Abstract
Background
Women with endometriosis are more likely to have migraine. The mechanisms underlying this co-morbidity are unknown. Prolactin, a neurohormone secreted and released into circulation from the anterior pituitary, can sensitize sensory neurons from female, but not male, rodents, monkeys and human donors.
Methods
We used a syngeneic model of endometriosis to determine whether elevated prolactin levels can sensitize trigeminal ganglion neurons and increase vulnerability to migraine pain.
Results
Mice with endometriotic lesions showed increased serum prolactin levels and developed persistent abdominal, but not cephalic, allodynia. However, inhalation of a transient receptor potential ankyrin 1 agonist, umbellulone, a known environmental trigger of headache in some patients, elicited cephalic allodynia in mice with endometriosis but not sham controls, suggesting that endometriosis can promote sensitization of trigeminal neurons and migraine attacks. Endometriosis dysregulated the expression of prolactin receptor isoforms in trigeminal neurons and increased their excitability measured by
Conclusions
These data demonstrate that circulating prolactin in endometriosis promotes vulnerability to migraine through sensitization of trigeminal afferents. Clinically available dopamine receptor agonists or novel monoclonal antibodies targeting prolactin signaling may be effective for migraine prevention in women with endometriosis.
This is a visual representation of the abstract.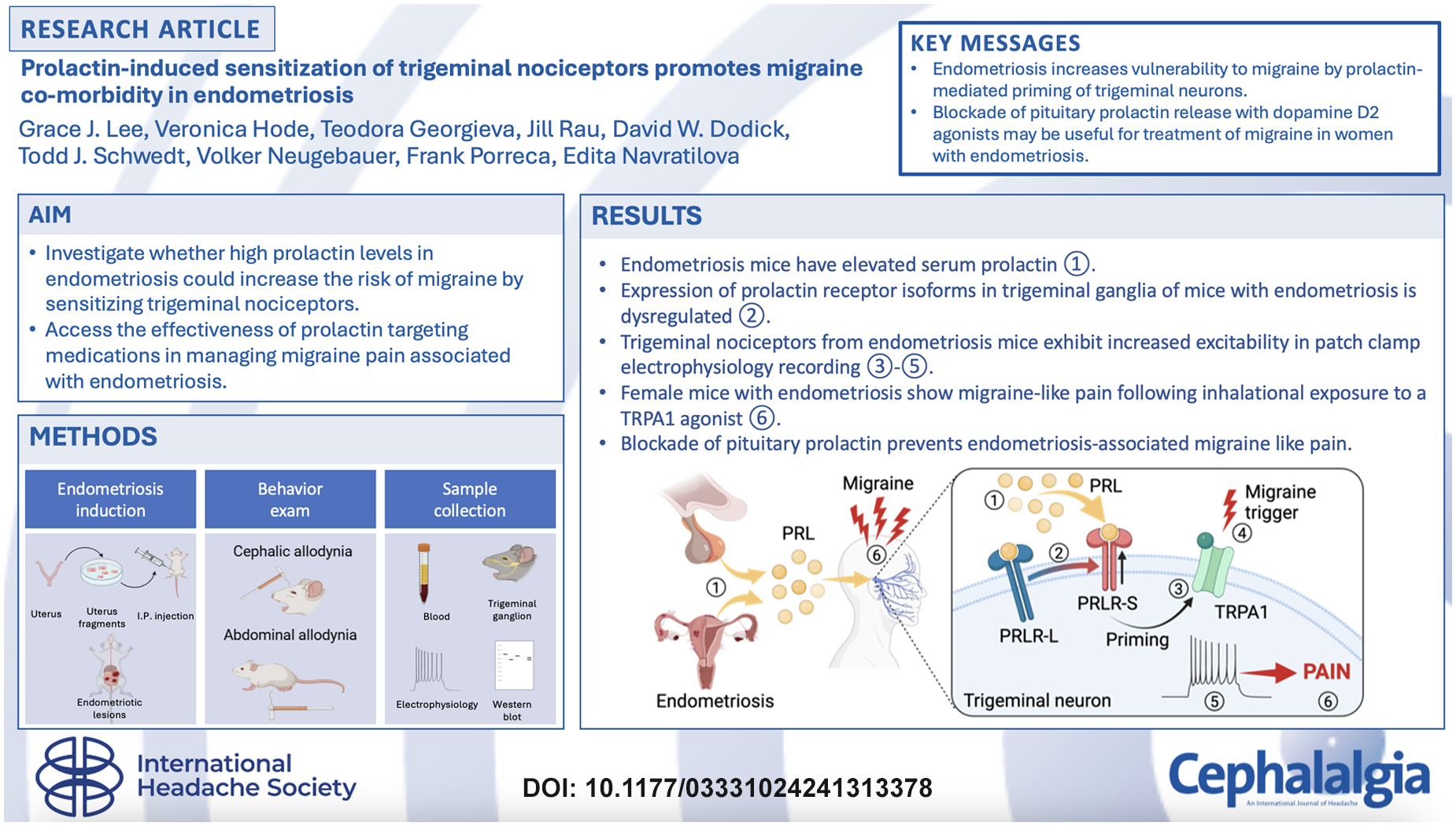
Keywords
Introduction
Endometriosis is an often-painful condition characterized by the growth of endometrial tissue outside the uterus (1). Endometriosis is estimated to impact 10–15% of women of reproductive age, and up to 70% of women experiencing chronic pelvic pain (2). Endometriosis-associated pain likely arises from local inflammatory events and stimulation of peritoneal and visceral sensory nerve fibers that innervate peritoneal endometriotic lesions (3),(4). During disease progression, it is proposed that endometriosis leads to systemic inflammation as well as peripheral and central sensitization that may contribute to co-occurrence of other female-prevalent chronic overlapping pain conditions (COPC), including chronic tension headache, migraine, temporomandibular joint disorder and fibromyalgia, among others (5),(6). Current treatments for endometriosis include pain management medications, hormonal therapies and surgery. However, these treatments often have limited effectiveness, are associated with many unwanted side-effects and usually do not target co-morbid pain conditions (7).
