Abstract
Objectives
To describe long-term treatment patterns in migraine patients initiating prophylactic therapy and to evaluate acute medication use and adverse events associated with opioids.
Methods
This study used the 2005–2014 IBM MarketScan® databases to evaluate migraine patients initiating prophylactic medication. Outcome measures included persistence with prophylactic migraine medications over 2–5 years. Acute medication use and gastrointestinal-related adverse events and opioid abuse following opioid use were evaluated. Cox proportional hazards models were used to evaluate predictors of non-persistence and predictors of gastrointestinal-related AEs and opioid abuse associated with long-term opioid use.
Results
In total, 147,832 patients were analyzed. Non-persistence was observed in 90% of patients; 39% switched, 30% restarted, and 31% discontinued treatment. Over the follow-up, 59.9% of patients received triptans, 66.6% non-steroidal anti-inflammatory drugs, 77.4% opioids, and 2.6% ergotamines. Among opioid users, 16.6% experienced nausea/vomiting, 12.2% had constipation, and 10.4% had diarrhea. Opioid abuse was reported in <1% of opioid users. Gastrointestinal-related adverse events increased with increasing number of days’ supply of opioids.
Conclusions
Non-persistence to prophylactic treatment was frequent among migraine patients. Opioid use was common in migraine patients and the risk of gastrointestinal-related adverse events and opioid abuse increased with long-term use of opioids. These results suggest a need for more effective prophylactic migraine treatments.
Introduction
Migraine is a chronic neurological disorder that affects 38 million people in the US (1). Evidence suggests migraine substantially reduces productivity and impairs functionality, which in turn translates into poor health-related quality of life (2). More than half of all individuals with migraine are never diagnosed, and of those who are diagnosed, the majority do not seek medical treatment (1).
A number of acute and daily prophylactic therapies are available to treat or prevent migraine, and treatment approach depends on the severity and frequency of headaches (3–5). Acute medications, like over the counter and prescription analgesics, triptans, and ergotamines, are used on a short-term basis (2–3 days per week), with or without background prophylactic migraine medication, to reverse or abort an ongoing attack and stop progression to severe pain (4). Opioid analgesics are widely prescribed as acute medication for treating migraine (6). Statistics show that as many as 65% of individuals with migraine receive short-term opioid therapy (<3 weeks) and approximately 3% to 4% of the US adult population receive long-term opioid therapy for chronic pain (7,8). Although acute therapies provide necessary pain relief for migraineurs, there is mounting evidence that their overuse can lead to worse and more frequent headaches, decreased responsiveness to other migraine medications, and poorer long-term outcomes (9,10). Furthermore, both short- and long-term opioid use lead to multiple adverse events (AEs) (11). As many as 80% of patients were found to experience at least one opioid-related AE, with gastrointestinal AEs being the most common (12–14).
Prophylactic migraine therapies are usually indicated in patients with recurrent and frequent migraine attacks (≥2 disabling attacks/month, or ≥2 attacks/week, or increasing attacks over time), in patients who ineffectively respond to or experience AEs from acute treatments, or in patients at risk of acute medication overuse (4,15). Treatment guidelines recommend use of prophylactic migraine medications like topiramate, other anticonvulsants, tricyclic antidepressants, serotonin-norepinephrine reuptake inhibitors (SNRIs), calcium channel blockers, anti-hypertensive and other migraine medications for a continued period of at least 2–3 months; however, evidence recommending long-term use is lacking (16). Additionally, guidelines published by the American Academy of Neurology and American Headache Society cite a lack of clinical evidence regarding long-term safety, efficacy, and compliance for current prophylactic migraine medications (17).
Despite availability of multiple prophylactic treatment options, only 12–20% of patients with migraine currently use prophylaxis (18–21), whereas it is estimated that approximately 38% of migraineurs may meet clinical criteria for receiving prophylactic therapy (20). Of those who receive prophylactic therapy, data from several real-world studies report poor adherence and persistence to these medications (19,22–27), and AEs were the most common reason for discontinuation (23). While previous studies have examined treatment patterns of prophylactic therapy among migraine patients with <12 months’ follow-up period (19,23,24,26–28), there is a paucity of data on the long-term treatment patterns and persistence among migraine patients initiating prophylaxis in the US. Therefore, the objective of this study was to describe long-term treatment patterns in migraine patients initiating prophylactic therapy and to evaluate the use of acute migraine medication and the prevalence of opioid-related AEs in real-world clinical practice.
Methods
Study design and data source
This retrospective cohort analysis used administrative claims data between 2005–2014 from the IBM MarketScan® Commercial Claims and Encounters (Commercial) and the Medicare Supplemental and Coordination of Benefits (Medicare) databases. These databases contain inpatient and outpatient medical, and outpatient prescription drug experience of individuals with employer-sponsored primary or those with Medicare supplemental health insurance. The study databases satisfy the conditions set forth in Sections 164.514 (a)-(b)1ii of the Health Insurance Portability and Accountability Act of 1996 privacy rule regarding the determination and documentation of statistically de-identified data. As this study used statistically de-identified patient data, Institutional Review Board review was not required in accordance with the policy of our institution.
