Abstract
Introduction
Pituitary adenylate cyclase-activating polypeptide (PACAP) is found in two functional isoforms, namely PACAP38 and PACAP27. The migraine-inducing properties of PACAP38 are well studied. However, it is not known whether the lesser-known and under-studied protein isoform, PACAP27, can also induce migraine attacks. Here, we studied the effect of human PACAP27 infusion on induction of migraine in a provocation model.
Methods
In a crossover study, 20 migraine without aura patients were randomly assigned to receive human PACAP27 (10 picomol/kg/min) or saline (placebo) infusion over 20 min. We recorded the migraine and associated symptoms.
Results
All patients completed the study. PACAP27 provoked migraine-like attacks in 11 patients (55%) and two developed attacks after placebo (10%) (p = 0.022). The headache intensity and duration after PACAP27 was significantly greater compared to placebo (p = 0.003).
Conclusion
PACAP27 triggers migraine attacks without aura. These novel data strengthen the role of PACAP and its receptors in migraine pathogenesis.
Keywords
Introduction
The novel multifunctional neuropeptide pituitary adenylate cyclase-activating polypeptide (PACAP) is highly conserved and found in two major isoforms: PACAP38 and PACAP27 (1,2). PACAP38 is the major isoform (3) and the lesser-known and under-studied isoform, PACAP27, is an amidated form of PACAP38, representing 10% of total PACAP in the body (4). PACAP isoforms and vasoactive intestinal polypeptide (VIP) are member of the secretin peptide superfamily (1). PACAP27 shows 68% homology in amino acid sequences with VIP (1). PACAP isoforms and their receptors are expressed in anatomical structures related to migraine (5,6), specifically in the trigeminal ganglion (7–9), perivascular nerve fibers and trigeminal nucleus caudalis (TNC) (10,11).
Interestingly, both isoforms have many similarities; however, some pharmacological differences have been reported (11–17). PACAP38’s migraine-provoking properties are well established (18–21). In contrast, VIP provoked no migraine in one study (22) and migraine attacks in few patients in another study (19). PACAP isoforms and VIP share three G-protein-coupled receptors (GPCRs), namely PAC1, VPAC1, and VPAC2 (4). PACAP isoforms bind with PAC1R with higher affinity (15) and activate the receptor approximately 1000-fold greater than that of VIP (2). PACAP38 provocation studies in migraine patients suggested PACAP or the PAC1 receptor as a possible novel target for migraine prevention (18,23). PACAP isoforms share several key pharmacological features, particularly their equipotent high affinity to the PAC1 receptor (24), yet PACAP27’s migraine-provoking properties have not been studied. Thus, we hypothesised that PACAP27 would provoke migraine-like attacks and induce flushing in a similar manner to PACAP38 in patients. To investigate this, we designed a double-blind crossover, placebo-controlled study using intravenous infusion of human PACAP27 in migraine without aura patients. Additionally, we used laser speckle contrast imaging to quantify and assess the effect of PACAP27 on the facial microvasculature.
Material and methods
Migraine without aura patients of both sexes were recruited via forsoegsperson.dk, a website for recruitment of volunteers to clinical research. All patients were invited by phone for further screening at the Rigshospitalet-Glostrup. All migraine patients were interviewed and physically examined by a trained physician (HG). Inclusion criteria were: age 18–60 years old; a minimum of two migraine without aura attacks per month as defined according to the ICHD-3 (beta) criteria (25). All female patients were allowed to take oral contraceptives. Exclusion criteria were: any medical conditions (except tension-type headache under five days per month and migraine without aura).
The Capital Region Ethics Committee of Denmark (H-17016232) approved the study protocol and the study was registered at Clinicaltrials.gov (ID: NCT03471039). In accord with the Declaration of Helsinki 2013 version, informed consent was obtained after oral and written information were provided. The study was performed between January 2018 and August 2018.