Migraine is a common comorbidity of endometriosis. Migraine is 1.7 times more prevalent in women with endometriosis than in those without the disease (8),(9). Pelvic pain, often experienced by patients with endometriosis, was also found to be a predictor of migraine (9). Both migraine and endometriosis pain are frequently exacerbated during the menstrual cycle (10),(11). Most women with both conditions started to experience migraine attacks after their endometriosis diagnosis rather than before (78.8% vs. 21.2%) (9), suggesting that endometriosis may be a priming condition predisposing patients to migraine. These observations suggest that combating specific endometriosis-induced priming mechanisms may be effective for prevention of migraine co-morbid in patients with endometriosis.
Prolactin, in addition to its role in lactation and other reproductive processes, has been implicated in multiple biological functions including pain. Prolactin is a peptide hormone, primarily produced by pituitary lactotrophs (12). Secretion of prolactin from lactotrophs is negatively regulated by dopamine released from tuberoinfundibular dopamine neurons located within the arcuate nucleus of the hypothalamus (13),(14). Additionally, prolactin can be produced by extra-pituitary tissues such as adipocytes, immune cells and stromal cells of the uterus (15)–(17) where its synthesis and release are regulated by autocrine/paracrine mechanisms independently from regulation of pituitary prolactin (15). The physiological effects of prolactin are mediated by its binding to and activation of prolactin receptor long (PRLR-L) and short (PRLR-S) isoforms. Prolactin selectively sensitizes nociceptors from female animals and from post-mortem female human donors, suggesting that this sexually dimorphic mechanism may contribute to increased pain in women (18)–(22). Both PRLR isoforms are found on sensory neurons in female mice and prolactin-induced nociceptor sensitization has been shown to occur through dysregulation of the expression of PRLR isoforms to bias prolactin signaling to the pronociceptive PRLR-S isoform (23). Preclinical studies have shown that prolactin promotes pain selectively in female animals (18),(23)–(26).
Prolactin has been linked to the pathophysiology of both endometriosis and migraine. Serum prolactin levels were found to be higher in women with endometriosis (27) and in women with migraine (28)–(30). Preclinical migraine models have also demonstrated that prolactin can elicit cephalic pain selectively in female, but not male, rodents. Dural administration of prolactin produced migraine-like pain behaviors in female, but not in male, rodents (31). A previous study from our laboratory showed that stress-induced prolactin release triggers acute cephalic pain in mice and promotes long-lasting priming (i.e. latent sensitization) of trigeminal nociceptors leading to migraine-like pain from normally innocuous stimuli (25). Collectively, these observations led us to hypothesize that high levels of circulating prolactin in endometriosis may dysregulate expression of PRLR isoforms to sensitize trigeminal ganglion (TG) neurons promoting comorbid migraine-like pain.
Methods
Animals
Female C57BL/6 mice aged 5–6-week-old were purchased from Jackson Laboratories (Bar Harbor, ME, USA). All experimental procedures were performed in accordance with the ARRIVE guidelines, the ethical guidelines of the International Association for the Study of Pain regulations on animal welfare and the NIH guidelines for the care and use of laboratory animals. The experimental procedures were approved by the Institutional Animal Care and Use Committee of the University of Arizona. Animals were housed under standard husbandry conditions with no more than five mice per cage under a 12:12-hour light/dark photocycle (lights on at 7:00 am) with
Mouse model of endometriosis
We implemented a syngeneic inoculation mouse model of endometriosis (32),(33) with some modifications. This protocol induces the growth of endometriotic lesions in the abdominal cavity as typically observed in patients with endometriosis (34)–(36). After one week of acclimatization, donor mice received a subcutaneous injection of β-estradiol 3-benzoate (3 μg per mouse; Sigma-Aldrich, St Louis, MO, USA) diluted in corn oil (Sigma-Aldrich) to stimulate the growth of the endometrium. Seven days later, the donor mice were anesthetized with 2% isoflurane, and their uterine horns were removed and placed in a dissection dish containing ice-cold Hank's Balanced Salt Solution (HBSS, 1X; Gibco, Life Technologies Corporation, Grand Island, NY, USA). The uterine horns were cut longitudinally and minced with a pair of scissors into fine fragments to expose the endometrium. Two recipient mice were implanted from each donor mouse. Each endometriosis mouse was injected intraperitoneally with the uterine horn fragments in 500 μl of HBSS using an 18-G × 1 ½ʺ needle (#305185; BD, Franklin Lakes, NJ, USA) attached to a 1-ml syringe (BD) while under 2% isoflurane. The sham mice were injected with 500 μl of HBSS. Mice were group-housed after recovery from the procedure.
