wei Chen, Cheng Peng, Brett Loop, Daniel T Mytych and Gabriel Paiva da Silva Lima in Cephalalgia
Footnotes
Acknowledgements
Medical writing and editorial assistance for this article was provided by Jon Nilsen, PhD (Amgen Inc.) funded by Amgen Inc.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors are employees and shareholders of Amgen Inc.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by Amgen Inc. Erenumab is codeveloped by Amgen Inc and Noartis.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