Experimental design
In a placebo-controlled study, 20 patients were randomly allocated to receive infusion of human PACAP27 (10 picomol/kg/min) (Bachem AG, Bubendorf, Switzerland) or placebo (saline) over 20 min in a double-blinded fashion on two different experiment days separated by a minimum of one week. This concentration was chosen based on earlier studies with PACAP38 that showed this dose induces migraine-like attacks and is well tolerated (18,19,21). A balanced randomisation was performed by the Capital Region Central Pharmacy to allocate patients equally to receiving infusion of PACAP27 or saline (placebo) on the first study day.
The experiment was canceled if the patient reported a migraine attack within 5 days, had taken any analgesics, or experienced headache two days prior to the experiment. Twelve hours before the start of the experiment, all patients were prohibited from consuming alcohol, cocoa, tea, coffee and caffeine, and smoking. Pregnancy tests were conducted on female patients on both study days.
Patients were placed in a supine position and a venous catheter was inserted into the right or left antecubital vein for PACAP27 or placebo infusion. The study was conducted in a temperature and light (dimmed) controlled lab at the same time of day ±1 h. After a rest, the baseline values were recorded in the following order: a) A well-validated headache questionnaire was used to record headache/migraine intensity and characteristics; b) an electrocardiogram was obtained; c) facial flushing was measured using a speckle contrast imager (moorFLPI, Moor Instruments, Devon, UK); d) mean arterial blood pressure (MAP) and heart rate (HR) were measured. After assessment of all baseline measurements, the infusion of PACAP27 was initiated from 0–20 min (Figure 1). All recordings were repeated every 10 min until 120 min. The study was completed after 2 hours and the patients were discharged from the hospital with a headache questionnaire to fill in every hour for 13 h post infusion. Patients were instructed to use their usual migraine medication as needed after completion of the experiment.
(a) Study design diagram. (b) Work flow chart of the methodological procedures during hospital phase (0–120 min) and post-hospital phase (3–13 hours).
Migraine-like attack criteria
According to the ICHD-3 (26), migraine attacks are spontaneous and last a minimum of 4 hours, if untreated. A migraine attack typically evolves slowly, and it may take hours before the criteria are met. However, in migraine provocation studies, the participants must be allowed to take rescue medication if needed. Therefore, as previously described, well-validated criteria for experimentally induced migraine-like attacks were used (27).
Headache and associated symptoms
The headache and migraine-associated symptoms, including premonitory symptoms, were recorded in the following order using a standard questionnaire: Nausea, vomiting, photophobia, phonophobia, unusual fatigue, craving, yawning, thirst, mood swings, facial flushing and difficulty in concentrating. We used a numerical rating scale (NRS) from 0 to 10 to obtain headache and migraine pain intensity, as published previously (28).
Facial skin blood flow
We measured blood flow intensity and fluctuation using the laser speckle contrast technique (moorFLPI, Moor Instruments, Devon, UK) as previously described (29–31). The contrast imager was manually optimised and placed 30 cm perpendicularly above the face of the patients. The laser speckle contrast imager measured facial skin blood flow automatically every 10 seconds. Approximately 1 minute before each of the chosen measurement time points, the patients were instructed to remain still for a minute. All patients were instructed to stay still in a supine position with closed eyes during the measurements.
Statistics and data analysis
All data are presented as mean ± standard deviation (SD) except the headache intensity scores, which are presented with individual data sets and median score. Furthermore, data shown in percentage changes are presented with a 95% confidence interval (CI).
We have calculated the sample size by determining the risk of type 1 error at 5% and defined a power at 80%. According to the study protocol, the type 2 error was fixed at 20%. Therefore, we estimated a requirement to include 20 patients in the study. In accordance with the trapezium rule (32), we applied the area under the curve (AUC) as a summary measure for analysing the difference in response between PACAP27 and placebo. We have set the difference between incidence of migraine headache and AUC for headache score (0–13 h) between PACAP27 and placebo as the primary endpoints of the study. The differences in secondary endpoints were in AUC (0–120 min) for facial skin blood flow, HR and MAP. Prior to the experiment initiation, the baseline was defined at time t0. In order to limit within-participant variation between experiment days, the baseline values were subtracted before calculating AUC. The non-parametric Wilcoxon signed rank test was used to compare AUC and baseline differences of all variables between PACAP27 and placebo. The Mann-Whitney test was used to test for period and carry-over effects for all baseline variables.
