Abstract
Background
Neurogenic inflammation, mediated by the activation of primary neurons, is thought to be an important factor in migraine pathophysiology. Programmed cell death ligand-1 (PD-L1) can suppress the immune response through the Programmed cell death-1 receptor. However, the role of PD-L1/PD-1 in migraine remains unclear. In this study we evaluated the expression and role of PD-L1/PD-1 in the trigeminal ganglia in an animal model of acute migraine.
Methods
Acute nitroglycerin induces acute mechanical hyperalgesia that can be used as a readout of migraine-like pain. We investigated the expression of PD-L1 and PD-1 in the trigeminal ganglia in a mouse model by means of immunofluorescence labeling, quantitative reverse transcription-polymerase chain reaction and western blotting. We explored the effects of PD-1 in a migraine model by the von Frey test and by analyzing the expression of calcitonin gene-related peptide, interleukin-1β (IL-1β), interleukin-18 (IL-18), Tumor Necrosis Factor-α (TNF-α), interleukin-6 (IL-6) and transient receptor potential vanilloid (TRPV4) after the intravenous injection of a PD-1 inhibitor.
Results
PD-L1 and PD-1 immunoreactivity were present in healthy trigeminal ganglia neurons. The mRNA levels of PD-L1 and PD-1 were significantly elevated 2 h, 4 h and 6 h after acute nitroglycerin treatment (p < 0.05). The protein levels of PD-L1 were significantly increased 2 h, 4 h and 6 h after treatment, and PD-1 was significantly increased at 2 h and 6 h. The blockade of PD-1 increased acute nitroglycerin-induced hyperalgesia, and this effect was accompanied by a more significant increase in calcitonin gene-related peptide, IL-1β, TNF-α, IL-6 and IL-18 in the trigeminal ganglia.
Conclusion
These findings suggest that PD-L1 and PD-1 might inhibit migraine-like pain by downregulating CGRP and inflammatory factors in the trigeminal ganglia. The use of PD-L1 and PD-1 as analgesics should be further studied.
Introduction
Migraine is a common neurological disorder that manifests as recurrent attacks of unilateral and throbbing headache accompanied by nausea, vomiting, photophobia and phonophobia (1). Understanding the underlying neurobiology of migraine will improve therapy and benefit migraineurs.
The role of the trigeminovascular system in migraine pathogenesis is widely accepted (2). Neurogenic inflammation, which is mediated by the activation of primary neurons including trigeminal ganglion (TG) neurons, is thought to be an important factor in the generation and modulation of migraine pain (3,4). Trigeminal nerves can be stimulated by cytokines and inflammatory chemokines produced after stimulation with nitroglycerin (NTG) (5), the response to NTG is characterized by the release of neuropeptides, including calcitonin gene-related peptide (CGRP), from the trigeminal innervation. Cross-talk between neurons and immune cells contributes to pain, in which neurotransmitters play a crucial role (6). In innate immunity and inflammation, TLR4 is a very important receptor that is expressed by microglia and astrocytes. The activation of toll-like receptors (TLRs) leads to an increase in the transcription of genes encoding cytokines in the IL-1 and TNF families (7), including IL-1β, IL-6, IL-18 and TNF-α. Moreover, transient receptor potential vanilloid (TRPV4) is also an important signaling molecule and its expression increases in irritation-evoked trigeminal pain (8–11).
However, the regulatory mechanism of these factors is not yet clear, and the negative feedback mechanism has been disregarded. Immune checkpoint proteins play a crucial role in the regulation of cellular immunity, while endogenous programmed death ligand 1 (PD-L1), which can be detected in healthy neural tissues including the dorsal root ganglion, the spinal cord, nerves and the skin, acts as an immune checkpoint inhibitor (12). PD-L1 can suppress the function of T cells and induce immune tolerance via its selective receptor programmed death 1 (PD-1) (13,14). A previous study showed that PD-L1 can inhibit acute and chronic pain by suppressing the activity of nociceptive neurons via PD-1 in the dorsal root ganglion (DRG) in melanoma and normal neural tissues (12). However, the role and the underlying regulatory mechanisms of PD-L1/PD-1 in migraine pain remain unknown. The administration of NTG can induce migraine-like pain with symptoms such as sensory hypersensitivity and light-aversive behaviors. A model of NTG-induced pain has already been widely used for both basic and clinical research for migraine-like pain (15–18).
