Abstract
The presence of cranial autonomic symptoms in migraine is well known and thought to represent activation of the trigeminal parasympathetic reflex pathway similar to trigeminal autonomic cephalalgias. However, studies regarding the prevalence of these symptoms are few. The characteristics of migraineurs with cranial autonomic symptoms and the association of cranial autonomic symptoms with laterality of headache have never been studied in a clinic population. Seventy-eight consecutive subjects with migraine were recruited from the Headache Clinic of the Department of Psychiatry after exclusion of subjects with secondary headache. Their demographic data and detailed history of headache were noted and leading questions were asked regarding cranial autonomic symptoms. χ2 test and Fisher's exact test was used for categorical variables, whereas an independent sample t-test was applied on numerical data. Spearman's correlation was used for correlational analysis of categorical variables. Female subjects (78.2%) outnumbered males and the average duration of illness in the whole sample was 3.81 years. Migraine without aura (53.8%) was the commonest diagnosis, followed by migraine with aura (24.4%). Cranial autonomic symptoms were present in 73.1% of subjects and, commonly, they were ipsilateral to headache. Moreover, strictly unilateral cranial autonomic symptoms were reported by only 32% of patients. The anatomical side of headache did not affect the presence of autonomic symptoms. Those with or without autonomic symptoms did not differ with respect to gender, diagnosis, laterality of headache or associated symptoms except phonophobia, which was more common in subjects with autonomic symptoms (P = 0.05). Those with autonomic symptoms had longer duration of illness (P = 0.03) and longer headache episodes (P = 0.04). In addition, sleep was ineffective in relieving their headache (P = 0.02). Cranial autonomic symptoms are frequent in migraineurs and are common in subjects with long duration of illness and longer headache episodes. Clinical evidence in the present study suggests that subjects with cranial autonomic symptoms have a hyperactive efferent arm of trigeminal autonomic reflex. The connections of trigeminal nucleus with the locus coeruleus and dorsal raphe nucleus may account for the observed phenotypic differences between the two groups. Further research, however, is required to elucidate the underlying neural mechanisms of cranial autonomic symptoms in migraine.
Keywords
Introduction
Activation of the trigeminovascular system is the most accepted explanation for migraine generation according to recent developments. Other headaches that develop with the activation of the trigeminovascular system, e.g. short-lasting, unilateral, neuralgiform headache attacks with conjunctival injection and tearing (SUNCT), paroxysmal hemicrania and cluster headache, present with cranial autonomic symptoms (CAS), e.g. lacrimation, conjunctival injection, nasal congestion and periorbital swelling (1) and differ from each other with regard to duration mainly. Since migraineurs suffer trigeminovascular system abnormality and autonomic disturbances have also been described in these patients (2–4), it is not illogical to expect CAS in migraineurs.
Few reports have been published describing the presence of CAS in migraine patients. Kaup et al. (5) and Al-Din et al. (6) described four and three cases of migraine, respectively, that showed conjunctival injection, tearing, nasal symptoms and ptosis. Furthermore, laterality of the headache may influence the occurrence of CAS and they may be conspicuous during unilateral headache (7) or during strictly unilateral pain with right side predominance (5) or in left-sided migraineurs (8). Laterality of CAS may also be correlated to the laterality of headache and they may be ipsilateral, contralateral or bilateral (7). Kaup et al. (5) have suggested that side-locked migraineurs with CAS may comprise a different group and, pathophysiologically, may lie between migraine and trigeminal autonomic cephalalgias (TAC). CAS have been reported only at the height of the pain (6, 9), and are usually short lasting (6).
To best of our knowledge there is only one systematic report that has examined the differences in clinical features of migraine subjects with or without CAS (7). Since this issue contributes to the understanding of the pathophysiology of migraine, the present study was designed to determine (i) the prevalence CAS in migraineurs, (ii) their laterality in relation to headache, and (iii) the clinical characteristics of patients with CAS.