Drugs
Cabergoline (Tocris Bioscience, Bristol, UK) was diluted in 10% dimethyl sulphoxide, 10% Tween-80 and 80% saline and administered i.p. at 1.2 mg/kg (24). Umbellulone (UMB; AdipoGen, San Diego, CA, USA) was prepared as a stock solution of 0.1
Western blot analysis
Sample collection and western blotting was performed as previously described (23),(24). Mice were euthanized and samples of trigeminal ganglia ophthalmic V1 region were obtained. Samples were homogenized in a lysis buffer (20 m
Mouse prolactin enzyme-linked immunosorbent assay (ELISA)
Whole blood was collected between 7:00
Histological analysis
Endometriotic lesions were dissected at 6 weeks post-implantation for histopathological analysis using hematoxylin and eosin (H&E). Mice were anaesthetized by isoflurane and transcardially perfused with 4% paraformaldehyde. The samples were post-fixed in 4% paraformaldehyde overnight and were then cryoprotected in 30% (w/v) sucrose and embedded in optimal cutting temperature compound (Sakura Finetek, Alphen aan den Rijn, The Netherlands). Next, 20-μm thick sections were cut on a cryostat (Microm, HM 525) and mounted on the slide glass (Surgipath X-tra microscope slides or Apex Superior Adhesive Slides; Leica Biosystems, Wetzlar, Germany). Lesions were processed for H&E staining to confirm endometriosis pathology. The sections were examined under Echo Revolve microscope (Echo Laboratories, San Diego, CA, USA).
Mouse behavioral testing
Abdominal and migraine-like pain behavior was assessed by measuring withdrawal responses to normally non-noxious tactile stimulation (i.e. mechanical allodynia) using calibrated von Frey filaments (Stoelting, Wood Dale, IL, USA). The mice were placed in clear Plexiglas chambers (4ʺ × 4ʺ × 8ʺ, length × width × height) on top of a wire mesh stand (0.635 cm2 grid) for 2 hours during 3 consecutive days to habituate. For abdominal allodynia a 0.6-g von Frey filament was applied to the abdominal region ten times for each animal, with an interval of approximately 20–30 seconds between each application, until the filament slightly arched. The 0.6-g filament strength was selected based on pilot studies where it produced approximately 10% responses in naïve mice. Positive responses (a flinch, a jump or a retraction of the abdomen) were noted and recorded in a frequency chart. Cephalic (i.e. periorbital) allodynia was evaluated by measuring the frequency of response to tactile stimulation of the periorbital region with a 0.4-g filament, as previously described (37). Measurements were performed before, and for up to 5 hours after UMB exposure. The 0.4-g von Frey filament was applied ten times to the periorbital region until the filament slightly arched. Behaviors scored as positive responses were facial grooming, head shaking and/or turning away after filament application. Abdominal and cephalic Frequency of Response was calculated as (number of positive responses/10 × 100%).