Patient selection
A previously published administrative-claims based algorithm published by Woolley et al. (29) was used to identify patients with migraine. Selected patients had evidence of migraine based on a) ≥1 inpatient claim, ≥1 emergency room claim or ≥2 outpatient claims (7–180 days apart) with a diagnosis of migraine (International Classification of Diseases, Ninth Revision, Clinical Modification [ICD-9-CM] diagnosis code 346.xx), b) ≥1 outpatient claim with a diagnosis of migraine and ≥1 outpatient pharmacy claim for a prophylactic migraine medication (either a triptan or ergotamine) (7–180 days apart), or c) ≥2 outpatient pharmacy claims for a triptan or ergotamine and initiation of treatment with a prophylactic migraine medication (other anticonvulsants, topiramate, tricyclic antidepressants, serotonin-norepinephrine reuptake inhibitors [SNRIs], calcium channel blockers, anti-hypertensive and other migraine medications) between 1 January 2006 and 31 December 2012. The initial prophylactic migraine medication prescription date was the index date. Patients initiating treatment were required to have a washout period with no prescription claims for a prophylactic migraine medication in the 12 months prior to the index date. All patients were required to be 18 years of age or older and to have continuous health plan coverage with medical and pharmacy benefits for at least 12 months before and 24 months following initiation of prophylactic medication. Patients who had a diagnosis of epilepsy during the 12-month baseline period were excluded. The follow-up period was of variable length and patients were followed for at least 24 months and up to 60 months from their index date until disenrollment, death, or end of study (31 December 2014), whichever occurred first.
Study outcomes
Treatment patterns were evaluated during the variable-length follow-up period and included proportion of patients initiating prophylactic migraine medication, treatment persistence (continuous use of any prophylactic migraine medication without a gap of >60 days after the previous prophylactic migraine prescription supply ran out, and separately by continuous use of index prophylactic medication class), and non-persistence (presence of a gap of >60 days in supply after the previous prescription ran out). Treatment modifications such as switching (adding a new prophylactic medication while discontinuing the initial medication), restarting (restarting a prophylactic migraine medication following a gap of >60 days in supply after the previous prescription ran out), discontinuation (ending treatment with an index medication without evidence of another refill within 60 days of exhausting the drug supply for the prior prescription), and augmentation (initiation of a prophylactic migraine medication prior to a 60-day gap in the supply of the previous prophylactic migraine prescription claim) were evaluated.
Utilization of acute migraine medications (triptans, ergotamines, NSAIDs, and opioids) during the baseline period and up to five years following the initiation of migraine prophylactic medications was evaluated. Incidence rates of a gastrointestinal (GI)-related AE and opioid abuse following opioid use were reported among opioid users in the follow-up period and among opioid users stratified by the number of days’ supply of opioids received during the follow-up period. GI-related AEs were identified by a medical claim with a diagnosis of nausea/vomiting, diarrhea, constipation (ICD-9-CM diagnosis codes 564.xx, 787.0x, 787.91) or by a prescription claim for the constipation medications lubiprostone or linaclotide. Opioid abuse was identified by a medical claim with a diagnosis of opioid abuse (ICD-9-CM diagnosis codes 304.0x, 304.7x, 305.5x, 965.00, 965.02, 965.09).
Covariates
Patient demographic characteristics, including age, gender, geographic region (US census division), urbanicity (based on urban or rural metropolitan statistical area (MSA)), and payer type, were measured on the index date. Other clinical characteristics were measured in the 12-month baseline period. Clinical characteristics included headache, chronic pain, hypertension, low back pain, depression, anxiety, osteoarthritis, inflammatory bowel disease, nausea/vomiting, asthma, fibromyalgia, menopause or menopause-related symptoms, constipation, cardiac arrhythmia, chronic migraine, diabetes, diarrhea, endometriosis, irritable bowel syndrome, menorrhagia, myocardial infarction, osteoporosis, and stroke.
Statistical analyses
Descriptive statistics were used to summarize all study variables, including demographic and clinical characteristics and treatment patterns. Categorical variables were summarized as counts and percentages while those for continuous variables were presented as means and standard deviations. Cox proportional hazards models were used to evaluate predictors of first-line treatment discontinuation (>60-day gap), switching and restarting. In addition, Cox proportional hazard models were used to assess the hazard of clinical events associated with the increasing number of opioid days supplied. Clinical events included opioid abuse and GI-related events, including constipation, nausea/vomiting, or diarrhea. All models were controlled for patients’ demographic (e.g. age, sex, payer type, geographic region), clinical characteristics (e.g. comorbid conditions), and first-line prophylactic medication (other anticonvulsants, tricyclic antidepressants, antihypertensives, beta blockers, calcium channel blockers, other migraine medications, and SNRIs). Hazard ratios (HRs) and
Results
Patient disposition
A total of 147,832 patients met the study eligibility criteria and were included in the study (Figure 1). Of these, 95,793 (64.8%) patients had at least 3 years of follow-up post-prophylactic medication initiation and 39,999 (27.1%) patients had at least 5 years of follow-up.