Therefore, in this study, we evaluated the expression and role of PD-L1/PD-1 in the TG in a mouse model of acute migraine.
Materials and methods
Mice
Male C57BL/6 J mice (6–8 weeks, 18–23 g) were purchased from Lingchang Biotechnology Co. Ltd. (Shanghai, China) and were housed in microisolator cages in the SPF facility of Fudan University. Mice were housed in plastic boxes in groups of five with water and food available ad libitum and kept on a 12:12 hours light-dark cycle. The studies adhered to the ARRIVE guidelines. The animal care and experimental protocols were approved by the Animal Care Committee of Fudan University.
Mouse model of migraine
Mice were randomly divided into two groups: The normal saline (NS) control and the NTG groups. NTG (10 mg/kg) was intraperitoneally (i.p.) injected to induce migraine-like pain. In the control group, mice were treated with the same volume of NS (i.p). At three time points (2 h, 4 h, 6 h) after NTG treatment, the mice were anesthetized by chloral hydrate (5 mg/10 g i.p.). Then, the temporal bones and the brain hemispheres were removed, and the TG tissues were separated from the cranial base. The tissue samples were collected for western blot analysis, quantitative reverse transcription-polymerase chain reaction (qRT-PCR) and immunofluorescent staining.
Tactile sensory testing
The mechanical thresholds in the face and hind paw regions were detected by the von Frey test (n = 8). The behavioral measurements were conducted in a blinded manner during the daytime (light cycle), from 2 pm to 5 pm. The room temperature and humidity were controlled for each experiment. All animals were habituated to the testing environment daily for 2 ∼ 3 days before testing. Tactile sensory testing was performed before and after the mice received NS, NTG or a PD-1 inhibitor. To test mechanical sensitivity, the mice were confined to boxes placed on an elevated metal mesh floor, and their hind paws and periorbital regions were tested separately using a series of von Frey hairs with logarithmically increasing stiffness (0.07–2.00 g, Stoelting). The von Frey hairs were applied perpendicularly to the central plantar surface and the periorbital region for each test. The threshold was recorded when the mouse quickly retracted its hind paw or head away from the rigid tip. The thresholds were determined based on at least three retractions over five stimulations, with an interval of at least 20 sec. The percentage of withdrawal was the percentage of retractions over 10 stimulations. The results are presented as g ± SEM. Data were recorded for each time point.
Administration of the PD1 inhibitor
For intravenous injection, a PD-1 inhibitor (5 mg/kg in 100 µl of PBS, MCE, USA, HY-101093) was administered into the tail vein of the mice (i.v.). The PD-1 inhibitor is a compound that can inhibit the function of PD-1. The mice in the control group were left untreated, the mice in the NS group were injected (i.p.) with NS, the mice in the inhibitor (IN) group were injected with the PD-1 inhibitor (i.v.), and the mice in the NTG + IN group were pretreated with NTG for 1 h and then injected with the PD-1 inhibitor (i.v.) for 1 h. Then, the von Frey test was conducted, or the trigeminal ganglia were dissected for analysis.
Quantitative reverse transcription-polymerase chain reaction
Forward and reverse primer sequences used in real-time PCR.

