Abstract
Background
The role of the trigeminal autonomic reflex in headache syndromes, such as cluster headache, is undisputed but sparsely investigated. The aim of the present study was therefore, to identify neural correlates that play a role in the initiation of the trigeminal autonomic reflex. We further aimed to discriminate between components of the reflex that are involved in nociceptive compared to non-nociceptive processing.
Methods
Kinetic Oscillation Stimulation (KOS) in the left nostril was applied in order to provoke autonomic symptoms (e.g. lacrimation) via the trigeminal autonomic reflex in 26 healthy participants using functional magnetic resonance imaging. Unpleasantness and painfulness were assessed on a visual analog scale (VAS), in order to assess the quality of the stimulus (e.g. pain or no pain).
Results
During non-painful activation, specific regions involved in the trigeminal autonomic reflex became activated, including several brainstem nuclei but also cerebellar and bilateral insular regions. However, when the input leading to activation of the trigeminal autonomic reflex was perceived as painful, activation of the anterior hypothalamus, the locus coeruleus (LC), the ventral posteriomedial nucleus of the thalamus (VPM), as well as an activation of ipsilateral insular regions, was observed.
Conclusion
Our results suggest the anterior hypothalamus, besides the thalamus and specific brain stem regions, play a significant role in networks that mediate autonomic output (e.g. lacrimation) following trigeminal input, but only if the trigeminal system is activated by a stimulus comprising a painful component.
Keywords
Introduction
The trigeminal autonomic reflex plays an important role in the pathophysiology of trigeminal autonomic cephalalgias (TACs) (1) and it becomes activated by painful stimulation of the trigeminal nerve (2). Both painful and non-painful mechanical stimulation of the trigeminal nerve result in a robust activation of the parasympathetic reflex arc in cluster headache patients (3) and healthy subjects (4), which can be quantified measuring lacrimation with a standardized Schirmer II test. However, the differences between central processing of a nociceptive and non-nociceptive mechanical trigeminal input, both activating the parasympathetic arc of the reflex, have not been investigated. This is of special interest and may shed light on the question of whether the parasympathetic symptoms in TACs are secondary to the excruciating trigeminal pain, or whether both components, the pain and the autonomic dysfunction, are in fact driven by central structures such as the hypothalamus.
In a recent study, using intranasal Kinetic Oscillation Stimulation (KOS), we reliably provoked lacrimation in cluster headache patients (3) and healthy subjects (4) by a mechanical trigeminal input and quantitatively assessed this in a standardized Schirmer II test. If this experimental design is used for more than 20 minutes, the mechanical input for some of the participants becomes more painful. Our experimental set-up therefore allows discriminating between processing of a non-painful and a painful mechanical trigeminal input, without changing the stimulating parameters. In the present study, we used KOS in an fMRI experiment to trigger the trigeminal autonomic reflex in healthy volunteers. We aimed to specifically disentangle the role of brain regions known to be involved in autonomic and trigeminal pain processing, especially the hypothalamus, the nucleus of the solitary tract and the spinal trigeminal nucleus with regard to nociceptive versus a non-nociceptive trigeminal input leading to parasympathetic output. We expected some trials to be perceived as slightly painful in some participants.
Methods
Participants
The study was approved by the local ethics committee (Ärztekammer Hamburg; PV5004) and participants gave written informed consent. Volunteers were remunerated for participation. In total, 31 healthy participants without any headache or other pain conditions were recruited via announcements on a local internet platform in telephone interviews. Exclusion criteria included suffering from any primary headache disease as well as suffering from psychiatric or neurologic disorders. Only participants who regarded themselves as free from any headache were included and then particularly asked for mild or short-lasting headaches. The cutoff criterion for participation was a maximum of five non-specific headache days within three months prior to the start of the experiment. Detailed characteristics of the participants are presented in the Supplemental material. Furthermore, it was ensured that participants did not suffer from nasal polyps, a deviated nasal septum or other anatomical restrictions or infections of the upper airways. Prior to the experimental sessions, all participants were asked to fill in questionnaires including a general headache questionnaire, and the Patient Health Questionnaire (PHQ, German version) (5).