Patient selection.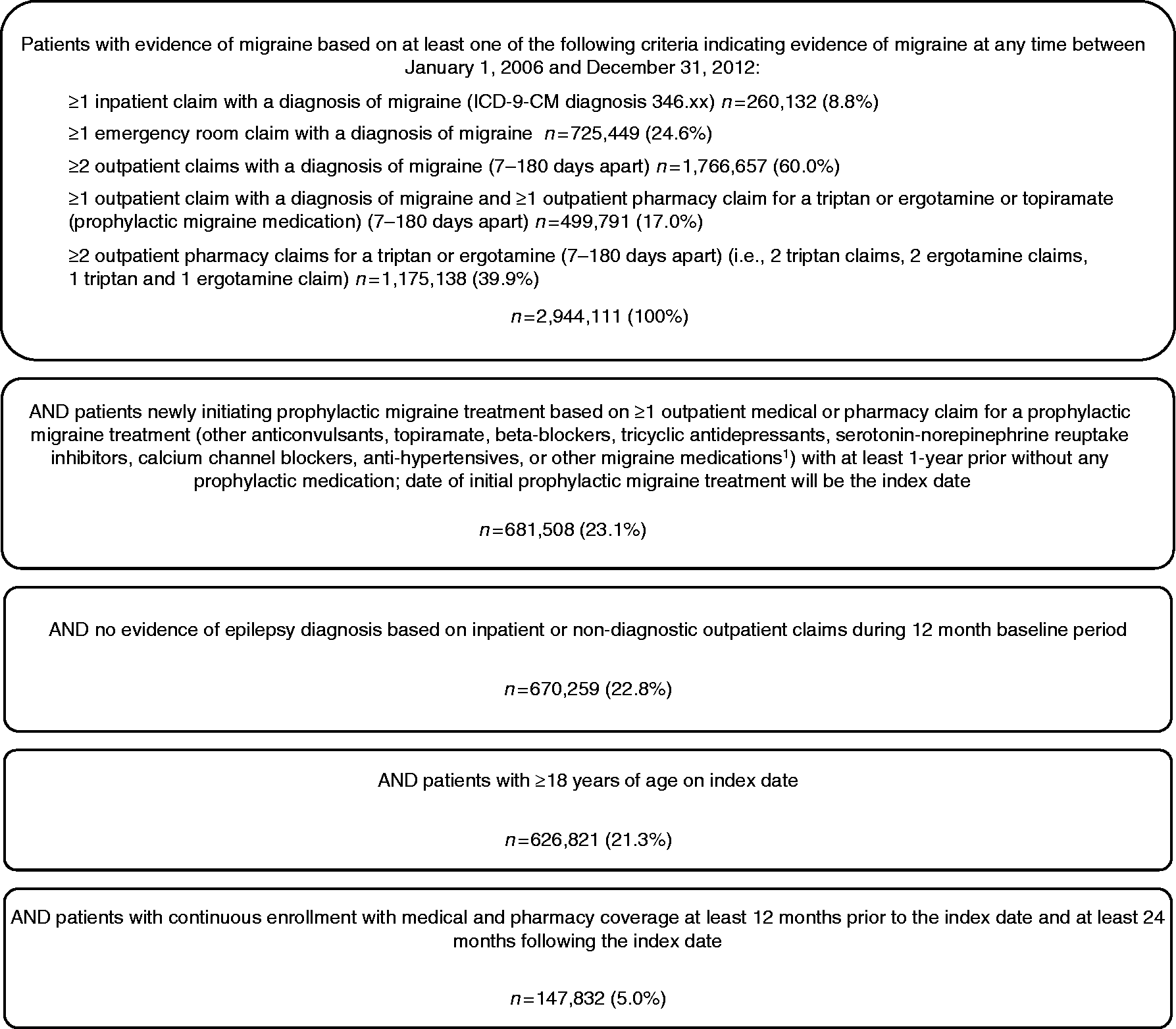
Patient characteristics
Demographic and clinical characteristics of patients initiating first prophylactic migraine medication.
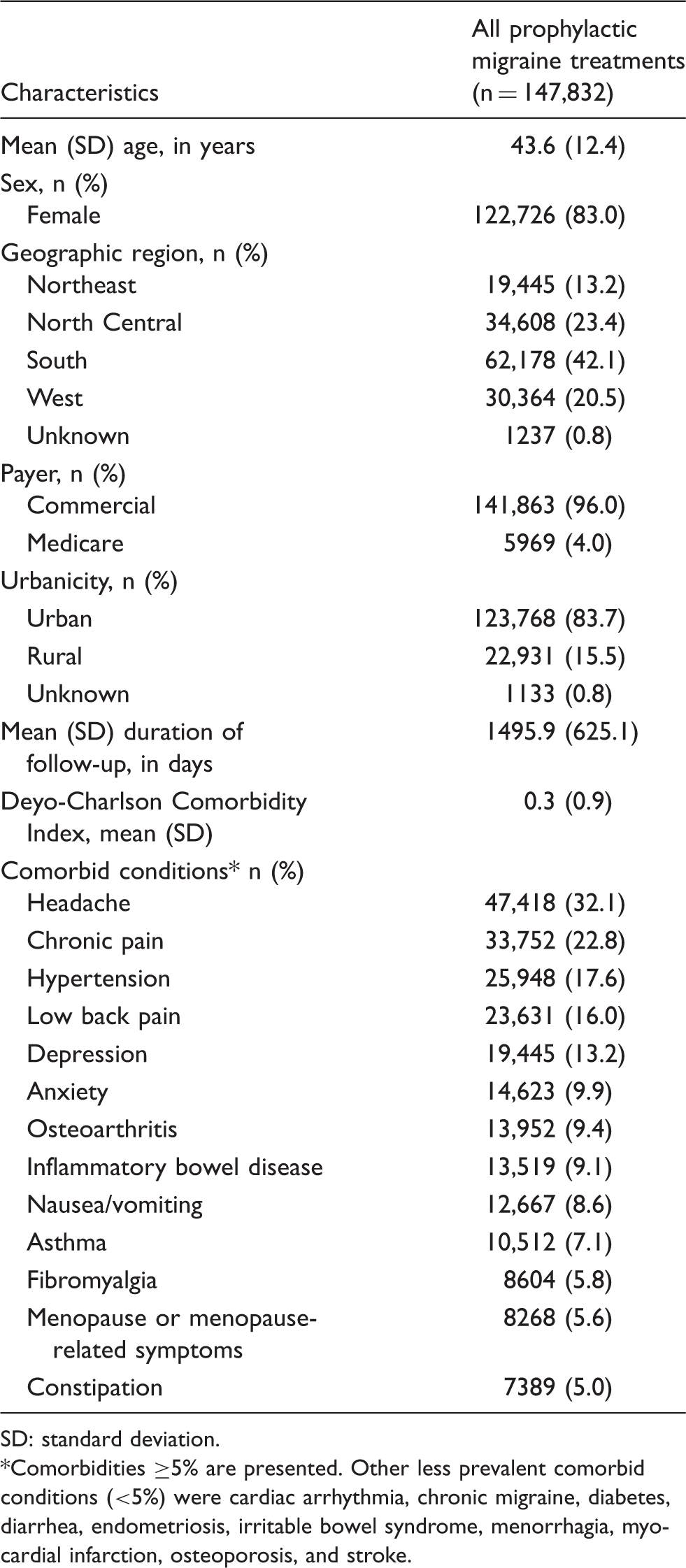
SD: standard deviation.