All statistical analyses were carried out using SPSS version 23.0 (Chicago, IL, USA) and no adjustments were made for multiple analyses. Five percent (p < 0.05) was accepted as the level of significance.
Results
Twenty patients completed both days of the experiment. The mean age of patients was 29 (range 18–52 years) and mean weight was 70 kg (range 52–91 kg). We tested the baseline values of MAP and HR (p > 0.05) for carry-over or period effect. Laser speckle data from seven patients were blindly excluded from analysis due to technical issues and artifacts.
Migraine-like attacks and associated symptoms
Eleven patients (55%) reported migraine-like attacks after PACAP27 infusion while only two patients reported this after placebo (10%) (p = 0.022). Median time for initiation of migraine-like attacks was 3 h (range 30 min – 7 h) and median time for headache onset was after 10 min (range 10 min – 3 h). Furthermore, the headache intensity score AUC0-13h was larger after PACAP27 compared to placebo (p = 0.003) (Figure 2). The peak headache intensity score was 2.5 (range 0–7) and the median headache duration was 2 h (range 10 min – 7 h) after PACAP27 and 0 (range 0–6) and 3 h (range 20 min – 11 h) after placebo.
Individual (black lines) and median (red line) headache intensity. The median peak headache score was 2.5 (range 0–7) after PACAP27 compared to 0 (range 0–5) after placebo.
