Abstract
Abstract
Background
Serum histamine, immunoglobulin E, and tryptase are markers of allergic diseases. Despite the reported association between migraine and allergic diseases, differences in these marker levels between episodic and chronic migraines remain unelucidated.
Methods
We investigated serum histamine, immunoglobulin E, and tryptase levels in 97 and 96 participants with episodic migraine and chronic migraine, respectively, and 56 controls according to the presence of allergic diseases.
Results
Serum histamine levels in episodic migraine (median and interquartile ranges, 0.78 [0.65–1.25] ng/mL, p < 0.001) and chronic migraine (0.89 [0.67–1.28] ng/mL, p < 0.001) participants were significantly lower than those in healthy controls (1.19 [0.81–2.08] ng/mL) among the 160 participants without allergic diseases. Serum immunoglobulin E levels in episodic migraine and chronic migraine participants with allergic diseases negatively correlated with headache frequency (correlation coefficient = −0.263, p = 0.017). Serum histamine levels in participants with allergic diseases and serum immunoglobulin E levels in participants without allergic diseases were not significantly different among episodic migraine, chronic migraine, and control groups. Serum tryptase levels did not significantly differ among episodic migraine, chronic migraine, and control participants with and without allergic diseases.
Conclusions
Altered serum histamine and immunoglobulin E levels in episodic migraine and chronic migraine and different profiles concerning allergic diseases suggest the involvement of allergic mechanisms in migraine pathogenesis.
Introduction
A close association between migraine and allergic diseases has been reported. Individuals with allergic diseases have a higher prevalence of migraine than those without allergic diseases and vice versa (1,2). Calcitonin gene-related peptide (CGRP) is a crucial mediator of migraine. Furthermore, elevated plasma CGRP levels have been observed in patients with allergic rhinitis (3). Moreover, many individuals with migraine experience attacks through exposure to allergens (4). Immunotherapy for allergic diseases may improve migraine frequency and disability in patients with allergies and migraines (5).
Histamine and immunoglobulin E (IgE) are pivotal in allergic and anaphylactic responses. Immunoglobulin E binds to mast cells, among the main inflammatory cells involved in allergic reactions, and sensitizes them to release inflammatory mediators, including histamine (6). Histamine is crucial in migraine pathophysiology, including mediating the interaction between mast cells and CGRP, maintaining neurogenic inflammation, and sensitizing the trigeminal ganglia (7–9). Histamine infusion can induce migraine-like headache attacks (10). In addition to being the major effectors in allergic reactions, mast cells are critical contributors to the pathogenesis of migraine. Activation of meningeal mast cells located around the perivascular space near neurons has been observed during migraine attacks (11).
Histamine and IgE levels are reportedly altered in the body fluids of individuals with migraine (12–14). Migraine is categorized into episodic migraine (EM) or chronic migraine (CM). EM and CM differ in prevalence, comorbidities, treatment responses, and biomarkers (15,16). Nevertheless, we lack reports on possible differences in the levels of histamine and IgE between EM and CM. To date, only one study has reported migraine-type-specific differences in histamine and IgE levels (12). Furthermore, serum tryptase levels, a marker of mast cell activation, in individuals with migraine remain unreported. We hypothesized that serum histamine, IgE, and tryptase levels were altered in individuals with EM and CM compared with control participants. These differences might vary according to the presence of allergic diseases.
Therefore, according to allergic diseases, we aimed to evaluate the serum histamine, IgE, and tryptase levels in participants with EM and CM.
Methods
Participants
The inclusion criteria were as follows: a) adults aged 19–65 years; b) fulfilling the criteria of migraine (code 1.1 or 1.2) or CM (code 1.3) in the third edition of the International Classification of Headache Disorders (ICHD-3) (17); c) at least 72 h after the cessation of an episode of headache attack for EM, and having less than mild headache intensity for CM; and d) a complete understanding of the study protocol. The exclusion criteria were as follows: a) secondary headache(s) other than medication-overuse headache (MOH) (17), b) chronic pain conditions other than fibromyalgia (FM), and c) current treatment for medical or psychiatric disorders. We enrolled participants who were taking antidepressants for migraine prevention but excluded those who were taking antidepressants for the treatment of depression. CM was diagnosed according to CM criteria in ICHD-3. EM was diagnosed if a participant fulfilled the criteria of migraine but did not fulfill the criteria of CM with <15 headache days per month during the previous three months. The healthy controls, recruited through advertisements, were considered eligible if they had no headache attacks in the last year and no migraine attacks during their lifetime. We did not enroll individuals under medications for allergic diseases.
