Abstract
Background
Intravenous infusion of pituitary adenylate cyclase-activating polypeptide-38 (PACAP38) provokes migraine attacks in 65–70% of migraine without aura (MO) patients. We investigated whether PACAP38 infusion causes changes in the endogenous production of PACAP38, vasoactive intestinal polypeptide (VIP), calcitonin gene-related peptide (CGRP), tumour necrosis factor alpha (TNFα), S100 calcium binding protein B (S100B), neuron-specific enolase and pituitary hormones in migraine patients.
Methods
We allocated 32 previously genotyped MO patients to receive intravenous infusion PACAP38 (10 pmol/kg/minute) for 20 minutes and recorded migraine-like attacks. Sixteen of the patients were carriers of the risk allele rs2274316 (
Results
PACAP38 infusion caused significant changes in plasma concentrations of VIP (
Conclusion
PACAP38 infusion elevated the plasma levels of VIP, prolactin, S100B and TSH, but not CGRP and TNFα. Development of delayed migraine-like attacks or the presence of the
Keywords
Introduction
Pituitary adenylate cyclase-activating polypeptide-38 (PACAP38) is a 38-amino acid neuropeptide belonging to the secretin/glucagon superfamily (1). It was first discovered in 1989 and has in recent years gained considerable interest in the migraine field (2). An intravenous infusion of PACAP38 induces migraine attacks in 65–70% of migraine without aura (MO) patients and long-lasting dilatation of extra-cerebral arteries (3,4). In contrast, vasoactive intestinal peptide (VIP), which belongs to the same family as PACAP38, causes a short-lasting dilatation of extra-cerebral arteries and does not provoke migraine attacks (5). The exact pathophysiological mechanisms behind PACAP38-induced migraine attacks are still unknown. Given that PACAP38 is a multifunctional peptide that is involved in various cellular and physiological responses, non-vascular mechanisms of PACAP38 contributing to a migraine-triggering effect cannot be ruled out (4,6). Interestingly, genome-wide association studies have identified a single-nucleotide polymorphism (SNP) rs2274316 localized within the
In the present study, we hypothesized that PACAP38 infusion causes increased endogenous production of: PACAP38, calcitonin gene-related peptide (CGRP) and VIP as markers for release from parasympathetic and sensory perivascular nerve fibres (9); the inflammatory cytokine tumour necrosis factor alpha (TNFα) as a marker for mast cell degranulation (10); S100 calcium binding protein B (S100B) and neuron-specific enolase (NSE) as markers of neuronal damage, glial cell activation or leakage of the blood–brain barrier (BBB) (11,12); and pituitary hormones such as prolactin (PRL), thyroid-stimulating hormone (TSH), follicle-stimulating hormone (FSH), luteinizing hormone (LH) and adrenocorticotropic hormone (ACTH) as markers of hypothalamic activation.
The main purpose of this study is threefold: 1) to investigate the effects of PACAP38 on measured biochemical variables in migraine patients compared to controls who received placebo infusion; 2) to investigate whether there are different concentration patterns of the biochemical variables after infusion of PACAP38 between migraine patients who developed migraine attacks compared to those who did not; and 3) to investigate whether there are different concentration patterns of the biochemical variables between patients with and without the
Materials and methods
Blood samples were collected during the first part of the study, in which we examined the association between incidence of PACAP38-induced migraine-like attacks and familial predisposition (family load) of migraine. The materials and methods have already been described in Part I. Briefly, the patients were recruited from a cohort of 1010 unrelated MO patients from the Danish Headache Center who were genotyped for the 12 identified SNPs associated with migraine, including the risk allele C of rs2274316 (
An exploratory analysis in a recent study by our group reported elevated levels of PACAP38 in patients who reported delayed attacks (4). We found that plasma levels of PACAP38 (but not VIP) at 60 minutes after PACAP38 infusion were higher among patients who developed delayed attacks (
Design
We conducted a double-blinded study with 32 genotyped MO patients who all completed the study. Half of the patients (
Women of childbearing potential used adequate contraception. Exclusion criteria were any other type of headache (except episodic tension-type headache ≤8 days per month), intake of any preventative medication (including migraine preventives) and serious somatic or psychiatric diseases. A full medical examination and electrocardiography were performed on the day of the study. We informed patients that PACAP38 might induce headache in some individuals, but the timing or the characteristics of such headaches were not discussed.