Western blot (WB) analysis
Total protein was extracted by protein lysis buffer (Shanghai KeJie Biotechnology Co. Ltd., China) (n = 6), and the protein content was detected using a BCA protein assay kit (Shanghai KeJie Biotechnology Co. Ltd., China). The protein samples were denatured and separated on a 10% SDS–PAGE gel and transferred to nitrocellulose membranes for 1.5 h at room temperature. Then, the membranes were incubated with the following primary antibodies: Rabbit anti-PD-1 (1:1000, Abcam Systems, EPR20665, USA), mouse anti-PD-L1 (1–2 µg/ml, Abcam Systems, Ab238697, USA), and mouse anti-β-actin (1:3000, Shanghai KeJie Biotechnology Co. Ltd., China) overnight at 4℃. The membranes were incubated with secondary donkey anti-rabbit or anti-mouse IgG antibodies (1:5000, Shanghai KeJie Biotechnology Co. Ltd., China) at 37℃ for 1 h. Finally, the immunoblots were detected with an ECL kit (Shanghai KeJie Biotechnology Co. Ltd., China) and visualized with X-ray films. Image J software was used to quantify the bands.
Immunofluorescence staining
Mice were anesthetized and perfused intracardially with 20 ml of PBS followed by 40 ml of 4% paraformaldehyde (PFA). The TG was collected and post-fixed in 4% PFA at 4℃ overnight, followed by gradient dehydration in sucrose. Then, the tissues were embedded in optimum cutting temperature compound at 4℃ overnight, and frozen in a −80℃ freezer. Tissue blocks were cut into 10 µM sections with a cryostat (Leica CM, Germany). For immunofluorescence, sections (n = 3) were randomly chosen to be blocked in PBS containing 0.3% Triton X-100 (Sigma, USA) and 10% donkey serum (Sigma, USA) for 1 h at room temperature and then incubated at 4℃ overnight with primary antibodies. The antibodies were as follows: Rabbit anti-PD-1 (1:100, CST, EPR20665,), mouse anti-PD-L1 (1:100, Abcam, Ab238697), mouse anti-Tuj1 (1:200, CST, D71G9), and rabbit anti-CGRP (1:200, CST, D5R8F). The sections were then incubated with secondary antibodies (1:1000, Invitrogen, USA) for 2 h at room temperature, and 49,6-diamidino-2-phenylindole (DAPI) (1:5000, Molecular Probes, USA) was used for nuclear staining. Immunofluorescence samples were visualized and photographed with an inverted DMI 400CS confocal microscope (Leica, Germany). To calculate the fluorescence intensity of CGRP in the different groups, three slices were chosen randomly from the control group, IN group and NTG+IN group, and the mean fluorescence intensity of the cells was analyzed by Image J software.
Statistical analysis
The statistical analyses were performed by SPSS 17.0 software (SPSS Inc., USA). The data are shown as the mean ± standard error of the mean (SEM). An a priori power analysis was conducted to determine the minimal sample size needed to obtain a statistical power of 0.80 at an alpha level of 0.05. On the basis of our previous studies evaluating the mechanical thresholds between NTG and the control group, the difference was 0.44 and the polled standard deviation was 0.23, and the minimal sample size was six mice for each group to analyze the statistical significance (GPower 3.1.9.2). The data fit the normal distribution and the homogeneity of variance based on tests of normality and variance. The behavioral data were analyzed using one-way analysis of variance (ANOVA) followed by Bonferroni post-hoc test. The PCR and WB data were analyzed using one-way ANOVA. p < 0.05 was considered statistically significant.
Results
Expression of PD-1 on TG neurons
We performed double immunofluorescence to evaluate the expression of PD-1 and Tuj1 in wild type mice with no induction of migraine like pain. Tuj1 is a marker of neurons. Double immunofluorescence showed that PD-1 and Tuj1 were coexpressed in the TG of control mice (Figure 1, n = 3), which indicated that PD-1 is expressed in the cytoplasm of healthy untreated TG neurons.
Immunofluorescence analysis of the expression of PD-1 showed that it was colocalized with Tuj-1 in the TG. Representative images of PD-1 (red), Tuj-1 (green) and DAPI (blue) in the control group; the merged images show the colocalization of DAPI, PD-1, and Tuj-1.
