Abstract
Background
*These authors are shared first authors.
Methods
Consecutive patients with CH and probable cluster headache (PCH) were prospectively recruited from 15 hospitals. We analysed the validity of the revised ICHD-3 criteria for CH against the beta version of the third edition of the ICHD (ICHD-3β).
Results
In total, 193 patients were enrolled: 140 (72.5%), 5 (2.6%) and 22 (11.4%) had episodic cluster headache (ECH), CCH, and PCH, respectively. The remaining 26 (13.5%) had CH with undetermined remission periods. One patient with ECH and one with PCH had only forehead and facial flushing and were diagnosed with PCH and non-cluster headache, respectively, according to the ICHD-3. Four participants with ECH according to the ICHD-3β had remission periods of > 1 month and between 1 and 3 months and were newly diagnosed with CCH according to the ICHD-3.
Conclusion
The change from ICHD-3β to ICHD-3 resulted in few differences in the diagnoses of CH and PCH.
Keywords
Introduction
Cluster headache (CH) is the most prevalent trigeminal autonomic cephalalgia, typically characterised by recurrent unilateral severe headache accompanied by ipsilateral autonomic features (1). Owing to the pain it causes, patients with CH usually experience a significant amount of distress and decrease in quality of life (2–4). Recent advances in the treatment of CH, however, have made it possible to better treat CH (5–11). Therefore, accurate diagnosis of CH is an important step to reduce the burden of CH.
Currently, there is no reliable biological marker of CH. A diagnosis of CH is made based on the patient's history and clinical presentation according to five diagnostic criteria of CH listed in the International Classification of Headache Disorders since its first edition: Total attack number, severe unilateral pain lasting 15–180 minutes, accompanying symptoms, frequency of attack, and not better accounted for by other disorders (1,12–14). The third edition of the International Classification of Headache Disorders (ICHD-3) was recently published and removed forehead and facial flushing and the sensation of fullness in the ear from the diagnostic criteria of CH (1); these two accompanying symptoms were first introduced in the beta version of the third edition of the International Classification of Headache Disorders (ICHD-3β) (14). Further, the ICHD-3 revised the time window of the remission period of chronic cluster headache (CCH) from less than one month to less than 3 months. Such changes underscore the need to validate the diagnostic criteria of CH in the ICHD-3. The present study aimed to establish this validation by using data from the Korean Cluster Headache Registry (KCHR), which includes patients with CH and probable cluster headache (PCH).
Methods
Study and participants
The KCHR study is a prospective multicentre registry study that consecutively enrolled patients with CH and PCH from 15 hospitals in Korea from September 2016 to July 2018. We included episodic cluster headache (ECH), chronic cluster headache (CCH) and probable cluster headache (PCH) based on the third edition beta version of the International Classification of Headache Disorder (ICHD-3β). Inclusion criteria were: a) Having CHs; b) age ≥ 19 years, and c) full understanding and agreement to the study protocol. Exclusion criteria were: a) inability to communicate in the Korean language, b) current enrolment in other clinical studies and c) an investigator's judgment of cognitive or psychological difficulty to complete the questionnaire. Informed consent was obtained from all participants. A detailed description of the study process was previously described (15). This study was approved by the Institutional Review Board of 15 participating hospitals. All participants provided written informed consent before entering the study. All clinical investigations were conducted according to the principles expressed in the Declaration of Helsinki.
Case definition of CH according to ICHD-3β
The diagnosis of ECH, CCH and PCH were based on the ICHD-3β criteria. Diagnoses of ECH (code 3.1.1) and CCH (code 3.1.2) were based on criteria A to E of CH (code 3.1): (A) At least five attacks fulfilling criteria B–D; (B) severe or very severe unilateral orbital, supraorbital and/or temporal pain lasting 15–180 minutes when untreated; (C) either or both of the following, 1. at least one of the following symptoms or signs ipsilateral to the headache: Conjunctival injection and/or lacrimation, nasal congestion and/or rhinorrhoea, eyelid oedema, forehead and facial sweating, forehead and facial flushing, sensation of fullness in the ear and miosis and/or ptosis; or 2. a sense of restlessness or agitation; (D) attacks have a frequency of between one every other day and eight per day for more than half of the time when the disorder is active and (E) not better accounted for by another ICHD-3β diagnosis. If a headache fulfilled criteria A–E of CH and had been occurring for more than 1 year without remission, or with remission periods lasting less than 1 month, the diagnosis of CCH was rendered. If a headache fulfilled criteria A–E of CH and was not classified as CCH, the diagnosis of ECH was rendered. If a participant's CH was his/her first episode and persisted, it could not be diagnosed as ECH or CCH and was classified as CH (code 3.1).