Sample size
The sample size was computed based on previous studies on serum histamine and IgE levels in individuals with migraine (12,14). The ratio of participants with the disease to healthy controls was 1:2. Assuming a 5% significance level and 80% power, the minimal sample sizes of the disease and control groups were computed as 10 and 20, respectively. We aimed to admit 45, 90, and 90 participants as control, EM, and CM, respectively, to investigate the association of serum histamine, IgE, and tryptase levels with clinical characteristics.
Serum preparation
A 5 mL blood sample was obtained from the right antecubital vein between 9:00 and 12:00 a.m. using a Vacutainer® SST™ tube (Becton Dickinson, NJ, USA). The collected whole blood was left undisturbed at room temperature for 15 min to facilitate coagulation. The clot was removed by centrifugation at 1,500 × g for 15 min at 4°C. Following centrifugation, the resulting supernatant was collected and stored at −70°C until further use.
Measurement of histamine, IgE, and tryptase
Serum histamine levels were quantified using an enzyme-linked immunosorbent assay (ELISA) kit (ab213975) (Abcam, Cambridge, UK) following the manufacturer’s instructions. The linear range of the histamine ELISA kit was 0.098–25 ng/ml. The estimated inter- and intra-assay accuracies were 5.2% and 4.6% coefficient of variation, respectively. Histamine ELISA was carried out in duplicate.
Total IgE and tryptase levels were quantified based on a fluorescence enzyme immunoassay using ImmunoCAP® total IgE and tryptase systems, respectively, of Phadia™ 250 Immunoassay Analyzer (Phadia AB, Uppsala, Sweden). Unless otherwise described, IgE and tryptase levels refer to total IgE and tryptase levels, respectively. The measuring range for IgE and tryptase were 2–5000 kU/L and 1–200 ng/mL, respectively. The estimated inter- and intra-assay accuracies for IgE were 3.6% and 3.9% coefficient of variation, respectively. Those for tryptase were 2.7% and 3.2% coefficient of variation, respectively.
Assessment of allergic diseases
Allergic diseases were identified based on the participants’ self-report. Participants with allergic eczema, conjunctivitis, rhinitis, atopic dermatitis, food allergy, or anaphylaxis were considered allergic diseases.
Assessment of anxiety, depression, MOH, and FM
Anxiety and depression were evaluated using the Generalized Anxiety Disorder-7 (GAD-7) and Patient Health Questionnaire-9 (PHQ-9), respectively. A GAD-7 score ≥8 and a PHQ-9 score ≥10 were classified as having anxiety and depression, respectively (18,19). MOH (code 8.2) was diagnosed based on the ICHD-3 guidelines. Fibromyalgia was diagnosed based on the 2016 American College of Rheumatology criteria (20).
Statistical analyses
Binary and ordinal scales are presented as numbers and percentages. The Kolmogorov–Smirnov test assessed the normality of continuous variables. Variables with normal distribution were analyzed using an independent t-test or analysis of variance. The results of the tests are expressed as mean ± standard deviation. Conversely, those with non-normal distribution were assessed using the Mann–Whitney U or Kruskal–Wallis test. The test results are presented as medians (interquartile ranges). Categorical variables were compared when appropriate using the chi-square or Fisher’s exact test.
We used correlation analyses to evaluate the relationship between IgE, histamine levels, and monthly headache frequency. If variables were normally distributed, we used Pearson’s correlation analyses. If variables were not normally distributed, we used Spearman’s rank correlation analyses (21).
Statistical significance was set at a two-tailed p < 0.05. Post hoc analyses were performed using Bonferroni’s method. Consequently, we applied p < 0.017 when post hoc analyses were performed among the three groups. IBM Statistical Package for Social Sciences software (IBM SPSS version 25.0; IBM Corp., Armonk, NY, USA) was used for statistical analyses.
Ethical approval
This study was reviewed and approved by the Institutional Review Board of Severance Hospital, Yonsei University (approval no. 2019-1403-005). This study’s design and all processes were conducted following the Declaration of Helsinki and its subsequent amendments. All participants provided written informed consent before enrollment.
Results
Participants and distribution of anxiety, depression, FM, and allergic diseases
We enrolled 249 participants (EM [n = 97], CM [n = 96], and healthy controls [n = 56]) in the Department of Neurology outpatient clinic at a tertiary university hospital between October 2019 and October 2020. The age, sex, and body mass index distribution of the participants with EM and CM, and healthy controls were not significantly different (Table 1). Phonophobia, anxiety, depression, FM, and MOH were more prevalent in participants with CM than in those with EM. In contrast, unilateral pain and migraine with aura were more prevalent in the participants with EM than in those with CM. All participants with EM and CM underwent acute treatment. 16 (16.5%) and 35 (36.5%) participants with EM and CM took preventive medications, respectively. No participant with EM and CM used onabotulinumtoxinA or anti-CGRP monoclonal antibody for migraine treatment.
