Abstract
Background
Deep brain stimulation in the ventral tegmental area (VTA-DBS) has provided remarkable therapeutic benefits in decreasing headache frequency and severity in patients with medically refractory chronic cluster headache (CH). However, to date the effects of VTA-DBS on cognition, mood and quality of life have not been examined in detail.
Methods
The aim of the present study was to do so in a case series of 18 consecutive patients with cluster headache who underwent implantation of deep brain stimulation electrodes in the ventral tegmental area. The patients were evaluated preoperatively and after a mean of 14 months of VTA-DBS on tests of global cognition (Mini Mental State Examination), intelligence (Wechsler Abbreviated Scale of Intelligence), verbal memory (California Verbal Learning Test-II), executive function (Delis–Kaplan Executive Function System), and attention (Paced Auditory Serial Addition Test). Depression (Beck Depression Inventory and Hospital Anxiety and Depression Rating Scale-D), anxiety (Hospital Anxiety and Depression Rating Scale-A), apathy (Starkstein Apathy Scale), and hopelessness (Beck Hopelessness Scale) were also assessed. Subjective pain experience (McGill Pain Questionnaire), behaviour (Pain Behaviour Checklist) and quality of life (Short Form-36) were also evaluated at the same time points.
Results
VTA-DBS resulted in significant improvement of headache frequency (from a mean of five to two attacks daily, p < .001) and severity (from mean Verbal Rating Scale [VRS] of 10 to 7, p < .001) which was associated with significant reduction of anxiety (from mean HADS-A of 11.94 to 8.00, p < .001) and help-seeking behaviours (from mean PBC of 4.00 to 2.61, p < .001). VTA-DBS did not produce any significant change to any tests of cognitive function and any other outcome measures (BDI, HADS-D, SAS, BHS, McGill Pain Questionnaire, Short Form-36).
Conclusion
We confirm the efficacy of VTA-DBS in the treatment of medically refractory chronic cluster headache. The reduction of headache frequency and severity was associated with a significant reduction of anxiety. Furthermore, the result suggests that VTA-DBS for chronic cluster headache improves pain-related help-seeking behaviours and does not produce any change in cognition.
Introduction
Cluster headache (CH) is a rare trigeminal autonomic cephalalgia (TAC) characterized by attacks of severe cranial pain associated with ipsilateral autonomic symptoms such as conjunctival injection, lacrimation, nasal congestion, ptosis or eyelid oedema (1). CH is one of the most excruciating and disabling conditions and it has a severe impact on the patient’s quality of life. Patients with CH show greater anxiety, depression and worse quality of life compared with healthy populations (2). The exact pathophysiology in cluster headache (CH) is a matter of debate (3–10). The seasonality and periodicity of CH attacks are indicative of a possible hypothalamic role (7,11). This has been further supported by neuro-endocrinological studies (12,13) and neuroimaging studies (14,15). Early imaging studies pointed to increased activity and neuronal density in what was thought to be ‘the posterior hypothalamic region’ (16). Further studies have shown this region to lie not in the hypothalamus but in the ventral tegmental area (VTA) (17–19). In the course of an attack, pathological activation of the trigemino-parasympathetic brainstem reflex is considered to cause trigeminal nerve and craniofacial parasympathetic activation resulting in distinctive ipsilateral cranial pain and autonomic features (7,20). The trigemino-hypothalamic tract, which connects the posterior hypothalamic region to the trigeminal nucleus in the brainstem has been proposed as an important pathway in this process (21). Standard medical treatments of CH include acute therapy aimed at aborting individual attacks and preventative medications, which reduce the frequency of attacks. These treatments are effective for a proportion of the patients (22). On the other hand, in a small but significant number of highly disabled individuals, standard medical therapy is not sufficiently effective and CH attacks are intractable. For these patients, deep brain stimulation (DBS) is considered as a therapeutic option. DBS is a surgical treatment in which electric pulses are continuously applied via stereotactically implanted electrodes. The first DBS procedure for CH in 2001 (23) targeted the so-called “postero-medial hypothalamus” with positive results. Subsequent investigators (18,19,21) have highlighted that the region being targeted is more accurately termed the VTA and have also reported beneficial effects of DBS of the VTA with significant reduction in the severity and the frequency of CH attacks (17). The aim of the present study was to investigate the effect of VTA-DBS for CH on cognition, mood, pain experience and behaviour, and quality of life in most of the patients who were also included in the clinical report of Akram et al. (17).