Kinetic Oscillation Stimulation
For the KOS procedure, a balloon catheter with an inflatable tip was coated with paraffin and placed into the participant’s left nostril, then securely fixed to the nose to prevent the catheter from moving during the scanning procedure. Prior to the experimental trials, the balloon catheter was inflated to 80 mbar for one minute without oscillation to allow for habituation to the mechanical perception of a “stuffed nose”. The balloon was kept inflated during the whole experiment. During stimulation, an oscillation at a frequency of 85 Hz was switched on, which stimulates trigeminal nerve terminals and leads to the activation of the trigeminal autonomic reflex. This in turn leads to a dose-dependent lacrimation (unpublished data). The lacrimation increases with increasing pressure and oscillation frequency, which can be quantified in a Schirmer II test (4).
Pre-testing measurements
As a proof of concept, for pre-testing measurements outside the MRI scanner, KOS (85 Hz/80 mbar) was applied in 12 additional healthy participants (mean age ± SEM (standard error of the mean) = 25.45 ± 0.99, four female) prior to the fMRI study. For anesthesia during the Schirmer II test, one drop (1 ml) Oxybuprocain (Conjuncain, EDO, Frankfurt, Germany) was applied to both eyes and incubated for 5 minutes. Subsequently, lacrimation was measured and documented during rest for a duration of 5 minutes. For the experimental condition, the participants lay down in supine position and underwent an almost identical experimental procedure to that applied for the fMRI experiment, including 15 trials (40 minutes) with alternating stimulation and rest in a behavioral lab. Three Schirmer II tests were performed 1 minute, 15 minutes and 35 minutes after the start of the experiment.
The experiment was presented on a laptop screen placed 20 cm above the chest on an oblique table and ratings of the intranasal perception on a visual analogue scale (VAS) were performed via button presses. The same KOS stimulation devices were used for the fMRI experiment, which was conducted a few weeks later.
Besides lacrimation, other autonomic symptoms including redness of the left eye and rhinorrhea were observed in a previous study in both cluster headache patients (3) and in the sample of healthy subjects; however, these were not quantified due to missing standardized methods.
Experimental protocol inside the scanner
The experimental procedure inside the MRI scanner was performed over a period of approximately 48 minutes, including 21 trials of KOS. The stimulation (oscillation at 85 Hz) was switched on for 32 seconds. The rather long stimulation period of 32 seconds was chosen in order to trigger the trigeminal autonomic reflex and to allow the system to reliably initiate parasympathetic output/lacrimation. During the resting periods, the oscillation was switched off for 90 seconds. We considered a long resting period of 90 seconds necessary for the trigeminal system to attenuate activation and to stop lacrimation. These resting periods were used to evaluate the individual perception of the previous stimulation for every trial. Participants were asked if they perceived the previous stimulation as painful and to choose “yes” or “no” via button press. If the stimulation was perceived as painful, the participants were instructed to rate the perceived pain intensity of the vibration via button presses on a visual analog scale (VAS) (6) from 0–100. If the trial was rated “yes” for pain, the trial was categorized as a pain trial; if rated “no”, it was categorized as a non-pain trial. Unpleasantness was assessed after every stimulation, independent from perceived pain. On the VAS, the label 0 referred to no pain at all or not unpleasant at all, and 100 referred to maximal possible pain or unpleasantness.
Image acquisition
Images were acquired in a 3 Tesla MRI scanner (TrioTim syngo, Siemens) with a 32-channel head coil. For anatomical images, we used an MPRAGE sequence (voxel size 1 × 1 × 1 mm3, TR 2.3 s, TE 2.98 ms, field of view 192 × 256 × 240 mm3 and a flip angle of 9°). Since we focused on the thalamus-hypothalamus-brainstem network, we used our brainstem-specific imaging protocol, which includes a volume from C1 up to the thalamus. Consequently, some regions of interest like the insula could not be investigated to their full extension. During the experimental procedure, 385 functional images (EPI, echo planar imaging) were acquired using a GRAPPA sequence (voxel size 1.25 × 1.25 × 2.5 mm3, no gap, TR 2.61 s, TE 27 ms, field of view 216 × 108 mm2 and a flip angle of 80°). Pulse and breathing were simultaneously recorded with an InVivo Expression Monitoring device (InVivo Corp, USA).