Umbellulone inhalation exposure
Inhalational exposure to a subthreshold dose of UMB that does not promote cephalic allodynia in naïve mice was performed according to a previously described protocol (37). A half square gauze was placed in each nose cone of the multi-station isoflurane anesthesia board (Parkland Scientific, Coral Springs, FL, USA) and 500 µl of UMB at 0.01
Primary cell culture
TG neurons from the ophthalmic V1 region were isolated and digested with media containing 3.125 mg/ml neutral protease (Dispase) (LS02104; Worthington, Lakewood, NJ, USA) and 5 mg/ml collagenase type 1 (LS004194; Worthington) for 30–40 min. Dissociated neurons were maintained in neurobasal-A cell culture media (#10888022; Thermo Fisher Scientific) supplemented with 2% B-27 (#17504044; Thermo Fisher Scientific), 1% penicillin/streptomycin (#16777-164; Hyclone, Logan, UT, USA), 1% GlutaMax (#35050061; Thermo Fisher Scientific), 10% fetal bovine serum (#F0926-50ML; Sigma-Aldrich), 25 ng/ml GDNF (#10788-106; VWR, Radnor, PA, USA) and 100 ng/ml mouse NGF (#76046-694; VRW). Dissociated neurons were plated onto 12 mm poly-
Electrophysiology
Whole-cell patch clamp recordings were performed at room temperature after approximately 14 oursh in culture as previously described (22). The patch pipettes were pulled from standard wall borosilicate glass capillaries (Sutter Instruments, Novato, CA, USA) with a horizontal puller (Model P-1000; Sutter Instruments) and had resistance of 4–6 MΩ when filled with internal solution. Cells were held at −60 mV for current clamp measurements of intrinsic excitability. Cells with a resting membrane potential (RMP) more depolarized than −40 mV were excluded from the analysis. Action potentials were evoked by current injection steps from 0–600 pA with an increment of 50 pA in 300 ms steps. RMP was measured without current injection. Rheobase was measured by injecting current stepwise starting from −10 pA in increments of 10 pA for 50 ms, until an action potential was observed. All experiments had a series resistance that was kept below 15 MΩ. Signals were acquired using a HEKA EPC10 USB amplifier (HEKA, Lambrecht, Germany), filtered at 10 kHz, and digitized at 20 kHz. Analyses were performed using Fitmaster software (HEKA) and Origin 9.0 software (OriginLab, Northampton, MA, USA). To evaluate sensitization of TG neurons from endometriosis animals to transient receptor potential ankyrin 1 (TRPA1) activation, we added into the external solution a subthreshold concentration of UMB (1 µ
Internal solution contained (in m
Statistical analysis
Data are expressed as the mean ± SEM. Statistical analyses were performed using Prism, version 7 (GraphPad Software Inc., San Diego, CA, USA). Data for prolactin levels were analyzed using Student’s
Results
Endometriosis is associated with abdominal pain and increased vulnerability to migraine
In a syngeneic mouse model of endometriosis (Figure 1a), we found that approximately 90% of animals receiving uterine tissue developed endometriotic lesions. Most of the lesions were clear fluid filled cysts (Figure 1b, top) and H&E staining showed the presence of endometrial-like glands and stromal cells (Figure 1b, bottom). We assessed abdominal tactile allodynia in endometriosis mice and sham controls weekly for 6 weeks by measuring response frequency to abdominal probing with von Frey filaments. Mice with endometriotic lesions developed abdominal allodynia starting from 1 week post-implantation, whereas there was no change in pain behavior in sham animals (Figure 1c). Response frequency remained significantly increased in the endometriosis group compared to sham controls at weeks 1–6 post induction. Animals that did not grow lesions after uterine tissue implantation (approximately 10%) were identified
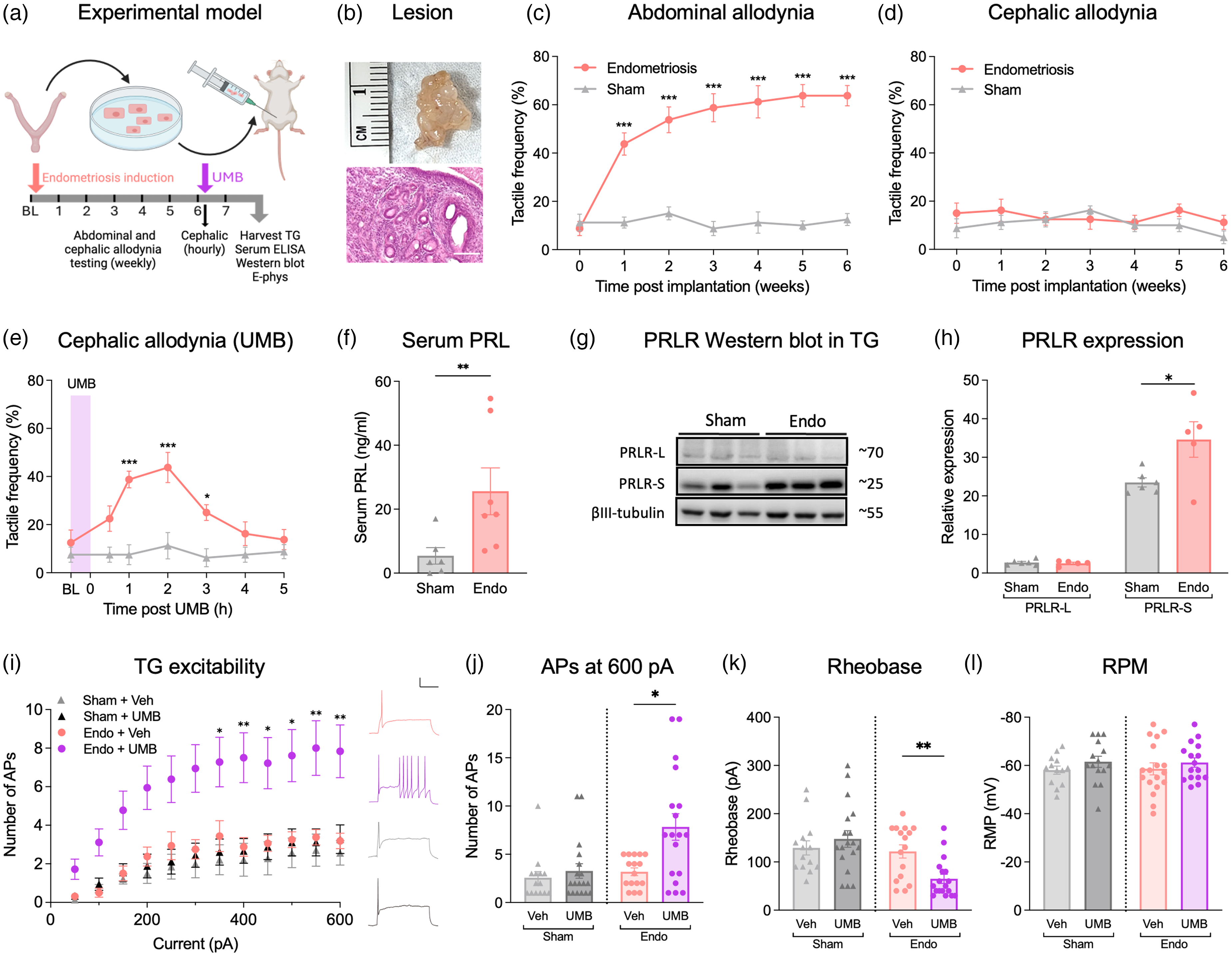