Methods
Subjects presenting with the complaint of headache in the psychiatry outpatient department were screened for the presence of migraine headache according to International Headache Society 2004 criteria (10) during a predefined study period. The diagnosis of headache was based upon history, clinical examination and, wherever required, laboratory investigations. Subjects with non-migraine primary headache or secondary headache, or who could not provide a reliable history, were excluded from the study, as were patients with other comorbid primary headaches.
Informed consent was obtained from the subjects and details of demographic data were gathered. The following data were recorded: total duration of illness, average number of attacks per month, time to attain maximum severity after onset, laterality, side of pain, location (temporal, frontal, occipital, parietal, orbital, generalized, neck), radiation of pain, quality (aching, pulsating/throbbing, pressing, tightening, band like, etc.), average duration of episode, usual time of onset, precipitating and relieving factors, premonitory symptoms (mainly yawning) and lastly, associated symptoms, e.g. photophobia, nausea, vomiting, photophobia, worsening with exertion, dizziness, ptosis, etc.
Depending upon the laterality of headache, patients were divided into the following categories: (a) bilateral, (b) bilateral but right-sided more severe than left-sided pain, (c) bilateral but left-sided more severe than right-sided pain, (d) unilateral right, (e) unilateral left, (f) unilateral, right side more frequent than left, (g) unilateral, left side more frequent than right, (h) unilateral left more frequent than bilateral, (i) unilateral right more frequent than bilateral, (j) bilateral more frequent than unilateral right, and (k) bilateral more frequent than unilateral left. For the sake of analysis, categories ‘a’ to ‘c’ were combined under bilateral headaches; categories ‘d’ to ‘i’ were combined under unilateral; and ‘j’ and ‘k’ were combined under bilateral more than unilateral.
Based on the side of headache, two groups were made: predominantly right-sided pain (b + d + f + i) and predominantly left-sided pain (c + e + g + h).
Cranial autonomic symptoms
Leading questions were asked regarding reddening of the eye during migraine, lacrimation, nasal blockade/running and periorbital swelling. In those patients with these symptoms, laterality of individual CAS was also ascertained in relation with the laterality of headache (ipsilateral or bilateral or bilateral with more intense towards severe pain side). We also asked patients about the frequency of these symptoms (whether they occurred during every attack or only sometimes) and their relation to intensity of headache. The duration of these symptoms was also ascertained. However, most patients could not respond satisfactorily regarding frequency and duration, therefore these factors were excluded from analysis.
Statistical analysis was performed using SPSS version 11.0 for Windows (SPSS Inc., Chicago, IL, USA). Multiple response catagories for two variables – ‘laterality of headache’ and ‘laterality of autonomic symptoms’ – interfered with analysis due to the small number of subjects in each category; therefore, few categories have been clubbed together (as already described).
Similarly, for the sake of simplicity of analysis, patients with bilateral headache and bilateral CAS were kept in the ‘CAS ipsilateral to headache’ category (Table 1).
Prevalence and pattern of cranial autonomic symptoms (CAS) in migraine patients (N = 78; F = 61, M = 17)

Cases without CAS were excluded.
IL, Ipsilateral to headache side – involves bilateral CAS in bilateral migraineurs and ipsilateral to headache in unilateral migraineurs; BL, bilateral CAS in unilateral migraineurs; UL, unilateral.
For the analysis, χ2 test and Fisher's exact test were used for categorical variables, while the independent sample t-test was applied for numerical variables. Spearman's correlation was used to determine the correlation between the laterality of headache and CAS and among different autonomic symptoms.
