Abstract
Background
Intravenous infusion of 10 pmol/kg/min pituitary adenylate cyclase-activating polypeptide-38 (PACAP38) induces migraine-like attacks in migraine patients without aura (MO). Here, we conducted a pilot study and investigated if lower doses of PACAP38 exert similar migraine-inducing abilities.
Methods
We randomly allocated six MO patients to receive intravenous infusion of 4, 6, and 8 pmol/kg/min of PACAP38 over 20 minutes in a double-blind, three-way cross-over study. Headache and migraine characteristics were recorded during hospital (0–2 hours) and post-hospital (2–13 hours) phases.
Results
PACAP38 induced migraine-like attacks in one out of six patients with 4 pmol, two out of six patients with 6 pmol and three out of six patients with 8 pmol (
Conclusion
A trend of a dose-response relationship between dose of PACAP38 and incidence of migraine was observed. We suggest that 10 pmol/kg/min PACAP38 is the most optimal dose to induce migraine-like attacks.
Keywords
Introduction
Intravenous infusion of pituitary adenylate cyclase-activating polypeptide-38 (PACAP38) induces migraine attacks and long-lasting dilatation of extracerebral arteries in animals and humans (1–3). Additionally, recent studies indicate elevated plasma levels of PACAP-38 during attacks (4,5). To date, 10 pmol/kg/min is the only dose that has been used in provocation studies with PACAP38 in migraine patients. This dose induces migraine attacks in 58%–73% of migraine patients and causes transient side effects such as flushing, palpitations, warm sensations and lowering of blood pressure (1,2,6). To use PACAP38 as an ideal migraine model, it would be relevant to investigate if lower doses exert the same migraine-inducing properties and with fewer side effects. No data on the variability and reproducibility of PACAP38-induced responses have been reported.
Here, we conducted a double-blind, three-way cross-over pilot study to investigate the migraine-inducing properties and side effects of three lower doses of PACAP38 (4, 6 and 8 pmol/kg/min) in migraine without aura (MO) patients.
Materials and methods
Six MO patients were randomly allocated to receive three different pharmacological doses of PACAP38 intravenously over 20 minutes on three study days, separated by at least one week.
Participants were enrolled via the websites sundhed.dk and forsoegsperson.dk and gave their written informed consent before inclusion. The study was approved by the Research Ethics Committees of the Capital Region of Denmark (Journal no.: H-4-2014-103) and was conducted according to the Helsinki II Declaration. The study was registered at ClinicalTrials.gov (NCT02364453).
Inclusion criteria were: MO patients who fulfilled the International Headache Society (IHS) criteria of headache classification (7), ages 18–55, weighing 50–100 kg, fertile women only if using birth control. Exclusion criteria: tension-type headache more than five times a month, other primary headache, headache (48 hours) or migraine (72 hours) prior to trial days, daily intake of any medication (including prophylactic migraine medication) other than oral contraceptives, intake of any medication other than oral contraceptives less than four times the half-life of the medication, pregnant or breastfeeding women, any other illnesses.
Experimental protocol
Participants arrived headache (48 hours) and migraine free (72 hours) at the clinic between 9:00 and 10:00 a.m. Each infusion began at T0 and lasted 20 minutes. Vital signs (blood pressure, heart rate (HR) and electrocardiogram (ECG)) and the headache questionnaire were recorded at baseline, during the infusion and every 10 minutes until 120 minutes after the start of each infusion.
Headache intensity and questionnaire
Headache intensity was recorded every 10 minutes from T–10 to T120 on a verbal rating scale (VRS) from 0 to 10 (0 = no headache, 1 = a very mild headache (or otherwise painless altered sensation in the head), 5 = headache of moderate intensity, 10 = the worst headache imaginable (8)). Headache localization, characteristics and associated symptoms were also recorded. After two hours of continuous recording by the investigator, the patients were instructed to record their headache characteristics, associated symptoms, and side effects by a self-administered questionnaire every hour until 13 hours post-administration or until they had gone to bed. Simple analgesics or triptans were allowed when patients experienced their usual migraine-like pain.
Migraine-like attack criteria
The following definition was used for a migraine-like attack:
Either (1) or (2):
Headache fulfilling criteria C and D for MO according to the IHS criteria (7).
C. Headache has at least two of the following characteristics:
Unilateral location Pulsating quality Moderate or severe pain intensity (moderate to severe pain intensity is considered ≥4 on VRS) Aggravation by cough (in-hospital phase) or routine physical activity (out-hospital phase).
D. During headache at least one of the following:
Nausea and/or vomiting Photophobia and phonophobia. Headache mimicking usual migraine attack and treated with acute migraine medication.
Statistical analyses
Clinical characteristics of the participants are presented as mean and range. Headache intensity scores were presented as median (range).
We calculated area under the curve (AUC) according to the trapezium rule to obtain a summary measure. Baseline values were subtracted before calculating AUC to reduce variation between sessions within patient. Difference in AUC for headache intensity scores, HR and mean arterial pressure (MAP) between the three doses was tested using the Friedman test, and the incidence of migraine-like attacks was analyzed as categorical data with Cochran’s Q test. Exploratory analysis for difference in AUC for headache intensity scores between two doses were tested using the Wilcoxon signed ranks test.
All analyses were performed with GraphPad Prism 5 for Windows. A
Results
Six patients (four females/two males) with median attack frequency 2.6 days per month (range 1–4) completed the study. Mean age was 27.5 years (range 19–45) and mean weight was 66.8 kg (range 58–82). None of the patients had previously participated in similar provocation studies.
Incidence of migraine-like attacks
Clinical characteristics and associated symptoms of the spontaneous (Spontan.) and provoked migraine-like attacks and/or headaches in the six MO-patients on three study days given IV infusions of 4, 6, and 8 pmol/kg/min of PACAP38.