Image preprocessing and denoising
Due to technical issues, five participants had to be excluded, since swallowing and movement caused artifacts in brainstem regions that were of pivotal interest in the present study. In total, the analysis of the imaging data included 26 healthy participants (age = 34.95 ± 2.56 (mean ± SEM); 10 females; 16 males). Brain activation was analyzed using MATLAB and SPM12 (version 6685, Wellcome Trust Centre for Neuroimaging, University College, London, UK). Preprocessing steps in SPM included denoising with a spatially adaptive nonlocal means algorithm (7), implemented in the CAT12 toolbox (http://www.neuro.uni-jena.de/cat/), slice time correction, realignment, coregistration with the structural image, normalization into Montreal Neurological Institute (MNI) space using the unified segmentation routine implanted in SPM12 and smoothing with a 4 mm3 isotropic Gaussian kernel (8). Due to a rather long inter-stimulus interval of 122 seconds on average, the high-pass filter was set to 256 s.
General linear model analysis
To assess neuronal activation caused by the intranasal vibration, we used general linear model (GLM) analyses on single subject level. Here we defined two regressors, one for intranasal oscillation and one for rest, by convolving their onsets and durations with a standardized hemodynamic response function (HRF). Besides that, six movement regressors, resulting from the realignment preprocessing step, and up to 20 regressors correcting for physiological noise, such as cardiovascular changes or breathing, or movement of the cerebrospinal fluid (CSF) (9), were included.
In order to discriminate between the vibration and the perceived pain induced by the KOS, we additionally included pain as a binary parametric modulator (10,11), as suggested by Henson and Friston (12), in a second GLM analysis. Consequently, all participants who perceived pain in either none of the trials (n = 4) or in every trial (n = 4) had to be excluded from this analysis. In order to investigate the effect of pain intensity, a third GLM was calculated, using pain intensity as a parametric modulator of the HRF similar to the second analysis.
Statistics
Contrast images from the aforementioned GLM analyses were included in the statistical analysis at group level. For all results, small volume corrections (SVC) at α level of 5% family-wise error (FWE) corrected within regions of interest were applied. We defined 10 regions of interest (ROI) known to be involved in trigeminal pain and autonomic processing: Right nucleus of the solitary tract (NTS) at [2, −46, −58] (mm/xyz/MNI) (13), left spinal trigeminal nucleus (sTN) (sphere with 6 mm radius) at [−4, −45, −53] (mm/xyz/MNI) (14), left locus coeruleus (sphere with 6 mm radius) at [−6, −36, −33] (mm/xyz/MNI) (15), hypothalamus (6 mm radius) at [0, 2, −6] (mm/xyz/MNI) (16), ventral posteriomedial nucleus of the thalamus (VPM) (Wake Forest University PickAtlas, release 3.0.4), lobule VIII of the cerebellum (Wake Forest University PickAtlas, release 3.0.4), bilateral parietal operculum (sphere with 10 mm radius) at [−56, −18, 12] (mm/xyz/MNI) and [54, −2, 8] (mm/xyz/MNI) (17), bilateral insula (10 mm radius) at [36, −4, 6] (mm/xyz/MNI) and [−40, 0, −2] (mm/xyz/MNI) (17). Results were not further corrected for the number of ROIs. As this is the first report on imaging for KOS, we further report results observed at a voxel-wise statistical threshold of p < 0.001 (uncorrected), which should be interpreted with caution.