Methods
Study sample
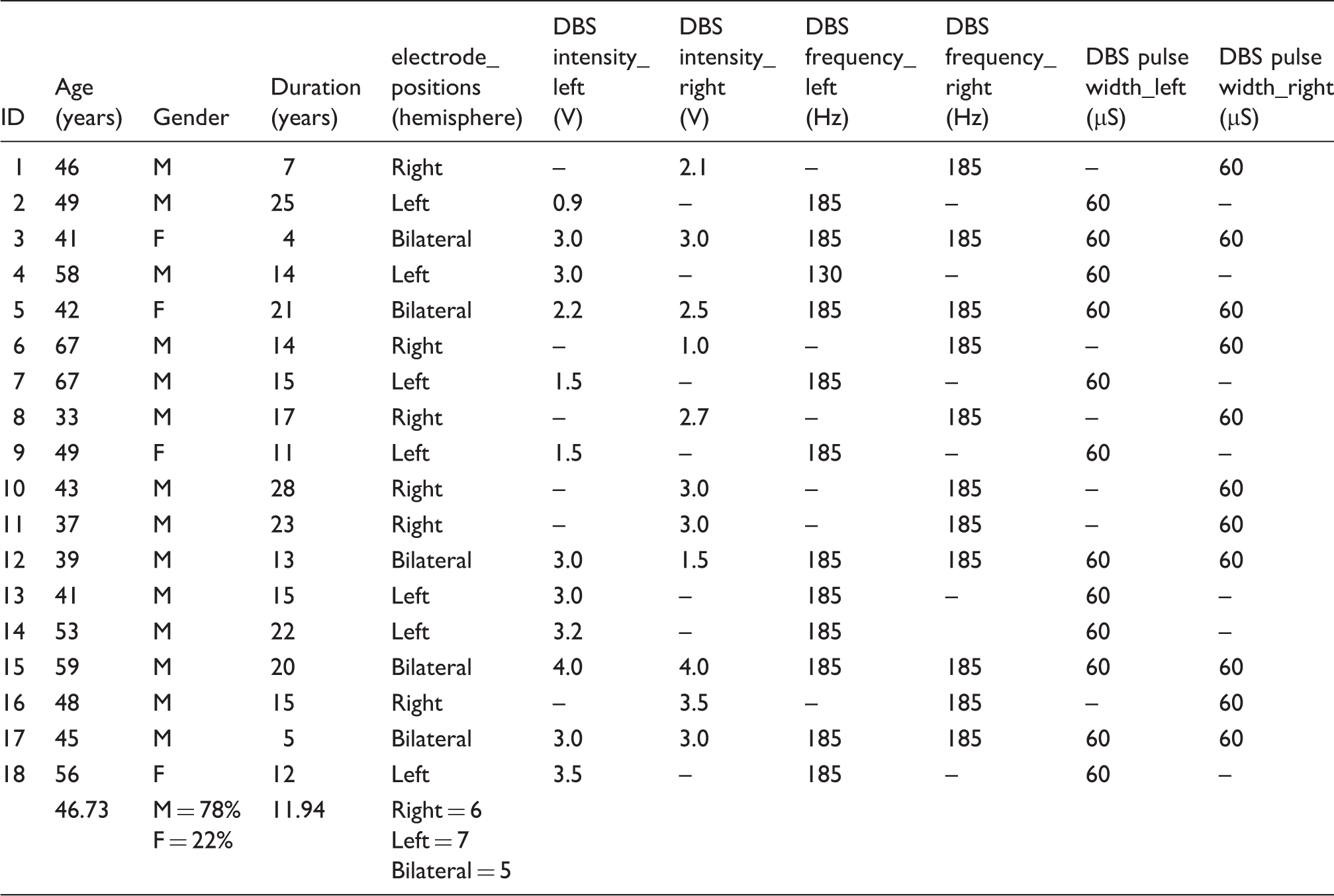
Demographic and deep brain stimulation (DBS) parameters of the patients with chronic cluster headache.
Procedure
All enrolled participants underwent clinical examination and neuropsychological assessment prior to surgery. Clinical and neuropsychological assessments were repeated 14 ± 4.5 months postoperatively. The surgical procedure has been described previously (24) and involved DBS lead (model 3389, Medtronic Inc.) implantation in the ipsilateral VTA or bilaterally (if symptoms were side alternating) under local or general anesthesia. The most deep contact location of the quadripolar lead was determined on a 1.5T T2 axial stereotactic MRI at a level directly above the mammillary bodies, anteromedial to the hypointense red nucleus and posterolateral to the hypointense mammillothalamic tract (24). In the weeks following surgery, open label programming was conducted to define optimal stimulation parameters. All devices were programmed with a frequency of 185 Hz and pulse width of 60 µs, gradually increasing the intensity up to 4.0 volts (V) on weekly intervals according to occurrence of side effects (diplopia, vertigo, oscillopsia, and ophthalmoplegia). Stimulation parameters are provided in Table 1.
Assessment of headache frequency, severity and load
Headache severity was evaluated on a verbal rating scale (VRS) for pain (0, no pain, to 10, the worst pain imaginable). Headache frequency was described as the number of CH episodes per day. Headache load (HAL) is a composite score to simultaneously measure frequency, severity and duration of cluster headache episodes. It was calculated as ∑ (severity [verbal rating scale] × duration [in hours]) of all headache attacks experienced over a 2-week period (17). These measures were assessed using headache diaries and were used as the clinical outcome measure.
Neuropsychological assessment
Participants were evaluated within one month prior to the DBS procedure and one year or more post-operatively (mean 14 months ± 4.5). The neuropsychological battery of tests was selected to be comprehensive and assess various cognitive domains, and involved assessment of global cognition, estimates of premorbid and current IQ, verbal and non-verbal memory, executive function, attention and language (for details please see (2)).
Global cognition and intelligence
The Mini Mental State Examination (MMSE) (25) was used as a measure of global cognitive functioning. The National Adult Reading Test (NART) (26) was used to obtain an estimate of premorbid intelligence quotient (IQ), and the Wechsler Abbreviated Scale of Intelligence (WASI) (27) was administered to obtain an estimate of current Full Scale IQ. The NART assesses reading and vocabulary skills. The participants are asked to read aloud 50 irregular English words. The total error score is used to obtain an estimate of premorbid IQ. The WASI is an abbreviated scale to provide a measure of current intelligence quotient (IQ). It includes four subscales: Vocabulary, similarities, block design, and matrix reasoning. The vocabulary scale is indicative of verbal knowledge and verbal concept formation. Matrix reasoning measures non-verbal skills of fluid and abstract reasoning. In the short form of the WASI, the vocabulary and matrix reasoning scores are used to obtain an estimation of the current Full Scale IQ.