Results
Demographic characteristics of the sample
Of this sample 78.2% were females, the mean age was 32 years and the average duration of illness was 3.81 years. According to ICHD-2004 (10), 53.8% suffered from migraine without aura (MoA), 7.7% probable MoA, 6.4% chronic MoA, 24.4% migraine with aura (MA) and 7.7% chronic MA. The average duration of each attack was 25.95 h and subjects suffered an average of 7.88 attacks per month. Laterality of headache as reported by subjects was as follows: (a) bilateral 39.7%, (b) bilateral but right-sided more severe than left-sided pain 3.8%, (c) bilateral, but left-sided more severe than right-side pain 9%, (d) unilateral right 17.9%, (e) unilateral left 12.8%, (f) unilateral, right-sided more frequent than left-sided pain 3.8%, (g) unilateral, left-sided more frequent than right-sided pain 6.4%, (h) unilateral left more frequent than bilateral 2.6%, (i) unilateral right more frequent than bilateral 1.3%, (j) bilateral more frequent than unilateral right 1.3%, and (k) bilateral more frequent than unilateral left 1.3%.
Differences between those with autonomic symptoms and those without it
The relationship between laterality of CAS and headache is depicted in Table 1. Anatomical side (right or left) of the headache did not correlate with any of the CAS. However, laterality of different CAS was significantly correlated with each other (red eye with lacrimation, P = 0.001; red eye with nasal symptoms, P = 0.003; lacrimation with nasal symptoms, P = 0.04), suggesting that a single pathophysiological mechanism was responsible for all symptoms.
Tables 2 and 3 depict differences between subjects with and without CAS based on the symptoms. Frequency of precipitating factors, e.g. stress (P = 0.59), hunger (P = 1.0), hot environment (P = 0.7), sleep deprivation (P = 0.26) and exacerbating factors, e.g. exertion and movement (P = 1.0), jerk (0.72), bending (P = 0.43), neck movement (P = 1.0) was not different between the two groups. Diurnal variation in the onset of headache was not observed (P = 0.91) between the two groups. The most common location for the onset of headache was temporal (57.1% in without CAS and 61.4% in with CAS) followed by frontal (14% in without CAS and 14.3% in with CAS), and other locations (P = 0.98). Pain was non-radiating in 28.6% subjects without CAS and in 15.8% with CAS, yet orbital radiation was seen exclusively in subjects with CAS (17%; P = 0.29). Non-steroidal anti-inflammatory drugs relieved pain in 28.6% without CAS and in 42.1% with CAS (P = 0.30); sleep relieved headache in 47.6% without CAS and in 21.1% with CAS (P = 0.02); local pressure was a relieving factor in 14.3% without CAS and in 24.6% with CAS (P = 0.53); and vomiting relieved the headache in 4.8% with CAS and 5.3% without CAS (P = 1.0).
Differences between subjects with and without cranial autonomic symptoms (CAS)

Number of positive cases; figures in parentheses show percentages.
MoA, Migraine without aura; MA, migraine with aura; BL, bilateral CAS in unilateral migraineurs; UL, unilateral.
Characteristics of migraine subjects with and without cranial autonomic symptoms (CAS)
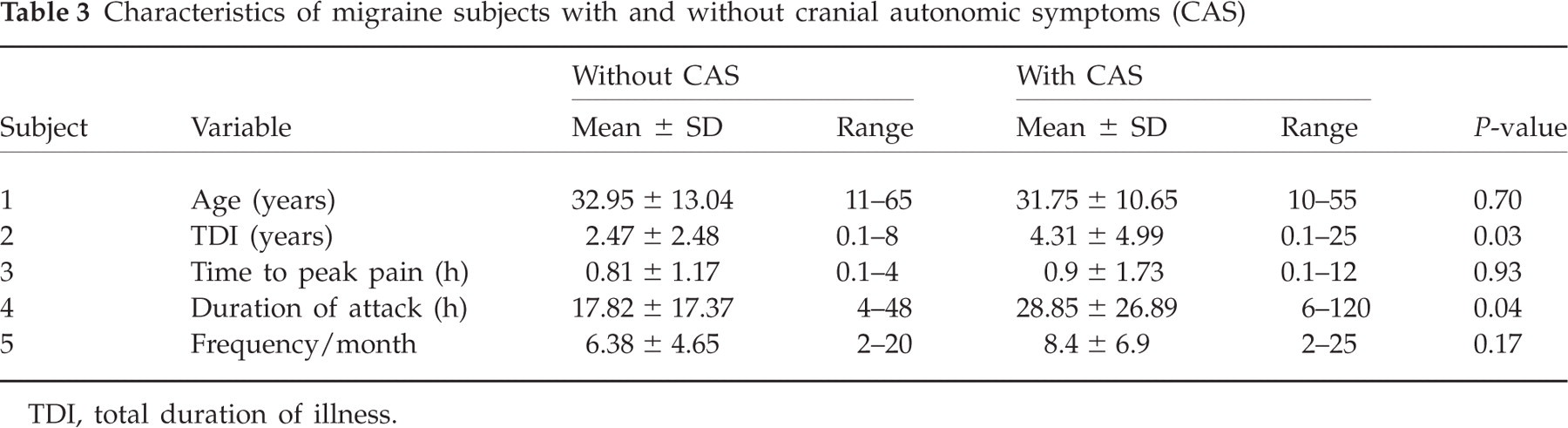
TDI, total duration of illness.
