Abstract
Purpose
The present study aimed to determine the effects of combined supplementation of Coenzyme Q10 with L-carnitine on mitochondrial metabolic disorders marker and migraine symptoms among migraine patients.
Methods
A total of 56 men and women, between 20–40 years of age with migraine headache, participated in this randomized, double-blind, placebo-controlled, parallel study. The subjects were randomly assigned to receive either 30 mg/day Coenzyme Q10 and 500 mg/day L-carnitine at the same time and/or placebo tablets for 8 weeks. The measurements were completed at the beginning and end of the study. The primary outcome was severity of headache attacks. The secondary outcomes included duration, frequency of headache attacks, the headache diary results (HDR), and serum levels of lactate.
Results
A significant reduction was obtained in serum levels of lactate (−2.28 mg/dl, 95% CI: −3.65, −0.90; p = 0.002), severity (−3.03, 95% CI: −3.65, −2.40; p ≤ 0.001), duration (−7.67, 95% CI: −11.47, −3.90; p ≤ 0.001), frequency (−5.42, 95% CI: −7.31, −3.53; p ≤ 0.001) and HDR (−103.03, 95% CI: −145.76, −60.29; p ≤ 0.001) after 8 weeks.
Conclusion
This double-blind parallel study provides evidences supporting the beneficial effects of Coenzyme Q10 and L-carnitine supplements on serum levels of lactate and migraine symptoms.
Trial registration
IRCT20121216011763N21.
Introduction
Migraine is a chronic and progressive brain disorder that is ranked among the causes of disability, according to the World Health Organization reports (1,2). Migraine is a common important cause of primary headache worldwide and our country (3,4). The prevalence of migraine in Western countries was estimated as 5–9% and 12–25% for men and women, respectively (5,6). The disorder is less prevalent in Asia; however, it is estimated that at least 3% of men and 10% of women experience this disorder (7). The prevalence of migraine is reported as 9.5% in the adult population in Iran (8).
Migraine can be a disabling disorder and affects the patients’ quality of life (4). It is also associated with other disorders such as cardiovascular disease, stroke, asthma, epilepsy, allergies and sleep disorders (9).
The exact etiology of migraine is not fully understood, but the relevance of mitochondrial dysfunction in the development of migraine has been suggested in this regard (10). This disorder causes deficiency in oxygen metabolism and affects the processing of neural information (10).
Based on the results of various studies, nutrition approaches are safer and more cost-effective than pharmacological approaches in the required treatments of this disorder (11). Several dietary factors such as riboflavin (12), niacin (13), and magnesium (14) have been reported to be helpful in the treatment of migraine.
One contributing explanation to the health benefits of these nutrients is related to their potential effects on mitochondrial metabolism. With regards to these effects, the association between mitochondrial dysfunction and migraine disease seems completely rational (15).
Among the nutritional factors affecting migraine, recently Coenzyme Q10 (CoQ10) and L-carnitine have received particular attention (16). CoQ10, also known as ubiquinone, is beneficial for migraine treatment by two active mechanisms. CoQ10 is a vital factor in the electron transport chain of mitochondria, which improves energy metabolism and oxygen utilization in the brain and muscles (16). Its antioxidant properties have also been confirmed by different studies (16).
L-carnitine is an essential substrate that aids the transporting of fatty acids across the mitochondrial membrane and metabolism of fatty acid in mitochondria (17).
The effectiveness of these compounds on migraine treatment has been indicated in earlier studies. Several investigations have reported the positive effects of CoQ10 and L-carnitine supplementation on migraine headache (18–21). However, whether the beneficial effects of CoQ10 and L-carnitine on migraine treatment are mediated through their effect on mitochondrial function still remains unclear.
Given the conflicting evidence of the effects of CoQ10 and L-carnitine on migraine, it was hypothesized that concurrent supplementation of CoQ10 and L-carnitine had better effects on migraine outcomes.