Verbal and non-verbal memory
The California Verbal Learning Test (CLVT-II) (28) is a test of episodic verbal memory for words that provides data on immediate memory span, verbal learning, short/long term free and cued recall and delayed recognition of 16 words, which belong to four semantic categories.
The Recognition Memory for Faces-short form (SRMF) (29) was used to assess non-verbal recognition memory for faces. The SRMF consists of two phases. In the first phase, 25 photographs of male faces are shown at the speed of one every 3 seconds. The participant is asked to indicate if they consider the face to be pleasant or not pleasant. In the second phase, 25 pairs of faces are shown and the participant is asked to identify which of the two faces had been previously presented. The score is the total number of correctly recognized faces.
Executive functions and inhibition
The Delis–Kaplan Executive Function System (D-KEFS) (30) is a set of tests sensitive to verbal and nonverbal executive functions. In our study, the following subtests were used to assess executive function: The Trail Making Test (TMT), the phonemic, semantic and alternating categories Verbal Fluency (VF), and the Colour-Word Interference Test (Stroop). In the D-KEFS verbal fluency, the participant has 60 seconds to orally produce as many words as possible. There are three conditions: In the first condition (letter fluency), the participants are presented with a letter (F, A and S) and asked to say as many words as possible starting with that letter. In the second phase (category fluency), the participants are asked to say as many words as possible in the category of animals and boys’ names. In the third phase (switching), the participant is asked to say as many words as possible alternating between a fruit and furniture category. The score for each condition is the total number of correct items produced. Based on the original test designed by Stroop (1935), the Delis-Kaplan Colour-Word Interference test consists of four subtests. The “colour naming” (naming colour of ink of coloured rectangles) and “word reading” (reading colour words printed in black ink) subtests serve as control conditions. The main ‘Stroop’ test is the “inhibition” subtest, which presents the names of colour words such as red, green and blue on the page printed in an incongruent colour of ink. For example, the word red is printed in blue ink. The patient has to name the colour of the ink as fast as they can. This requires inhibition of the more habitual and prepotent response of reading the word. Executive functions employed in this subtest are response inhibition, cognitive control and flexibility (30). The final inhibition/switching condition also involves colour words printed in incongruent ink and requires switching between naming the colour of ink they are printed in and reading the word when the word is surrounded by a box. This condition has the additional requirement of switching between two task sets. The Trail Making Test includes five conditions (30): Visual scanning, number sequencing, letter sequencing, number and letter switching and motor speed. In the visual scanning subtest, the participants were shown letters and numbers and asked to strike off all “3 s” presented in a group of letters and other numbers. In the number sequencing subtest, the participants were asked to join all the numbers in progressive order ignoring the letters. In the letter sequencing subtest, the participants were asked to connect all the letters in alphabetical order and to ignore the numbers. In the number-letter switching subtest, the participants were asked to join numbers and letters in two alternating sequences of a number and then a letter (eg 1-A-2-B-3-C … ). The motor speed test required the participants to be as quick as possible to trace dashed lines connecting circles. The executive functions believed to be important for the successful completion of this test are: Behavioural regulation, cognitive flexibility and inhibition of perseverative responding.
Attention
The Paced Auditory Serial Addition Test (PASAT) (31) involves presentation via a tape recorder of a series of 31 numbers between 1 and 9. The patient is required to add each number read out to the immediately preceding one and say out their sum. Performance engages working memory and sustained attention. The percent correct is calculated.
Depression and anxiety
The Beck Depression Inventory (BDI-II) (32) is a self-report inventory assessing the severity of depression with regard to the cognitive, affective, somatic, or behavioural symptoms. Scores range from 0 to 63, with higher scores denoting higher depression. The Hospital Anxiety and Depression Rating Scale (HADS) (33) is a self-report measure assessing depression and anxiety. The sum of items in each subscale represents a total score indicating global anxiety (HADS-A) or depression (HADS-D). On both Depression and Anxiety subscales scores range from 0 to 21, with higher scores indicating more severe depression or anxiety and scores above 11 considered ‘caseness’.
Hopelessness
The Beck Hopelessness Scale (BHS) (34) is a self-report measure of three major aspects of hopelessness: Feelings about the future, loss of motivation, and expectations. The sum of items ranges from 0 to 20, and higher scores are indicative of higher hopelessness.
Apathy
The Starkstein Apathy Scale (SAS) was used to assess reduction of interest, motivation, affective responsivity, and engagement in goal-directed behaviours. The SAS consists of 14 items that are answered on a four point Likert scale (scores: “not at all”, “slightly”, “some”, “a lot”). The total scores range from 0-42 and greater scores indicate more severe apathy (35).
Pain experience and behaviour
The McGill Pain Questionnaire (MPQ) (36) contains 78 pain descriptors assigned to three categories of pain qualities: Sensory, affective, and evaluative. There is also a miscellaneous category of pain-related words. The Pain Behaviour Checklist (PBC) (37) is a self-report assessment to quantify three classes of pain behaviours: Help seeking, avoidance, and complaint.
Quality of Life
The Short Form-36 (SF-36) is a 36-item questionnaire which measures Quality of Life (QoL) across eight domains (physical and social functioning, physical and emotional role limitations, mental health, energy, pain, and general health perceptions). Eight different subscores, and a physical and mental summary score, can be derived. The maximum score ranges from 0 (lowest or worst possible level of functioning) to 100 (indicates the best possible health state).