The study was approved by the Regional Committee on Health Research Ethics of Copenhagen (H-2-2013-033) and the Danish Data Protection Agency, and was conducted according to the Helsinki II Declaration of 1964, as revised in 2008. The study was also registered at ClinicalTrials.gov (NCT01841827). All participants gave their written informed consent before inclusion.
Experimental protocol
All participants arrived non-fasting at the clinic at between 8:30 and 10:30 a.m. They had to be without any kind of headache or intake of analgesics for 48 hours before the study day. Venous catheters (Venflon®) were inserted into the left and right antecubital vein for the administration of PACAP38 and for the drawing of blood samples, respectively. All patients were kept in the supine position in quiet surroundings for at least 15 minutes before baseline blood samples were taken. Immediately after this, we started the PACAP38 infusion in migraine patients or saline in healthy controls for 20 minutes. We collected blood samples at baseline and 20, 30, 40, 60 and 90 minutes after the start of the infusion. The patients were observed for at least 90 minutes and were then discharged.
Blood sampling and assays
In order to avoid haemolysis of the blood during sampling, we conducted a small test with two healthy subjects prior to the study using different methods of blood sampling, showing that blood drawn from the venous catheter by syringe caused the least haemolysis compared to other standard blood sampling methods. Thus, for the study, we withdrew blood from the venous catheter into two 20-mL plastic syringes after we aspirated and discarded the first 3 mL of blood. The catheter was flushed with isotonic salt water after each sample was taken. The blood from the syringes was then transferred into different tubes: serum tubes for TNFα, S100B, NSE and TSH measurements; pre-cooled lithium heparin tubes containing aprotinin (Trasylol®) for VIP; standard ethylenediaminetetraacetic acid (EDTA) tubes for CGRP, PRL, FSH and LH; and finally, pre-cooled EDTA tubes containing aprotinin for PACAP38 and ACTH measurements. All tubes were inverted several times immediately after the blood was collected. Apart from the serum tubes, which stood at room temperature (23℃) for 20 minutes, all other tubes were stored at 5℃ for 20 minutes. All samples were centrifuged together at 4℃ at 1851
For measurement of PACAP38 and VIP, the radio-immunoassays were performed as previously described (5,17). Serum TNFα was measured by a highly sensitive human immunoassay (R&D Systems Europe, Ltd, Abingdon, UK) for the Bio-Tek ELx808™ microplate reader. S100B, NSE, PRL, FSH, LH and ACTH were measured using automated electrochemiluminescence immunoassays (cobas® 6000 or cobas® e411, Roche, Basel, Switzerland), whereas TSH was measured using a chemiluminescence immunoassay (Vitros® 5600, Ortho Clinical Diagnostics, Rochester, NY, USA). Plasma CGRP concentrations was determined by radioimmunoassay using antibody AB 4-2905 and α-CGRP as a calibrator. The tracer was prepared by iodination of [Tyr0] α-CGRP(25–37) amide and purification was by high performance liquid chromatography (HPLC). In brief, the samples and calibrators were incubated with the antiserum at 4℃ for approximately 90 hours prior to the addition of the tracer and subsequent incubation for 24 hours. Free and antibody-bound tracers were separated by a solid-phase separation system (SAC-CEL, IDS, Boldon, UK), as described in detail by Schifter (18). For all methods, the inter-assay coefficient of variation was below 10%.
Migraine-like attacks and questionnaire
We recorded migraine headache characteristics and associated symptoms using a validated questionnaire (19). After discharge from the hospital, the patients were carefully instructed to continue recording their headache by a self-administered questionnaire every hour until 12 hours after the start of infusion or until they went to bed. The questionnaire recorded headache characteristics and associated symptoms according to the International Headache Society criteria (16), but also included questions concerning adverse events and whether the reported headache mimicked the spontaneous migraine attacks. The definition of a migraine-like attack has been described in several previous studies (3,4,20). The patients were allowed to take their usual acute migraine medication at any time.
Statistical analysis
The clinical characteristics of the participants are presented as means and ranges. The outcome variables are presented as median values with interquartile ranges.
The primary endpoints were: 1) differences in plasma concentrations over time (0–90 minutes) of PACAP38, CGRP, VIP, TNFα, S100B, NSE, PRL, TSH, FSH, LH and ACTH between migraine patients who received a PACAP38 infusion and healthy volunteers who received a saline infusion; 2) differences in plasma concentrations over time of measured biochemical variables after PACAP38 infusion between migraine patients who developed migraine attacks and those who did not; and 3) differences in changes in plasma concentrations over time of the measured biochemical variables between patients with and without the
The secondary endpoint was the difference in blood levels of PACAP38 and VIP at 60 minutes between patients who developed migraine compared to patients who did not develop migraine.