Based on prior knowledge, there has been no investigation for evidence of the effect of combined CoQ10 with L-carnitine supplementation on migraine headache.
Therefore, the present study will specifically examine the effect of combined CoQ10 with L-carnitine supplementation on mitochondrial metabolic disorders marker and migraine symptoms of migraine patients.
Materials and methods
Participants
The present study was designed as a randomized, double-blind, placebo-controlled, parallel trial. A total of 56 patients with migraine, 49 females and seven males, were consecutively enrolled in the study based on inclusion and exclusion criteria and were randomly assigned into intervention and placebo groups (Figure 1). Enrollment was done by the researchers.
Patient flow diagram.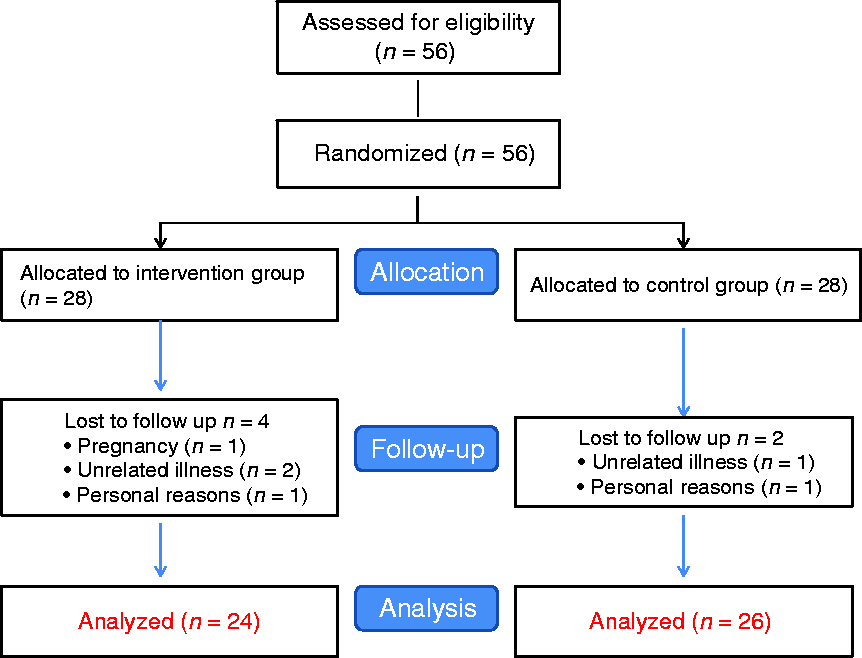
Random assignment was carried out using computer-generated random numbers created by statisticians. Randomization was undertaken by blocks of two and random allocations were carried out by a technician in the study. All the investigators, participants and laboratory technicians were blinded to the random allocation during the study, with the exception of the study technician who undertook the randomization. All the volunteer participants were recruited from Imam Mousa Sadr Clinic, belonging to Isfahan University of Medical Sciences, Isfahan, Iran, from May to August 2017. Migraine was confirmed by a neurologist according to the International Headache Society criteria (IHS) (2). The primary measure included severity of headache attacks and the secondary measures included the duration, frequency of headache attacks, HDR, and serum levels of lactate.
Inclusion criteria were: Patients with migraine (with or without aura), at least two attacks per month, chronic headache lasting from 4 to 72 h, 20–40 years of age, male and female gender, non-pregnant, non-lactating, non-smoker, and non-alcoholic patients, who were willing to participate in the study. Exclusion criteria were: chronic diseases, chronic tension-type headache or secondary headaches and the use of anti-migraine medication or supplements for a month before the experiment, headache medication overuse during the last six months, following a diet or exercise program during the past six months.