Comorbidities ≥5% are presented. Other less prevalent comorbid conditions (<5%) were cardiac arrhythmia, chronic migraine, diabetes, diarrhea, endometriosis, irritable bowel syndrome, menorrhagia, myocardial infarction, osteoporosis, and stroke.
Treatment patterns of first-line prophylactic migraine medications
Prophylactic migraine treatment patterns with first-line medication.
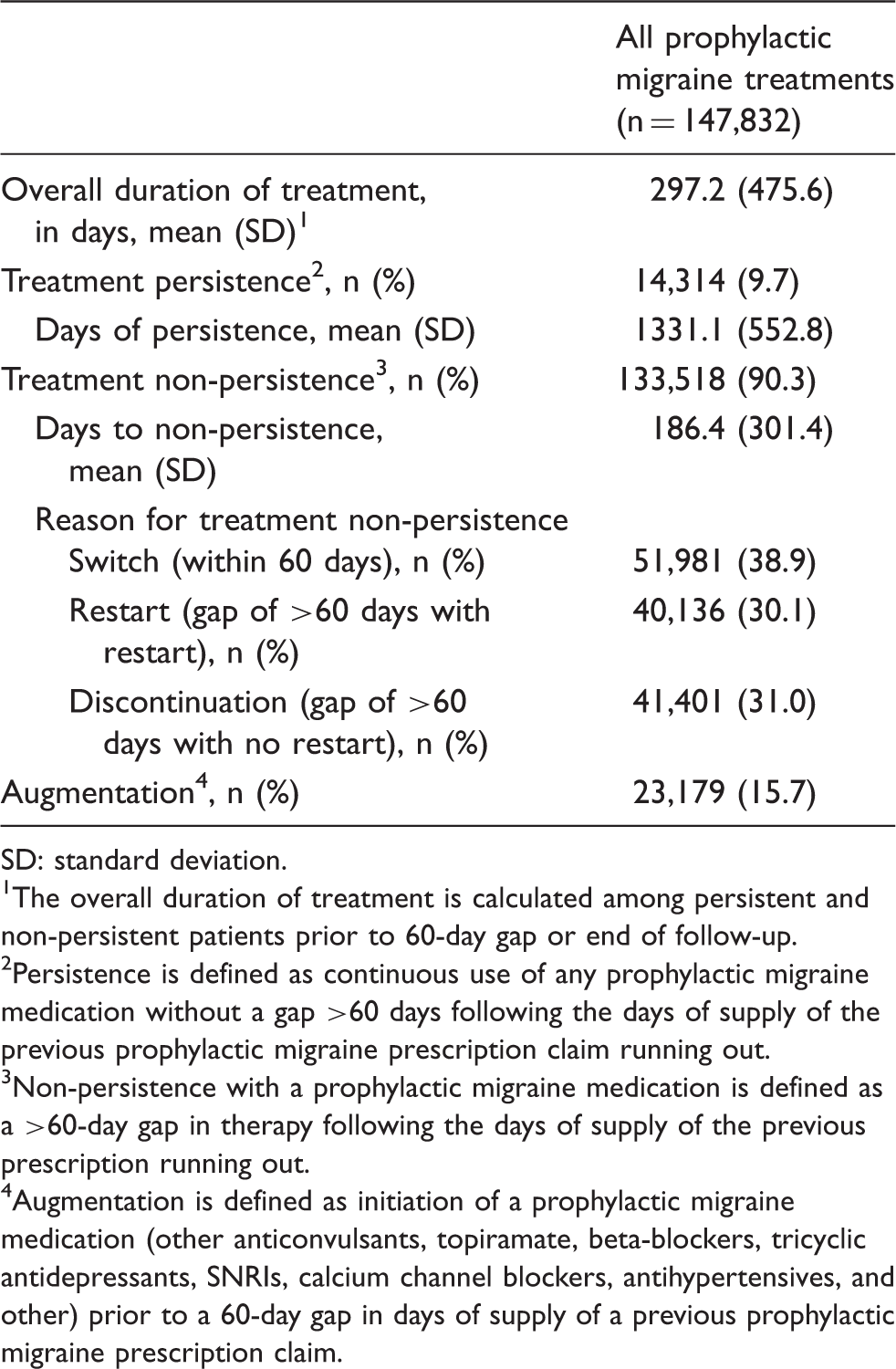
SD: standard deviation.
The overall duration of treatment is calculated among persistent and non-persistent patients prior to 60-day gap or end of follow-up.
Persistence is defined as continuous use of any prophylactic migraine medication without a gap >60 days following the days of supply of the previous prophylactic migraine prescription claim running out.
Non-persistence with a prophylactic migraine medication is defined as a >60-day gap in therapy following the days of supply of the previous prescription running out.
Augmentation is defined as initiation of a prophylactic migraine medication (other anticonvulsants, topiramate, beta-blockers, tricyclic antidepressants, SNRIs, calcium channel blockers, antihypertensives, and other) prior to a 60-day gap in days of supply of a previous prophylactic migraine prescription claim.
Hazard ratio of switching, restart, and discontinuation among migraine patients initiating prophylactic medication.