The difference between the two groups over time was assessed by repeated-measurements analysis of variance (RM-ANOVA), with the interaction between time and group (time × group) being the term of interest.
The
Results
Clinical characteristics of migraine patients and healthy volunteers (controls).

NA: not applicable.
Chi-square test.
Unpaired
Migraine responses after PACAP38 infusion have been reported in the first part of the study (Part I). Briefly, 23 out of 32 (72%) migraine patients developed a migraine-like attack after PACAP38 infusion. Eleven out of 16 patients with the bi-allelic
Patients versus controls
We found significant differences in plasma concentrations for PACAP38, VIP, PRL, S100B and TSH between migraine patients who received PACAP38 infusion and controls (who received saline infusion over 20 minutes) (Figure 1). No differences were found in the baseline values between migraine patients and healthy volunteers ( Effects of PACAP38 on plasma concentrations of measured neuropeptides, TNFα and pituitary hormones in migraine patients compared to saline infusion in healthy volunteers. Data are shown as medians with interquartile ranges. Filled triangles represent migraine patients who received PACAP38 infusion (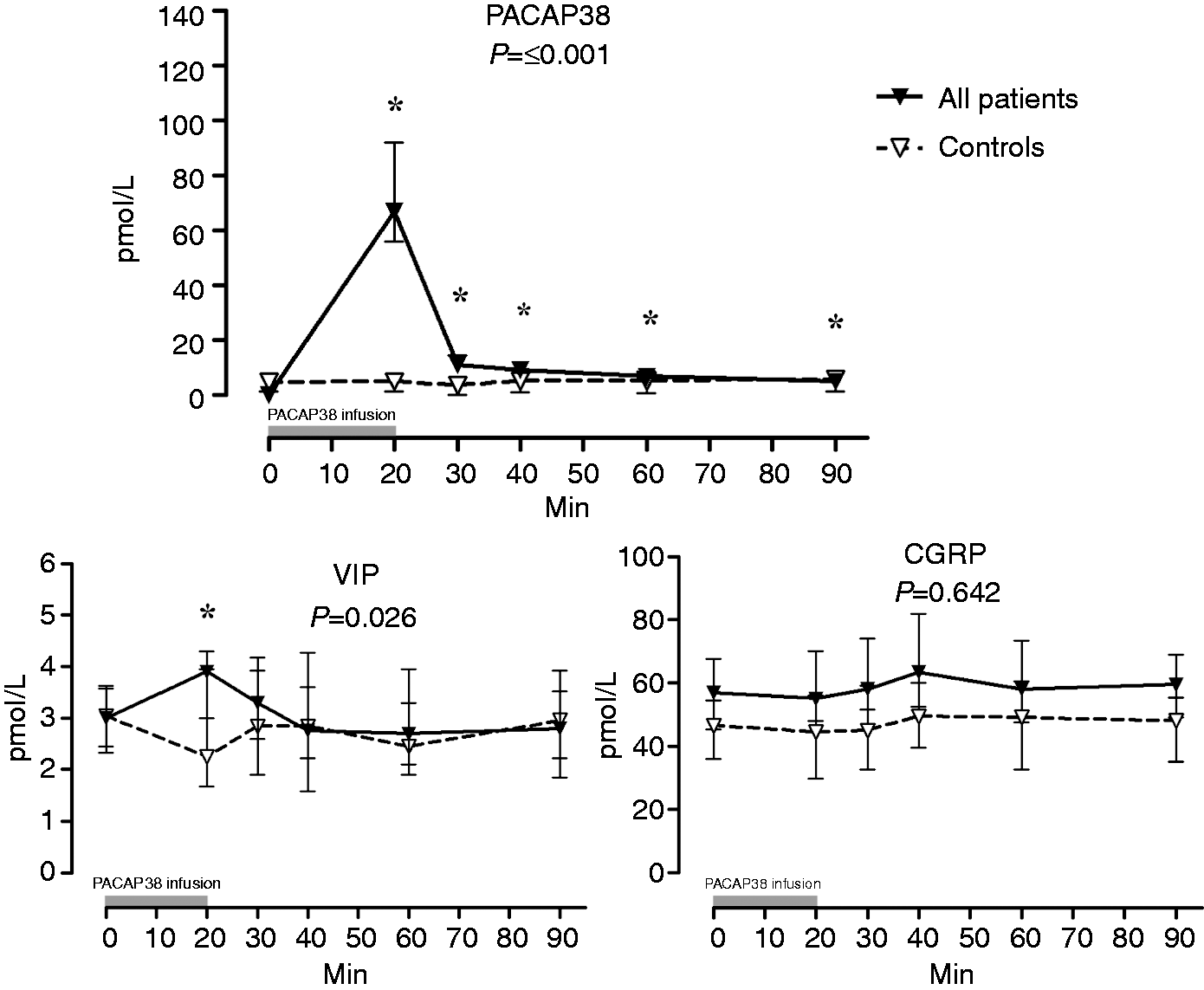
Patients who report migraine versus patients who did not report migraine
We found no differences in the biochemical variables between patients who developed migraine-like attacks ( Plasma concentrations of measured neuropeptides, TNFα and pituitary hormones in patients who developed PACAP38-induced migraine like-attacks compared to those who did not. Data are shown as medians with interquartile ranges. Filled circles represent patients who developed migraine attacks (