The sample size was calculated according to the sample size formula suggested for the parallel trial. Type 1 error of 5%, type 2 error of 20% (power = 80%), and migraine severity were considered as key variables (18). Assuming possible exclusions within the study, the total of 56 migraine patients based on the above-mentioned inclusion criteria were considered for this study. During the interventions, six patients (four in the treatment and two in the placebo group) were excluded from the study due to pregnancy, unrelated illness and personal reasons. The protocol of the study was clearly explained to all the participants, and their written informed consent was obtained. The study was approved by the Research Council and Ethical Committee of Isfahan University of Medical Sciences, Isfahan, Iran, and has been registered at the Iranian website for registry of clinical trials (IRCT20121216011763N21).
Study design
The intervention group (n = 28) received 30 mg/day CoQ10 (MIX natural, Canada) and 500 mg/day L-carnitine (Karen, Iran) at the same time, for 8 weeks, and subjects in the control group (n = 28) were also given the placebo tablets in the same form, color, package, dosage and administration as the active supplements to ensure a blinded design. Individuals in both groups were given a list of tyramine-containing foods and were asked not to consume any of these during the study. All measurements were completed at the beginning and the end of the study. Adherence was defined as having taken the tablets and was measured using self-reports and supplement counts. Compliance with the consumption of supplements was monitored once a week by the researchers through phone contact interviews.
During the study, all participants provided dietary and physical activity records once every 2 weeks. Therefore, each participant had 3 days of dietary and physical activity records (two weekdays and one weekend day). The days were allocated by the dietitian. Then, the reported portion sizes in the records were converted to grams using household measures. All the dietary data were based on the average value of three dietary records. The grams of food intake data were linked with Nutritionist IV software to derive the nutrient intake data. The nutrient database of Nutritionist IV software was based on the USDA food composition database, which has been modified for Iranian foods. The physical activity level was evaluated using a 3-day physical activity record and expressed as the metabolic equivalent (MET) hour/week (22). To calculate the MET, the compendium list of physical activity was used as follows: MET × duration of physical activity (minutes).
Finally, the average value for 3 days MET was reported for each subject as the total MET. The dietary records were based on estimated values in household measurements.
Anthropometric assessment
Measurement of height was undertaken in a standing position, without shoes and with shoulders relaxed, using a tape rule. Weight was measured to the nearest 100 g using a digital scale (Seca, Hamburg, Germany), in light clothing. Body mass index (BMI) was calculated as weight in kilograms divided by height in meters squared. Waist circumference was measured at the smallest circumference. To avoid subjective error, all measurements were taken by the same person. Blood pressure was measured in a sitting position using a digital sphygmomanometer with the appropriate cuff, using the standard method. Three readings were recorded at 5-minute intervals with complete deflation of the cuff between each reading. The average value of the three readings was calculated.
Biochemical assessment
Fasting blood samples (10 mL) were taken at baseline and after the 8-week intervention at Baradaran laboratory in Isfahan, Iran (which is under external quality control of the National Reference Laboratory).
Plasma lactate levels were measured with RANDOX L-Lactate kits (Crumlin, United Kingdom). Patient samples and standard samples were incubated for 5 minutes at 37℃ using N-ethyl-N-(3 methyl phenyl)-N acetylethylendiamine solution. The mixture was allowed to rest for 4.5 minutes at 37℃ after addition of lactic oxydase. Color intensity was measured using a dual-beam spectrophotometer at 660/600 nm.
The inter- and intra-assay CVs for all biochemical indicators were < 10%. All the biochemical measurements were carried out in a blinded fashion, in duplicate, in pairs (before/after intervention) at the same time, in the same analytical run, and in a random order to reduce systematic error and inter-assay variability.
Migraine assessment
Diagnosis of the migraine patients was conducted by an experienced neurologist according to the international headache society (IHS) criteria (headache classification committee of the IHS 2004) (2). Characteristics of migraine attacks such as headache severity, frequency and duration were determined. The visual analog scale was used to measure the migraine severity (23). In this method, the headache severity is ranked from 1 to 10, and each patient is expected to select a number between 1 and 10 for severity of their headache. Frequency was considered as the number of attacks per month. Mean duration (hour) of migraine attacks was considered as the duration. Moreover, the headache diary result (HDR) was measured as duration of headache × frequency of headache (23).