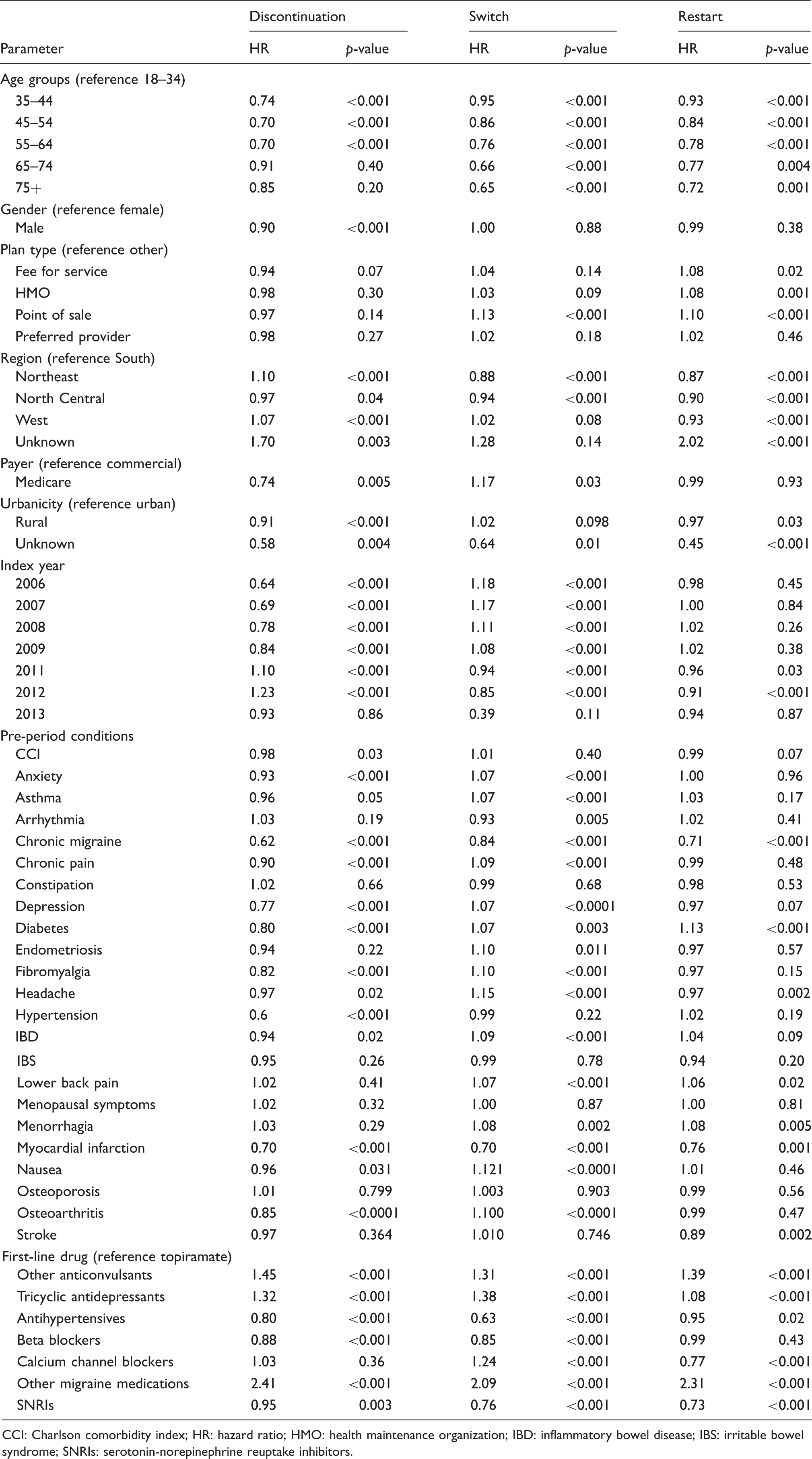
CCI: Charlson comorbidity index; HR: hazard ratio; HMO: health maintenance organization; IBD: inflammatory bowel disease; IBS: irritable bowel syndrome; SNRIs: serotonin-norepinephrine reuptake inhibitors.
Treatment with acute migraine medications
Acute migraine treatment patterns in the follow-up period.