Statistical analysis
All data were analysed using the computing environment R (38). The data for all tests were first assessed for presence of outliers, the parametric assumptions of normality (Shapiro-Wilk Test), and homogeneity of variance (Bartlett’s Test). When the assumptions for parametric analysis were met, paired samples t-tests were used to compare cognitive, mood, pain and quality of life measurements before and after VTA-DBS. When the assumptions were violated, nonparametric Wilcoxon signed ranks test was used and the median was given instead of the mean. To protect against Type I error, for each outcome measure domain (Cognitive, Mood, Pain and Quality of Life) a Bonferroni correction was applied. Thus the corrected p-value was used for determination of significance of the results in each domain. The corrected p-value for the measures of cognitive function is p = .001 (that is αaltered = .05/30 = .001). For the measures of Mood, the corrected p = .01; for Pain-related Behaviours it is p = .006; for the Quality of Life measure it is p = .006. Furthermore, to determine if any of the post-operative changes were statistically reliable, we calculated the reliable change index (RCI). RCI verifies if variation in a score is statistically strong and reliable (39). The formula for calculating it is:
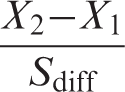
That is, mean of post operation score minus mean of pre operation score divided by the standard error of the difference. The formula for calculating the standard error of difference is:
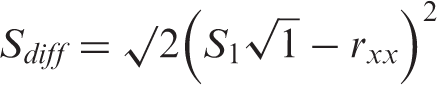
The RCI cut off is 1.96, RCI < −1.96 are considered a reliable decline, RCI > + 1.96 are considered a reliable improvement, RCI between −1.96 and + 1.96 are not considered as reflecting reliable change. We also calculated the confidence intervals of reliable changes for specific measures. The formula for calculating this is:
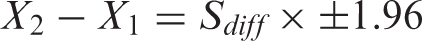
Based on this score, the results are then regrouped into three classes representing the percentages of the participants with reliable decline, no change and reliable improvement.
Results
Headache frequency, severity and load before and after surgery
Means and standard deviations for the measures of cognitive function before and after VTA-DBS and reliable change indices (RCI) showing percent with decline, no change and improvement on the cognitive measures.
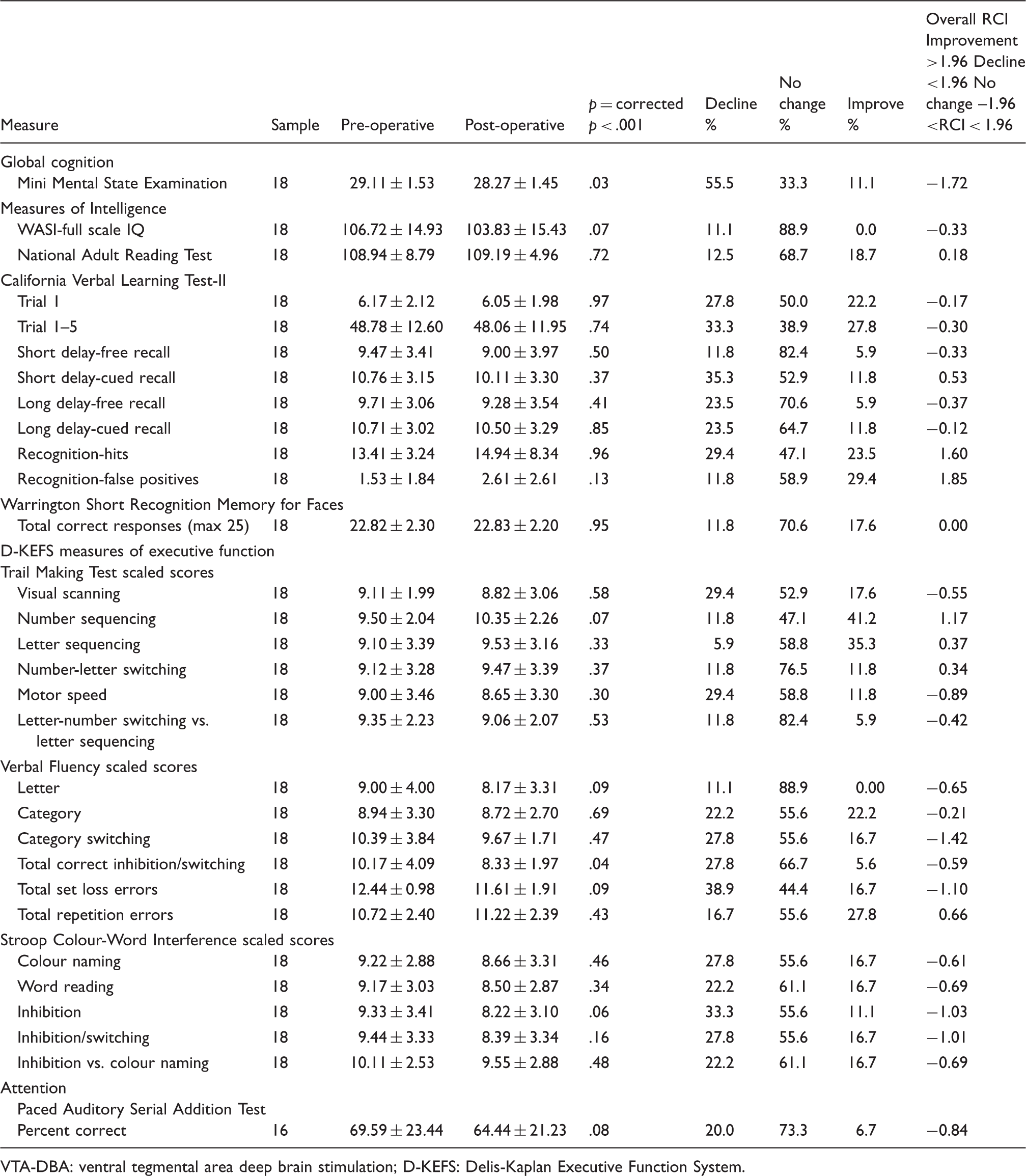
VTA-DBA: ventral tegmental area deep brain stimulation; D-KEFS: Delis-Kaplan Executive Function System.