PD-L1/PD-1 is increased in a mouse model of acute migraine
We investigated the expression of PD-L1 and PD-1 in the TG in the mouse model by quantitative reverse transcription-polymerase chain reaction and western blotting. A von Frey test was used to test the mechanical threshold in the hindpaw region of the mice to ascertain the successful establishment of the migraine model 2 h after the injection of NTG. The mechanical threshold was decreased in the NTG model (Figure 2). The mRNA expression levels of PD-L1 and PD-1 in the TG were detected 2, 4, and 6 h after NS and NTG administration. The mRNA levels of PD-1 and PD-L1 were significantly elevated 2, 4 and 6 h after NTG administration compared to the levels in the NS-treated control mice (Figure 3(a), (b), n = 6). The same three time points (2, 4 and 6 h) were chosen to evaluate the protein levels of PD-L1 and PD-1 in the TG after NTG or NS injection. The protein levels of PD-1 and PD-L1 were increased from 2 h to 6 h after treatment, specifically, compared to the expression in NS-treated control mice; PD-1 was significantly increased 2 h and 6 h after treatment, and PD-L1 was most robustly increased 6 h after treatment (Figure 3(c)–(e), n = 6).
The thresholds of mechanical stimulation are decreased in the mouse model of NTG-induced migraine. Behavioral responses following the mechanical stimulation of the right hind paw region with different von Frey filaments (0.07, 0.16, 0.4, 0.6, 1.0, 1.4 and 2 g) were assessed in mice before and 2 h after the injection of NTG (n = 8) or NS (n = 8). The withdrawal thresholds and the percentage of retractions over 10 stimulations were analyzed. The mice in the control group were left untreated, the mice in the NS group were injected (i.p.) with NS. There was no significant difference in the thresholds of mechanical stimulation between the control and saline group. Data are shown as mean ± SEM. One-way analysis of variance for repeated measures was used, followed by Bonferroni post-hoc test (**p < 0.01, *p < 0.05). The mRNA and protein levels of PD-1 and PD-L1 are upregulated after NTG treatment. ((a),(b)) The expression of PD-1 and PD-L1 mRNA was significantly increased 2 h, 4 h and 6 h after treatment in the NTG group compared with the control group. ((c)–(e)) The protein levels of PD-1 and PD-L1 were increased from 2 h to 6 h after treatment; specifically, compared with the expression in the saline-treated mice, PD-1 was significantly increased 2 h and 6 h after treatment, and PD-L1 was most remarkably increased 6 h after treatment (**p < 0.01, *p < 0.05). Data are shown as mean ± SEM of three separate experiments.

Immunofluorescence labeling for PD-L1 and PD-1
Double immunofluorescence showed that PD-1 was present in the cytoplasm of TG neurons, and PD-L1 was present around the cytomembrane of TG neurons (Figure 3, n = 3). Additionally, the PD-L1 and PD-1 immunopositive signals observed 6 h after the injection of NTG were obviously enhanced compared to those observed in NS-treated control mice (Figure 4).
The expression of PD-1 and PD-L1 was upregulated 6 h after NTG treatment, as determined by immunofluorescence. (a) Representative images of PD-1 (green), PD-L1 (red), and DAPI (blue) in the control group; the merged image shows the colocalization of DAPI, PD-1 and PD-L1. (b) Representative images of PD-1 (green), PD-L1 (red), and DAPI (blue) in the NTG-treated group.
PD-1 inhibits pain in a mouse model of migraine
The von Frey test revealed a significant decrease in paw and head withdrawal thresholds 2 h after NTG i.p. injection compared with those of the NS-treated group (Figure 2; Figure 5). The mice were pretreated with NTG 1 h before the i.v. injection of a PD-1 inhibitor (5 mg/kg), and the paw withdrawal threshold was significantly decreased compared to that of the NTG-injected group (Figure 5(a), (b)); the head withdrawal threshold was remarkably decreased compared to that of the NTG-treated group, but there was no statistically significant difference (Figure 5(c)) (n = 8, *p < 0.05, **p < 0.05).
Inhibition of endogenous PD-1 induces more significant mechanical allodynia in a mouse model of migraine. (a) Percentage of paw retractions over 10 stimulations. (b) Paw withdrawal threshold. (c) Head withdrawal threshold. There was no significant difference in the thresholds of mechanical stimulation between the control and saline group, while the inhibition of endogenous PD-1 induces more significant mechanical allodynia in a mouse model of migraine. The data are the mean ± SEM, n = 8 mice/group. ((a),(b)).