Demographic and clinical characteristics of the participants with episodic migraine, chronic migraine, and controls.
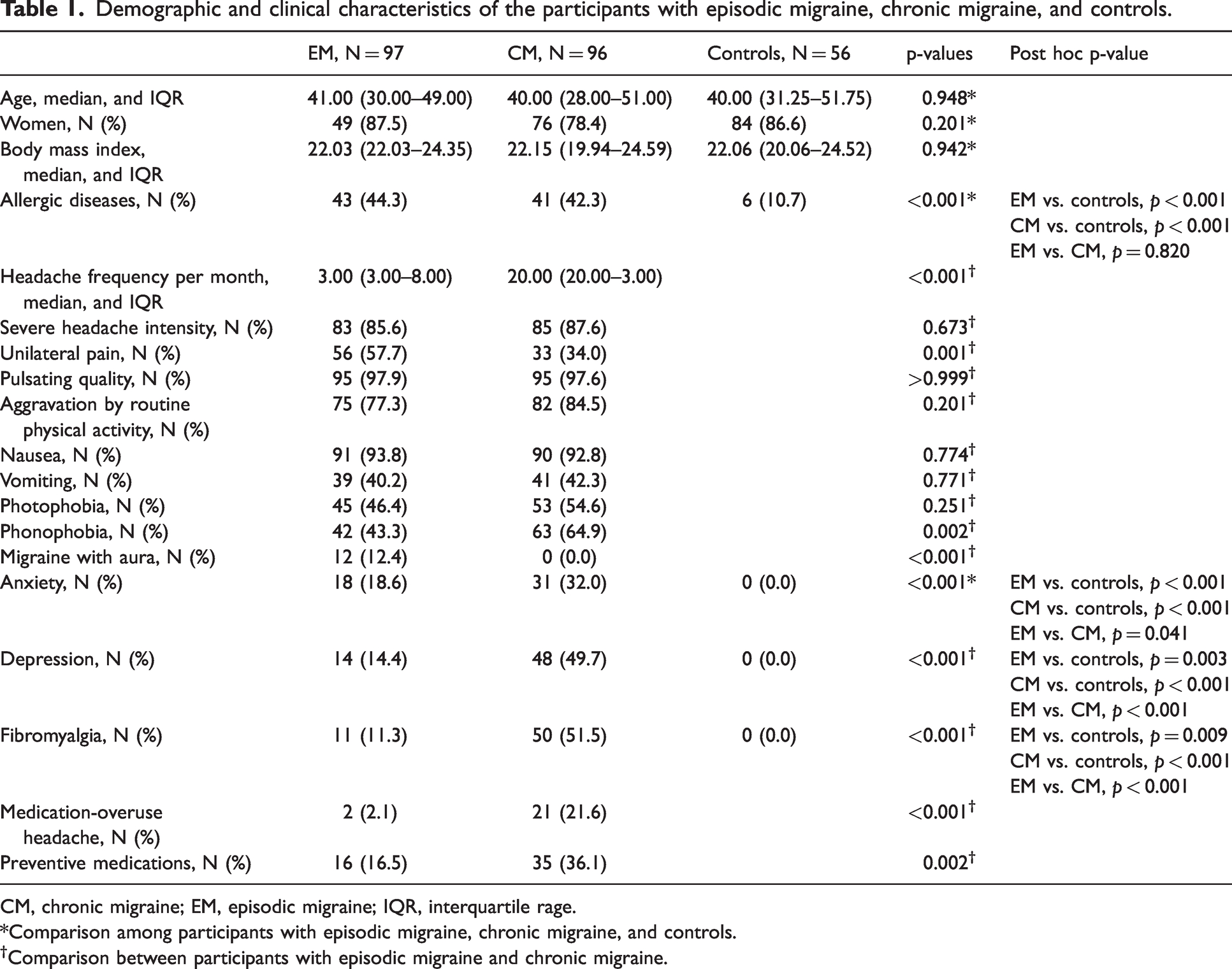
CM, chronic migraine; EM, episodic migraine; IQR, interquartile rage.
*Comparison among participants with episodic migraine, chronic migraine, and controls.
Comparison between participants with episodic migraine and chronic migraine.
Among the 249 participants, 89 (EM [n = 43], CM [n = 40], and healthy controls [n = 6]) had allergic diseases, and the remaining 160 participants (EM [n = 54], CM [n = 56], and healthy controls [n = 50]) did not have allergic diseases. The distribution of allergic diseases is summarized in Online Supplementary Table 1. The frequency of allergic diseases was significantly different among the EM, CM, and healthy control groups (44.3% [43/97] vs. 41.7% [40/96] vs. 10.7% [6/56], p < 0.001). Post hoc analysis revealed that allergic diseases were more prevalent in the EM (p < 0.001) and CM (p < 0.001) groups than in the healthy control group. Nevertheless, the frequencies of allergic diseases between the EM and CM groups did not differ significantly (p = 0.820).