Means and standard deviations for the measures of mood, pain, disability and quality of life before and after VTA-DBS surgery.
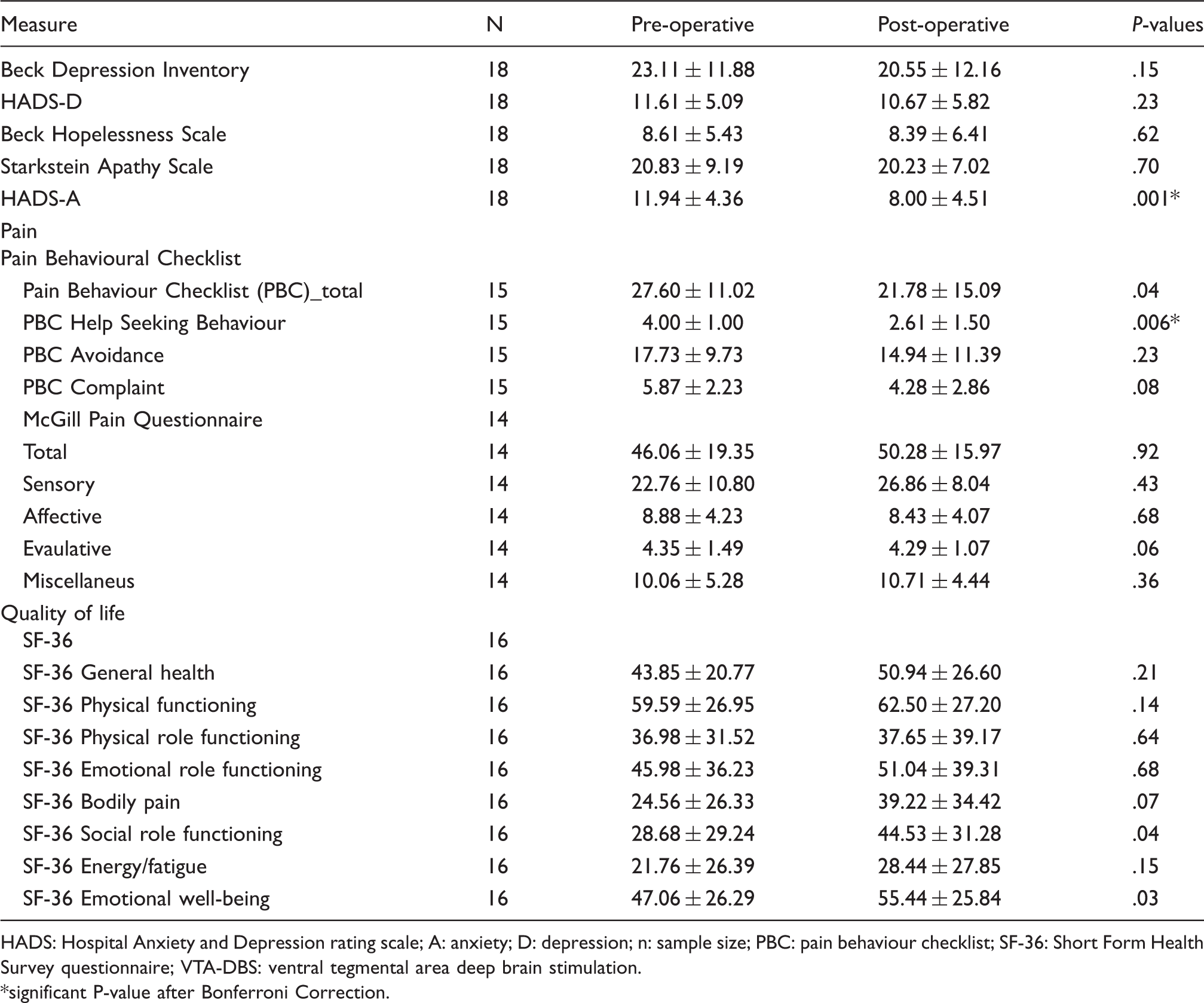
HADS: Hospital Anxiety and Depression rating scale; A: anxiety; D: depression; n: sample size; PBC: pain behaviour checklist; SF-36: Short Form Health Survey questionnaire; VTA-DBS: ventral tegmental area deep brain stimulation. *significant P-value after Bonferroni Correction.
Cognitive function before and after VTA-DBS
Measures of global cognition and intelligence
Similar to the pre-operative MMSE score, the group median MMSE score at follow-up was also within the normal range. With the corrected p-value of .001, the differences between pre and post DBS surgery in the MMSE scores were not significant. There were no significant differences between pre- and post-DBS measures of the WASI estimate of current Full Scale IQ. There were no significant differences between pre- and postoperative measures of estimated premorbid IQ (NART), and between current and estimated premorbid IQ either before (p = .57) or after (p = .38) surgery.
