Abstract
The objective was to assess the efficacy of coenzyme Q10 as a preventive treatment for migraine headaches. Thirty-two patients (26 women, 6 men) with a history of episodic migraine with or without aura were treated with coenzyme Q10 at a dose of 150 mg per day. Thirty-one of 32 patients completed the study; 61.3% of patients had a greater than 50% reduction in number of days with migraine headache. The average number of days with migraine during the baseline period was 7.34 and this decreased to 2.95 after 3 months of therapy, which was a statistically significant response (P < 0.0001). Mean reduction in migraine frequency after 1 month of treatment was 13.1% and this increased to 55.3% by the end of 3 months. Mean migraine attack frequency was 4.85 during the baseline period and this decreased to 2.81 attacks by the end of the study period, which was a statistically significant response (P < 0.001). There were no side-effects noted with coenzyme Q10. From this open label investigation coenzyme Q10 appears to be a good migraine preventive. Placebo-controlled trials are now necessary to determine the true efficacy of coenzyme Q10 in migraine prevention.
Keywords
Introduction
At present there are very few efficacious migraine preventives and fewer without significant side-effects. In medicine there has been a trend towards the development of natural therapies that can be safely taken by all who suffer with a disease regardless of age and past medical history. Migraine in particular is a disorder of young people and this patient population is very resistant to taking any form of medication, especially when it needs to be used on a daily basis. Migraine can be a disabling disorder and migraine preventives have been shown to not only reduce headache frequency, intensity and duration but also improve quality of life. Coenzyme Q10 is a naturally occurring substance and essential element of the mitochondrial electron transport chain. It has been the most extensively studied agent for the treatment of mitochondrial disorders and has been shown to have almost no identifiable side-effects in humans. There has been a recent interest in the role that mitochondria may play in migraine pathogenesis. Clues from magnetic resonance spectroscopy (MRS) (1) studies and DNA analysis (2) suggest that migraine, at least in a subset of individuals, may be the result of mitochondrial impairment. If indeed migraine results from mitochondrial dysfunction, then coenzyme Q10 could be used as a successful migraine preventive. The objective of this investigation is to assess the efficacy of coenzyme Q10 as a preventive treatment for migraine headaches.
Coenzyme Q10
Coenzyme Q10 is an essential element of the mitochondrial electron transport chain. (Fig. 1) It is a naturally occurring, small hydrophobic substance, that freely moves throughout the inner mitochondrial membrane transferring electrons from the NADH dehydrogenase complex (complex I) and the succinate-Q-reductase complex (complex II) to cytochrome C. In addition to its actions as an electron carrier, coenzyme Q10 may also act as an antioxidant (3) and help protect the myocardium from post-ischaemic re-perfusion injury (4).

Mitochondrial electron transport chain.
The treatment of mitochondrial cytopathies has focused on improving defects in respiratory metabolism. Coenzyme Q10 has been the most extensively studied agent in this setting. Coenzyme Q10 therapy has led to improvement of cardiac conduction defects in a patient with Kearns–Sayre syndrome (5). Muscle weakness and fatigue has been improved in patients with chronic muscular dystrophies and neurogenic atrophies after coenzyme Q10 therapy (6). Coenzyme Q10 administration has improved exercize tolerance, strengthened muscle weakness (7), lowered serum pyruvate and lactate levels, and quickened post-exercize recovery of phosphocreatine (8) in patients with mitochondrial encephalomyopathies. Exogenous coenzyme Q10 also appears to act on the central nervous system directly by causing a marked reduction in CSF lactate and pyruvate levels (9), as well as an improvement in somatosensory evoked potential latencies (8) in patients with mitochondrial cytopathies. Barbiroli et al. (10), utilizing 31P MRS, have identified improvement in both brain and muscle energy metabolism after coenzyme Q10 administration. Coenzyme Q10 has not only been used in the treatment of mitochondrial disorders, but has also found success in the treatment of chronic congestive heart failure (11) and as an aid for myocardial preservation in patients undergoing cardiac surgery.
Coenzyme Q10 can be administered orally or parenterally. Peak blood levels occur 5–10 h after oral administration. Elimination half-life is 34 h. Endogenous coenzyme Q10 levels normally range from 0.7 to 1 mcg/ml and can usually be increased to two times normal levels with oral doses of 100–150 mg per day (12). Koroshetz et al. (13) have shown that oral administration of coenzyme Q10 in adult rats will elevate cortical concentrations by 30%.
In most instances coenzyme Q10 administration has been very well tolerated in doses up to 600 mg per day, with an excellent side-effect profile. The most common side-effects pertain to the gastrointestinal system and include nausea, diarrhoea, appetite suppression, heartburn and epigastric discomfort. In large studies the incidence of gastrointestinal side-effects is less than 1%.