Statistical analysis
Normal distribution of variables was examined using the Kolmogrov-Smirnov test. To properly evaluate difference between groups, a two-sided, two-sample equal variance t-test was utilized. A total of 56 (n = 28 placebo and n = 28 active) achieved 80% power to reject the null hypothesis of equal means assuming a mean of zero and standard deviation of 1 for both groups (Cohen’s D = 0.8) when alpha = 0.05. Comparison of data on dietary intakes and physical activity was done using a paired t-test. The association between primary serum levels of lactate with primary migraine symptoms was examined using correlation. p-values < 0.05 were considered significant. All statistical analyses were conducted using SPSS (version 18) statistical software (SPSS, Inc., Chicago, IL, USA).
Results
Out of the 56 subjects (female = 49, male = 7) recruited for the study, six subjects (four in the treatment and two in the placebo group) dropped out. One subject withdrew due to pregnancy, three subjects withdrew due to unrelated illnesses, and two other subjects withdrew due to personal reasons. Therefore, 50 participants completed the study (the intervention group (n = 24) and the control group (n = 26)) (Figure 1).
Baseline characteristics of participants 1 (n = 56).
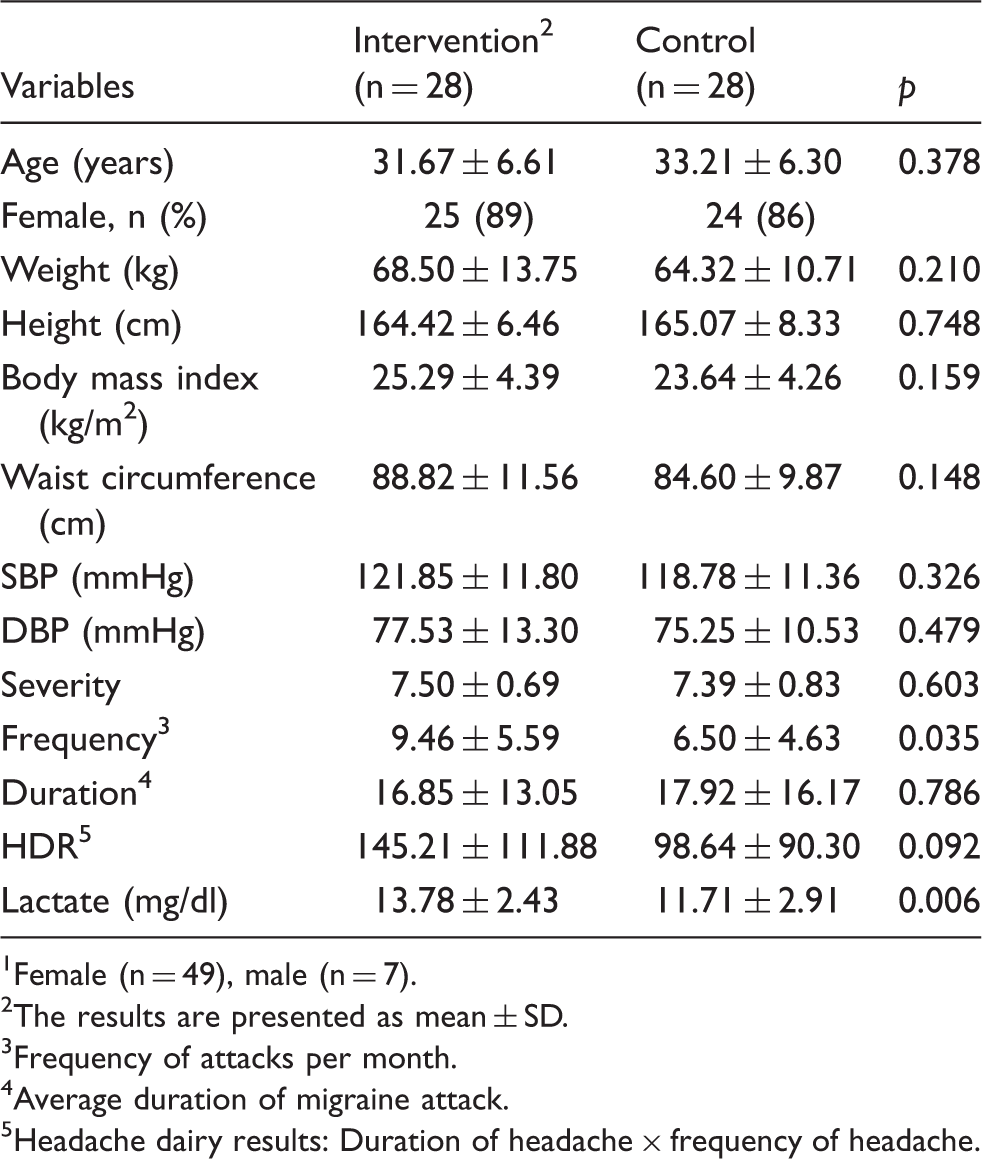
Female (n = 49), male (n = 7).