Prolactin (PRL) mechanism of trigeminal nociceptor sensitization and migraine-like pain in a mouse model of endometriosis. (a) Endometriosis model and experimental design. (b) Gross morphology and histological features of endometriotic lesions. Scale bar = 90 µm. Abdominal (c) and cephalic (d) tactile frequency of response was collected before endometriosis induction (time = 0) and repeated weekly for 6 weeks after induction to assess allodynia (n = 8 per group). (e) Six weeks after endometriosis induction, baseline cephalic responses were collected, followed by inhalation of umbellulone (UMB) and cephalic measurements collected hourly for 5 hours. (f) Serum prolactin levels in sham and endometriosis mice (Sham, n = 6; Endo, n = 7). (g) Representative western blots and (h) quantification of prolactin receptor (PRLR) isoform protein expression in trigeminal ganglion (TG) from sham and endometriosis mice (Sham, n = 6; Endo, n = 5). (I–L) V1 region of the trigeminal ganglia were collected and dissociated neurons cultured for patch clamp electrophysiological assessment. (i) Average number of action potentials (APs) in response to incremental 50 pA current steps in TGV1 neurons from sham and endometriosis mice treated with either vehicle or UMB (Sham + Veh, n = 14, Sham + UMB; n = 18, Endo + Veh, n = 16; Endo + UMB, n = 18. (I, right) Representative action potential traces at 600 pA. Scale bars = 20 mV/100 ms. Comparisons of (j) the number of APs at 600 pA, (k) rheobase and (l) resting membrane potential (RMP). Results are expressed as the mean ± SEM of measurements. *
Clinical studies show high comorbidity between endometriosis and migraine (41). Therefore, we investigated whether the mice in our experimental model of endometriosis would develop cephalic allodynia as a surrogate measure of headache pain. The same mice that exhibited abdominal allodynia 1–6 weeks after endometriosis induction (Figure 1c) did not show increased sensitivity in the cephalic region (Figure 1d). Our group has previously utilized a mouse model of hyperalgesic priming with repeated restraint stress to establish a state of increased sensitivity to a subsequent trigeminal stimulation with inhalation of a TRPA1 activator, UMB (37). While a subthreshold dose of UMB has no effect in naïve animals, it elicits cephalic allodynia in restraint stress primed mice, modeling the condition of increased vulnerability to TRPA1 agonists, known to trigger headache in vulnerable patients (38). We therefore explored whether endometriosis would produce hyperalgesic priming of the trigeminal pathways to increase sensitivity to inhalational UMB. We exposed endometriosis and sham animals to UMB for 30 minutes and measured periorbital tactile responses for the following 5 hours. UMB exposure elicited cephalic allodynia in the endometriosis, but not the sham, group that lasted for approximately 3 hours (Figure 1e). These finding suggest that endometriosis induces systemic changes that result in hyperalgesic priming of the trigeminal pathways, increasing the vulnerability to migraine triggers.
Endometriosis increases serum prolactin levels and primes trigeminal nociceptors
Clinically, both endometriosis and migraine are associated with increased concentrations of prolactin (27)–(29),(42). Hence, we evaluated the possible involvement of the prolactin/PRLR pain mechanism in sensitization of trigeminal neurons in our model of endometriosis. After the behavioral experiment, we collected blood from endometriosis and sham mice to assess prolactin levels in the serum as well as the V1 region of the trigeminal ganglia (TGV1) for PRLR-L/S western blot evaluation. Serum prolactin was significantly elevated in the endometriosis group compared to shams (Figure 1f). The expression of PRLR-S was upregulated in the TGV1 of endometriosis mice compared to shams, while PRLR-L expression did not change (Figure 1g,h). The increased proportion of the PRLR-S isoform relative to the PRLR-L isoform in TGV1 nociceptors may bias prolactin signaling to promote sensitization resulting in increased responses to normally subthreshold stimuli such as UMB.