Diagnosis of PCH was based on criteria A to C of probable trigeminal autonomic cephalalgia according to the ICHD-3 β (code 3.5.1): (A) headache attacks fulfilling all but one of criteria A–D for 3.1 CH; (B) not fulfilling ICHD-3β criteria for any other headache disorder and (C) not better accounted for by another ICHD-3β diagnosis. If a participant's headache fulfilled all but one of criteria A–E for CH (code 3.1), they were classified as having PCH (code 3.5.1).
Case definition of CH according to ICHD-3
Diagnoses of ECH (code 3.1.1 in ICHD-3) and CCH (code 3.1.2 in ICHD-3) were based on criteria A to E of ICHD-3. Criteria A, B, D and E are the same as those of ICHD-3β. Criterion C is as follows: Either or both of the following. 1. At least one of the following symptoms or signs ipsilateral to the headache: Conjunctival injection and/or lacrimation, nasal congestion and/or rhinorrhoea, eyelid oedema, forehead and facial sweating, and miosis and/or ptosis. 2. A sense of restlessness or agitation. If a headache fulfilled criteria A–E of CH and occurred for more than 1 year without remission, or with a period of remission lasting <3 months, the diagnosis of CCH was rendered. If a headache fulfilled criteria A–E of CH and was not classified as CCH, the diagnosis of ECH was rendered.
Diagnosis of PCH in the ICHD-3 was based on criteria A to C of probable trigeminal autonomic cephalalgia in the ICHD-3 (the same as ICHD-3β). If a headache fulfilled all but one of criteria A–E of CH (code 3.1 in ICHD-3), they were classified as having PCH (code 3.5.1).
Validation of the ICHD-3 diagnostic criteria of CH
We compared the performances of each criterion of CHs as defined by the ICHD-3 and the ICHD-3β by using the data of the KCHR study. Evaluation of performance was conducted in two stages according to the main changes in the diagnostic criteria of CH from the ICHD-3β to the ICHD-3: Accompanying symptoms and time window for remission period of CCH.
To assess the change in accompanying symptoms in the ICHD-3, we included participants with ECH, CCH and PCH. We also included patients with CH who could not be identified as ECH or CCH on account of their having had only one CH episode and consequently an undetermined period of remission.
To validate the change in the remission period of CCH in the ICHD-3, we included participants with ECH, CCH and PCH. The remission periods were assessed in patients with recurrent episodes or 1 year or more of follow-up. We did not include participants with CH (code 3.1) who could not be identified as either ECH or CCH.
Analysis
The distribution of normality was evaluated by the Kolmogorov-Smirnov test. After ensuring the normality of the data's distribution, Student's t-tests or analyses of variance were adopted for analysing the difference of continuous variables. If normality was not confirmed, Mann-Whitney U tests or Kruskal-Wallis tests were used. For comparing categorical variables, we used Chi-square tests. Statistical significance was set to p < 0.05. The Statistical Package for Social Sciences version 23.0 (SPSS 23.0; IBM, Armonk, NY, USA) was utilised for all statistical analysis.
Results
Participants
A total of 213 participants with CH and PCH were invited; 193 were finally enrolled in the present study during the study period. According to the ICHD-3β, 140 (72.5%), 5 (2.6%) and 22 (11.4%) participants were classified as having ECH (code 3.1.1), CCH (code 3.1.2) and PCH (code 3.5.1), respectively. The remaining 26 participants who met the criteria of CH but had the first episode of CH without a determined remission period were classified as having CH (code 3.1) (Figure 1). Reasons for the diagnosis of PCH were as follows: three (13.6%) for total attack number; 10 (45.4%) for typical duration of attack (seven [31.8%] for longer attack duration and three [13.6%] for shorter attack duration), one (4.5%) for headache intensity, one (4.5%) for unilateral pain, one (4.5%) for location of pain, three (13.6%) for accompanying symptoms, and three (13.6%) for lower attack frequency. Distribution and clinical characteristics of participants are summarised in Table 1.