The results are presented as mean ± SD.
Frequency of attacks per month.
Average duration of migraine attack.
Headache dairy results: Duration of headache × frequency of headache.
Dietary intakes of participants, obtained from three dietary records, throughout the study 1 (n = 50).

All values are means ± SD.
By independent t-test.
A significant correlation was found to exist between primary serum levels of lactate and primary severity of migraine (0.09, 95% CI: 0.05, 0.55; p = 0.038). No significant associations were found between primary serum levels of lactate and primary frequency and duration (0.064, 95% CI: −0.20, 0.32; p = 0.638) and (−0.195, 95% CI: −0.44, 0.07; p = 0.151), respectively).
The effects of Coenzyme Q10 with L-carnitine supplementation on mitochondrial metabolic disorders marker and migraine symptoms 1 .

All data are means ± SD.
Results of two independent sample t-tests.
Frequency of attacks per month.
Average duration of migraine attack.
Headache dairy results: Duration of headache × frequency of headache.
Except for constipation and stomach ache (10% in the intervention group versus 3% in the placebo group), no other serious adverse side effect was observed for supplement use during the study.
Discussion
The results of this double-blind, randomized controlled trial among migraine patients revealed that the combined supplementation of CoQ10 with L-carnitine for 8 weeks could significantly affect serum levels of lactate and migraine symptoms. To the best of our knowledge, this is the first study that has examined the effects of concurrent supplementation of CoQ10 and L-carnitine on mitochondrial metabolic disorders marker and migraine symptoms in patients with migraine. In a case series, Richard G Boles (24) suggested that a protocol consisting of mitochondrial-targeted cofactors (CoQ10 and L-carnitine) is highly effective in the prevention of vomiting episodes. Since mitochondrial dysfunction is hypothesized to be involved in the development of both cyclic vomiting syndrome (CVS) and migraine. It is hypothesized that these substrates may also be beneficial in prophylaxis of migraine.
A positive relationship was found to exist between primary serum levels of lactate, marker of mitochondrial metabolic disorders, and primary severity of migraine headache. Mitochondria are small organelles involved in energy metabolism, especially in high energy-consuming organs like the brain, heart, liver and muscles, reactive oxygen species (ROS) production, regulation of apoptosis, and control of calcium (Ca2+) homeostasis (11). The role of mitochondrial dysfunction in the development of migraine has been recently suggested. Mitochondria play a central role in the functions of neurons by producing sufficient ATP and regulating intracellular calcium levels (25). Therefore, any abnormal mitochondrial function affecting energy metabolism and ion homeostasis in neurons might lead to several downstream imbalances consequently resulting in increased susceptibility to migraine (25). Oxidative stress might play a role in the pathogenesis of migraine. Recent studies have shown an impairment of the brain oxidative energy metabolism in migraine attacks (10). Various studies have demonstrated that lactic acid levels are increased in patients with migraine. Therefore, the increased levels of lactate are suggested as a marker of defective oxidative metabolism (15,25). Thus, the results in this study supported the concepts of the association between mitochondrial dysfunction and migraine disease.