To evaluate this possibility, we cultured TGV1 neurons from endometriosis and sham animals at 7 weeks after induction and measured their excitability in the presence of either vehicle or a subthreshold concentration of UMB (1 µ
Pituitary prolactin mediates endometriosis-associated migraine-like pain
We next investigated whether elevated serum prolactin levels observed in mice with endometriosis may contribute to sensitization of TG neurons to UMB and UMB-induced cephalic allodynia. Seven weeks after endometriosis induction, we treated the animals with cabergoline (1.2 mg/kg, i.p.) or vehicle daily for 2 weeks (Figure 2a). No effect on abdominal (data not shown) or periorbital (Figure 2b) responses was observed during the two-week treatment with saline or cabergoline. Following the 2-week treatment period, vehicle-treated endometriosis mice developed cephalic allodynia elicited by UMB exposure, confirming the results in Figure 1e. However, UMB did not induce cephalic allodynia in endometriosis mice treated with cabergoline (Figure 2c), suggesting that increased levels of pituitary prolactin promote sensitization of trigeminal pathways to migraine triggers. After termination of the experiment, we collected the blood and confirmed that cabergoline significantly decreased levels of serum prolactin (Figure 2d).

Systemic cabergoline reverses umbellulone-induced cephalic allodynia and endometriosis-induced priming of trigeminal neurons. (a) Experimental timeline. (b) Cephalic tactile allodynia was assessed once a week for 6 weeks and every 3 days after starting cabergoline (CBG) or vehicle (Veh) treatment (n = 8 mice/group). (c) After baseline (BL) measurement on week 9, mice were exposed to inhalation of umbellulone (UMB), and periorbital allodynia was assessed hourly for 5 hours. (c) Serum prolactin (PRL) levels were measured after termination of the experiment in vehicle and CBG treated endometriosis mice. Quantification (e) and representative western blots (f) of prolactin receptor (PRLR) isoform expression in TGV1 tissues from sham and vehicle or CBG treated endometriosis mice (sham, n = 4; Endo + Vehicle, n = 5; Endo + CBG, n = 5). (g) Representative action potential (AP) traces at 600 pA. Scale bars = 20 mV/100 ms. (h) Average number of APs in response to 50 pA current steps in TGV1 neurons from vehicle or CBG treated endometriosis mice with
Additionally, we examined the effects of cabergoline on the expression of PRLR isoforms in TGV1. Consistent with the previous experiment (Figure 1h), vehicle-treated endometriosis animals showed a significantly higher expression of PRLR-S than the sham group; treatment of endometriosis mice with cabergoline significantly decreased PRLR-S expression in TGV1 (Figure 2e,f). PRLR-L expression was not significantly different between the groups. Decreased expression of PRLR-S in cabergoline treated endometriosis mice is consistent with lack of UMB-induced cephalic allodynia in these animals.
Lastly, to investigate whether inhibition of UMB-induced cephalic allodynia by cabergoline is associated with decreased excitability of TGV1 neurons, we performed whole-cell patch-clamp electrophysiology in TGV1 neurons cultured from cabergoline and vehicle treated endometriosis mice. Action potential firing following UMB treatment was lower in TGV1 neurons from the cabergoline-treated compared to the vehicle-treated endometriosis mice (Figure 2g–i). Note that TGV1 neurons from vehicle treated endometriosis animals showed a similar number of action potentials following UMB challenge as observed in Figure 1(h,i). Rheobase was elevated in the cabergoline treated endometriosis group (Figure 2j), but RMP did not change with cabergoline treatment (Figure 2k). Collectively, these results suggest that increased levels of circulating prolactin and dysregulation of PRLR isoforms in TG neurons likely play a role in endometriosis-associated sensitization of trigeminal neurons and increased vulnerability to migraine-like pain.
Discussion
Endometriosis is a common chronic gynecological disease which is associated with other female-prevalent COPC including headache disorders such as chronic tension headache and migraine (5),(6). Clinical observations indicate that migraine attacks often start after an endometriosis diagnosis rather than before (9), suggesting that endometriosis may increase the risk for developing migraine. Identification of the mechanisms promoting comorbidities associated with endometriosis is important for development of new classes of personalized medications for COPCs such as migraine in women with endometriosis.