Executive functions and inhibition
The pre- and post-DBS scores on the measures of executive function on the Trail-making, Stroop and verbal fluency tasks are presented in Table 2. The scaled score for the total correct category switches on the alternating category VF test was lower after (M = 8.33, SD = 1.97) than before (M = 10.17, SD = 4.09) surgery, but this change was not significant at the corrected p value of p < .001. The reliable change index (RCI = −0.59) also indicated that this was not a reliable change. There were no other significant differences between pre- and post-measures on the D-KEFS subtests of the Trail-Making Test, verbal fluency or Stroop Colour-Word interference.
Memory
As evident from Table 2, there were no significant differences between pre and post VTA-DBS measures derived from the CVLT-II or on the short Recognition Memory for Faces (all p > .001).
Pain experience and behaviour before and after VTA-DBS
After Bonferroni correction, there were no significant differences on the Pain-Behaviour Checklist (PBC) total scores after (M = 21.78, SD = 15.09) compared to before (M = 27.60, SD = 11.02), t(14) = 2.17, p = .047, r = .50 surgery. However, the PBC Help Seeking behaviours were significantly reduced after (M = 2.61, SD = 1.50) compared to before (M = 4.00, SD = 1.00), t(14) = 4.36, p < .001, r = .76 surgery, suggesting less engagement in help-seeking behaviours for their CH pain from patients after surgery (Figure 1(a)). The reliable change index (RCI = −4.43) indicates that this change is reliable. At post-surgical follow-up, a larger proportion of the participants showed improvement (80%) than no change (13%) or decline (6%) in help-seeking behaviours. The avoidance and complaints subscales of the PBC were not significantly altered by DBS. There were no statistically significant differences between pre- (M = 46.06, SD = 19.35) and post-surgery (M = 50.28, SD = 15.97), t(12) = 0.10, p = 0.92 scores on the McGill Pain Questionnaire or any of the sensory, evaluative, affective or miscellaneous subscores (see Table 3).
Boxplots showing the distribution of values before and after VTA-DBS for (a) Pain Behaviour Checklist Help Seeking Behaviours, (b) Hospital Anxiety and Depression Scale, Anxiety subscale. Black dots represent patients’ raw scores. The solid line represents zero change. Values above the top dotted line represent a reliable increase in scores from baseline to follow-up after VTA-DBS. Values below the lower dotted line represent a reliable decrease in scores from baseline to follow-up after VTA-DBS, values between the dotted lines represent no reliable change.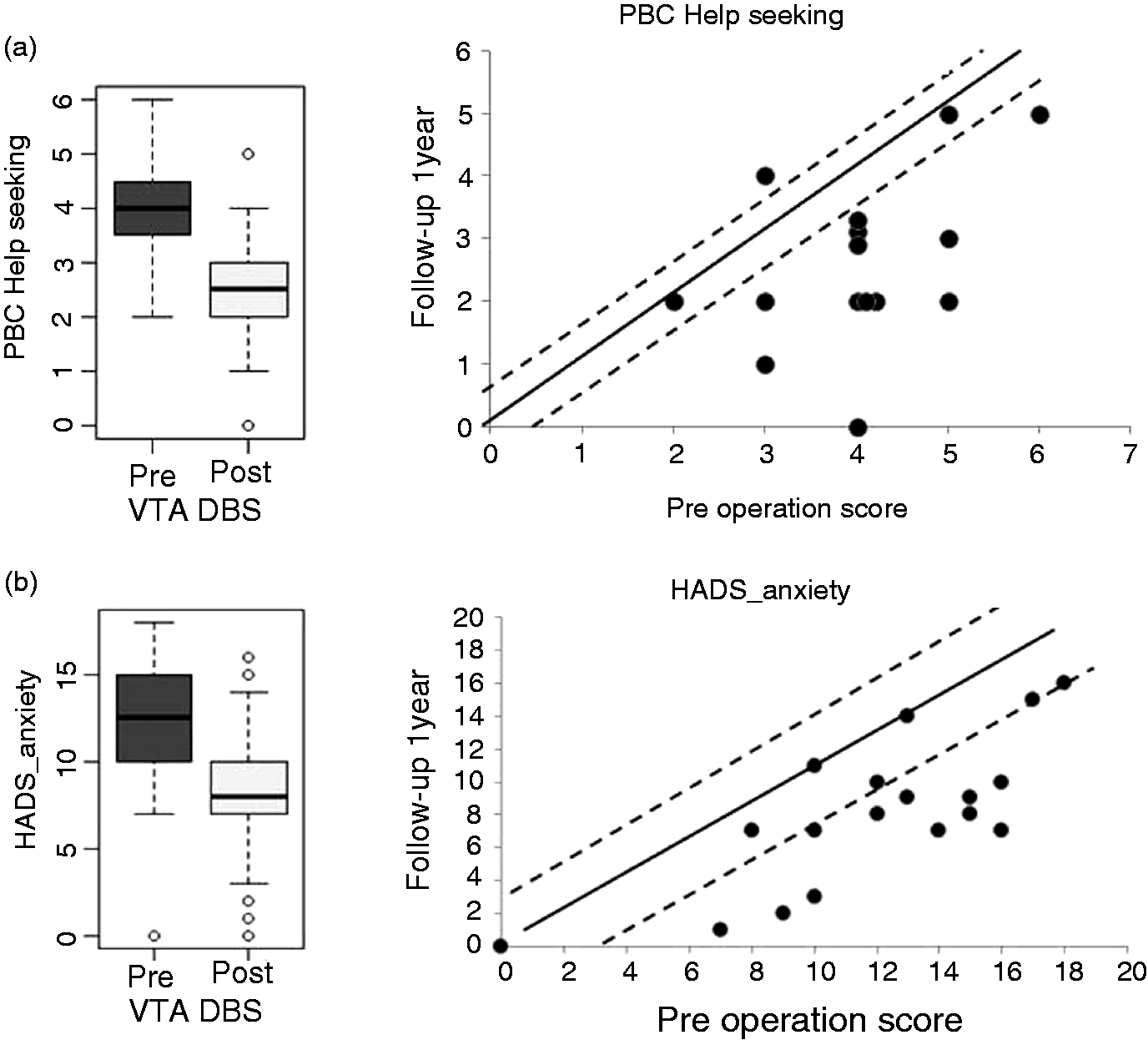
Measures of mood and quality of life before and after VTA-DBS
After Bonferroni correction, on the HADS-A scale, self-reported anxiety levels were significantly reduced after (M = 8.00, SD = 4.51) compared to before VTA-DBS (M = 11.94, SD = 4.36), t(17) = 5.43, p < .001, r = .79. (Figure 1(b)). The reliable change index (RCI = −2.86) indicated a reliable reduction of anxiety. At follow up, 61% of patients had a reliable reduction of anxiety, 39% had no change and no patient had an increase of anxiety. While the means suggest some improvement in depression after surgery, there were no statistically significant differences between pre (M = 23.11, SD = 11.88) and post (M = 20.55, SD = 12.16), t(17) = 1.50, p = .15 surgery measures of the Beck Depression Inventory. Similarly, there were no statistically significant differences between pre (M = 11.61, SD = 5.09) and post (M = 10.67, SD = 5.82) surgery scores on the HADS-D, t(17) = 1.22, p = .23. Scores on the Hopelessness Scale and the Starkstein Apathy Scale were not altered by surgery (see Table 3 for the pre and post-operative means). On the SF36 measure of quality of life, the score on the Social Role Functioning subscale was higher after (median = 43.75) compared to before (median = 25.00), indicating better social functioning of patients after VTA-DBS; however, after Bonferroni correction this was not significant. Similarly, on the SF36, the score on the emotional wellbeing subscale was higher after (M = 55.44) compared to before (M = 47.06), surgery, indicating greater emotional wellbeing of patients after VTA-DBS; however, after Bonferroni correction the improvement was not significant. Nevertheless, at follow up 46 % of the patients had better social functioning.
