Abstract
In migraine and other primary headaches there is a strong vascular component. Besides the trigeminovascular components some of the associated symptoms point to the involvement of brain stem regions. The central limb of the trigeminal vascular pathway is its projection to the trigeminal nucleus caudalis (TNC) and to the C1-C2 levels of the spinal cord. The aim of the present study was to demonstrate the occurrence of some neurotransmitters in these regions in man. In both the TNC and in the Rexed's laminae I and II of the dorsal horns at the C1 and C2 levels there were numerous substance P immunoreactive fibres. Fibres containing calcitonin gene-related peptide (CGRP) and pituitary adenylate cyclase-activating peptide (PACAP) were moderately dense in number. Fibres containing vasoactive intestinal peptide (VIP) or nitric oxide synthase (NOS) were not seen in the TNC or at the C1 and C2 levels of the spinal cord.
Introduction
Much of the associated symptoms in attacks of primary headaches, e.g. pain, nausea and vomiting, suggest the involvement of brain stem regions. With positron emission tomography, Weiller et al. (1) demonstrated a centre in the brain stem that was activated in migraine attacks, thus linking the intracranial pain-sensitive blood vessels with central aspects of the migraine attack in man.
Studies in laboratory animals and in humans have revealed that the afferent innervation of the large intracranial blood vessels and the dura mater is primarily nociceptive and originates in the trigeminal ganglion (2–4). The bipolar trigeminal cells project centrally to the trigeminal nucleus caudalis (TNC) and its reciprocal parts at the C1 and C2 levels of the spinal cord. Recently, it has been observed that stimulation of the trigeminal ganglion results in the accumulation of the immediate early genes c-fos and c-jun in the Rexed's laminae I and II of the TNC (5, 6). Stimulation of the superior sagittal sinus, used as a model for excitation of craniovascular pain, mimics several of the changes seen during a migraine attack in man (7, 8). Activation of the superior sagittal sinus by electrical or mechanical means increases the metabolic activity in the TNC and in the dorsal horns of the cervical spinal cord (8). In order to study which neurotransmitters might be involved in mediating the signalling in the central projections of the trigeminal nerve we have examined the occurrence of neurotransmitters previously shown to be expressed in the human trigeminal ganglion (9) in the human TNC and C1 and C2 regions.
Methods
Materials
Brain stem and cervical spinal cord were obtained at autopsy from adult subjects in accordance with the Hungarian University Medical School Guidelines for Ethics in Human Tissue Experiments. At autopsy the tissues were removed from six subjects (four male, two female) with an average age of 58.5 years (31–77 years). None of the subjects suffered from any central nervous system diseases. Tissues were collected within 18–24 h of death.
Fixation
The brain stem was cut into 0.5 cm parts beginning at the level of the obex and ending below the spinal segment of C3. For immunocytochemistry, the tissues were immersed overnight in a fixative consisting of 2% paraformaldehyde and 0.2% picric acid in 0.1 mol/L phosphate buffer, pH 7.2. After fixation the tissues were rinsed and stored in Tyrode solution with 10% sucrose for 4 days.
Freezing
The tissue was put in a test tube filled with 50 ml of isopenthane that had been stored in dry ice in a −85°C freezer for 20 min. After 10 s the tissue was removed and stored at −85°C until it was cut into 10-µm sections in a cryostat and mounted on chrome-alum coated slides.
