Abstract
Background
In trials of monoclonal antibodies against calcitonin gene-related peptide or its receptor for prevention of episodic migraine, we observed two problematic aspects: a) The graphic presentations; b) the methods of calculating “response rates” (≥50% decrease of monthly migraine days from baseline).
Observations
Decrease in monthly migraine days is presented, over time, in figures on a downward (negative) scale from zero at baseline, with the ordinate stopped just beyond the maximum effect of the active drugs. In one trial, decreases in monthly migraine days were −1.8 after placebo, −3.2 after erenumab 70 mg and −3.7 after erenumab 140 mg, with the ordinate stopped at −4.5. The reader can perceive only a relative 2-fold benefit of erenumab versus placebo. If, however, treatment periods are compared with baseline in bar charts, MMDs persisting after treatment in the same trial can be illustrated as follows, creating a different perception: 78% for placebo, 61% for erenumab 70 mg, and 55% for erenumab 140 mg.
In the nine trials, “response rates” defined as above were calculated in five different ways, taking different numbers of treatment months into account in comparisons with the one-month baseline. This makes comparisons impossible.
Suggestions for improvements
Mean monthly migraine days before and after treatment should be presented in a bar chart. Such figures, presenting persisting MMDs, are more clinically relevant and less misleading than decreases from baseline.
The definition and methods of calculating and presenting “50% response rates” should be standardized by the Drug Trial Committee of the International Headache Society.
Introduction
After a long period with little progress in the drug-prevention of migraine, the recent development of monoclonal antibodies against CGRP or its receptor, based on improved understanding of the pathophysiology of migraine, offers a new treatment option (1,2). Assuming at least equal efficacy, a likely advantage of these specific treatments is better tolerability than is experienced with the range of drugs currently used for migraine prevention. For example, in randomized, controlled trials (RCTs) of topiramate 