Methods
Thirty-two patients (26 women, 6 men) with a history of International Headache Society (HIS) classified episodic migraine with or without aura were treated with coenzyme Q10 at a dose of 150 mg per day in an open-label fashion. Coenzyme Q10 was given at breakfast time. Study subjects had to have a history of migraine for at least 1 year. Study participants experienced between two and eight migraine attacks per month. The number of days of headache experienced with each individual migraine attack varied from one to several days. No study subject was on preventive therapy 2 months before recruitment for the trial. Prior to involvement in the study each patient was interviewed and examined by a neurologist with experience in headache management. An adequate headache history was ascertained including: age at migraine onset, headache attack frequency, severity, duration, presence or absence of aura and if aura what type (visual, motor, sensory) and normal duration, associated symptoms (nausea, vomiting, photophobia, phonophobia), and family history of migraine. Other questions on personal and family medical history included: history of seizures (age at onset), exercise intolerance, stroke (age at onset), dementia (age at onset), short stature, deafness (unilateral or bilateral, age at onset), learning disabilities, cardiac conduction defects (dysrhythmias), cardiomyopathy/congestive heart failure, diabetes, blindness (age at onset) and motor strength weakness (age at onset).
Total study length was 4 months, with a 1-month baseline period and 3-month therapy phase. A study diary was provided to each study participant. Diary results included assessing each migraine attack for duration (days with headache per migraine attack), severity (0–3 scale) and associated symptoms (nausea, vomiting, photophobia or phonophobia). The diary was started 1 month prior to administration of coenzyme Q10 and continued throughout the study. Laboratory studies included fasting blood levels during the baseline month and after 3 months of coenzyme Q10 administration: CBC with differential and platelets, BUN, creatinine, GGT, lactate and pyruvate levels.
The primary outcome measure for this open label investigation was calculating the percentage of patients who achieved a greater than 50% reduction in number of days with migraine headache after coenzyme Q10 administration. To determine the primary outcome measure we compared the mean number of days with migraine headache during the baseline phase with the mean number of days with migraine headache during the last 2 months of treatment combined. Reduction in migraine attack frequency was also recorded. Secondary outcome measures looked at reduction in migraine headache intensity compared with baseline.
Statistical analysis
An
Results
Thirty-one of 32 patients completed the study. One patient was lost to follow-up. No adverse events were associated with coenzyme Q10 therapy in any of the patients. With regard to the primary outcome measure, 61.3% of the patients treated had a greater than 50% reduction in number of days with migraine headache; 93.5% of the patients had at least a 25% reduction in number of days with migraine. Only two patients showed no improvement with coenzyme Q10 therapy (Table 1). The average number of days with migraine headache during the baseline non-treatment phase was 7.34 and this decreased to 2.95 days by the end of the trial (mean number of days with migraine headache in the last 60 days of treatment), which is a statistically significant response (P < 0.0001) (Fig. 2). The mean reduction in migraine frequency after 1 month of treatment was 13.1% and this increased to 55.3% by the end of 3 months of therapy.
Treatment response

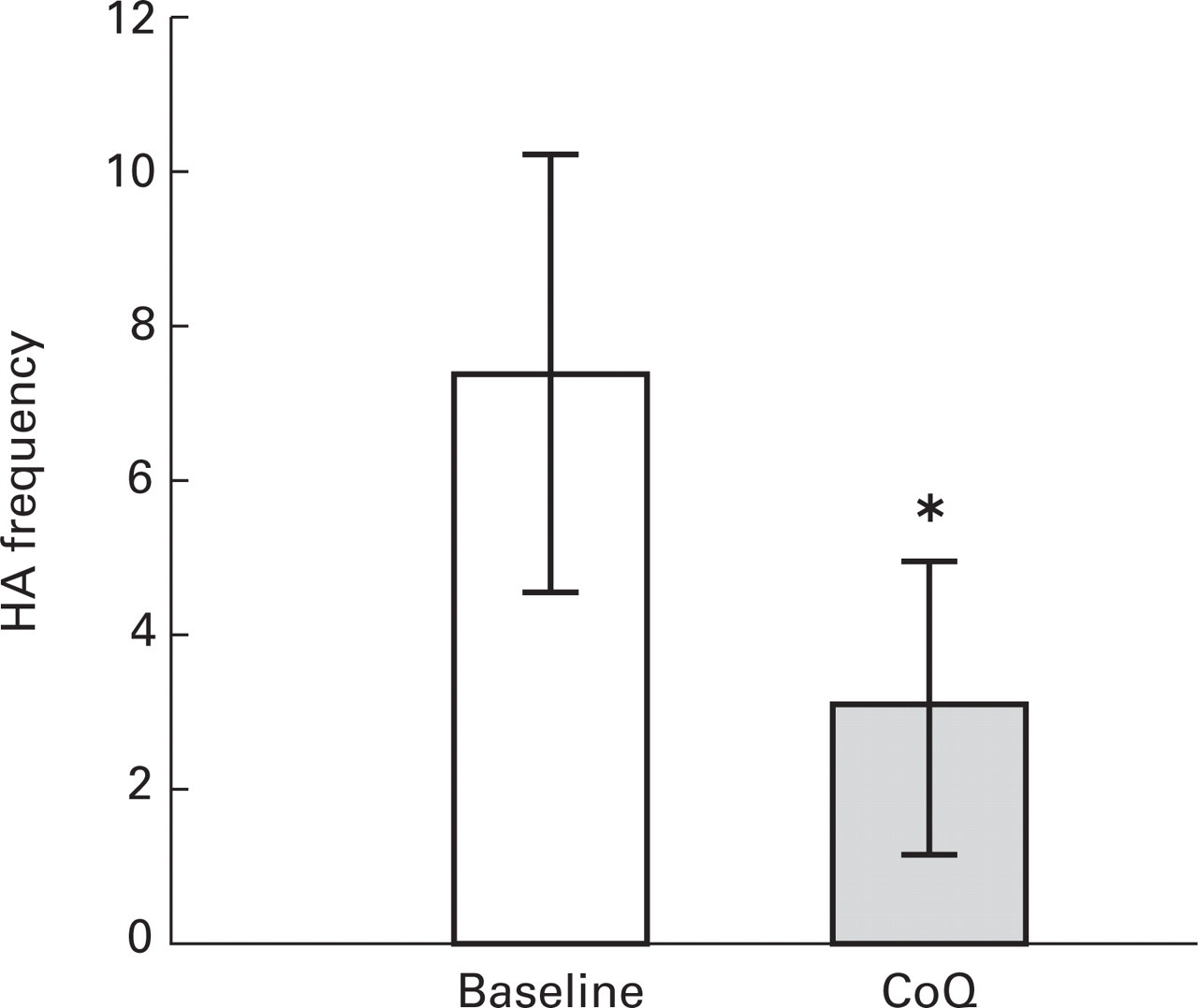