Immunocytochemistry
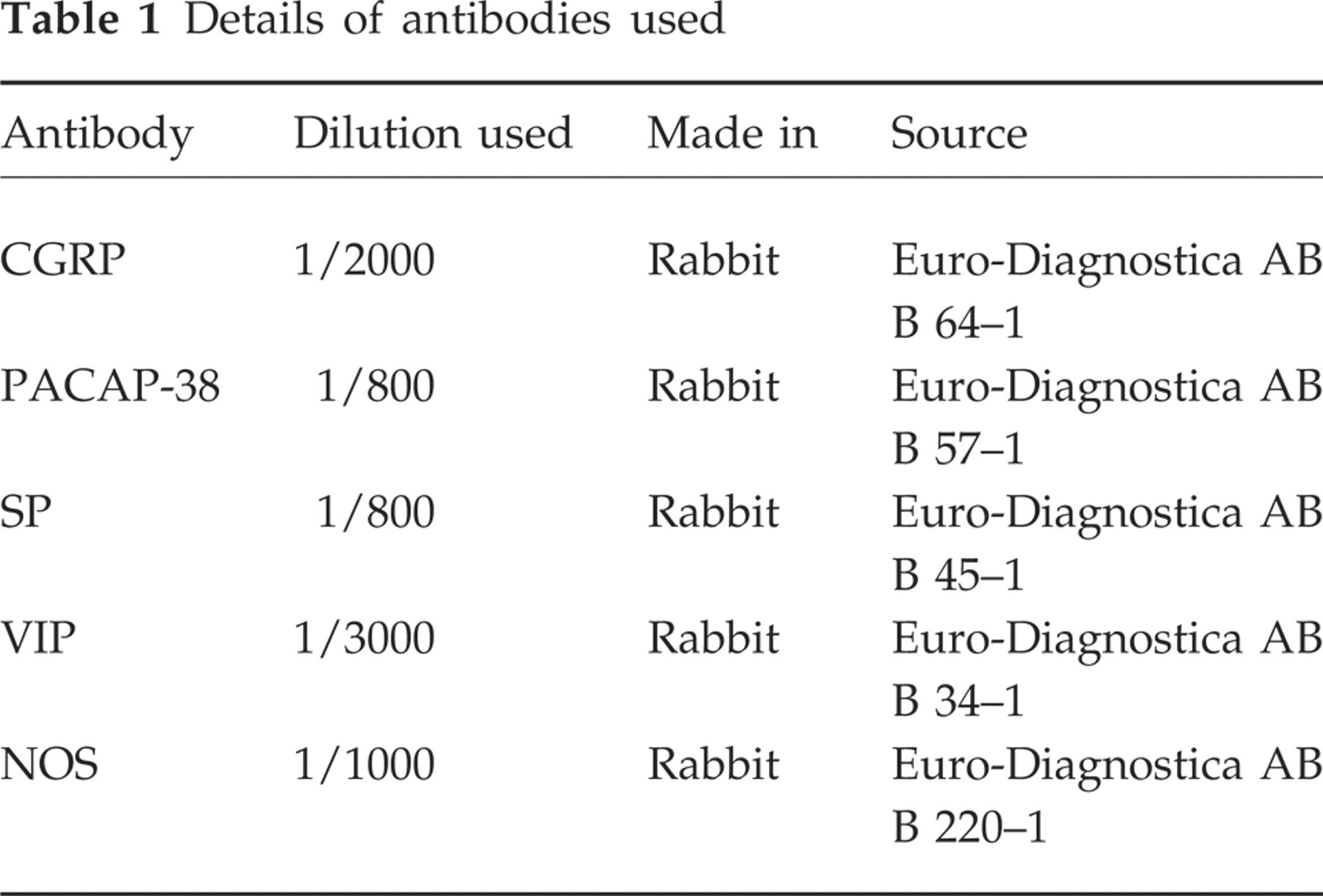
For immunocytochemical demonstration of neuropeptides and neuronal NOS indirect immunofluorescence was used. For details of the antibodies see Table 1.
Details of antibodies used
Briefly, the sections were incubated with the primary antibody overnight at +4°C. The site of the antigen–antibody complex was revealed by application of a secondary antibody (1 : 80; swine antirabbit IgG, DAKO, Copenhagen, Denmark) and conjugated to fluorescein isothiocyanate (FITC). In the control experiments the primary antibody was omitted.
The antibodies used were tested for specificity by preincubation with the respective antigen of related peptides (10–100 µg/ml diluted antibody).
Every twelfth section was stained with haematoxylin-eosin for neuroanatomical localization.
Processing
Immunocytochemical sections were compared at the corresponding anatomical level with haematoxylin-eosin stained sections. Sections from three regions were studied; subnucleus caudalis of TNC, and the C1 and C2 levels of the spinal cord. The cytoarchitecture of the trigeminal subnucleus caudalis can be subdivided into three layers. The first layer is the marginal layer corresponding to the Rexed's lamina I of the spinal dorsal horn. The second layer is the substantia gelatinosa corresponding to the Rexed's lamina II. The third layer is the magnocellular layer, which corresponds to Rexed's laminae III and IV of the spinal dorsal horn. The laminae V and VI of the spinal dorsal horn do not continue into the trigeminal subnucleus caudalis (10).