100 mg, 24% (90/380) of patients discontinued active treatment because of adverse events (AEs), while 10% (40/381) discontinued placebo (absolute difference 13% [95% CI: 8–18%) (3–5). In contrast, in five large RCTs (6–10) of antibodies against CGRP or its receptor, discontinuations due to AEs were 2% both for active treatments (58/2376) and for placebo (34/1764).
However, in the various presentations and evaluations of efficacy data in the recent antibody RCTs, two problems emerge. These are reviewed in the next paragraphs.
Review and comment
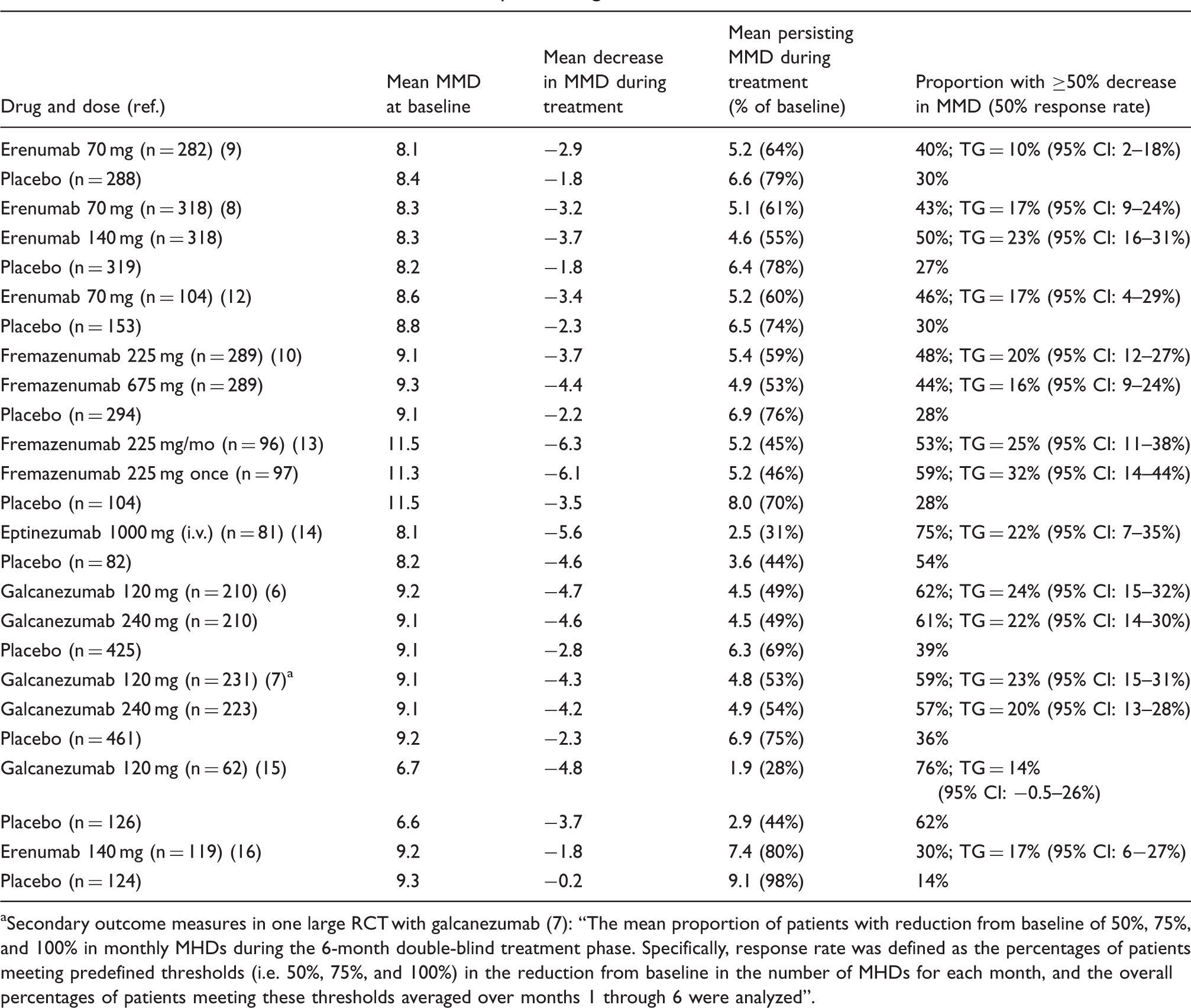
The first problem, well displayed in an RCT of erenumab 70 (10), is in the presentation of decrease in monthly migraine days (MMDs) over time on a downward (negative) scale from zero at baseline. In Figure 1 from the publication in Cephalalgia 2018 (6)), the visual impression is of migraine largely disappearing after 3 months of active treatment, as the curves flatten at MMD −2.7 to −2.9 while the ordinate is stopped at −3.5 (Figure 1). With the placebo curve levelling at MMD −1.8, and no anchors at either end of the scale, the reader can only perceive a considerable relative benefit. The reality is as shown in Table 1: Decreases in MMDs are from 8.1 (mean baseline) to 5.2 with erenumab 70 mg (i.e. with 64% of MMDs still persisting), and from 8.4 to 6.6 (79% persisting) with placebo.
Changes in mean monthly migraine days after treatment with erenumab 70 mg and placebo (monthly subcutaneous injection) (9). Reproduced from Dodick et al., Cephalalgia 2018; 38: 1026–1037, with permission from the publisher. Preventative effects of CGRP antibodies in episodic migraine RCTs. Secondary outcome measures in one large RCT with galcanezumab (7): “The mean proportion of patients with reduction from baseline of 50%, 75%, and 100% in monthly MHDs during the 6-month double-blind treatment phase. Specifically, response rate was defined as the percentages of patients meeting predefined thresholds (i.e. 50%, 75%, and 100%) in the reduction from baseline in the number of MHDs for each month, and the overall percentages of patients meeting these thresholds averaged over months 1 through 6 were analyzed”.