Serum histamine, IgE, and tryptase levels
Serum histamine (p = 0.094), IgE (p = 0.183), and tryptase (p = 0.356) levels did not differ significantly among the 249 participants (Figure 1 and Online Supplementary Table 2). However, serum histamine (1.17 [0.83–2.17] ng/mL vs. 0.96 [0.70–1.35] ng/mL, p = 0.005) and IgE (72.90 [29.00–202.50] kU/L vs. 44.30 [23.60–109.75] kU/L, p = 0.049) levels were significantly higher in the 89 participants with allergic diseases than those in the 160 participants without allergic diseases. However, tryptase levels (3.15 [2.38–3.91] ng/mL vs. 3.29 [2.50–4.05] ng/mL, p = 0.556) did not significantly differ between participants with and without allergic diseases.
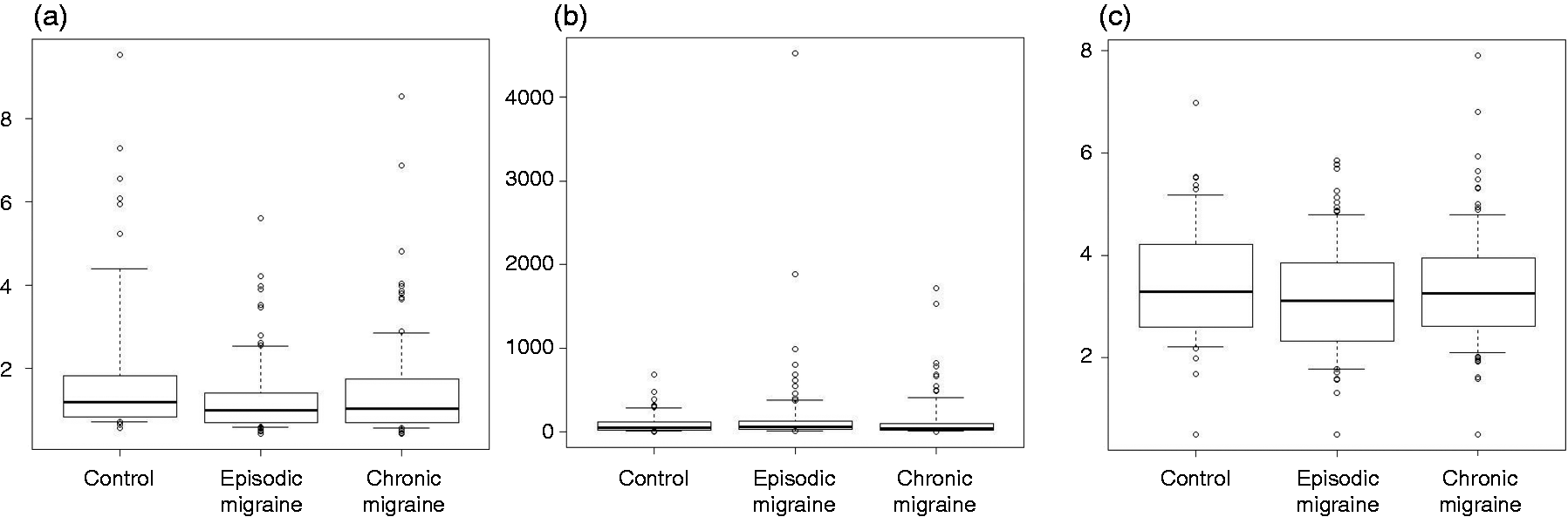
Serum histamine (a), immunoglobulin E (b), and tryptase (c) levels in participants with episodic migraine, chronic migraine, and controls.
Serum histamine, IgE, and tryptase levels in participants with allergic diseases
Of the 89 participants with allergic diseases, IgE levels significantly differed among the EM, CM, and control groups (p = 0.024). Post hoc analyses revealed no significant differences between the CM and control groups (p = 0.039), between the EM and control groups (p = 0.315), and between the EM and CM groups (p = 0.025). Conversely, histamine (p = 0.907) and tryptase (p = 0.611) levels were not significantly different among the three groups with allergic diseases (Figure 2 and Online Supplementary Table 2).