Localization/intensity/quality (throb: throbbing; pres: pressing)/aggravation (by cough during in-hospital phase and by movement during out-hospital phase).
Nausea/photophobia/phonophobia.
Migraine-like attacks are defined according to criteria, described in Methods.
Pain freedom or pain relief (≥50% decrease of intensity) within two hours.
Not taken during migraine-like attack.
MO: migraine without aura; IV: intravenous; PACAP38: pituitary adenylate cyclase activating peptide-38; ID: identification; Bilat: bilateral; Unilat: unilateral; N/A: not applicable.
We found a numerical increasing trend but no statistical difference in incidence of migraine-like attacks induced by PACAP38 among the three doses of 4 pmol/kg/min (one out of six patients), 6 pmol/kg/min (two out of six patients) and 8 pmol/kg/min (three out of six patients) ( Frequency of migraine-like attacks and headache after intravenous infusion of 4, 6, and 8 pmol/kg/min of pituitary adenylate cyclase activating peptide-38 (PACAP38) (
Incidence and intensity of headache
All patients reported headache after 8 pmol/kg/min, whereas five out of six participants reported headache after both 4 and 6 pmol/kg/min. We found no difference among the three doses in the AUC for headache intensity over the 12-hour observation period ( Median headache intensity on a 0–10 verbal rating scale (VRS) for six migraine without aura (MO) patients given 4, 6, and 8 pmol/kg/min of pituitary adenylate cyclase activating polypeptide-38 (PACAP38) and compared with historical data* (2) using 10 pmol/kg/min (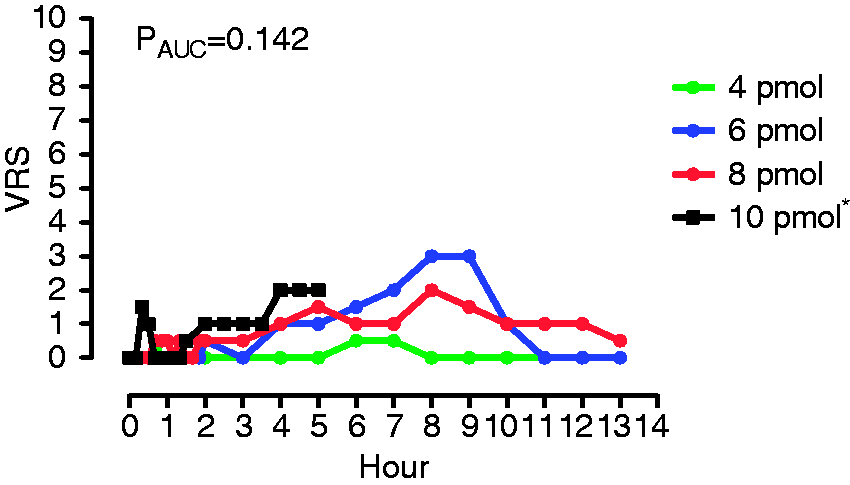
Side effects
We found no difference for the most common side effects among the three doses (
HR and MAP
There was no difference in AUC0–2 for HR (
Discussion
We conducted a pilot study and showed a numerical increasing trend in incidence of migraine-like attacks after three doses of PACAP38. A lack of statistical difference is likely due to small sample size. Interestingly, we saw no correlation between side effects and dose. One possible explanation could be that headache and side effects represent two different effects of PACAP38 that are not correlated to each other. Our historical data (2) on 10 pmol dose shows a 73% migraine response in patients, which is higher than we observed in the present pilot study after 4, 6 and 8 pmol (Figure 1). Whether a dose >10 pmol would result in higher percentages of patients reporting migraine attacks is unknown. Birk et al. reported a dose-response pilot experiment to compare 5, 10, 15, and 20 pmol PACAP38 in three healthy volunteers (9), but the infusions were stopped after 10 pmol/kg/min because of a 40%–50% increase in HR. Headache response was not described. Interestingly, a dose-response relationship of HR to PACAP38 was in accordance with the trend we found. In addition, we found a dose-dependent difference in HR during the initial 30 minutes of observation.
Reproducibility of PACAP38-induced migraine-like attacks has not been investigated before. We found that three patients developed migraine-like attacks on two experimental days and three patients did not develop migraine-like attacks on any of the experimental days. These data indicate that the PACAP38-induced migraine-like attacks seem to be reproducible, and that patients may have different dose thresholds for migraine induction.
We conclude that the dose of 10 pmol/kg/min is best suited for experimental studies. Firstly, because of the higher incidence of migraine-like attacks of 58%–73%. Secondly, it seems that lower doses of PACAP38 did not cause fewer side effects compared to 10 pmol/kg/min. Taken together, there is no clear advantage of using a reduced dose of PACAP38 in future provocation studies.
Footnotes
Clinical implications
We found a trend of a dose-response relationship between dose of pituitary adenylate cyclase activating polypeptide-38 (PACAP38) and incidence of migraine.
We suggest that 10 pmol/kg/min PACAP38 is the most optimal dose to induce migraine-like attacks.
Our study indicated reproducibility of PACAP38-induced migraine pointing toward different dose thresholds for migraine induction.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Messoud Ashina has received consulting fees, speaking/teaching fees, and/or honoraria from Alder, Allergan, Amgen, ATI and Eli Lilly. The other authors have nothing to declare.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: grants from Novo Nordisk Foundation (NNF11OC1014333) and Independent Research-Medical Sciences (FSS) (DFF-1331-00210A), Lundbeck Foundation (R155-2014-171) and the FP7-EUROHEADPAIN (602633). Anne Luise Vollesen received a scholarship grant from the Danish Neurological Society.
Acknowledgements
The authors thank all participating patients.