Results
SP immunoreactivity
The results are summarized in Table 2. There was a rich supply of substance P (SP) positive nerve fibres in the TNC that were located in the marginal layer and substantia gelatinosa of the subnucleus caudalis (Fig. 1). At the C1 and C2 levels a rich supply of SP-ir nerve fibres was also seen in Rexed's laminae I and II and in the tract of Lissauer (Fig. 2).
Schematic representation of staining in the various regions with the different antibodies studied

Arbitrary scale: – no staining, + faint, + + moderate, + + + rich staining.
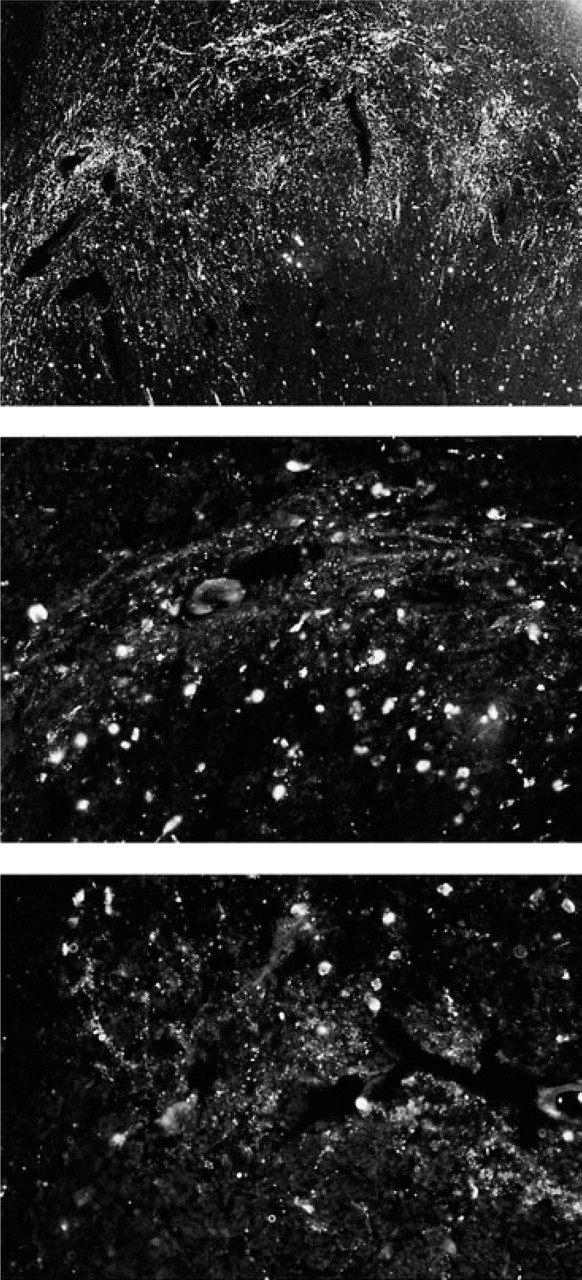
Numerous SP-immunoreactive nerve fibres (top) (× 150), a moderate supply of CGRP-immunoreactive nerve fibres (middle) (× 250) and a moderate supply of PACAP-immunoreactive nerve fibres (bottom) (× 250) can be seen in the human trigeminal nucleus caudalis.

The C1 level of the human cervical spinal cord. A rich supply of SP-immunoreactive nerve fibres (top) (× 250) and CGRP-immunoreactive fibres (middle) (× 250) and a moderate supply of PACAP-immunoreactive fibres (bottom) (× 275).
CGRP immunoreactivity
The marginal layer and the substantia gelatinosa in the subnucleus caudalis of the TNC contained a moderate supply of calcitonin gene-related peptide (CGRP)-ir fibres (Fig. 1). CGRP positive cells were not observed. At the C1 and C2 levels, Rexed's laminae I and II contained numerous CGRP-ir nerve fibres but no cell bodies, the inner layer being more heavily stained (Fig. 2). Also the tract of Lissauer had numerous CGRP-positive fibres at the C2 level.