Reduction in days with migraine comparing baseline month and last 2 months of therapy with coenzyme Q10.
Coenzyme Q10 also significantly reduced migraine attack frequency. Mean migraine attack frequency was 4.85 during the baseline period and this decreased to 2.81 attacks by the end of the study period (mean number of migraine attacks in the last 60 days of treatment), which was a statistically significant response (P < 0.001). With regard to secondary outcome measures, coenzyme Q10 did not appear to significantly reduce headache intensity compared with the baseline phase even though total attack days were less. Coenzyme Q10 appeared to work as well in migraine with aura patients as in those without a history of aura. Because of the small number of males in the study and individuals less than age 30 years, we are unable to comment on gender or age group differences in response to coenzyme Q10. All laboratory tests were normal, with no evidence of baseline elevated lactate or pyruvate levels.
Discussion
There has been a recent explosion in the number of new migraine abortive treatments but novel, safe and efficacious migraine preventive therapies have been lacking. Knowledge about migraine pathogenesis has helped in the development of the triptans and a better understanding of migraine pathophysiology will lead to the discovery of newer preventives. There has been recent interest in the role that mitochondria may play in migraine pathogenesis. It is clear from MRS studies that at least a subset of migraineurs have a dysfunction in mitochondrial energy metabolism. Coenzyme Q10 has been shown to improve mitochondrial oxidative phosphorylation in humans. If mitochondrial dysfunction is playing a role in migraine genesis then coenzyme Q10 could improve mitochondrial function and thus prevent migraine headaches. This belief is not without precedence as riboflavin, in an open label pilot study (14) and a placebo controlled trial (15), has been shown to reduce migraine frequency. Riboflavin is indirectly involved in the electron transport chain as a precursor of flavin mononucleotides (FMN). Coenzyme Q10 is an essential element of the electron transport chain, suggesting that it could also work as a migraine preventive. (Fig. 1)
From this open-label investigation coenzyme Q10 appears to be a good migraine preventive. Over 60% of coenzyme Q10-treated patients had a greater than 50% reduction in number of days with migraine headache. Our data suggest that coenzyme Q10 starts to work within 4 weeks of initiation of therapy (mean reduction in days with migraine was 13.1% after 1 month of therapy) but usually takes 5 to 12 weeks to yield a greater than 50% reduction in days with migraine (by the end of 3 months of therapy mean reduction in days with migraine was 55.3%). From this small sample population coenzyme Q10 appears to work equally well in migraineurs with and without a history of aura.
A very important finding from this open label study is that coenzyme Q10 administration appears to be associated with no significant adverse events and is extremely well-tolerated. Placebo-controlled trials are now necessary to determine the true efficacy of coenzyme Q10 in migraine prevention. Dose ranging studies will also be useful. Higher doses of coenzyme Q10 (up to 3000 mg) (16) have been utilized in mitochondrial encephalomyopathy patients without adverse events, so dosages above 150 mg may yield even higher 50% reduction rates than was demonstrated in this study. Coenzyme Q10 looks to be an excellent choice for initial therapy for prevention of episodic migraine if confirmed by controlled studies of efficacy. It can be given to almost any age group without fear of significant side-effects.
Footnotes
Acknowledgement
This study was completed with a MERCK Medical Student Education Grant.