In general, CAS were reported to coincide with the height and severity of attacks. They were shorter lasting than the pain episode except for eye symptoms, which were reported in both the ascending as well as the descending phase of the pain.
These results suggest that CAS are not unusual in migraine, although they are usually bilateral and are associated with a longer duration of illness and longer individual episodes. Patients with CAS have a greater chance of developing phonophobia and sleep is ineffective in terminating their headache.
Discussion
Although subjects in the present study have some phenotypic similarities with the TAC, this is not sufficient evidence to ‘broadly classify’ migraine with the TAC group as previously suggested (1). Despite the fact that CAS have been reported in migraine subjects (9, 11) and migraine symptoms, e.g. photophobia, phonophobia and vomiting, in cluster headache patients (12, 13), recent evidence suggests that these entities are different but with partial pathophysiological overlapping. Although they share the trigeminal vascular system and trigeminal autonomic reflex, as manifested by an increase in calcitonin gene-related peptide and vasoactive intestinal peptide during attacks in migraineurs and cluster-headache patients (1, 14), different neuroanatomical regions are involved in symptom development, as shown in functional imaging studies. While the brainstem is the pain generator region in migraine (15), the hypothalamic grey matter, on the other hand, is key region in the initiation of cluster headache (16). Diagnosis of TAC requires the presence of strictly unilateral autonomic symptoms (10), whereas in this study most subjects suffered bilateral autonomic symptoms and strictly unilateral symptoms were present in only of 32% of subjects. Moreover, patients with and without CAS were not different with regard to precipitating factors, exacerbating factors, location, radiation and quality of pain and most of the relieving factors. These findings confirm that these patients were indeed a subgroup of migraine. Additionally, to the best of our knowledge these factors have never previously been examined in relation to CAS in migraineurs.
The presence of CAS depends upon the activation of the trigeminal autonomic reflex (1) and its efferent arm may be more active in migraineurs with CAS (14). The afferent arm of reflex comprises the afferent fibres of the trigeminal nerve. These fibres stimulate ipsilateral peri-aqueductal grey and dorsal raphe nuclei as a unit (PAG + DRN) and a few fibres directly stimulate the ipsilateral and contralateral superior salivatory nucleus. PAG + DRN then, activate the ipsilateral locus coeruleus, which in turn stimulates the superior salivatory nucleus of the same side. Both the locus coerulei normally inhibit each other (17). Efferent fibres from the superior salivatory nucleus form the efferent arm of this reflex. They are responsible for CAS ipsilateral to pain. On the other hand, bilateral CAS in unilateral migraineurs can be attributed to crossing over of parasympathetic fibres in the brainstem (17, 18). In addition, symptoms that represent either afferent arm sensitization (increase of headache with coughing or bending or with physical activity and throbbing pain) or thalamic nuclei sensitization, e.g. scalp tenderness (19), were not different between the two groups, further substantiating the fact that only the efferent arm of reflex was sensitized in these patients (14).