PACAP immunoreactivity
A moderate amount of pituitary adenylate cyclase activating peptide (PACAP) positive fibres were found in the TNC, located in the marginal layer and substantia gelatinosa of the subnucleus caudalis of the TNC (Fig. 1). A scarce to moderate amount of PACAP-ir fibres was observed at the C1 and C2 levels in the Rexed's laminae I and II and in the tract of Lissauer (Fig. 2).
VIP and NOS immunoreactivity
Nerve fibres or cell bodies containing vasoactive intestinal peptide (VIP) or nitric oxide synthase (NOS) were not seen in the TNC or at the C1 and C2 levels in the Rexed's laminae I and II.
Discussion
To understand the pathophysiology of primary headaches it is essential to identify the regions in the brain that may process the signs of the disorder. This study has demonstrated that there is a rich supply of SP-ir fibres in the marginal layer and in the substantia gelatinosa of the subnucleus caudalis of the TNC and the Rexed's laminae I and II of the C1 and the C2 levels of the human cervical spinal cord. In addition, there was a moderate supply of CGRP-ir and PACAP-ir fibres in these areas, although NOS-ir or VIP-ir fibres were not seen. These findings are in accordance with previous studies showing a rich supply of sensory fibres in the TNC and in the spinal cord (11–14).
Migraine involves changes that are characterized by pain and nausea, symptoms that are mediated by the sensory system but also by centres in the brain stem. The vascular components of the disorder are mediated via the trigeminal nerve. Mechanical or electrical stimulation of the dura mater or of cranial blood vessels reproduces signs of migrainous pain (15). The nerve cell bodies of the human trigeminal ganglion express a number of neurotransmitters, such as SP, CGRP, PACAP and NO (9). Electrical stimulation of the trigeminal ganglion in man and cat results in increased plasma levels of SP and CGRP in the jugular vein (16, 17).
The central structures that process craniovascular pain have to some degree been mapped. Stimulation of the trigeminal ganglion in the rat induces a reduction in the immunoreactivities of SP and CGRP in the TNC, ipsilateral to the stimulated side (18, 19). Electrical stimulation of the superior sagittal sinus in the cat leads to increased metabolic activity in the TNC and in the C2 region of the spinal cord (20). A marked increase of the immediate early gene c-fos in laminae I and II of the TNC and in the superficial layers of the C1 and C2 regions can be seen upon stimulation of the middle meningeal artery, the superior sagittal sinus or the trigeminal ganglion in monkeys and cats (21–23). This response is reduced by anti-migraine drugs, such as triptans (24–27).
In man, evidence for a central site of action of the triptans has come from binding studies that demonstrate, both in vitro and in vivo, their association with the superficial laminae of the caudal part of the TNC and the cervical dorsal horn, as well as of the nucleus of the tractus solitarius. In an attempt to characterize the receptors involved it has been suggested that the 5-HT1B receptors are present in very low concentrations in all these nuclei in man (below 12% of total specific binding), while the 5-HT1D receptors account for about 50% of the total specific sumatriptan binding (28). In addition, a significant number of 5-HT1F-binding sites can be seen (29, 30). The 5-HT1F site has been examined using a specific agonist LY334370 (31). LY334370 had no contractile effect and did not inhibit CGRP release. However, it was observed that LY334370 could block the transmission of nociceptive impulses in the TNC. These data gave weight to the proposal that the anti-migraine actions could in part be exerted centrally on these nuclei.
The present study in man has revealed that immunocytochemical distribution of SP-, CGRP- and PACAP-ir coincides with the reported localization of the 5-HT1B/1D binding sites in the TNC and in particular with the distribution of 5-HT1B/1D receptor-ir. Thus, it is tempting to suggest that if the triptans can reach the TNC and the C1 and C2 levels, they may also here inhibit the activity of the central aspects of the sensory trigeminal fibres. It also suggests that the role of nitric oxide and VIP at this site is minor, although it cannot be ruled out that the effects of the post-mortem delay in combination with a long immersion period in the fixative may reduce the antigenicity of the NOS antibodies (32).
Footnotes
Acknowledgements
Supported by grants from the Swedish Medical Research Council (no 5958).