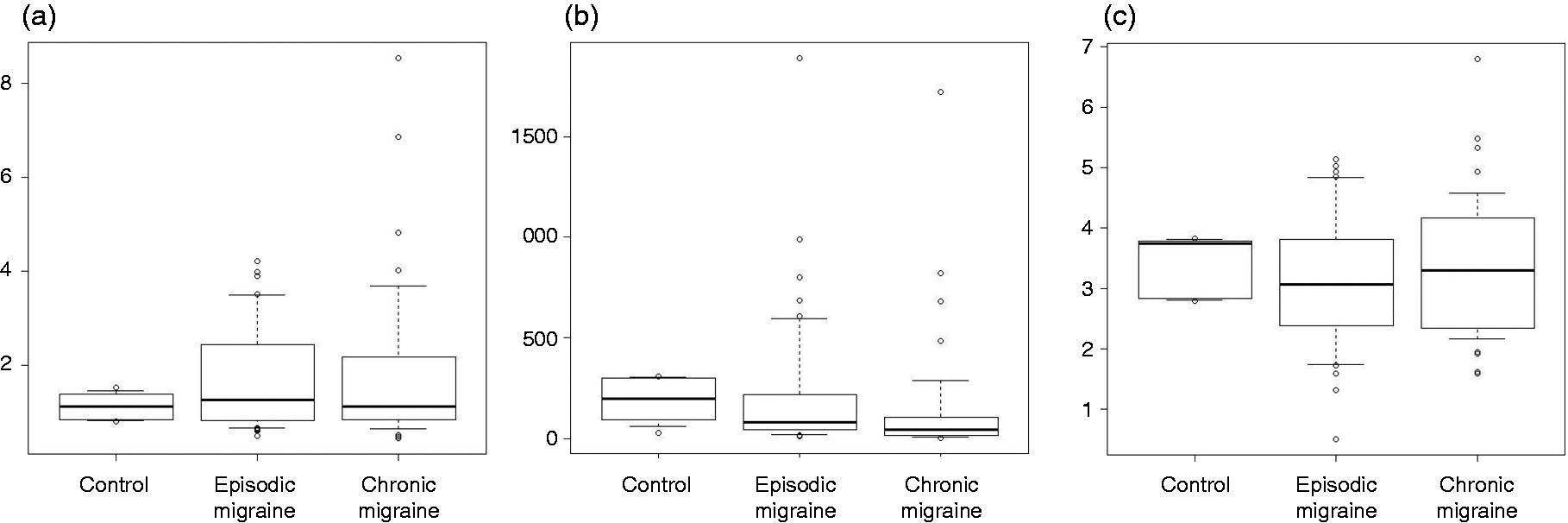
Serum histamine (a), immunoglobulin E (b), and tryptase (c) levels in participants with episodic migraine, chronic migraine, and controls with allergic diseases.
Serum histamine, IgE, and tryptase levels in participants without allergic diseases
Of the 160 participants without allergic diseases, histamine levels significantly differed among the EM, CM, and control groups (p = 0.002). Post hoc analyses revealed that histamine levels were significantly higher in the control group than those in the EM (p = 0.001) and CM (p = 0.011) groups. Nevertheless, histamine levels did not significantly differ between the EM and CM groups (p = 0.389). IgE and tryptase levels did not differ significantly among the three groups without allergic diseases (Figure 3 and Online Supplementary Table 2).

Serum histamine (a), immunoglobulin E (b), and tryptase (c) levels in participants with episodic migraine, chronic migraine, and controls without allergic diseases.
Association of IgE levels with clinical characteristics of EM and CM with allergic diseases
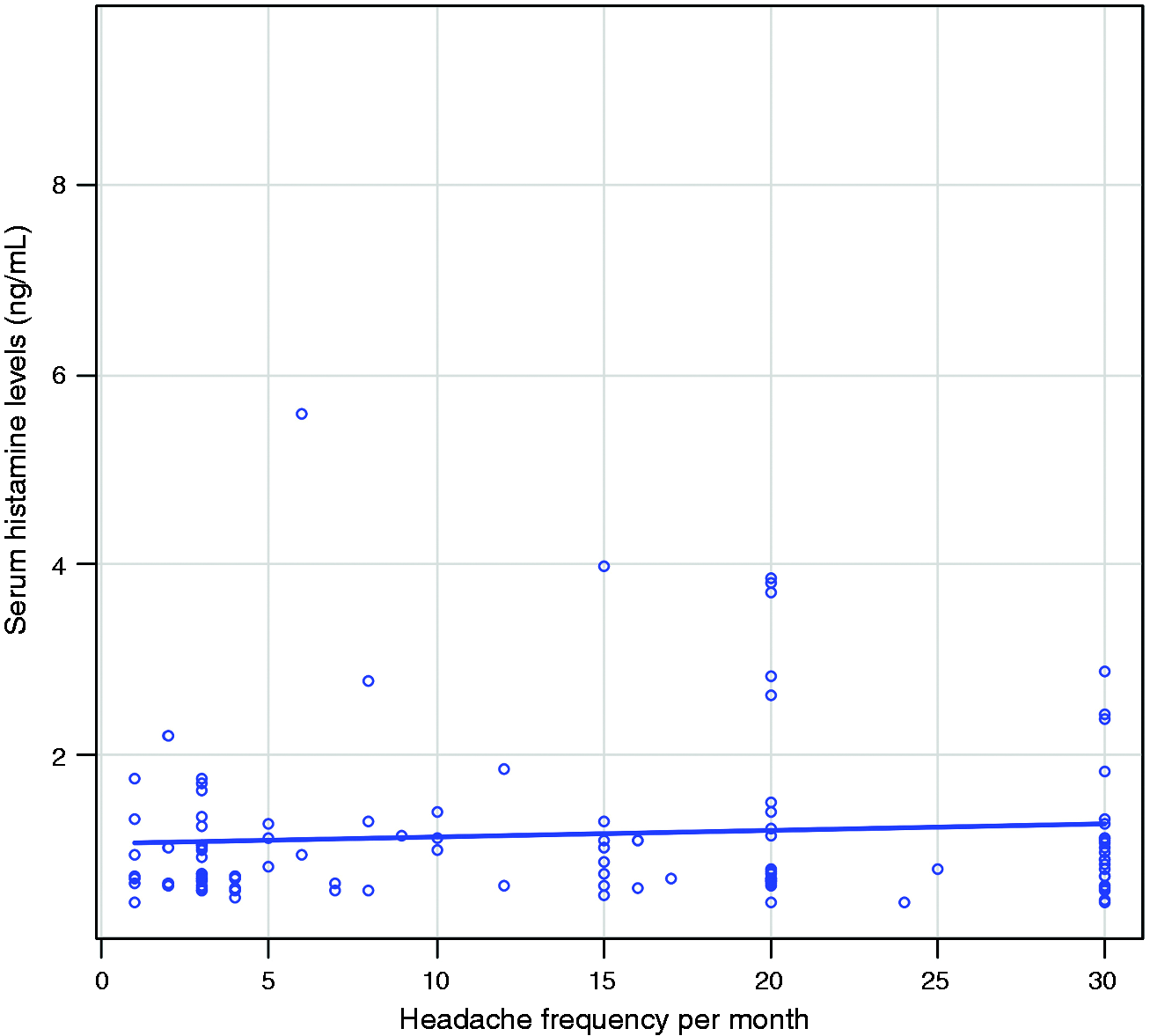
The combined results from 43 EM and 40 CM participants with allergic diseases indicated that IgE levels, except for FM and MOH, were comparable according to clinical characteristics and comorbidities. Serum IgE levels in participants with FM and MOH were lower than those without FM and MOH (Online Supplementary Table 3). IgE levels and headache frequency per month were not normally distributed; therefore, we conducted a Spearman’s rank correlation analysis. The analysis revealed that IgE levels were negatively associated with monthly headache frequency (correlation coefficient = −0.263, p = 0.017) (Figure 4).

Association between immunoglobulin E levels and monthly headache frequency in 43 and 40 participants with episodic and chronic migraines with allergic diseases, respectively.
Association of histamine levels and clinical characteristics of EM and CM without allergic diseases
The results of the 54 EM and 56 CM participants without allergic diseases revealed that serum histamine levels did not differ significantly according to clinical characteristics, comorbidities, or preventive treatment (Online Supplementary Table 4). As histamine levels and monthly headache frequency were not normally distributed, we conducted a Spearman’s rank correlation analysis. The analysis revealed no significant association between histamine levels and headache frequency (correlation coefficient = 0.112, p = 0.245) (Figure 5).
Association between histamine levels and monthly headache frequency in 54 and 40 participants with episodic and chronic migraines without allergic diseases, respectively.
Discussion
The present study’s main findings are as follows: 1) serum histamine, IgE, and tryptase levels were not significantly different among participants with EM and CM, and controls; 2) among participants without allergic diseases, serum histamine levels significantly decreased in those with EM and CM compared with healthy controls; 3) among participants with allergic diseases, serum IgE levels had a significant negative association with monthly headache frequency in participants with EM and CM; and 4) serum tryptase levels did not significantly differ among the EM, CM, and controls for participants with and without allergic diseases.