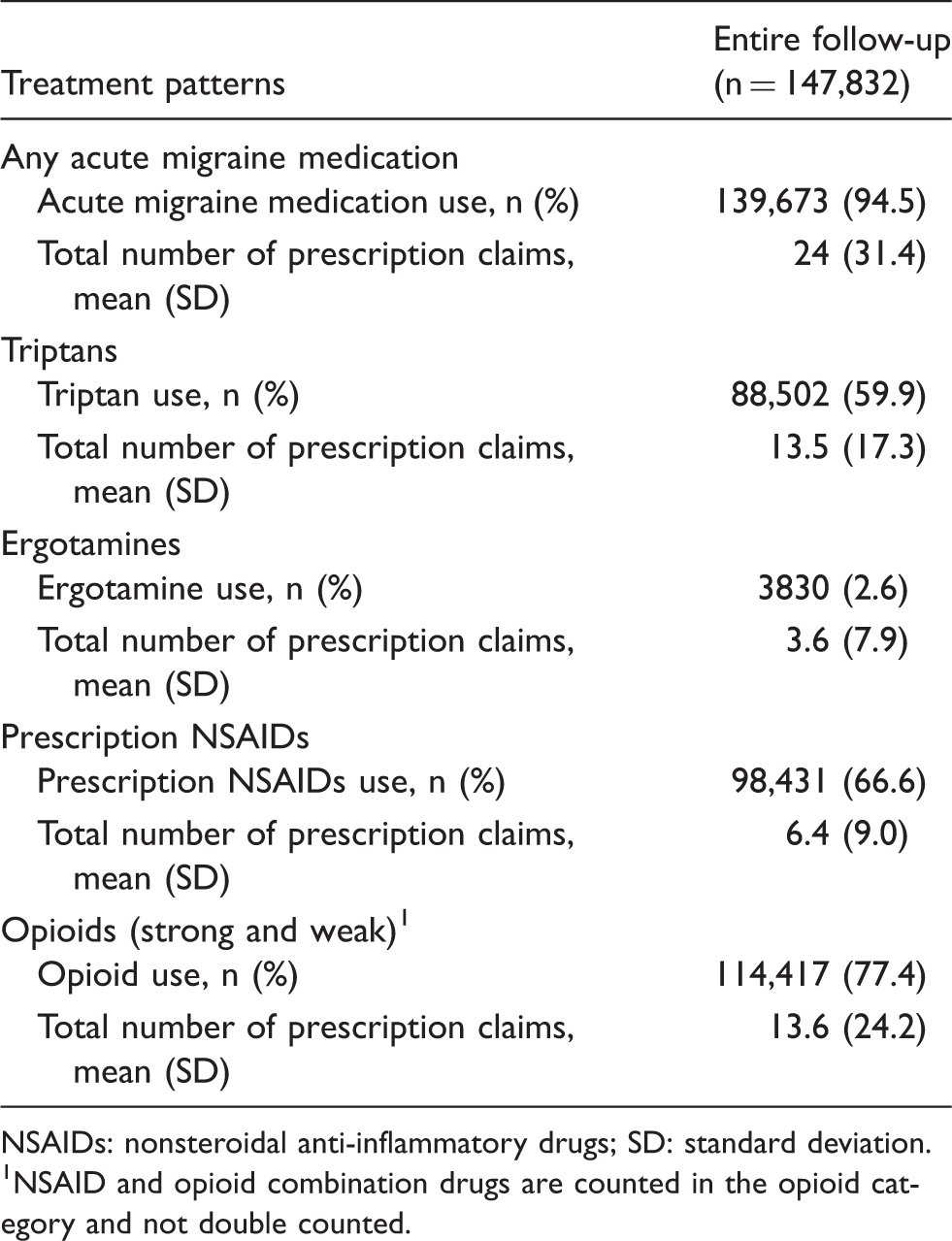
NSAIDs: nonsteroidal anti-inflammatory drugs; SD: standard deviation.
NSAID and opioid combination drugs are counted in the opioid category and not double counted.
GI-related AEs among opioid users
Potential incident opioid-related clinical events among prophylactic migraine patients.
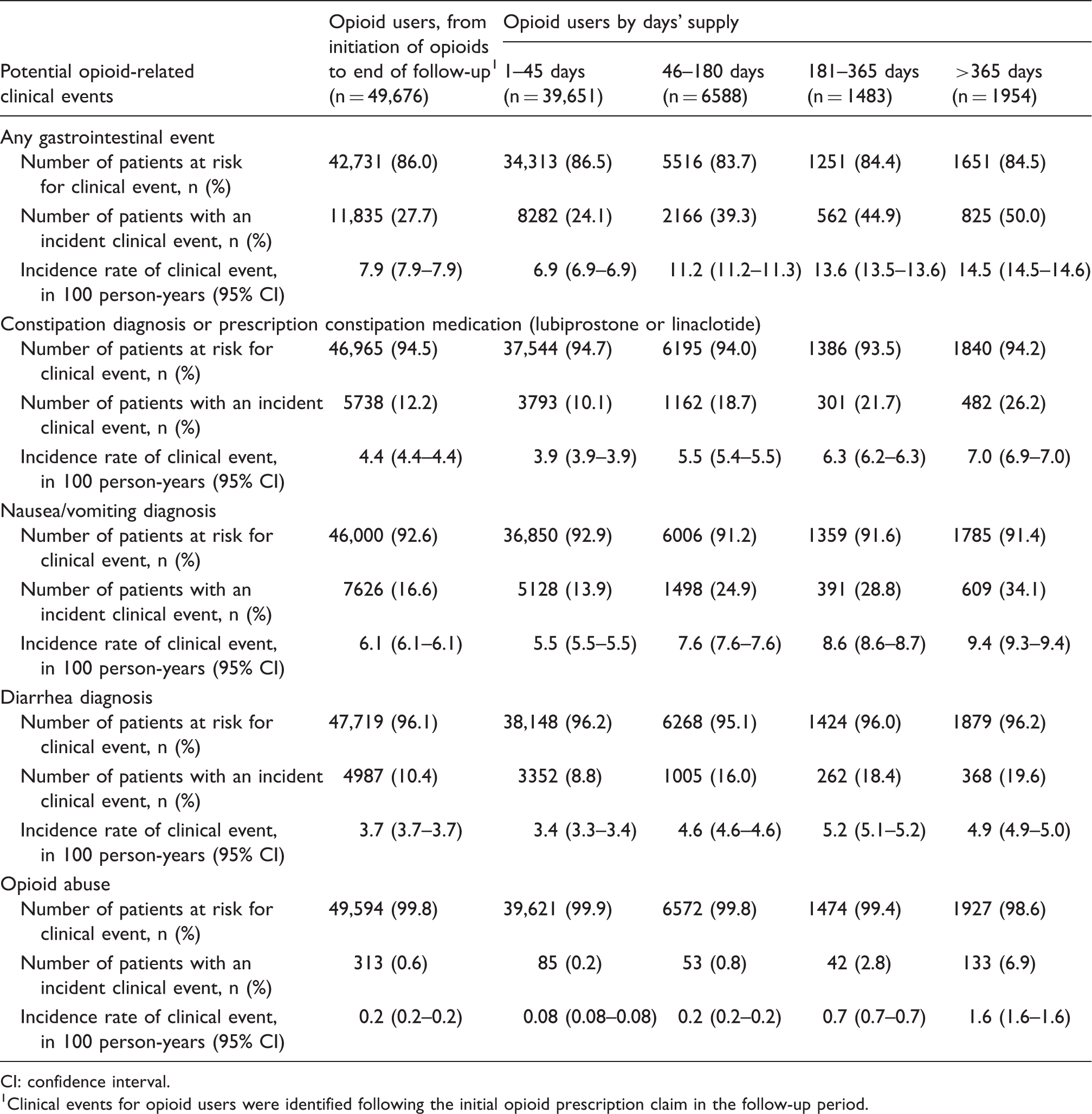
CI: confidence interval.