Patients with the MEF2D gene variant versus patients without the MEF2D gene variant
We found no differences in the biochemical variables over time (0–90 minutes) between patients with (
Pooled data of changes in PACAP38 concentration during the pre-ictal phase
When we pooled our present data with our previous data (4), changes in the plasma levels of PACAP38 and VIP at 60 minutes after PACAP38 infusion showed no differences between patients who developed migraine-like attacks ( Box-and-whisker plot of median changes (interquartile range) in plasma concentrations of PACAP38 and VIP at 60 minutes after PACAP38 infusion in patients who developed migraine-like attacks (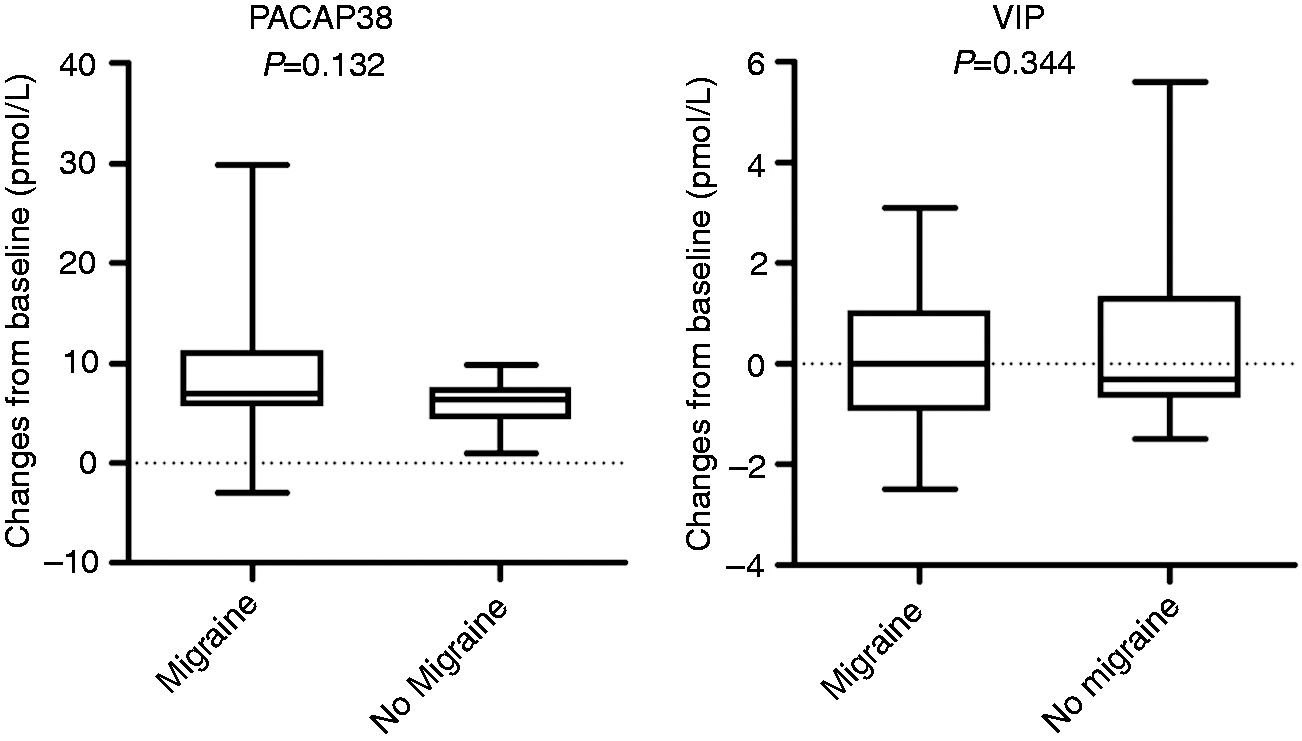
Discussion
The major finding of the present study is that PACAP38 infusion caused significant changes in plasma concentrations for VIP, PRL, S100B and TSH, but not CGRP and TNFα in migraine patients. Further analysis revealed no trends towards increased changes in the measured variables in patients who reported delayed migraine attacks or in patients with the
PACAP38 effect on VIP, CGRP and TNFα
It has been suggested that the migraine-inducing and vasodilatory properties of PACAP38 may be caused by: 1) activation of the parasympathetic nervous system (3); 2) activation of perivascular sensory nerve endings (2); or 3) mast cell degranulation (21–23).