PD1 suppresses the expression of TNF-α, IL-6, CGRP, IL-1β and IL-18
The mRNA expression of TNF-α, IL-6, CGRP, IL-1β, IL-18 and TRPV4 in the TG was significantly increased 2 h after treatment in the NTG-injected group compared with the control group (n = 6). The administration of a PD-1 inhibitor had no significant effects on the mRNA expression of CGRP, TNF-α, IL-6, IL-1β, IL-18 or TRPV4. One hour after the i.v. injection of a PD-1 inhibitor (5 mg/kg, p < 0.05), which was administered 1 h after NTG injection, the mRNA expression of CGRP, IL-1β, TNF-α, IL-6 and IL-18 was significantly increased compared with that observed 2 h after NTG injection (Figure 6 (a)–(e). However, TRPV4 was not affected by the PD-1 inhibitor (Figure 6 (f)). Double immunofluorescence of CGRP and PD-L1 showed that the expression of CGRP was elevated 2 h after the injection of NTG. The PD-1 inhibitor more remarkably increased the expression of CGRP, but based on statistical analysis, there was no significant difference between the NTG and NTG+IN groups (Figure 7(a), (b)). The fluorescence intensity of PD-L1 was enhanced in the NTG+IN and NTG groups compared to the control group (Figure 7(a), (b)).
PD-1 might inhibit pain sensitivity by downregulating the expression of TNF-α, IL-6, CGRP, IL-1β and IL-18 in a model of NTG-induced migraine. ((a)–(f)) The mRNA levels of CGRP, IL-1β, IL-18, TNF-α, IL-6 and TRPV4 were upregulated 2 h after NTG treatment. ((a)–(e)) The intravenous injection of a PD-1 inhibitor significantly increased the mRNA levels of TNF-α, IL-6, CGRP, IL-1β and IL-18. (f) The mRNA level of TRPV4 was not affected by the PD-1 inhibitor (5 mg/kg). The mice in the control group were left untreated, the mice in the NS group were injected (i.p.) with NS, the mice in the inhibitor (IN) group were injected with the PD-1 inhibitor (i.v.), and the mice in the NTG+IN group were pretreated with NTG for 1 h and then injected with the PD-1 inhibitor (i.v.) for 1 h. The immunofluorescence expression of CGRP was enhanced after the i.v. injection of a PD-1 inhibitor in a mouse model of migraine. The