Degranulation of mediators from mast cells is essential in developing neurogenic inflammation in migraine pathogenesis (22). Histamine and serotonin are key mediators of mast cells (23). Histamine levels are reportedly altered in the body fluids of individuals with migraine. Heatley et al. (13) reported elevated plasma histamine levels in 18 individuals with migraine compared with that in 19 controls during the pain-free period. Furthermore, the study revealed elevated histamine levels in 12 individuals with migraine during attacks. Haimart et al. (14) demonstrated elevated plasma histamine levels in 18 individuals with migraine during remission and attack periods compared with 28 controls. Gazerani et al. (12) observed elevated histamine levels in 70 individuals with migraine during pain-free and pain periods compared with 45 controls. Heatley et al. and Haimart et al. also examined the whole blood but observed no significant differences in histamine levels between individuals with migraine and controls (13,14). These findings suggest that the change in plasma histamine levels could be attributed to the altered distribution of histamine among blood components rather than an increase in histamine in specific blood components. Haimart et al. observed an increased proportion of plasma histamine levels in the whole blood of individuals with migraine, supporting this hypothesis (14).
Our study presented results contradictory to previous studies in that serum histamine levels were significantly decreased in participants with EM and CM compared with controls among participants without allergic diseases. One explanation for this discrepancy might be the difference in the body fluids used for the evaluation. Heatley et al. and Haimart et al. did not use serum but plasma, which is likely devoid of histamine released during platelet aggregation (13,14). Gazerani et al. used serum; however, they used acid-washed glass tubes, which might have influenced platelet aggregation (12). The increase in plasma histamine levels in previous studies can be explained by the activation of mast cells during a migraine attack, which results in the release of histamine from mast cells and an increase in the aqueous portion of the blood (22). An increase in histamine in the aqueous portion of the blood might occur in migraine; however, the contribution of decreased histamine release from platelet aggregation is plausibly more pronounced, resulting in decreased serum histamine levels. The plasma fraction was <3% of total blood histamine during the ictal period and <2% during the interictal period (14).
Histamine is a biogenic amine similar to serotonin. Furthermore, human platelets can take up preformed biogenic amines by a calcium-and-energy-dependent process (24). Therefore, to clarify the cause of the decreased serum histamine levels in the EM and CM groups without allergic diseases, it is necessary to confirm whether histamine storage in platelets decreases in individuals with EM and CM compared with healthy controls. Changes in platelets and their metabolism in migraine reportedly alter the number and volume of platelets, reduce platelet monoamine oxidase activity, increase the expression of fibrinogen and glycoprotein IIb receptors, and increase the interaction between platelets and leukocytes (25–29). A 30–45% reduction in platelet serotonin, another common monoamine in platelets, was observed during migraine attacks (30,31). Among participants with allergic diseases, serum histamine increased by allergic diseases, offsetting the decrease in EM and CM so that no significant difference was observed. The findings of our present study and previous studies collectively suggest the altered histamine metabolism in individuals with migraine and the potential involvement of histamine in the pathophysiology of migraine (32).
Another possible reason for the discrepancy between the present study and previous studies is a difference in ethnicity. The previous studies were conducted in France, Canada, and Iran, where many participants are likely Caucasians, with fewer Asians (12–14). In contrast, all of our participants were Korean, an Asian ethnicity.
Several studies have evaluated IgE levels in individuals with migraine (33,34). Nevertheless, only Gazerani et al. evaluated IgE levels in individuals with migraine compared with controls in the presence of allergic diseases (12). They observed elevated levels of IgE in individuals with migraine compared with controls. Furthermore, the elevation of IgE levels was more prominent in individuals with migraine and allergic diseases than in those without allergic diseases. However, we observed conflicting results; serum IgE levels were lower in individuals with EM and CM than in controls among participants with allergic diseases. This discrepancy between our findings and that of Gazerani et al. may be attributed to the use of different body fluids, measurement methods, distribution of the respective allergic diseases, and ethnicity of the participants. Decreased IgE levels in participants with EM and CM also suggest the possibility of a non-IgE-dependent allergic disease process in these populations. Nevertheless, further investigations according to migraine subtypes, clinical characteristics, and migraine occurrence will provide novel insights into the association between IgE, migraine, and allergic diseases.
In this study, we observed that the serum histamine levels of individuals with EM and CM significantly differed from those of controls among participants without allergic diseases but not those with allergic diseases. In contrast, serum IgE levels were highest in controls, followed by the EM group, and lowest in the CM group among participants with allergic disease. However, IgE levels in the EM, CM, and control groups were similar in participants without allergic diseases. These findings suggest that histamine and IgE are implicated in the pathogenesis of migraine. However, they play different roles depending on the presence or absence of allergic diseases.