Distribution of diagnostic codes of participants according to ICHD-3β and ICHD-3. Demographics and clinical characteristics of patients diagnosed according to the ICHD-3β. +Represented as mean ± standard deviation. ICHD-3β, the third edition beta version of the International Classification of Headache Disorders; all CH: cluster headaches (code 3.1, 3.1.1, 3.1.2, 3.5.1); ECH: episodic cluster headache (code 3.1.1); CCH: chronic cluster headache (code 3.1.2); PCH: probable cluster headache (code 3.5.1).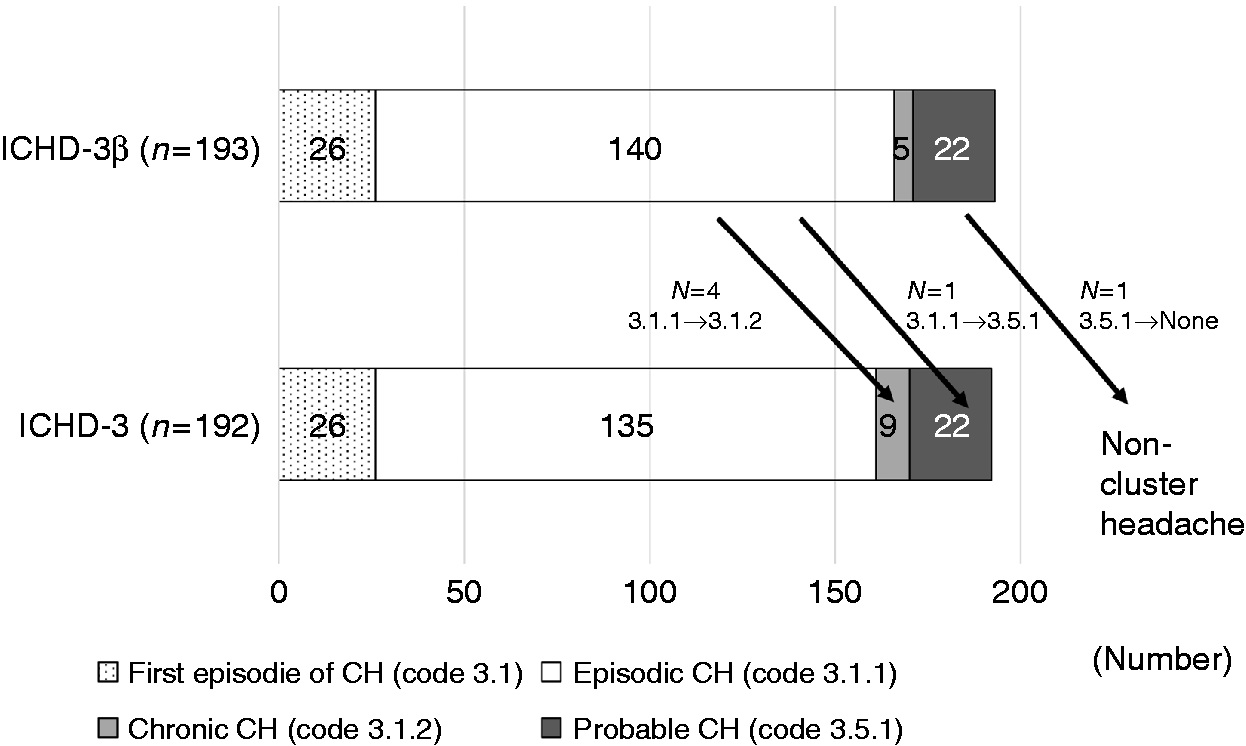

Of the 193 participants, 192 participants were also classified as having CH or PCH and one participant was classified as having non-cluster headache according to the ICHD-3. Among the 192 participants with CH or PCH, 135 (70.3%), nine (4.7%) and 22 (11.5%) were classified as having ECH (code 3.1.1), CCH (code 3.1.2) and PCH (code 3.5.1) according to ICHD-3, respectively. The remaining 26 (13.5%) were classified as having CH (code 3.1, Figure 1).