Main effect of intranasal activation of the trigeminal autonomic reflex (KOS): To detect brainstem activation in response to KOS stimulation, we performed a paired t-test comparing the stimulation and rest condition (KOS > rest), with betas from our first GLM analysis. Main effect of KOS corrected for the influence of pain: Based on the GLM including pain as a binary parametric modulator (second GLM), we first included contrast images for the vibration (not the parametric regressor) into a one-sample t-test to illustrate the effect of KOS in the absence of painful input. Additional effect of pain on activation of the trigeminal autonomic reflex: Based on the above-mentioned second GLM we further calculated a t-test for the parametric modulator, explaining variance caused by pain (in addition to KOS). Effect of pain intensity on activation of the trigeminal autonomic reflex: As mentioned above, for the analysis of brain regions displaying increased activation with increased perceived pain intensity, we performed a third GLM analysis, including a continuous parametric modulator of pain (similar to the binary modulator of the second GLM) and calculated a t-test for the parametric oscillator. Analysis of pain trials vs. no-pain trials: In order to support our hypothesis that the hypothalamus in particular is crucial for processing of a stimulus including a nociceptive component, we performed an additional analysis, including trials from four participants who perceived all trials as painful and four participants who perceived none of the trials as painful. We used the first GLM analysis at single subject level (first level), without any parametric modulator. For the second level analysis, we performed a two sample t-test (n = 4 vs. n = 4) and present results for the contrast pain > non-pain at a voxel-wise statistical threshold of p < 0.005, uncorrected, and a minimal extent of 20 voxels per cluster for visual inspection.
Analyses of the behavioral data
Thirty-one healthy participants (age = 35.48 ± 2.23 (mean ± SEM); 12 females; 19 males) were included in the analysis of unpleasantness and pain ratings using SPSS (IBM, version 21).
Results
Pre-testings as proof of concept
KOS reliably provoked lacrimation on the stimulated side (mean ± SEM = 15.6 ± 2.52 mm) that was significantly higher (p < 0.001) compared to the non-stimulated side (mean ± SEM = 4.3 ± 2.73 mm) in an additional cohort of 12 healthy participants in a behavioral study that was performed prior to the fMRI study (Figure 1). The results remained stable over time, which suggests that no habituation or sensitization takes place if KOS is applied in an alternating manner over 40 minutes.
Behavioral study: In a pre-testing session prior to the fMRI study, we observed a significant difference in lacrimation increase during intranasal oscillation (KOS) on the stimulated side compared to the non-stimulated side in 12 healthy participants. Error bars indicate the SEM (standard error of the mean), the asterisk indicates p < 0.001.
Behavioral results of the imaging sample
The assessment of unpleasantness on a visual analog scale (VAS) from 0–100 revealed stable results between 40.07 and 49.86 (mean: 45.56 ± 0.59). Assessment of painfulness revealed perceived pain in 48% percent of the trials with a mean value of pain intensity of 21.0 (±1.4 SEM) on a scale from 0–100. The lacrimation was accompanied by rhinorrhea and conjunctival injection; however, these results were not quantified. Detailed results for the participants are presented in the Supplemental material.
Imaging results
For the main effect (KOS > rest, first GLM) we observed increased activation in three different clusters of the operculum (see Figure 2, Table 1).
Main effect KOS. For the main effect of KOS, we observed activation of the ipsilateral operculum (OP) (a) and bilateral symmetrical opercula (b). For visualization purposes, the threshold was set to p < 0.001 uncorrected and a minimal cluster extent of 10 voxels. L and R indicate the side (neurological convention) and the color bar displays the t-values. Results of the imaging analyses: Activated regions with peak coordinates for the main effect (vibration > rest) including pain and non-pain trials as well as for the parametric modulation of pain, including or excluding the painful component of the mechanical trigeminal stimulus. Coordinates are with reference to MNI space. L: left; R: right. p < 0.05 voxel-wise FWE corrected (small volume); *p < 0.001 uncorrected.

The effect of KOS (second GLM) revealed, contralateral to the stimulated side, increased activation in two different pontine nuclei, activation of cerebellar lobule VIII, as well as bilateral activations of the anterior insulae (see Figure 3, Table 1) when corrected for nociceptive input (which was modelled as an additional regressor).
Non-painful activation of the trigeminal autonomic reflex. For the effect of vibration (variance explained by pain), we observed (a) activation located in the ipsilateral cerebellar lobule (lobule Vlll), (b) bilateral symmetrical insula (INS), and (c) two distinct pontine nuclei (PN), located on the right. For visualization purposes, the threshold was set to p < 0.005 uncorrected (a) and p < 0.001 uncorrected (b, c) and a minimal cluster extent of 10 voxels. L and R indicate the side (neurological convention) and the color bar displays the t-values.