Correlational analysis
Pearson correlational analyses were performed to explore the relationship between the change scores in Headache Load (before and after VTA-DBS) and change scores in mood, pain experience and behaviour, and quality of life (listed in Table 3) and cognitive measures (listed in Table 2). Variations in anxiety levels correlated positively and significantly with changes in Headache Load (r = 0.57, p < .01). None of the other correlations were of a notable magnitude or significant.
Discussion
A previous clinical study provided evidence for the positive impact of VTA-DBS on CH severity and frequency (24). In particular, Akram and colleagues demonstrated a significant decrease in headache frequency, severity and load after surgery, with an associated relative decrease of triptan medication intake. However, to our knowledge, the influence of VTA-DBS on measures of cognition, mood, pain experience and behaviour, and quality of life has not been previously examined in detail, which was the aim of this study. We found that VTA-DBS produced clinical benefits on CH frequency, severity and load and produced no adverse effects on cognitive function, and was associated with significant decrease of anxiety and significant improvement of pain-related help-seeking behaviours, indicating better coping with pain after surgery.
Akram and colleagues defined the target area for the DBS electrode as the ventral tegmental area (VTA). The VTA is the most anterior aspect of the midbrain tegmentum and encloses distinct types of neuronal cells. However, it is mainly distinguished by its abundant number of dopamine (DA) neurons.Together with the substantia nigra, the VTA is considered to be a main dopaminergic territory in the brain (40). Projections of dopaminergic neurons start from the VTA to reach various regions of the cortex through two main routes: The mesocortical and the mesolimbic dopaminergic pathways. The former transmits information from the VTA to prefrontal, orbitofrontal and cingulate cortices. The dorsolateral prefrontal cortex (DLPFC) (41,42), receives dopaminergic signals from VTA and it has been characterised to be an important node for a range of cognitive funtions such as working memory (43,44), behavioural flexibility and inhibitory control of willed actions (45,46), aspects of social cognition, emotional regulation, memory retrieval (47) and implicit temporal processing (48–50). Thus, dopaminergic projections to the forebrain, including the frontal lobes and the dorsal and ventral striatum, constitute a fundamental part of the neural circuits underlying a variety of cognitive and executive functions. A recent patch clamp electrophysiology study in rats demonstrated the functional importance of VTA terminals to driving intrinsic inhibition in the PFC (51). The mesolimbic path projects from the VTA to reach different limbic areas. In particular, there are numerous connections with the nucleus accumbens (NAc). Among the functions that have been attributed to this mesolimbic dopaminergic circuit, there is the regulation of the reward system (52). It has been shown that VTA dopamine neurons are responsive to anticipation of time of reward, unpleasant or new stimuli (53–55). Recently, a computational model of the afferents to the VTA that replicates many of the experimental observations has been proposed (56). In light of the connectivity of the VTA with the prefrontal cortex, in the present study we administered subtests of the Delis–Kaplan Executive Function System (D-KEFS) (30), a set of tests to assess verbal and nonverbal executive functions before and after VTA-DBS to test for any possible effects on ‘prefrontal’ executive functions. The results showed that VTA-DBS surgery did not significantly alter any of the measures of executive function included.