We used a mouse model of endometriosis to investigate the role of the prolactin/PRLR system in the increased vulnerability of women with endometriosis to migraine. The main goals were to determine (i) if endometriosis might be a risk factor for migraine and (ii) whether prolactin targeting medications would be useful for the treatment of endometriosis-related migraine pain. We found that induction of endometriosis was accompanied by abdominal, but not cephalic, allodynia. However, while endometriosis did not directly elicit cephalic allodynia, trigeminal nociceptors from mice with endometriosis exhibited hyperalgesic priming so that cephalic allodynia was triggered by inhalational exposure to a subthreshold concentration of a TRPA1 agonist, umbellulone. Hyperalgesic priming was accompanied by increased expression of PRLR-S in the ophthalmic division of the trigeminal ganglion and by increased excitability of TGV1 neurons with

Proposed mechanism underlying migraine co-morbidity in endometriosis. Endometriosis increases blood levels of prolactin (PRL) (1). High circulating PRL levels lead to upregulation of the prolactin receptor short (PRLR-S) isoform on trigeminal neurons that shifts PRL signaling to the pronociceptive pathways (2), sensitizing transient receptor potential ankyrin 1 (TRPA1) channels and priming the neurons to normally innocuous stimuli (3). Exposure of primed trigeminal neurons
Pituitary prolactin
Exogenous prolactin has been shown to sensitize sensory neurons in culture and promote pain responses
We investigated whether decreasing serum prolactin levels with cabergoline would relieve cephalic pain. Our results show that a 2-week course of cabergoline treatment produced an almost complete inhibition of circulating prolactin. Importantly, reducing serum prolactin reversed sensitization of TG neurons to UMB, as well as UMB-induced cephalic allodynia, suggesting that migraine-like pain resulting from a normally subthreshold stimulus in mice with endometriosis is due to increased pituitary prolactin. Cabergoline treatment, however, had no effect on abdominal allodynia, suggesting that other mechanisms likely contribute to sensitization of nociceptors in the pelvic region. Interestingly, dopamine D2 receptor agonists have shown beneficial effects in some patients with menstrual migraine and in patients with hyperprolactinemia (44)–(46). In a case series of patients with hyperprolactinemia caused by a prolactin-secreting pituitary adenoma 11 out of 12 patients had improvement or resolution of headache after treatment with a dopamine D2 receptor agonist (47). Our study is in agreement with these clinical findings and suggests that prolactin may play a more general role in the pathogenesis of migraines including migraine associated with endometriosis.
PRLR-mediated nociceptor sensitization
Preclinical studies have established that prolactin promotes pain behavior selectively in female rodents by signaling at dorsal root ganglia (DRG)/TG neurons through the PRLR-S homodimers, whereas signaling through the PRLR-L homodimers activates the JAK/STAT intracellular pathway and gene transcription (48). The formation of PRLR-S/L heterodimers results in silencing of prolactin signaling, providing a mechanism by which the net effect of prolactin depends on the relative ratio between the isoforms. We have previously shown that clustered regularly interspaced short palindromic repeats (CRISPR)/CRISPR-associated protein 9 (i.e. Cas9) editing down-regulates the expression of PRLR-L promotes hypersensitivity in naïve female mice through enhanced PRLR-S signaling (23). Importantly, PRLR-L downregulation can occur physiologically in conditions associated with high prolactin, including repeated restraint stress (25), or chronic exposure to migraine medications associated with medication overuse headache such as triptans (24). Dysregulation of PRLR isoform expression to favor PRLR-S may therefore represent a female specific mechanism of nociceptor sensitization. Our current results demonstrating upregulation of PRLR-S in the TGs of endometriosis mice are consistent with this pro-nociceptive mechanism (26). Reduction of pituitary prolactin levels with cabergoline normalized PRLR isoform expression in the TGs as was also observed in our previous studies (24),(25).
The pro-nociceptive prolactin signaling has been shown to involve downstream phosphorylation and sensitization of TRP channels including TRPA1 (48). Our findings that endometriosis mice have normal baseline cephalic sensory thresholds but develop cephalic allodynia following inhalation of a subthreshold dose of UMB are consistent with the prolactin-mediated sensitization of TG neurons to subsequent TRPA1 activation. This mechanism is also supported by increased excitability of TG neurons obtained from endometriosis, but not sham, mice following UMB treatment. TRPA1 activators, including UMB or environmental irritants such as cigarette smoke, promote migraine only in individuals with underlying headache disorders, possibly reflecting sensitization of TRPA1 mechanisms in patients (38),(49). Consistent with these clinical findings, we previously observed that mice subjected to repeated restraint stress exhibit transient cephalic allodynia followed by long-lasting latent sensitization of the trigeminal pathways that can be revealed by increased sensitivity to UMB (37). While latent sensitization is observed after restraint stress priming in both male and female animals, it is dependent on prolactin/PRLR signaling only in females (25). Likewise, in female mice with endometriosis, cabergoline treatment blocked both UMB-induced cephalic allodynia and increased TG excitability suggesting that endometriosis promotes latent sensitization of trigeminal sensory neurons through the prolactin/PRLR priming mechanism, increasing the vulnerability to migraine triggers.