The incidence of the migraine–associated symptoms nausea (p = 0.016), photophobia (p = 0.039) and fatigue (p = 0.008) was significant after PACAP27 compared to the placebo day. The time to onset of nausea after PACAP27 infusion was 4 h (range 2–13 h), corresponding to the median time for onset of migraine-like attack (3 h) (Figure 3). We found no difference in incidence of phonophobia (p = 0.125). Table 1 shows migraine and headache-associated symptoms and characteristics.
PACAP27-induced migraine associated symptoms and adverse events. (a) Onset time of each parameter. (b) Duration of each parameter. White box: Median. Red box: percentile 25–75%.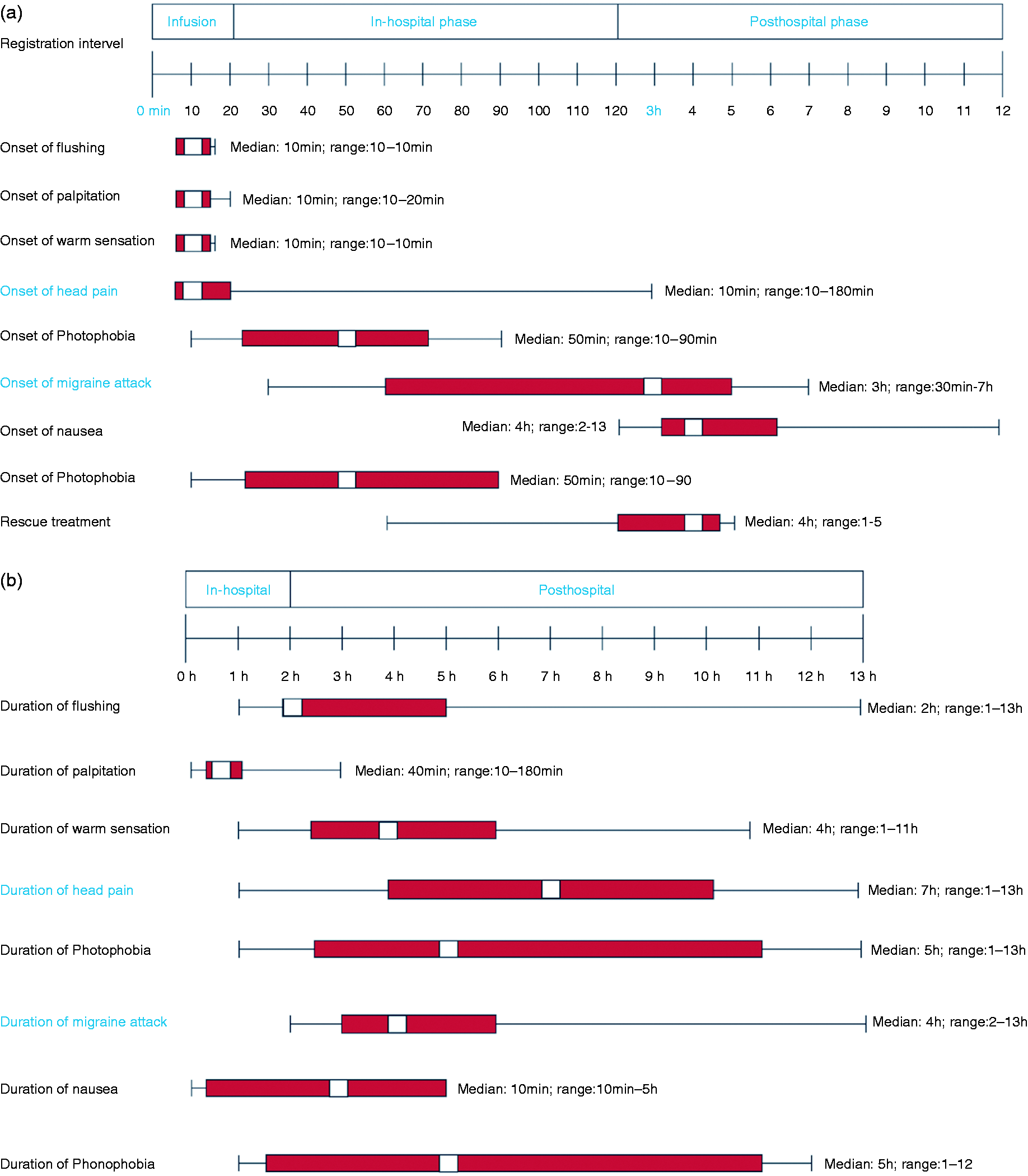
Vital signs and facial skin blood flow
PACAP27 induced increased facial skin blood flow (p = 0.001) (Figure 4). The AUC0–120 min for HR was higher after PACAP27 compared to placebo (p < 0.001). AUC0–120min for MAP (p = 0.305) was unchanged (Figure 5).
Facial skin blood flow changes after PACAP27 and placebo (0–120 min) shown in percentage change (n = 13) and direct flux measurement of an individual patient. The complete data sets (PACAP/placebo days) of seven patients were excluded due to movement artifacts, facial hair and other technical issues. Percentage change from baseline of heart rate and MAP after infusion of PACAP27 (red) and placebo (black).

Headache and adverse events
Eighteen out of 20 patients reported headache after PACAP27 infusion, with a median headache duration of 7 h (range 1–13 h) compared to six out of 20 after placebo (median 5.5 h, range 20 min – 13 h).
Characteristics of headache and associated symptoms in migraine without aura patients after PACAP27 and placebo (0–13 h).