Patients contemplating drugs for migraine prevention need information that is both reliable and realistic. The visual presentation may appear convincing (and, presumably, is intended to be), but it is a manipulation brought about by ordinate truncation. Numerical presentation of MMDs persisting after treatment better informs both clinicians and patients. In 12 treatments with CGRP antibodies in seven phase-3 RCTs, the median persisting MMDs were 55% (range: 45–64%), whereas on placebo (n = 7) the median persisting MMDs were 75% (69–78%) (Table 1). In summary, CGRP antibodies reduce MMDs by about a half, and placebo by about one quarter, which is a relative twofold benefit, as suggested by the visual presentation, but, importantly, a much smaller absolute benefit. An alternative visual presentation of the data in Figure 1 (10), simpler, realistic and therefore more informative, is presented in Figure 2.
Mean migraine days per month before and after treatment with placebo or erenumab 70 mg (monthly subcutaneous injection) for 3 months (9).
The second problem lies in the different ways of calculating “response rates” in MMDs: ≥50% and ≥75%; and, in some cases, in the directly erroneous way of calculating (or reporting) 100% response rate (11). The proportion of patients with MMD reductions of ≥50% (the response rate) has been variously estimated/calculated:
as ≥50% mean decrease in MMDs in month 3 of 3 months' treatment after baseline (in five RCTs (9,12,13,15,16)) as ≥50% decrease in MMDs calculated as the mean of monthly estimates during months 4–6 of 6 months' treatment after baseline (one RCT (8)) as ≥50% decrease in MMDs calculated as the mean of monthly estimates during all 3 of 3 months' treatment after baseline (one RCT (10)) as ≥50% mean decrease in MMDs during weeks 5–8 of 3 months' follow-up after a single intravenous injection (one RCT (14)) as ≥50% decrease in MMDs calculated as the mean of monthly estimates during all 6 of 6 months' treatment after baseline (two RCTs (6,7)).
Despite these different methodologies, the ≥50% response rate for placebo ranged relatively narrowly (27–39%) in the seven phase-3 RCTs, with a median of 30% (6–10,15,16). It is perhaps noteworthy that the two greatest placebo MMD reductions, 36% and 39%, were observed in the two galcanezumab RCTs (6,7), with the very special methodology of calculating response in each month over all 6 months of treatment (see notes to Table 1). In the two smaller phase-2 RCTs, the ≥50% placebo response rates were, for unknown reasons, 54% (14) and 62% (15).
The only recent, previous trial programme of a preventative drug for episodic migraine was conducted with topiramate (n = 1367), with similar methodology in three RCTs (3–5). The optimum dose of topiramate was 100 mg, which was tested in 378 patients who were compared with 378 treated with placebo. A 4-week baseline period was followed by an 8-week titration period and an 18-week maintenance period, with ≥50% decrease in MMDs from baseline evaluated over the whole treatment period. Response rates were 47.5% on topiramate and 22.5% on placebo; the therapeutic gain (TG) (active drug minus placebo) was 25% (95% CI: 18–32%). Thus, the best results from treatment with CGRP antibodies match the TG for topiramate, an established drug for migraine prevention, but the diverse calculations of ≥50% response rates in the CGRP RCTs exclude fair comparison. There is therefore a need for comparative RCTs between the CGRP-related antibodies and standard drug treatments.
Estimates and reporting of 75% response rate, defined as the proportion of treated patients with ≥75% decrease in MMDs (6,7,13,16), probably suffer from the same problems, while 100% response rate, defined as the proportion of treated patients with a 100% decrease of MMD (i.e. migraine-free), has been reported in a peculiar way in two studies (6,7) (see below).