Validation of criterion for accompanying symptoms
Distribution of accompanying symptoms according to cluster headache subtypes based on the ICHD-3β.

ICHD-3β: third edition beta version of the International Classification of Headache Disorders; All CH: cluster headache (code 3.1, 3.1.1, 3.1.2, 3.5.1); DCH: definite cluster headache (code 3.1, 3.1.1, 3.1.2); ECH: episodic cluster headache (code 3.1.1); CCH: chronic cluster headache (code 3.1.2); PCH: probable cluster headache (code 3.5.1).
Validation of criterion for remission period of CCH
Distribution of the available remission periods of 156 participants.
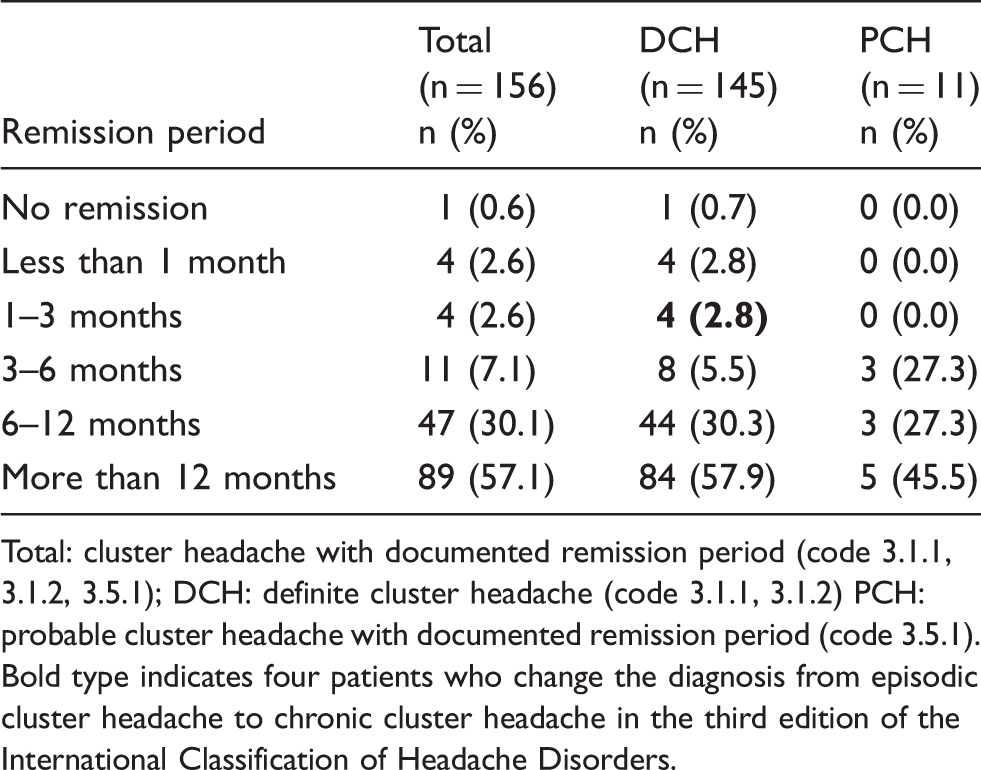
Total: cluster headache with documented remission period (code 3.1.1, 3.1.2, 3.5.1); DCH: definite cluster headache (code 3.1.1, 3.1.2) PCH: probable cluster headache with documented remission period (code 3.5.1). Bold type indicates four patients who change the diagnosis from episodic cluster headache to chronic cluster headache in the third edition of the International Classification of Headache Disorders.
Clinical characteristics of participants with CH and PCH
Comparison of demographic and clinical characteristics between patients with episodic cluster headache (ECH) and patients with chronic cluster headache (CCH) according to the ICHD-3.

+Represented as mean ± standard deviation.
ICHD-3: third edition of the International Classification of Headache Disorders; ECH: episodic cluster headache (code 3.1.1); CCH: chronic cluster headache (code 3.1.2); VAS: visual analogue scale.
Mann-Whitney test.