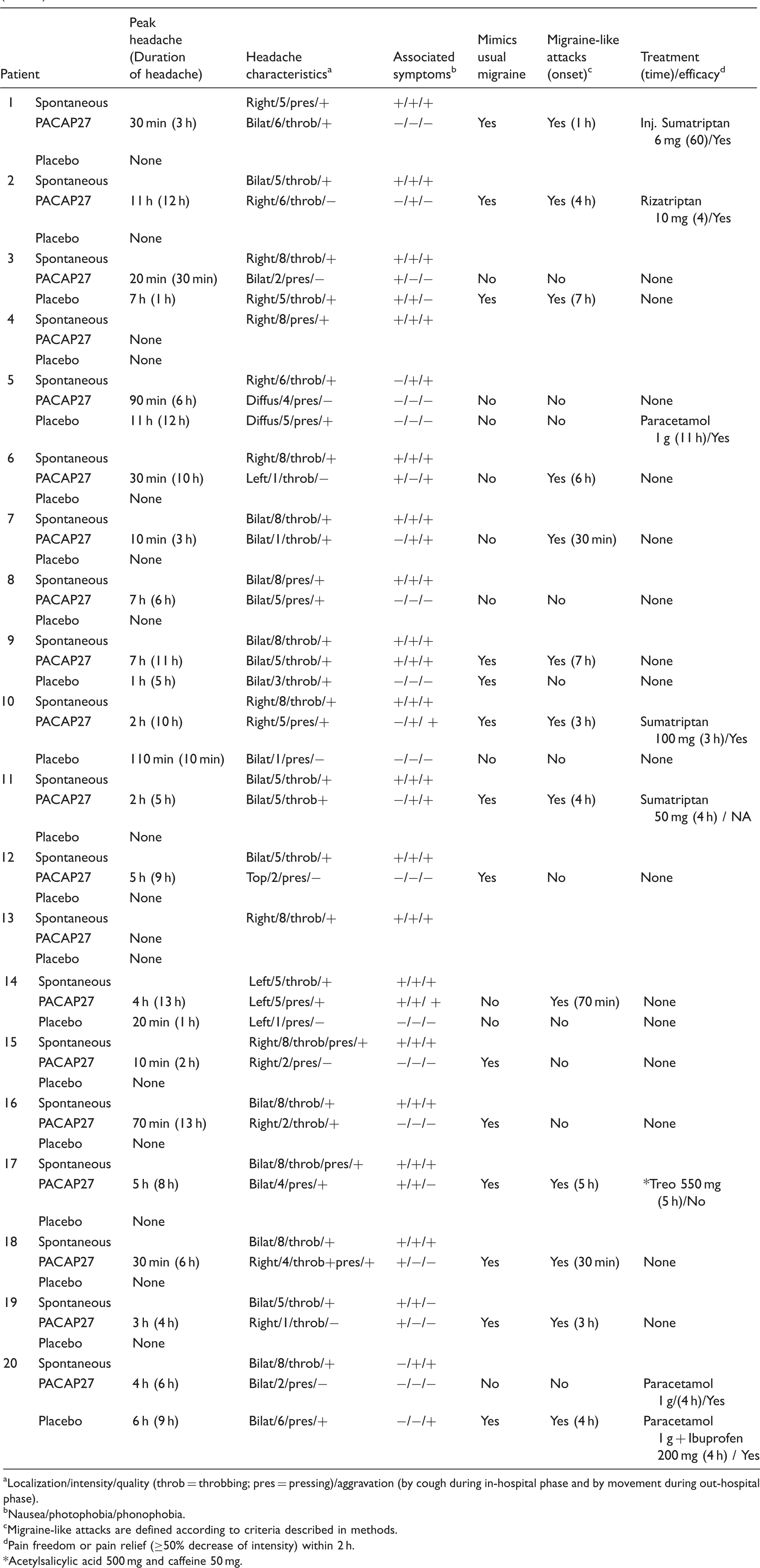
Localization/intensity/quality (throb = throbbing; pres = pressing)/aggravation (by cough during in-hospital phase and by movement during out-hospital phase).
Nausea/photophobia/phonophobia.
Migraine-like attacks are defined according to criteria described in methods.
Pain freedom or pain relief (≥50% decrease of intensity) within 2 h.
Acetylsalicylic acid 500 mg and caffeine 50 mg.
Discussion
Incidence of adverse events 0–13 h after PACAP27 or placebo.