In one large phase-3 galcanezumab RCT (6), 100% response rates were 6.2% (26/425) for placebo, 15.6% (33/210) for galcanezumab 120 mg (odds ratio [OR]: 2.8 [95% CI: 2.0–4.0]) and 14.6% (30/210) for galcanezumab 240 mg (OR: 2.6 [95% CI: 1.8–3.7]). In the other phase-3 galcanezumab RCT (7), 100% response rates were similar: 26/461 (5.7%) for placebo, 27/231 (11.5%) for galcanezumab 120 mg, and 31/223 (13.8%) for galcanezumab 240 mg (for how these secondary outcome parameters were evaluated, see note in Table 1). Apparently, there were consistent findings of a small but possibly relevant effect of the two galcanezumab doses. In a post hoc analysis of 100% response rates on galcanezumab 120 mg and 240 mg and placebo in these two phase-3 RCTS (17), the results were presented in an alternative way: As the number of months with 100% response (17). At least 1 month migraine-free during 6 months' treatment was observed in 171/875 (19.5%) of patients on placebo, 169/436 (38.8%) on galcanezumab 120 mg and 178/428 (41.6%) on galcanezumab 240 mg. Three months migraine-free were observed in 48/875 (5.5%) on placebo, 46/436 (10.6%) on galcanezumab 120 mg and 59/428 (13.8%) on galcanezumab 240 mg. Finally, 100% response during all 6 months was observed in 2/827 (0.2%) on placebo, 3/435 (0.7%) on galcanezumab 120 mg and 6/427 (1.4%) on galcanezumab 240 mg (17). In our view, “100% response” describes freedom from migraine attacks for a predefined period within or after the treatment period, not random months of migraine freedom dispersed among six months (11). The galcanezumab RCTs demonstrate that 100% response over all 6 months occurs very seldom (in approximately 1% of patients).
Conclusions
Changes in MMDs in RCTs of migraine preventative drugs should be presented graphically, in an appropriate way that provides realistic information to patients and their doctors, not manipulated to emphasize differences from placebo. This is best done by presenting baseline MMDs and MMDs persisting after a period of treatment.
The 50% response rate (proportion with ≥50% decrease in MMDs) is an important secondary outcome measure, recommended by the International Headache Society (IHS) since 2012 (18). When no RCTs directly comparing two drugs for migraine prevention are available, their relative effects can, with some caution, be estimated by comparing their TGs on this measure. This outcome has also been used in meta-analyses of propranolol (19) and topiramate (20). Generally, a prerequisite for using an outcome measure for comparisons is, of course, that it is similarly calculated in all RCTs, and this is not the case for CGRP antibody RCTs: Investigators and manufacturers of these drugs have used five different ways to calculate the 50% response rate. The reason for this is obscure, but the consequence is that it is very doubtful whether this measure can be used to compare the CGRP antibodies with each other or with other preventative drugs. Given the enormous investment in these RCTs, this is regrettable (and, arguably, unforgivable). An additional caveat is that in most earlier RCTs, with other agents, 50% response rates were estimated as ≥50% decrease in migraine attacks per month (regardless of attack duration), not migraine days (MMDs: All days affected by or during the course of attacks) as used in the CGRP antibody RCTs.
We hope that the IHS Drug Trial Committee will formulate new and more precise recommendations concerning the definition and reporting of response rates in RCTs investigating the prevention of (episodic) migraine.
Clinical implications
Current illustrations of effect of CGRP antibodies in migraine can give an exaggerated impression of the effect. This is due to the presentation only of changes in monthly migraine days (MMDs) for placebo and active drugs. Presentation of mean MMDs during baseline and persisting MMDs after treatment will provide more relevant information. In the trials with CGRP antibodies, ≥50% decrease in MMDs is calculated in five different ways, and a consensus on how this should be done is needed.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: H-CD received honoraria for participation in clinical trials, contribution to advisory boards or oral presentations from: Alder, Allergan, Amgen, Autonomic Technology, Bristol-Myers Squibb, CoLucid, Electrocore, Ipsen, Lilly, Medtronic, MSD, Novartis, Pfizer, Schaper and Brümmer, Teva and Weber & Weber. Financial support for research projects was provided by Allergan, Electrocore, MSD and Pfizer. Headache research at the Department of Neurology in Essen is supported by the German Research Council (DFG), the German Ministry of Education and Research (BMBF) and the European Union. H-CD has no ownership interest and does not own stocks of any pharmaceutical company. H-CD serves on the editorial boards of Cephalalgia and Lancet Neurology. H-C Diener chairs the Clinical Guidelines Committee of the German Society of Neurology and is member of the Clinical Trials Committee of the IHS. PT-H and TS have no conflicts of interests.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