Clinical events for opioid users were identified following the initial opioid prescription claim in the follow-up period.
Hazard ratio of risk of any GI event, constipation, nausea, diarrhea, and opioid abuse among migraine patients initiating prophylactic medication.
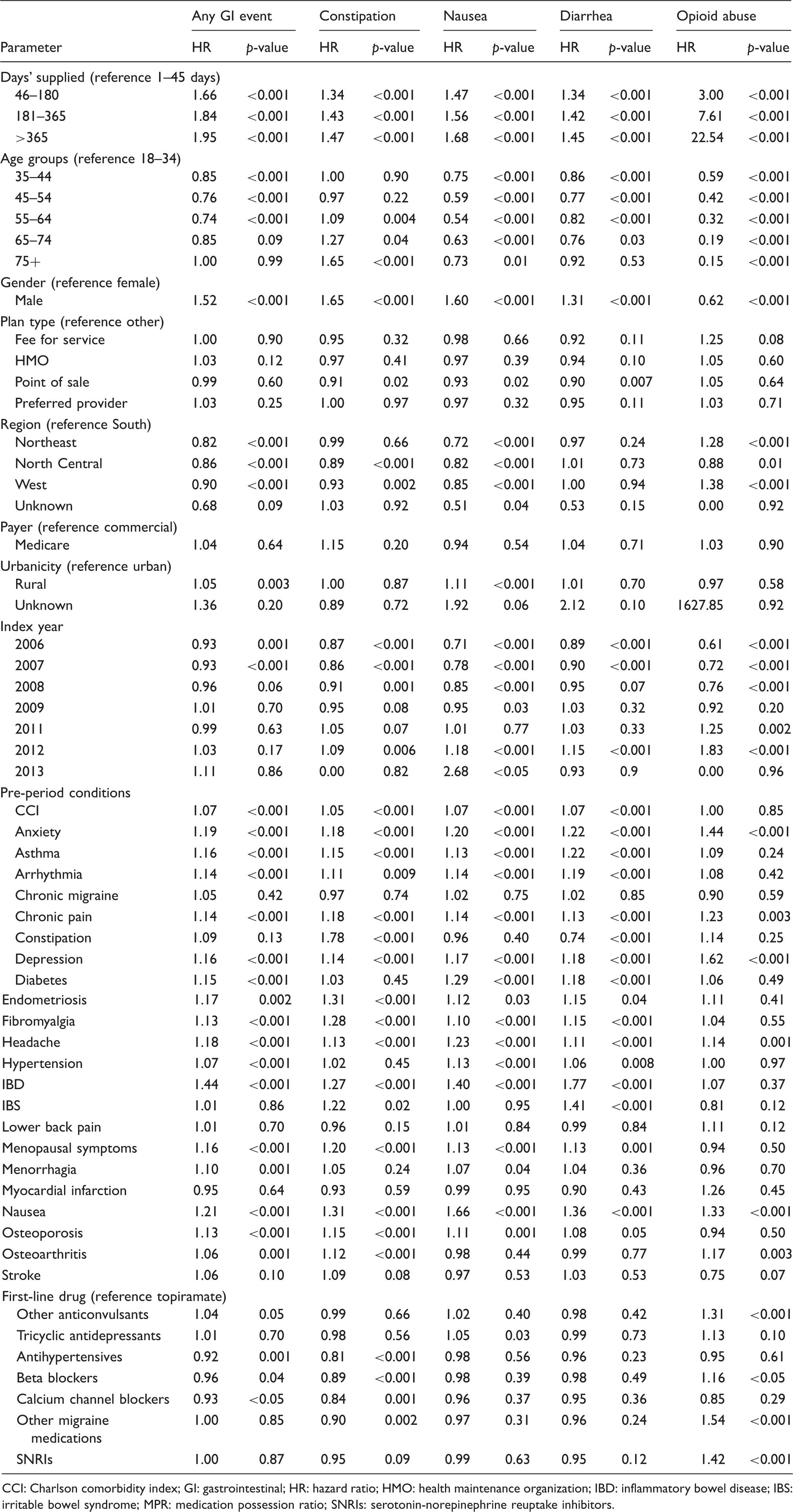
CCI: Charlson comorbidity index; GI: gastrointestinal; HR: hazard ratio; HMO: health maintenance organization; IBD: inflammatory bowel disease; IBS: irritable bowel syndrome; MPR: medication possession ratio; SNRIs: serotonin-norepinephrine reuptake inhibitors.
Discussion
In this US administrative claims-based analysis of adults with migraine who initiated prophylactic therapy, long-term persistence with first-line prophylactic medications was poor. Over 90% of the migraine patients either discontinued, restarted or switched from their initial prophylactic treatment a little after the first 6 months of initiation. Additional treatment with acute migraine medications (opioids, NSAIDs, triptans, and ergotamine) to reverse or abort an ongoing migraine was common among migraine patients that were receiving prophylactic therapy. Furthermore, GI-related AEs and opioid abuse related to use acute migraine medications increased with longer duration of treatment with opioids.