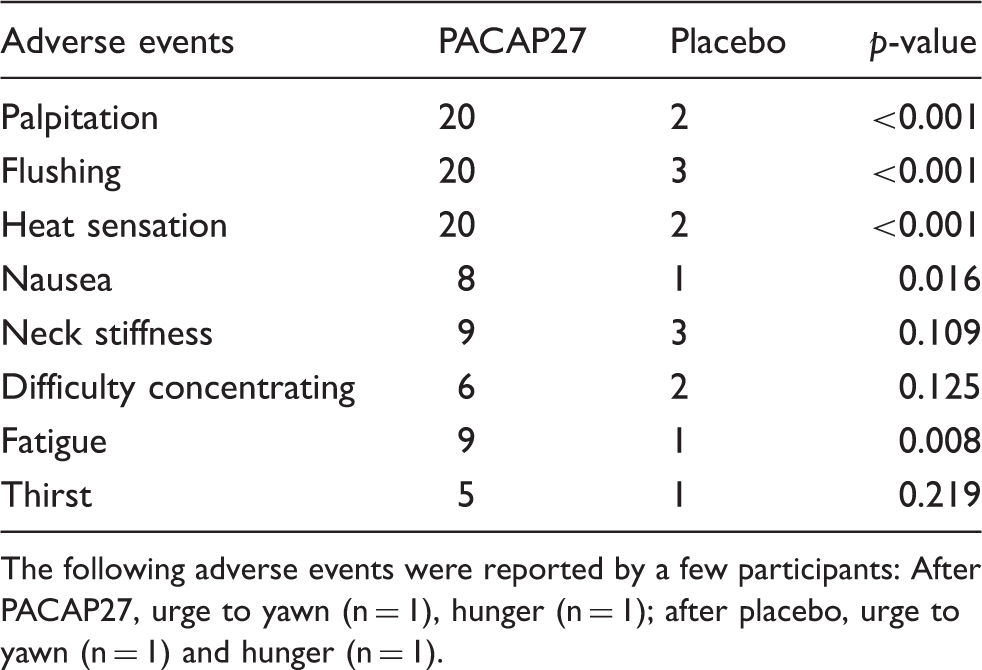
The following adverse events were reported by a few participants: After PACAP27, urge to yawn (n = 1), hunger (n = 1); after placebo, urge to yawn (n = 1) and hunger (n = 1).
Comparison of variables between PACAP27 and PACAP38.

Headache and migraine incidence in percentage.
Mean change from baseline.
PACAP is a potent local and systemic vasodilator across several species, and both isoforms dilate intracerebral and meningeal arteries with the same potency in animal models (24). In contrast to PACAP38, the vasorelaxant effect of PACAP27 shows regional differences, with a larger effect on the middle cerebral artery (MCA) and anterior cerebral arteries compared to the basilar artery in animals (33). In migraine (19) and healthy participants (34), PACAP38 caused a prolonged dilatation (>2 h) of extracranial arteries. We assessed facial skin blood flow intensity and showed that PACAP27 induced sustained dilatation of facial vasculature for up to >2 h (Figure 4). Three patients in the placebo group reported flushing in the post-hospital phase (3–13 h), including the two patients that developed migraine. However, we found no flushing in these patients during the hospital phase (0–2 h). To our knowledge, this is the first study to quantify facial flushing induced by a vasoactive peptide in migraine patients. Laser speckle can independently verify any hemodynamic change in facial vasculature. Therefore, we propose that future migraine studies should look at correlation between facial flushing intensity and migraine induction.
Distinct downstream signaling features of the PACAP isoforms are reported (15). Both PACAP isoforms increase cyclic adenosine monophosphate (cAMP) production, while only PACAP38 activated extracellular signal regulated kinase (ERK) in glia cells (15). It has been suggested that PACAP38 is more neuroprotective compared to PACAP27 (16). In an in vivo rat study, intrathecal administration of PACAP27 was able to suppress C-fiber-evoked flexion reflex at lower doses than PACAP38 (6). This study suggested that PACAP27 might be a more potent neurotransmitter or neuromodulator in c-fiber signaling than PACAP38. However, the role of PACAP27 in nociceptive signaling and cerebral hemodynamics in humans should be further studied.