Previous patch clamp electrophysiology studies from our laboratory have shown that cultured DRG neurons from female, but not male, mice are selectively sensitized by overnight exposure to prolactin (22). Importantly, female selective sensitization by prolactin was also demonstrated in neurons recovered from human donors, indicating that this mechanism is conserved in humans and may therefore be relevant to human pain conditions (22). We now show that, while TG neurons from endometriosis mice are not sensitized at baseline, they show increased excitability following
Outstanding questions
First, the mechanisms by which endometriosis could lead to increased serum prolactin levels observed in this mouse model as well as in women with endometriosis are not known but could be related to pain-induced activation of stress pathways. Second, as noted, female-selective dysregulation of PRLR isoforms has been previously demonstrated in rodent models of opioid-induced hyperalgesia, medication overuse headache and in a repeated stress priming model of migraine (23)–(25). In these models, we found down-regulation of the PRLR-L that increases pro-nociceptive signaling through the PRLR-S. In the endometriosis model, we also found a shift to the pro-nociceptive PRLR-S signaling, but through upregulation of the PRLR-S isoform. The differences between these findings could be a result of the chronicity of pain in different models or a result of prolactin increased tonically versus in cyclic surges. Third, our previous (23)–(25) and current data suggest that dysregulation of PRLR isoforms is observed with both sustained nociceptor sensitization promoting ongoing allodynia as well as with silent sensitization (i.e. priming) of nociceptors that requires an additional ‘trigger’ such as UMB to uncover the allodynic effect. It is likely that additional signaling mechanisms determine the extent of nociceptor sensitization. Future studies will address these questions.
Summary
Our study identified a mechanism that may explain high occurrence of comorbid pain conditions including migraine in women with endometriosis (Figure 3). Increased pituitary prolactin in endometriosis likely promotes generalized latent sensitization of nociceptors, including trigeminal neurons, through the dysregulation of PRLR isoform expression that may contribute to comorbid migraine following exposure to a normally innocuous stimulus. These findings provide avenues for therapy in endometriosis comorbid migraine and other pain conditions, including the use of dopamine D2 receptor agonists and future development of antibodies against prolactin or PRLR.
Endometriosis represents a risk factor for migraine, but the mechanisms underlying this comorbidity are unknown. Using a mouse endometriosis model, we demonstrated that circulating prolactin promotes vulnerability to migraine through sensitization of trigeminal neurons. Prolactin lowering medications may be effective for migraine prevention in women with endometriosis.
Supplemental Material
sj-docx-1-cep-10.1177_03331024241313378 - Supplemental material for Prolactin-induced sensitization of trigeminal nociceptors promotes migraine co-morbidity in endometriosis
Supplemental material, sj-docx-1-cep-10.1177_03331024241313378 for Prolactin-induced sensitization of trigeminal nociceptors promotes migraine co-morbidity in endometriosis by Grace J. Lee, Veronica Hode, Teodora Georgieva, Jill Rau, David W. Dodick, Todd J. Schwedt, Volker Neugebauer, Frank Porreca and Edita Navratilova in Cephalalgia
Footnotes
Acknowledgments
We acknowledge the National Institutes of Health and Department of Defense for providing the funding that supported this work in the form of grants to FP and EN.
Data availability
The data to support the findings of this study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
GJL, VH, SB, TG, VN and EN declare that they have no personal, financial or relational conflicts of interest with this work. FP has served as a consultant or received research funding from Voyager, SiteOne Therapeutics, Nektar, Amgen, Acadia, Blackthorn, Teva, Eli Lilly, Hoba, Allergan, Ipsen and Proximagen, and is a founder of Catalina Pharma and Axon Therapeutics. JR reports the following conflicts within the past 12 months, speaking engagements: Abbvie and advisory panel: Lundbeck. DWD reports the following conflicts within the past 12 months: Consulting: AEON, Amgen, Clexio, Cerecin, Ctrl M, Allergan, Alder, Biohaven, Linpharma, Lundbeck, Promius, Eli Lilly, eNeura, Novartis, Impel, Satsuma, Theranica, Vedanta, WL Gore, Nocira, XoC, Zosano, Upjohn (Division of Pfizer), Pieris, Revance and Equinox. Honoraria: CME Outfitters, Curry Rockefeller Group, DeepBench, Global Access Meetings, KLJ Associates, Academy for Continued Healthcare Learning, Majallin LLC, Medlogix Communications, MJH Lifesciences, Miller Medical Communications, Southern Headache Society (MAHEC), WebMD Health/Medscape, Wolters Kluwer, Oxford University Press and Cambridge University Press. Research Support: Department of Defense, National Institutes of Health, Henry Jackson Foundation, Sperling Foundation, American Migraine Foundation and Patient Centered Outcomes Research Institute (PCORI). Stock Options/Shareholder/Patents/Board of Directors: Ctrl M (options), Aural analytics (options), ExSano (options), Palion (options), Healint (Options), Theranica (Options), Second Opinion/Mobile Health (Options), Epien (Options/Board), Nocira (options), Matterhorn (Shares/Board), Ontologics (Shares/Board), King-Devick Technologies (Options/Board) and Precon Health (Options/Board). Patent 17189376.1-1466:vTitle: Botulinum Toxin Dosage Regimen for Chronic Migraine Prophylaxis. Within the past 24 months, TJS has received compensation for consulting with AbbVie, Amgen, Eli Lilly, Linpharma, Lundbeck, Scilex and Theranica, and royalties from Up To Date. He has held stock options in Aural Analytics and Nocira. Research support has been received from the American Heart Association, Henry Jackson Foundation, National Headache Foundation, National Institutes of Health, Patient Centered Outcomes Research Institute, Pfizer, Spark Neuro and United States Department of Defense.
Funding
This work was supported by R01 NS120395 and R01 NS129552 to FP and EN.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