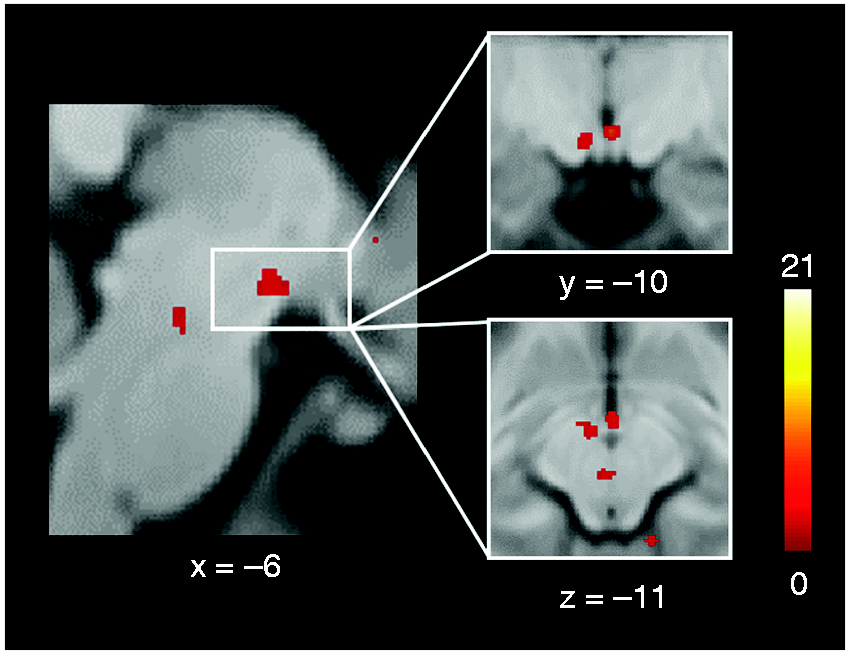
Painful KOS (second GLM) additionally activated the ipsilateral hypothalamus, the ipsilateral insula, the ipsilateral ventral posteromedial nucleus (VPM) of the thalamus, the ipsilateral locus coeruleus, the ipsilateral pontine nucleus as well as the contralateral nucleus of the solitary tract (see Figure 4, Table 1). Since n = 4 participants perceived all stimulations as painful, and n = 4 perceived none of the stimulations as painful, we additionally compared both groups as a proof of concept. Although this meant comparing low numbers of participants, we found again an increased activation of the hypothalamus in those participants who perceived all stimuli as painful compared to those who did not perceive pain at all (see Figure 5).
Painful activation of the trigeminal autonomic reflex. In trials perceived as painful, we observed significantly increased activation (as compared to vibration only) in the (a) ipsilateral hypothalamus (HT), (b) ipsilateral insula (INS), (c) ipsilateral ventral posteriomedial nucleus of the thalamus (VPM), and (d) the contralateral nucleus of the solitary tract (NTS). Moreover, an increased activation was observed for the (e) ipsilateral locus coeruleus (LC) as well as a pontine nucleus (PN). For visualization purposes, the threshold was set to p < 0.005 uncorrected (a–c, e) and p < 0.01 uncorrected (d) and a minimum cluster extent of 10 voxels. L and R indicate the side (neurological convention) and the color bar displays the t-values. Sub-analysis painful > non-painful trials (proof of concept) for the HT. Participants were included who perceived all trials as painful (n = 4), and were compared with those who did not perceive any trial as painful (n = 4). The colored bar shows the T-values.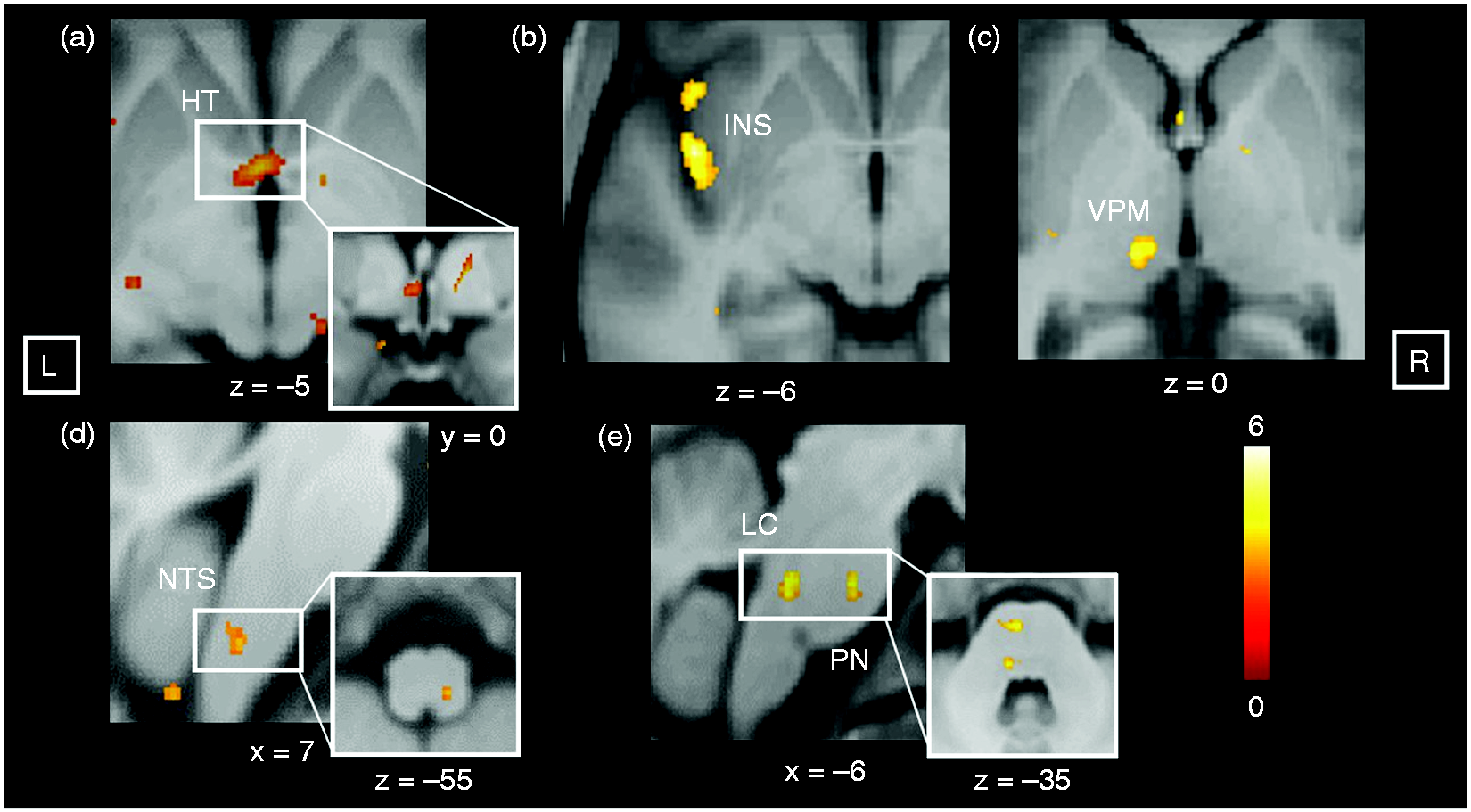
For the inverse contrasts of all above-mentioned imaging analyses, no significant results were observed in previously defined regions of interest in the brain stem (NTS, sTN, cuneiform nucleus), hypothalamus, thalamus, insula, and opercular regions.
We also performed a correlation analysis in order to investigate in which brain region we might observe a correlation with pain; however, we did not find any.
Discussion
The findings of this study investigating central processing of the trigeminal autonomic reflex fall into three broad groups: Areas activated as part of the trigeminal autonomic reflex independent from nociception, areas specifically activated during non-painful stimulation, and areas activated uniquely during painful mechanical trigeminal stimulation.
Non-painful activation of the trigeminal autonomic reflex
Structures activated during mechanical activation of the trigeminal autonomic reflex perceived as non-painful included the contralateral pontine nuclei (PN) as well as bilateral regions of the insula and lobule VIII of the cerebellum. We note that these regions were not significantly activated when the stimulus was perceived as painful. It needs to be pointed out that since we used a brainstem imaging protocol, the insula could not be investigated to its full extent. This is important, as it indicates different central pathways transmitting the non-painful versus painful mechanical information. Both inputs, however, result in parasympathetic discharge (3,4).