Possible mechanisms behind PACAP27 induced migraine-like attack
In the present study, PACAP27 induced migraine-like attacks in 55% of patients, similar to migraine induction after PACAP38 (58–75%) (18–20,35). We found that the incidence of the migraine-associated symptom nausea was numerically higher after PACAP27 compared to PACAP38. This might be due to the longer plasma half-life of PACAP27 (45 min) compared to PACAP38 (less than 5 min) (36). The migraine-inducing action of PACAP27 may originate from several different anatomic sites, as the PACAP isoforms and their receptor are expressed in trigeminovascular structures such as the trigeminal ganglion (7–9), perivasvular nerve fibers and TNC (10,11). Experimental human migraine models suggested that the PACAP38 migraine-provoking properties might be mediated via the PAC1 receptor (18), which in turn might lead to delayed sensitisation of the central trigeminovascular neurons (37). It is not known if PACAP27 sensitises central trigeminovascular neurons. In rats, PACAP isoforms were elevated in the trigeminal nucleus caudalis upon electrical stimulation of the trigeminal ganglion or by intraperitoneal glyceryl trinitrate injection (11). This suggests that PACAP27 may be involved in central pain processing. PACAP27 activates the PAC1 receptor with similar affinity to PACAP38 (4). The first study of the monoclonal anti-PAC1 receptor antibody in migraine prevention has been completed (NCT03238781), but data have not yet been reported. Interestingly, a recent study reported a new humanised monoclonal anti-PACAP antibody that was highly selective to PACAP isoforms (38). Future studies will clarify whether anti-PACAP or its receptors are effective in migraine prevention.
The VPAC1 and VPAC2 receptors’ role in migraine pathophysiology is not well studied. Both PACAP isoforms can activate VPAC1 and VPAC2 receptors (4). Activation of PACAP receptors stimulates cAMP accumulation in smooth muscle cells (1,2). PACAP isoforms increase cAMP at a similar rate (15,39). Furthermore, the activation of VPAC1 leads to anti-inflammatory processes (40,41), whereas VPAC2 stimulation induces peripheral vasodilation (42). However, VIP, which has the same affinity to VPAC1 and VPAC2 receptors as PACAP isoforms, did not provoke migraine in one study (22) and induced a minimal headache in healthy volunteers (43). This calls into question the role of VPAC1-2 receptors in migraine induction.
It has been suggested that mast cell degranulation in the dura mater (12) may be attributed in migraine pathogenesis (44) and may in part explain the long-lasting vasodilatation caused by PACAP38 (34). PACAP27 is a weak dural mast cell degranulator compared to PACAP38 (12). PACAP27 releases histamine from skin mast cells more effectivly than PACAP38 (45). Pretreatment with antihistamine (mepyramine) prevented migraine-like attacks induced by infusion of histamine in migraine without aura patients (46). A recent study reported that pretreatment with H1-antihistamine and an anticholinergic drug (Clamastine) was unable to prevent PACAP38-induced migraine. This study suggests that mast cell degranulation or histamine release might not play a major part in PACAP38-induced migraine (47).
Conclusion
PACAP27 induced migraine-like attacks in migraine without aura patients with similar incidence rate to PACAP38. These data reinforce PACAP and its receptors’ role in migraine pathogenesis and the therapeutic potential of targeting PACAP or its receptors for novel migraine treatment.
Key findings
PACAP27 induced migraine-like attacks in 55% of patients. PACAP27 provoked prolonged headache in 90% of patients. PACAP27 increased facial blood flow and heart rate.
Footnotes
Acknowledgements
Lab technicians Winnie Grønning and Lene Elkjær are acknowledged for their help in data collection.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Hashmat Ghanizada, Mohammad Al-Mahdi Al-Karagholi, Nanna Arngrim and Jes Olesen declare no conflicts of interest. Messoud Ashina is a consultant, speaker or scientific advisor for Alder, Allergan, Amgen, Eli Lilly, Novartis and Teva, and principal investigator for Alder, Amgen, ElectroCore, Novartis and Teva trials.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study received financial support from Candys Foundation, Liechtenstein and Lundbeck Foundation (R155-2014-171).