Mast cells are the primary inflammatory cells involved in allergic diseases. Furthermore, tryptase is the most abundant mediator stored in mast cell granules. Total tryptase serves as a marker for mast cell activation (35). Elevated levels of serum total tryptase occur during anaphylactic reactions, the active phase of allergic diseases, and hematologic disorders (36). The total serum tryptase concentration sharply rises during the first 90 min from the onset of reactions, followed by a steady decline, and takes at least 24 h to return to baseline levels (37). In this study, we demonstrated that the serum tryptase levels in the EM and CM groups were not significantly different from those in the control group, suggesting that mast cell activity did not differ between the EM, CM, and control groups during the interictal period. As serum tryptase levels and mast cell activities were not evaluated during the ictal period in the present study, we could not exclude the involvement of mast cells in migraine pathogenesis.
The present study has several limitations. First, we did not evaluate the platelet count of participants in the present study. We attributed the decreased serum histamine levels in participants with EM and CM to possible platelet number and metabolic alterations in these populations. Studies on platelet number in migraine have reported conflicting results; however, platelet number may affect serum histamine levels based on previous studies (24,38). Nevertheless, we did not examine the platelet counts of the participants in the present study. Second, participants with EM and CM were recruited from a tertiary university hospital. Therefore, the present study’s findings may not accurately and equitably reflect all EM and CM populations and may lack generalizability. Further studies on various subpopulations are required to provide more generalizable results. Third, the present study used data with a sufficient sample size based on the findings of previous studies; however, some subgroup analyses may not have had an adequate sample size. Specifically, our analysis revealed that the serum IgE levels among participants with EM and CM, and controls with allergic diseases were significantly different; however, the sample size of controls (n = 6) was too small. Therefore, the results should be interpreted cautiously, and our findings should be confirmed in a larger study. In addition, histamine and IgE levels according to clinical characteristics, comorbidities, and preventive treatment did not have sufficient sample power, suggesting an insufficient sample size. Nevertheless, we believe the results provide original information on histamine and IgE levels according to various conditions in the migraine population. Fourth, we assessed allergic diseases based on reports from participants rather than those from doctors using diagnostic criteria and objective methods, which could be a limitation of this study. Fifth, we assessed participants’ acute and preventive treatment; however, detailed names and dosages of medications were not evaluated. Acute treatment medications such as ibuprofen, naproxen, and aspirin can affect platelet aggregation (39). Given that our findings suggest that platelet aggregation influences histamine levels, not evaluating the detailed names and doses of acute treatment medications is a limitation of the present study.
In conclusion, serum histamine levels in individuals with EM and CM were significantly lower than those in controls among participants without allergic diseases. Among the participants with allergic diseases, serum IgE levels were not significantly different among the EM, CM, and control groups. However, they negatively correlated with monthly headache frequency. Serum tryptase levels did not significantly differ among the EM, CM, and control groups, regardless of allergic diseases.
Article highlights
Among participants without allergic diseases, serum histamine levels in participants with EM and CM were significantly lower than those in controls. Serum IgE levels in participants with EM and CM negatively correlated with headache frequency in participants with allergic diseases. Our findings suggest the involvement of different allergic mechanisms in migraine pathogenesis, according to the presence of allergic diseases.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024231178218 - Supplemental material for Altered interictal serum histamine and immunoglobulin E but unchanged tryptase levels in individuals with episodic and chronic migraine
Supplemental material, sj-pdf-1-cep-10.1177_03331024231178218 for Altered interictal serum histamine and immunoglobulin E but unchanged tryptase levels in individuals with episodic and chronic migraine by Chae Gyu Park, Hye Young Na, Da Eun Park, Ha Yan Kim and Min Kyung Chu in Cephalalgia
Footnotes
Author contributions
CGP conceptualized and designed the study, analyzed the data, and reviewed the manuscript. HYK, DEP and HYN analyzed the data and reviewed the manuscript. MKC conceptualized and designed the study, collected and analyzed the data, interpreted the data, and drafted the manuscript. All authors have read and approved the final manuscript.
Data availability statement
Anonymized data relevant to this study will be shared upon request by a qualified investigator after obtaining appropriate institutional review board approval.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MKC was a site investigator for a multicenter trial sponsored by Biohaven Pharmaceuticals, Allergan Korea, and Ildong Pharmaceutical Company. He received lecture honoraria from Eli Lilly and Company, Handok-Teva, and Ildong Pharmaceutical Company over the past 24 months. He received grants from Yonsei University College of Medicine (6-2021-0229), the Korea Health Industry Development Institute (KHIDI) (HV22C0106), and a National Research Foundation of Korea (NRF) grant from the Korean government (MSIT) (2022R1A2C1091767). Author HYN received a grant from the National Research Foundation of Korea (NRF) (2021R1I1A1A01043872). The other authors declare no conflicts of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by a grant from the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (Grant No.: HV22C0106), and a National Research Foundation of Korea (NRF) grant from the Korean government (MSIT) (2022R1A2C1091767 and 2021R1I1A1A01043872).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