fluorescence intensity of CGRP was enhanced 2 h after NTG injection. The PD-1 inhibitor more remarkably increased the expression of CGRP (arrows, (a)), but there was no significant difference between the NTG and NTG+IN groups (b). The fluorescence intensity of PD-L1 was enhanced in the NTG+IN and NTG groups compared with the control group ((a),(b)). The mice in the control group were left untreated, and the mice in the NTG+IN group were pretreated with NTG for 1 h and then injected with the PD-1 inhibitor (i.v.) for 1 h. CGRP (green), PD-L1 (red), and DAPI (blue).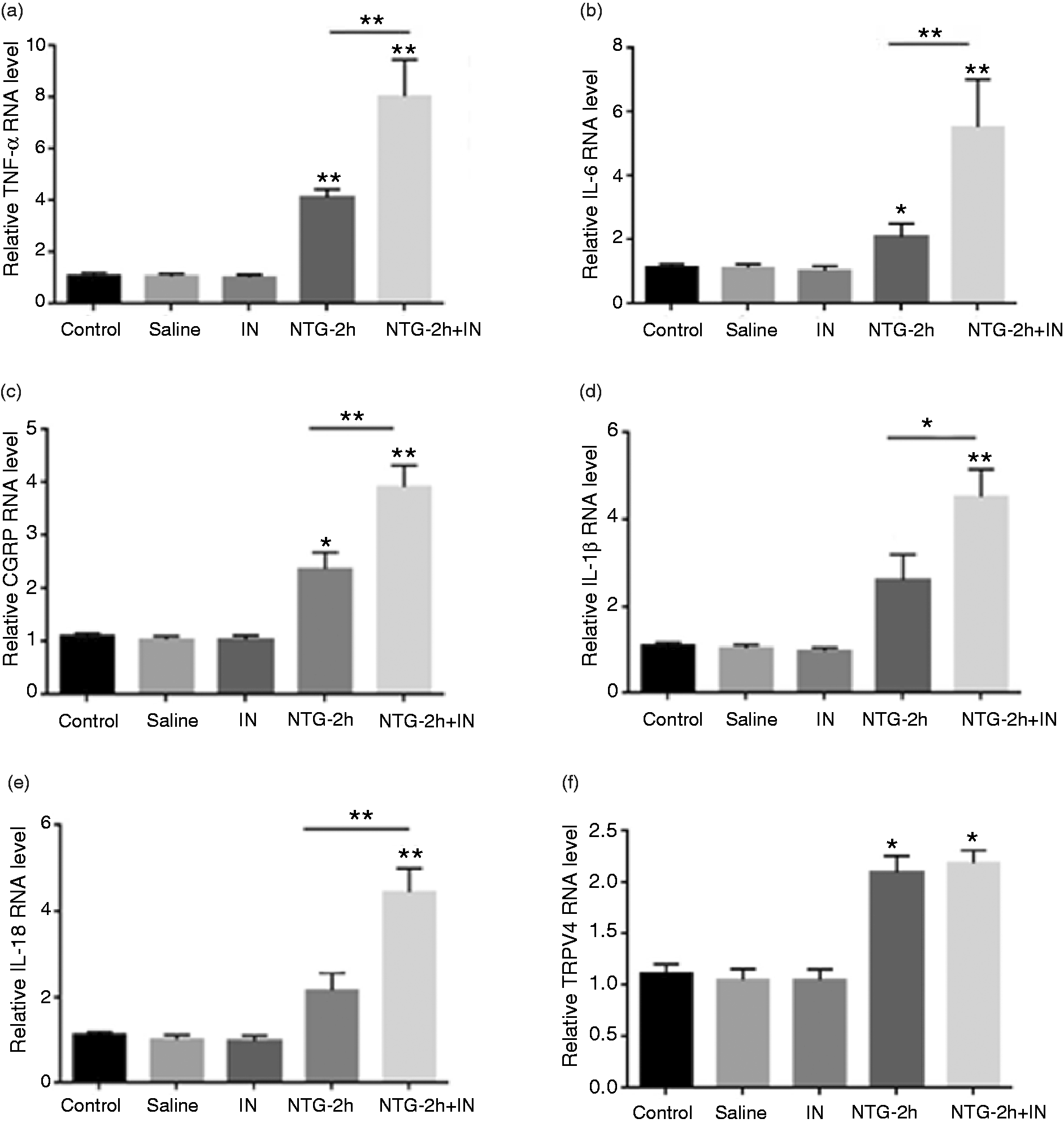

Discussion
In summary, PD-L1 and PD-1 are expressed in TG neurons. PD-L1 and PD-1 are upregulated in a model of NTG-induced migraine and may have a reversal effect on pain by preventing CGRP expression and other proinflammatory responses in the TG. The systemic administration of NTG has been widely used to study migraine, as it can cause migraine-like headache in migraineurs and rodents, and effectively mimic symptoms in migraineurs (19). NTG can induce the activation of primary trigeminovascular afferents (20), and it can induce the release of inflammatory neuropeptides and other pro-inflammatory mediators, promoting neurogenic inflammation and the development of peripheral sensitization of primary nociceptive neurons, including trigeminal ganglion nerves (21,22). PD-1 plays an important role in balancing immunopathology and protective immunity, homeostasis and tolerance. Our findings provide the first evidence that PD-1 is expressed in TG neurons in healthy tissue, which indicates that primary nociceptive neurons may play a role in immune balance. PD-L1, a checkpoint inhibitory protein, is the main functional ligand of the coinhibitory PD-1. Together with its cognate ligand PD-L2, PD-L1 plays a key role in maintaining peripheral and central immune cell tolerance through binding to the PD-1 receptor (23). In our research, we found that PD-L1 was also expressed in TG neurons, indicating that the release of PD-L1 may activate the peripheral trigeminal nerve via binding PD-1.