The pontine nuclei (PN) are located within the ventral portion of the pons and forward information mainly via mossy fibers from the cerebral cortex to the cerebellum, establishing a complex network between the cortex, the PN and the cerebellum. The majority of mossy fibers originating from the PN project to the contralateral cerebellum, which has been studied in rats (18) and monkeys (19). It is thought that pontine nuclei serve as an integrator of information that is received from various cortical regions (20). Besides input from cortical regions, input from subcortical regions, including the trigeminal nuclei, has been described in rats (21,22), suggesting, together with our findings, a potential role of the PN and lobule VIII of the cerebellum in the activation of the trigeminal autonomic reflex (23,24).
Painful activation of the trigeminal autonomic reflex
The most interesting finding of our study is an increased activation of the anterior hypothalamus during mechanical trigeminal stimulation perceived as painful, but not if the stimulus is perceived as non-painful. This finding is in line with earlier studies showing increased ipsilateral hypothalamic activation during nociceptive trigeminal stimulation in healthy subjects (25), increased in migraineurs (26,27) and cluster headache patients (28). Of note, previous imaging studies investigating the clinical state of acute attacks attributed to the hypothalamus a prominent role in the pathophysiology of TACs (1,29,30). Animal studies suggest reciprocal connections between nuclei of the hypothalamus and trigeminal nuclei (31,32) and demonstrated that several hypothalamic nuclei become activated during nociceptive dural stimulation (33). These data underline the modulating effects of the hypothalamus on nociceptive processing in the trigeminocervical complex (TCC) and, together with our data, these findings suggest the hypothalamus to be an important hub in mediating the nociceptive pathway of the trigeminal autonomic reflex.
Besides the activation of the hypothalamus, we observed activations of several other structures involved in the nociceptive transmission and autonomic output, namely the locus coeruleus (LC) and the ventral posteromedial nucleus (VPM) of the thalamus (34). These findings are similar to findings from earlier trigeminal nociceptive imaging studies (25,35,36). Given that findings revealed activation of the anterior hypothalamus and distinct brainstem nuclei only when the activation of the trigeminal autonomic reflex was perceived as painful strongly suggests that there are indeed different pathways for processing the mechanical and the nociceptive input, rather than one principal pathway that is expanded when the stimulus becomes painful.
Autonomic processing
It is important to point out that both the painless and painful conditions prompted the same autonomic output and therefore both use peripheral and central structures in processing of this output. Indeed the insula (37), LC (38), hypothalamus and mediodorsal thalamus (37) have been suggested to be involved in autonomic functions. Of note, the LC has been shown to control autonomic function via direct projections to the dorsal motor nucleus of the vagus, the nucleus of the solitary tract (NTS) and the paraventricular nucleus (38), structures that are important in sympathetic and parasympathetic processing. Our data also show that higher brain areas such as the opercular and insular cortices are involved in trigeminal processing, independent from the fact of whether the trigeminal input was perceived to be below or above pain threshold, due to activation of silent nociceptors (39).
We conclude that the hypothalamus is involved in the trigeminal-autonomic reflex, but only if the trigeminal input exhibits a nociceptive component. Furthermore, we were able to discriminate structures involved in non-nociceptive autonomic processing including the contralateral pontine nuclei, lobule VIII, and the bilateral anterior insulae. Activated pain modulating structures included the HT, LC, CN and thalamic regions. These results suggest that different central networks are involved, either processing the non-nociceptive or the nociceptive component of trigeminal input.
Article highlights
The trigeminal autonomic reflex can be activated mechanically and following nociceptive input. Using fMRI, we could show that specific regions involved in the trigeminal autonomic reflex became activated, including several brainstem nuclei but also cerebellar and bilateral insular regions. Our results suggest the anterior hypothalamus to play a significant role in networks that mediate autonomic output following trigeminal input, but only if the stimulus is painful.
Supplemental Material
Supplemental material for Hypothalamic activation discriminates painful and non-painful initiation of the trigeminal autonomic reflex – an fMRI study
Supplemental Material for Hypothalamic activation discriminates painful and non-painful initiation of the trigeminal autonomic reflex – an fMRI study by Maike Möller, Jan Mehnert and Arne May in Cephalalgia
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the German Research Foundation, SFB936/A5 to AM.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