Reports on short-term persistence with prophylactic therapy have been published previously (19,23,24,26–28,30). Data from our study showed that less than 10% of migraine patients persisted with index prophylactic therapy over a 4 year average duration of follow-up, which is consistent with the rates reported in previous studies (23,24,26,27,31). Furthermore, our study suggested younger age, lower pre-index CCI score, history of select comorbidities such as anxiety, asthma, chronic migraine, chronic pain, depression, diabetes, fibromyalgia, headache, hypertension, IBD, myocardial infarction, nausea, and osteoarthritis, and first-line medication use with other anticonvulsants, tricyclic antidepressants, antihypertensives, beta blockers, other migraine medications, and SNRIs in the pre-index period were significantly associated with lower risk of discontinuation from the initial prophylactic treatment. Taken together, these findings suggest that migraine patients receiving treatment with existing prophylactic medications have poor persistence in the real world. Although this study did not investigate the reasons for non-persistence, the relatively low persistence rate can possibly be due to a lack of perceived efficacy and/or undesirable side effects associated with the use of prophylactic medication in migraine, which appears to be even more distinct among patients who are older and have a higher disease burden (23,24,27,32,33).
Frequent and long-term use of acute therapies are not recommended; however, the majority of patients initiating migraine prophylaxis use both specific and non-specific acute medications to supplement prophylactic migraine medications to achieve rapid pain relief during a migraine (6). In an analysis of data from the 2005 American Migraine Prevalence and Prevention (AMPP) study, 91.7% of respondents meeting criteria for episodic migraine used acute treatment for their headaches (34). Similarly, in a recent US claims-based analysis that assessed the use of specific and non-specific acute migraine medication before and after prophylaxis, the majority of patients (84%) were found to use at least one acute medication within 1 year of initiating prophylaxis, which increased to 97% over 5 years of follow-up (35). Consistent with these findings, the use of acute migraine medication was widespread in this cohort of patients who initiated prophylactic treatment. Analysis of the data from the 2009 AMPP study demonstrated that opioids are widely used as acute treatment for migraine (36). Furthermore, 40–90% of opioid-treated patients experienced any GI-related AE (13,14,37–39), and the rate of AEs was found to increase with the duration of opioid prescriptions (40,41). Data from the current study extends support to these findings, as opioids were the most commonly used acute medications in this study cohort, and the risk of any GI-related AEs (constipation, nausea/vomiting, and diarrhea) and opioid abuse increased with increasing days of opioid supply.
This analysis is subject to several limitations. First, this analysis evaluated individuals with migraine initiating prophylactic treatments; therefore, the results may not be generalizable to migraine patients not treated with prophylactic treatments or those previously treated with prophylactic treatments. Furthermore, this analysis evaluated a patient sample that reflected the geographic distribution of the overall population in the MarketScan Research Databases population and was not balanced to control for differences in urban versus rural MSA or by geographic region. Moreover, incidence rates of AEs were evaluated within the subset of patients in the study population treated with opioids. This study did not describe whether opioid users differed from the overall patient sample or whether opioid users differed by increasing days’ supply of opioids.
Several limitations that are inherent to claims database analyses apply to this study as well. As with all administrative healthcare database, the MarketScan Research databases rely on administrative claims data for clinical detail; therefore, the data may be subject to coding limitations, potentially introducing measurement error. Furthermore, patients were selected based on the presence of diagnoses based on ICD-9-CM diagnosis codes and the receipt of prescription medication based on NDC codes and HCPCS codes. Under-diagnosis, as well as coding errors, may affect study selection to an extent that cannot be quantified. To limit the impact of migraine undercoding in administrative claims data, a previously published algorithm using the combination of a migraine diagnosis and prescription claims for prophylactic migraine medications was used to identify patients with chronic migraine. However, this database does not enable the characterization of migraine when medical care was not provided. Furthermore, patients who did not meet the continuous enrollment criteria, without any health coverage, or who died during the follow-up, were excluded. Therefore, it may be possible that the patients included in the analysis may have been healthier than the total population of patients in this claims database. Medication exposure was based on fill prescriptions. While we assumed these patients took medications as prescribed, confirmation of this assumption was not possible. Lastly, this study primarily included migraine patients enrolled with either Commercial or Medicare Supplemental health coverage, thus results of this analysis may not be generalizable to all migraine patients with other insurance or populations without health insurance coverage.
This study does offer some potential advantages – in contrast to most previous retrospective studies, the current study provides contemporary long-term data on the persistence of prophylactic treatment among patients with migraine from two large administrative claims databases. Furthermore, the wide range of information on patients’ demographic factors, medical and medication history allows multivariable adjusted analysis to include many important potential confounders.
In summary, the results of this study suggest that long-term persistence with prophylactic medications in migraine patients was poor, reflecting an unmet need for improved prophylactic therapies. Use of acute migraine medications, especially opioids, coupled with increased rates of opioid-related AEs, were common in migraine patients treated with prophylactic therapy. New treatment options with improved efficacy and tolerability are warranted to address these concerns.
Clinical implications
Long-term persistence with prophylactic medications in migraine patients is poor. Acute migraine medications, including opioids, are used frequently to manage migraine pain. Increasing use of opioids is associated with higher rates of opioid-related adverse events. There is an unmet need for improved prophylactic therapies to manage migraine.
Footnotes
Acknowledgements
Editorial/writing assistance for this manuscript was provided by Santosh Tiwari, who is an employee of IBM Watson Health.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: This study was conducted by IBM Watson Health. All listed authors meet the criteria for authorship set forth by the International Committee for Medical Journal Editors. Machaon Bonafede and Kathleen Wilson are employees of IBM Watson Health, which received compensation from Amgen Inc. to conduct the study. Fei Xue is an employee and stockholder of Amgen Inc.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Amgen Inc., USA.