VIP is a parasympathetic neurotransmitter that is structurally related to PACAP38 (6). In rats, PACAP38-positive nerve fibres have been shown to innervate VIP-expressing neurons (24,25). Furthermore, PACAP38 induces
CGRP is expressed in the sensory nerve fibres of the cranial vasculature (30,31), and studies in migraine patients reported increased plasma levels of CGRP during spontaneous attacks (27,32–34). However, one study using an intra-patient comparison design and two different CGRP assays collected from the jugular vein failed to reproduce these findings (35). We also reported increased inter-ictal CGRP levels in migraine patients (36), which could not be reproduced in the present study, most likely due to the small number of controls. Similarly to PACAP38, CGRP induces migraine attacks in MO patients (60–65%) (37), and both peptides activate the cyclic adenosine monophosphate-dependent signalling pathway (38,39). Immunofluorescence studies have shown that CGRP and PACAP38 are, to some extent, co-expressed in the trigeminal nucleus caudalis (40) and the trigeminal ganglion (41). In rats, PACAP38 causes the release of CGRP from the trigeminal nucleus caudalis but not from the trigeminal ganglion (41). In addition, both PACAP38 and CGRP are upregulated following inflammation in sensory neurons (42,43). To our knowledge, no studies have investigated plasma CGRP after the infusion of PACAP38 in migraine patients. The present study revealed that PACAP38 infusion did not cause changes in plasma CGRP concentrations, including in patients who later reported migraine-like attacks. However, we cannot exclude possible changes in plasma CGRP during PACAP38-induced delayed migraine-like attacks.
TNFα is an inflammatory cytokine that is released upon mast cell degranulation (44). Mast cells are located perivascularly in the dura (45) and have been suggested to be involved in migraine pathophysiology (22). Plasma levels of TNFα are reported to be elevated during spontaneous migraine attacks (46). In mice, PACAP38 has been shown to inhibit macrophage and microglia production and the release of TNFα (47). In a previous study, we investigated plasma levels of tryptase as a marker for mast cell activation after PACAP38 infusion in migraine patients and found no changes (4). The present study used TNFα as a marker of mast cell degranulation and failed to demonstrate any changes, including in patients who reported delayed attacks. Collectively, these data question the role of mast cells in PACAP38 responses, but we cannot rule out delayed ictal changes in plasma levels of TNFα.
PACAP38 effect on plasma levels of S100B and NSE
S100B is a calcium-binding protein that is produced and released mostly by glial cells in the central nervous system (48). It has been suggested that an isolated S100B increase may be an early marker of BBB opening and is not necessarily related to neuronal damage (12). A small study causing iatrogenic BBB disruption with mannitol showed that serum S100B increased (∼50%) significantly, while NSE did not (12). If PACAP38 causes leakage of the BBB, this would enable the passage of molecules that normally do not enter the brain. Present findings of increased serum levels of S100B (10–20%), but not NSE, over the observation period (0–90 minutes) suggest that infusion of PACAP38 alters the BBB’s permeability. However, we cannot rule out the notion that elevated S100B levels reflect a release from the peripheral nervous system (49) in response to the inflammation induced by PACAP38.