Discussion
The key findings of the present study are as follows: (a) The ICHD-3β and the ICHD-3 informed the same diagnosis of CH and PCH for most participants; (b) diagnosis of headache changed owing to revised criteria for accompanying symptoms in the ICHD-3 for a small proportion of the participants with CH and PCH; and (c) the number of CCH diagnoses increased owing to the new criterion for the remission period in the ICHD-3.
Detection of CCH is important to alleviate the significant burden that CCH imposes on daily activity (16,17). Although there is disagreement regarding the relative efficacy of therapeutic interventions addressing the two disorders, acute treatment of oxygen and non-invasive vagal nerve stimulation are considered to be less effective in attenuating CCH than ECH (9,10,18). Kudrow reported greater benefit of oxygen inhalation therapy among ECH patients using a facial mask at a rate of 7 L/min than CCH patients, although not using evidence-based 12–15 L/min via non-rebreather mask (18,19). Prophylactic treatment of CCH also features diminished success than when prescribed to treat ECH by the administration of lithium, verapamil and melatonin (7,17,20,21). Prophylactic treatment by sphenopalatine ganglia stimulation is only used to treat CCH (11). The present study showed that patients with CCH and ECH diagnosed based on the ICHD-3 differed in demographic and clinical characteristics (Supplemental Table 1).
Attempts have therefore been made to increase the sensitivity and specificity of the diagnostic criteria of headache disorders. As a part of this effort, two additional autonomic symptoms – forehead and facial flushing, and sensation of fullness in the ear – were added to the criterion for accompanying symptoms in ICHD-3β. The present study showed that of the 171 participants with CH, 29 (17.0%) and 16 (9.4%) had forehead and facial flushing, and sensation of ear fullness, respectively. Among the 22 participants with PCH, two (9.1%) and two (9.1%) patients exhibited forehead and facial flushing, and sensation of fullness in the ear, respectively. Though these two symptoms presented in a significant proportion of individuals with CH, they usually presented with the other already established accompanying symptoms in patients with CH and did not improve diagnostic accuracy (22); moreover, although the observed frequency of sensation of fullness in the ear was lower than that found by a previous study (33.5% vs. 9.4%), both studies demonstrated no improvement in diagnostic accuracy by considering the two aforementioned accompanying symptoms. Hence, the omission of forehead and facial flushing and sensation of fullness in the ear symptoms from the ICHD-3 did not diminish the sensitivity or specificity of CH diagnosis.
The first diagnostic criterion in the first edition of the International Classification of Headache Disorders defined CCH as attacks occurring for more than 1 year without remission or with remission lasting less than 14 days (23). The remission period for CCH increased in ICHD-2 and ICHD-3β as lasting < 1 month for at least one year (13,14). In ICHD-3, the remission period further increased to lasting <3 months (1). Accordingly, the number of CCH diagnoses based on ICHD-3 increased remarkably relative to the number diagnosed based on the ICHD-3β. The present study demonstrated that demographic and clinical characteristics between patients with CCH and a remission period of <1 month and those with a remission period between 1 month and <3 months were not different. The similarity of demographics and clinical characteristics between the two periods and the lack of significant differences in key clinical characteristics between ECH and CCH suggests that the remission period of CCH was well revised. Further studies on the differences in acute and preventive treatment response and impairment of daily activity between ECH and CCH, the diagnoses of which are based on the ICHD-3, will be needed to validate the suitability of the remission period of CCH in ICHD-3.
Extending the criteria of the remission period, however, might influence the duration and frequency of the cluster period. Some ECH patients were reported to have recurrent cluster attacks during the period of >1 week, especially when drinking alcohol (24). This suggests the possibility of a so-called “mini-bout of 1 week” that might not be an isolated period but rather a less active time during the same cluster period defined by the ICHD-3 (25,26). Practically, it is difficult to determine when a cluster period ends or when to stop preventive medications without waiting 3 months or for the provocation by calcitonin gene-related peptide (27). The duration and frequency of the cluster period in ECH require evaluation with a prospective headache diary based on ICHD-3.