The CAS were more prevalent in subjects with a long history of illness and long individual attacks and were reported to appear at the height of pain. Perhaps the activity of the trigeminal autonomic reflex is linked to trigeminal nucleus sensitization. Two facts must be considered here: (i) the temporal summation theory states that the frequency of stimulus correlates with the intensity of response; and (ii) frequent subthreshold/threshold stimuli facilitate the neuronal membrane (20). Long-lasting illness with frequent headaches (21) and possibly longer pain episodes induce central sensitization of the trigeminal nucleus and consequent facilitation of the trigeminal autonomic reflex. We assume that when a critical level of stimulation is attained, the autonomic reflex discharges and this may be the reason why CAS were reported only at the height of pain attacks.
Besides the frequency of headache, female gender and Indian ethnicity (22, 23) were also reported to influence central sensitization positively. These factors might have added to the factors described above, resulting in the higher prevalence of CAS in this report compared with previous findings (7).
Associated symptoms of migraine, e.g. phonophobia, photophobia, aura, nausea, etc., depend upon activation of different cortical and subcortical areas and these areas may form different modules, with each module for one major clinical symptom (24). Modules may be anatomically or functionally linked to each other and may be activated in a characteristic manner in an individual. Each module can be activated to a different degree and the level of symptoms may correlate with the degree of activation of the respective module (19). It is possible that the modules for autonomic symptoms and phonophobia were activated to a higher degree in subjects with CAS. The differential influence of duration and intensity of pain on different modules has been described recently. Phonophobia, photophobia, dizziness, nausea and vomiting were found to be related to headache intensity, whereas osmophobia and dysguesia correlated with headache duration in migraineurs (9). Phonophobia in migraineurs may be pathophysiologically similar to photophobia, which is secondary to the release of inhibitory processes in the thalamus through interaction with the trigeminal nucleus (11). Other theories suggest that serotonin dysfunction may cause phonophobia or central hyperacusis (25), probably by affecting the inferior colliculus, which receives projections from raphe nuclei (26). The trigeminal autonomic reflex includes dorsal raphe nuclei in its circuitry (17), which may induce phonophobia in these patients secondary to serotonin dysfunction. The presence of phonophobia in all our subjects, but with a higher prevalence in CAS sufferers, shows that more than one mechanism may be responsible for photophobia in these patients.
Sleep provided no relief in subjects with CAS. Sleep is regulated by the hypothalamus and brainstem nuclei, particularly the locus coeruleus, dorsal raphe nuclei and pedunculo-pontine nuclei, and anatomical connections between them exist. The hypothalamus is also linked to the periaqueductal grey, which is a pain-suppressing structure. The hypothalamus and interconnected brainstem areas may be the neural sites responsible for the chronobiological features of some headaches, in particular the sleep-related attacks typical of the TAC and migraines (27). The possibility of hypothalamic involvement in subjects with CAS cannot be ruled out and it may be the reason why sleep could not relieve pain in these subjects, similar to episodes of cluster headache. Functional imaging studies may throw light on this issue.
Laterality or the side of headache did not influence presence of CAS in this study. Although previous reports have suggested that unilateral pain is more common in subjects with CAS (5, 7) and CAS are more prevalent in bilateral migraineurs because of diminished or absent reciprocal inhibition between two locus coerulei (17), we found no effect of laterality or sidedness on the prevalence of CAS. Moreover, conflicting evidence demands further research.
In conclusion, cranial autonomic symptoms are not uncommon in migraineurs. This study shows that subjects with CAS are comparable to migraine patients without CAS in other characteristics, but are clinically different from TAC sufferers. Further studies are required to analyse whether pathophysiological mechanisms and the therapeutic response in these subjects are comparable to ‘pure migraineurs’ or TO TAC patients.
Acknowledgements
We thank Dr Aditya Ratnani for helping in the revision of the manuscript.