Several dietary factors have been suggested to beneficially influence mitochondria metabolism and migraine headache (12–14). CoQ10 and L-carnitine have been shown to be effective in treatment of mitochondrial disorders as well as migraine (16). It was discovered that supplement intake might reduce serum levels of lactate in patients with migraine. The findings are in line with the findings in earlier studies in this respect (26,27).
In the current study, it was also shown that concurrent CoQ10 and L-carnitine supplementation favorably affects migraine symptoms. The findings are also in agreement with the findings in earlier studies, that CoQ10 or L-carnitine supplementation might reduce migraine severity and frequency (17–20,28).
In contrast to the present findings, some studies did not find a significant effect (16,29). Hagen et al. (16) in a triple-blinded crossover trial indicated that acetyl-l-carnitine supplementation in 3 g dosage for 12 weeks had no effects on headache outcomes. No significant differences in headache outcomes between the CoQ10 and placebo groups were found in another crossover trial (29). The different between-study findings could be explained by the discrepancy in recruited subjects in terms of their health conditions, age, and gender. Type and dosage of supplements used as intervention along with duration of intervention might also explain some differences.
Several mechanisms have been suggested to underlie the protective effects of CoQ10 and L-carnitine on migraine outcomes. CoQ10 and L-carnitine play independent roles in mitochondrial metabolism of energy. Coenzyme Q10 act as an essential component of energy metabolism in mitochondria. It also protects mitochondria from oxidative stress (16,30).
L-carnitine is a shuttle of fatty acids across the mitochondrial membrane and thus it is necessary for fatty acid oxidation. Any dysfunction in fatty acid oxidation in brain mitochondria may cause accumulation of toxins (17,31). CoQ10 and Carnitine has been considered as a prophylactic medication for migraine.
This study had several advantages. This is the first clinical trial that has compared the effects of concurrent supplementation of CoQ10 and L-carnitine on migraine symptoms. Furthermore, dietary intakes and physical activity of subjects were assessed throughout the study, which enabled the exploration of the changes in these variables.
Despite the significant effects of CoQ10 plus L-carnitine supplements on serum levels of lactate and migraine symptoms, some points deserve considerable attention. Due to budget limitations, serum levels of CoQ10 and L-carnitine were not quantified at the study baseline to ascertain the percentage of patients who were deficient in that regard. In addition, it was not possible to measure the serum levels of CoQ10 and L-carnitine at the end of the study to ensure compliance with consumption of the supplements. Compliance was monitored once a week through phone interviews by researchers. However, the compliance with consumption of supplements seems relatively appropriate, based on self-reports and pill counts. It should be noted that most of our participants were female, so generalization of the study results with respect to men should be done by caution.
Finally, the current study had adequate power to detect the significant effects; however, further studies with longer duration of interventions might be needed to confirm the long-term health benefits of supplements in patients with migraine.
In summary, the findings suggest that CoQ10 plus L-carnitine supplements had beneficial effects on serum levels of mitochondrial metabolic disorders marker and migraine symptoms in migraine patients. Cross-over trial studies are required to further confirm these findings.
Key findings
A double-blind, randomized controlled trial was conducted. It was discovered that CoQ10 plus L-carnitine supplements had beneficial effects on migraine symptoms. This study supported the evidence for efficacy of CoQ10 plus L-carnitine supplementation as a prophylactic medication for migraine.
Footnotes
Acknowledgements
The authors would like to thank all participants and their parents. This paper is based on the PhD thesis (No. 396054) approved by the Food Security Research Center, School of Nutrition and Food Science, Isfahan University of Medical Sciences, Isfahan, Iran.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The present study was supported by a grant from the Food Security Research Center, IUMS, Isfahan, Iran.