PACAP38 effect on plasma levels of PRL and TSH
We found that PACAP38 caused increases of PRL and TSH compared to controls. It is unclear whether this effect is caused by actions on the pituitary level or on the hypothalamic level, or both. The effect of PACAP38 on the pituitary gland has previously been reviewed in detail (50,51). In rats, PACAP38 stimulates hormone release in essentially all pituitary cells. In addition, PACAP38 enhances plasma PRL levels in hypothalamus-lesioned animals (52). The most abundant population of PACAP38-containing neurons and the highest density of PACAP38 binding sites are found in the hypothalamus (53). Since the pituitary gland and the median eminence of the hypothalamus lack an effective BBB (54), they are rather reachable for intravenously administered PACAP38. A small study in seven healthy men reported that infusion of low PACAP38 doses (4 and 8 pmol/kg/minute) stimulated the release of PRL and ACTH, but not TSH and LH (55). In contrast, the present study, which primarily had female participants, showed an increase of TSH, but not ACTH after a high dose of PACAP38 (10 pmol/kg/minute). It is also possible that the plasma concentration of PACAP38 was not sufficiently high to activate the receptors, signalling pathways and secretion of all of the pituitary cells. It would be relevant to examine plasma levels of growth hormone-releasing hormone and LH-releasing hormone as markers for more specific hypothalamic activation.
The risk allele of rs2274316 (MEF2D) and biochemistry after PACAP38
The risk allele is localized intronically within the
Pre-ictal plasma levels of PACAP38
The present study analysed pooled plasma PACAP38 data in order to confirm or refute (due to possible a type 1 error) our previous report of increased endogenous production of PACAP38 in patients who later reported migraine-like attacks. This strategy allowed us to increase the power and sample size in order to ascertain our finding. Apart from the fact that we collected blood samples inside the magnetic resonance imaging scanner in our previous study (4), in both studies, patients received the same dose of PACAP38 infusion over 20 minutes. Furthermore, identical blood sampling and analysis methods and headache questionnaires were used.
The pooled data analysis showed no increase of PACAP38 plasma levels in the pre-ictal phase of PACAP38-induced attacks. Nonetheless, recent studies reported elevated plasma levels of PACAP38 during migraine attacks, which were reduced by 1 hour after treatment with sumatriptan (32,59).
Conclusion
PACAP38 infusion caused changes in plasma concentrations of VIP, PRL, S100B and TSH compared to controls, but not CGRP and TNFα. Our findings indicate that PACAP38 might activate parasympathetic nerve endings causing VIP release, rather than activation of perivascular sensory nerve endings and mast cell degranulation.
Article highlights
Pituitary adenylate cyclase-activating polypeptide-38 (PACAP38) infusion caused changes in plasma concentrations of vasoactive intestinal polypeptide, prolactin, S100 calcium binding protein B and thyroid-stimulating hormone. PACAP38 may activate parasympathetic nerve endings rather than sensory nerve endings or mast cell degranulation. Pre-ictal levels of PACAP38 are not elevated in patients who reported delayed attacks.
Footnotes
Acknowledgements
The authors thank all participating patients. Special thanks are given to Majken Gudmundsson Vandbæk, Niklas Rye Jørgensen, Mette Nors, Lars Schack Kruse and Charlotte Sewerin Larsen for assistance in performing the biochemical analyses.
Declaration of conflicting interest
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Jes Olesen has received grants and/or research support from, has been a consultant and/or scientific adviser for and has been in the speakers’ bureaus of Allergan, Inc., AstraZeneca Pharmaceuticals LP, Boehringer Ingelheim, Eli Lilly, GlaxoSmithKline, Janssen Pharmaceutical Products, Lundbeck, Merck, Amgen, Alder and Pfizer. Messoud Ashina has received honoraria for lecturing from Allergan and is a consultant and/or scientific adviser/speaker for the ATI, Allergan, Amgen, Alder and Eli Lilly.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: grants from Novo Nordisk Foundation (NNF11OC1014333) and Independent Research-Medical Sciences (FSS) (DFF-1331-00210A), the Lundbeck Foundation (R155-2014-171) and FP7-EUROHEADPAIN (602633).
Supplementary article
Part I: Pituitary adenylate cyclase-activating polypeptide-38-induced migraine attacks in patients with and without familial aggregation of migraine. DOI: 10.1177/0333102416639516.