This paper was subject to several limitations. First, this study used the data of the Korean Cluster Headache Registry, which includes only Korean CCH patients. Previous studies in Asian countries revealed a lower frequency of CCH, restlessness, or aura (28–31); the present study confirmed this observation. Therefore, the demographics and clinical profiles identified by the present study could differ from those of Western countries. Second, there was a relatively small number of PCH and CCH patients. Although we tried to enrol all patients with CCH and PCH in 15 headache clinics over the course of 2 years, the number for PCH and CCH cases might have yielded insufficient results in some subgroup analyses. In other words, the statistical significance was possibly compromised by the limited sample size. Third, this study is cross-sectional and therefore did not investigate the validity of CH diagnosis in a longitudinal design. Therefore, the CH diagnosis may change after follow-up.
The present study featured several strengths. First, this is the first report that attempted to validate the diagnostic criteria of CH in ICHD-3 based on the prospective collections of patient information by headache experts. Second, the present study enrolled a relatively large number of CH patients considering the short period of its scope. The estimated 1-year prevalence of CCH ranged from 10–150 per 100000 (32). Although several CH studies in Western countries observed a higher number of CH patients than did the present study, our investigation has the largest number of participants compared to previous studies performed in Asian countries (28,30,33,34). Third, our study included PCH participants in addition to CH participants and identified a significant number of the former: Primary cluster-like headache that fulfilled all but one criterion of CH.
Clinical implications
The revised criteria in the ICHD-3 for accompanying symptoms changed headache diagnoses in a small proportion of participants with CCH and PCH. The number of CCH cases increased on account of the revised remission period in the ICHD-3. There were no differences in clinical characteristics between CCH participants with a remission period of < 1 month and those with a remission period between 1 month and < 3 months.
Supplemental Material
Supplemental material for Field testing the diagnostic criteria of cluster headache in the third edition of the International Classification of Headache Disorders: A cross-sectional multicentre study
Supplemental Material for Field testing the diagnostic criteria of cluster headache in the third edition of the International Classification of Headache Disorders: A cross-sectional multicentre study by Heui-Soo Moon, Soo-Jin Cho, Byung-Kun Kim, Mi Ji Lee, Pil-Wook Chung, Jong-Hee Sohn, Soo-Kyoung Kim, Yun-Ju Choi, Tae-Jin Song, Jae-Moon Kim, Jeong Wook Park, Kwang-Yeol Park, Jae-Myun Chung, Jin-Young Ahn, Byung-Su Kim, Kyungmi Oh, Kwang-Soo Lee, Chin-Sang Chung and Min Kyung Chu in Cephalalgia
Footnotes
Acknowledgements
The authors would like to express appreciation to all participants in the present study.
Author contributions
Conception and coordination of the study: SJ Cho, BK Kim, MK Chu; design of ethical issue: PW Chung, JH Sohn, YJ Choi, JW Park; acquisition of data: HS Moon, SJ Cho, BK Kim, MJ Lee, PW Chung, JH Sohn, SK Kim, YJ Choi, TJ Song, JM Kim, JW Park, KY Park, JM Chung, JY Ahn, BS Kim, KM Oh, CS Chung, MK Chu; analysis and interpretation of data: HS Moon, SJ Cho, KY Park, MK Chu; data review and statistical analysis: SJ Cho, MJ Lee, BS Kim, MK Chu; manuscript preparation: HS Moon, SJ Cho, MK Chu; critical revision of the manuscript: HS Moon, SJ Cho, MK Chu. Manuscript approval: all authors.
Ethical statement
The study protocol was reviewed by the Independent Ethics Committee or Institutional Review Board for each centre. The study was conducted according to the ethical principles of the Declaration of Helsinki. All patients gave their written informed consent to participate in this study.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SJC was involved as a site investigator of a multi-centre trial sponsored by Otsuka Korea, Eli Lilly and Company, and Novartis and worked as an advisory member for Teva, and received research support from Hallym University Research Fund 2016 and a grant from Korean Neurological Association (KNA-16-MI-09). MKC was a site investigator for a multi-centre trial sponsored by Otsuka Korea, Novartis International AG and Eli Lilly and Company. He worked an advisory member for Teva, and received lecture honoraria from Allergan Korea, Handok-Teva and Yuyu Pharmaceutical Company in the past 24 months. The other authors declared no potential conflicts of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