Effect of VTA-DBS on cognitive function
Cognitive function in CH has been examined in previous studies. While some have reported impairments in verbal memory in CH (57), others did not find any significant differences in memory or executive function in CH patients relative to healthy controls (58). In a previous study by our group, Torkamani et al. (2) found no impairment in IQ, verbal memory or executive functions in patients with chronic or episodic CH relative to matched healthy controls and only deficits in working memory and self-reported cognitive failures were documented. Other evidence has suggested that cognitive performance in CH is altered only during headache attacks (59), due to the fact that pain can also make demands on attentional resources (60). In this study, we examined the impact of VTA-DBS on global cognitive function and more specific cognitive domains of intelligence, executive function, memory and attention. The effective site of the DBS for CH in the ipsilateral hypothalamus was shown to have high probability connectivity to the frontal cortex (21,61). In a similar vein, it is likely that stimulation through electrode contacts located in the VTA modulates distant neuronal dynamics through its electrical and chemical influences. However, we observed no significant cognitive changes in executive function after VTA-DBS (see Table 2). Previous research (2) showed no major deficits on tests of executive function in CH patients relative to healthy controls, including the Stroop and verbal fluency tests also employed here, and the results of the present study provide unique data that indicate that surgical implantation of electrodes and chronic stimulation of the VTA is generally safe from a cognitive perspective.
Effect of VTA-DBS on pain experience and behaviour, mood and quality of life
The impact of CH on mood, daily functioning and QoL has been measured in a number of previous studies. These have reported that patients with CH had elevated levels of depression, suicidal ideation, anxiety, hopelessness, and increased disability and poorer QoL relative to healthy controls (2,11,57,62–64). Anxiety symptoms, as measured by the HADS-A, significantly improved after DBS-VTA surgery. The RCI confirmed these changes showing that one year postoperatively the majority of patients showed improvement (61%) and none of the participants showed worsening of anxiety after VTA-DBS. It is difficult to determine whether this beneficial effect on anxiety is generated by VTA stimulation directly altering the activity in the mesolimbic circuits, or indirectly because VTA-DBS reduced headache frequency and severity. In current experimental paradigms, anxiety has been operationalized as an emotional response to potential threats. VTA is a crucial region in the anxiety circuit and receives both excitatory glutamatergic and inhibitory GABAergic inputs from the ventral bed nucleus stria terminalis (vBNST). The current concept is that two parallel vBNST-to-VTA pathways mediate either anxiogenic or anxiolytic behavioural response. Specifically, activation of glutamatergic vBNST inputs to the VTA increased anxiety and induced avoidance, whereas activation of GABAergic inputs enhanced anxiolytic effects (65). Therefore, a possible explanation for the reduction of anxiety following VTA-DBS might be that the stimulation disrupts the glutamatergic inputs from vBNST. Another possibility is that stimulating VTA leads to a similar effect to the GABAergic inputs that produce anxiolytic effects. A second explanation is that anxiety reduction is secondarily related to reduced headache frequency and severity. Further work is required to establish the mechanisms of the reduction of anxiety following VTA-DBS.
On the Pain Behaviour Checklist, the help-seeking behaviours were significantly reduced after VTA-DBS compared to before surgery, suggestive of better adjustment and coping with pain. The RCI endorsed these changes, revealing that more patients showed improvement (80%) than deterioration (13 %) or no change (6%) on the Pain Behaviour Checklist after VTA-DBS. However, the changes in the severity or qualitative aspects of pain experience on the McGill Pain Questionnaire were not altered following VTA-DBS. Hopelessness and apathy were also not significantly altered by surgery. Depression showed a trend toward reduction (pre-operative, M = 23.11, post-operative, M = 20.55) but was not significantly changed by surgery. With VTA-DBS, the social role functioning and emotional wellbeing components of QoL on the SF36 improved, albeit non-significantly, after Bonferroni correction.
A major limitation of this study is the small sample size. However, given the infrequency of the VTA-DBS procedure for refractory CH, the current sample is satisfactory. A second limitation is that multiple comparisons were conducted, which increases the risk of type 1 error. To overcome this limitation, we conducted a Bonferroni correction. Despite these limitations, the study provides new information about DBS-VTA safety and efficacy. In conclusion, VTA-DBS did not produce any adverse cognitive effects on the measures of cognition assessed and was associated with significant improvement of anxiety and help-seeking pain-related behaviours and non-significant improvement of the social role functioning and emotional wellbeing aspects of quality of life one year after surgery in our sample of CH patients.
Article highlights
Deep brain stimulation of the ventral tegmental area (VTA-DBS) in patients with medically refractory chronic cluster headache (CH) did not induce any significant adverse cognitive effects. There was significant improvement of anxiety symptoms after VTA-DBS. There was significant reduction of help seeking pain-related behaviours after VTA-DBS, suggestive of better adjustment and coping with pain after surgery.
Footnotes
Acknowledgements
The Unit of Functional Neurosurgery is supported by the Parkinson Appeal UK and the Monument Trust.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: AR was funded by 3 + 1 PhD studentship, Queen Mary College, London University; HA was funded by the Brain Research Trust.