The activation of PD-1 by PD-L1 can modulate neuronal excitability in the dorsal spinal cord of the peripheral nervous system and synaptic transmission in the spinal cord of the central nervous system both in humans and mice (28). Neurogenic inflammation and peripheral sensitization of primary nociceptive neurons are important mechanisms in migraine pathophysiology, and pro-inflammatory factors have been shown to induce the upregulation of the expression of PD-1 and PD-L1 (24–27), which suggests that PD-L1/PD-1 may be involved in migraine pathophysiology. NTG induces a migraine attack, which is delayed 2–6 h after infusion (29,30). Therefore, we analyzed the expression of PD-L1/PD-1 between 2 and 6 h after i.p. injection. The immunofluorescence intensities of PD-L1 and its selective receptor PD-1 were enhanced in a mouse model of migraine at 6 h. The mRNA levels of PD-1 and PD-L1 were significantly elevated 2, 4 and 6 h after NTG administration compared to those observed in NS-treated control mice. The protein levels of PD-1 and PD-L1 were increased from 2 h to 6 h after treatment; specifically, compared to the expression in NS-treated control mice, PD-1 was significantly increased 2 h and 6 h after treatment, and PD-L1 was most robustly increased 2 h, 4 h and 6 h after treatment, indicating that the activation of PD-L1/PD-1 in the peripheral trigeminal nerve may play a role in migraine. While our data highlights the effects of PD-L1 and PD-1 on NTG-induced migraine, it should be noted that the current study was conducted on only male mice. Given that migraine is significantly more common in females and clear gender-dependent effects are observed in mouse experimental traits (31), the impact of modulation of PD-L1 and PD-1 receptors in females remains to be confirmed. Moreover, to understand the specific mechanisms involved, further investigation is required.
Pretreatment with a PD-1 inhibitor decreased the pain threshold and increased the expression of CGRP and related inflammatory factors, including TNF-α, IL-6, IL-1β and IL-18, but had no effect on the expression of TRPV4. Sensitivity to mechanical stimuli was tested by the von Frey test, and the pain threshold was decreased in the migraine model (32). In the TG, CGRP, an important inflammatory mediator, can modulate pain transmission through neuron-glia signaling, contributing to various orofacial pain conditions. Many previous clinical trials have proven that CGRP receptor antagonists are effective for treating migraine, and the increase in CGRP in the TG induced by the PD-1 inhibitor indicates that PD-1 may inhibit sensitivity to pain in the peripheral nervous system. CGRP contributes to both neurogenic inflammation and peripheral sensitization of nociceptive neurons (33,34), and PD-1 can inhibit both CGRP and inflammation in migraine, contributing to inhibiting pain sensitivity. Although TRPV4 is abundantly expressed in TG neurons and is necessary for trigeminal irritant pain (9), it is not modulated by PD-1. These results may indicate that PD-1 inhibits CGRP and inflammatory factors in the TG and that PD-L1/PD1 may act as a negative feedback mechanism in migraine to dampen inflammation in the TG.
Conclusion
In conclusion, our study revealed that the PD-L1 and its receptor PD-1 were expressed on TG; PD-L1/PD-1 could be acting on migraine pathophysiology by down-regulating CGRP and inflammatory factors in the TG. However, the underlying mechanism of PD-L1 and PD-1 on migraine need further research.
Article highlights
PD-L1 and its receptor PD-1 are expressed on TG neurons. PD-L1 and PD-1 down-regulate the expression of CGRP and inflammatory factors in the TG. PD-L1 and PD-1 might be involved in migraine pathophysiology.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by National Natural Science Foundation of China (No. 81670933).
