Abstract
Objectives
Currently, there is no scoring system for assessing the complexity of migraine aura. Our goal was to develop a Migraine Aura Complexity Score that synthesizes the quantity and quality of aura symptoms and to test its applicability in neuroimaging studies.
Methods
Patients with migraine aura were interviewed in order to obtain characteristics of migraine aura. Explorative and confirmatory analyses were used to develop the Migraine Aura Complexity Score. Median values were derived from 10 consecutive migraine auras in each patient. The Migraine Aura Complexity Score was correlated with an average cortical thickness of different brain areas in studied patients. The Surface-based Morphometric Analysis approach was used to estimate cortical thickness.
Results
This study included 23 (16 females and seven males) migraineurs with aura. Confirmatory factor analysis suggested the second-order model with three-factor measurement for grading migraine aura. The first factor is linked to higher cortical dysfunction during migraine aura, while the second is associated with the degree of involvement of primary visual and somatosensory cortices; the third linked symptoms of somatosensory aura and hand and head involvement. Positive correlation of Migraine Aura Complexity Score and averaged cortical thickness were found in the left and right hemispheres overall (r = 0.568, p = 0.007; r = 0.617, p = 0.003) and in some of their regions.
Conclusions
This study demonstrates that the Migraine Aura Complexity Score could be a valuable tool for assessing migraine aura. The score could be used in neuroradiological studies in order to achieve a stratification of patients with migraine aura.
Introduction
Migraine is a well-studied type of primary headache, whose worldwide prevalence has been estimated as 14.7% (1). In the Global Burden of Disease Study 2015, it was ranked as the third-highest cause of disability worldwide in patients under the age of 50 years (2). Of those who suffer from migraine, about 30% have migraine aura (MA) (3), which can manifest very heterogeneously (4). According to the International Classification of Headache Disorders (3rd edition), a typical manifestation of MA includes visual phenomena, followed by somatosensory and language disturbances, but no motor weakness, brainstem or retinal symptoms (5). However, visual phenomena can be multiple, including positive and negative symptoms, and range from simple visual phenomena such as flashes of bright light or zig-zag lines to more complex disturbance of visual perception, such as looking through cracked glass, perceptual distortion of the size or shape of an object or change in color perception (4,6,7). Similar to visual phenomena, somatosensory symptoms can also be positive, such as tingling, negative, such as numbness, or a combination of both; creating “odd” unilateral sensations in the hands, arms, face or tongue (4,5). Dysphasia and other higher cortical dysfunctions (HCDs), such as disturbances of memory, apraxia and ideational agnosia, although less common, are not rare phenomena during aura (8). Longer duration aura may be associated with a significantly increased number of HCDs (8). The symptoms of HCD could contribute to the patient’s disability during an attack.
Magnetic resonance imaging (MRI) studies have suggested altered gray and white matter structures in people with migraine (9,10). Previous studies regarding cortical thickness in migraine patients with aura, however, had found different results with respect to cortical areas involved in the pathophysiology of migraine aura (10–12). We hypothesize that such differences are due to heterogeneity of characteristics of MA of selected patients. It is well known that aura characteristics can vary between patients and also in the same patient (6,8). Such data suggest a variable involvement of different brain regions during different auras. We believe that these data require further research in order to establish an endophenotypes subdivision in patients with MA.
Thus, in order to improve the quality of future neuroradiological studies in MA, it is necessary to perform a stratification of attacks according to their clinical features. The only existing scale, VARS (The Visual Aura Rating Scale), just assesses visual symptoms of MA, and there is no scale for assessing non-visual phenomena of MA (13). Moreover, VARS does not assess the complexity of visual aura but just the likelihood that the visual symptoms are an MA. Indeed, the development and validation of a scoring system for typical MA is difficult due to intra-individual differences in attacks.
Our aim is to estimate the individual quality and quantity of symptoms that occur during MA in each selected patient and try to develop a system for scoring the complexity of a typical aura. We used structural MRI findings of these patients to explore the applicability of this new scoring system in a clinical-neuroradiological association study.
Materials and methods
This study was approved by the Medical Ethics Committee of the Neurology Clinic, Clinical Center of Serbia, and was conducted in accordance with the Declaration of Helsinki. Informed consent forms were completed by all the participants after receiving an explanation of the study.
Participants
All participants had migraine with aura according to the International Classification of Headache Disorders criteria (3rd edition) (5). The inclusion criteria were as follows: a) 21 to 60 years of age, b) the participants suffered from episodic migraine for five years or longer, c) no migraine 72 h prior to the scan and no symptoms of developing one during or 48 h after the MRI scan, d) at least five migraines with aura per year, e) only attacks of a typical migraine with aura (except <2/month migraines without aura and infrequent tension-type headaches), and f) all subjects were right-handed (in order to avoid possible differences of brain regions, such as Broca’s region). The exclusion criteria were: a) patients with T2 hyperintensities on their previous MRI scans, and b) patients treated with a migraine preventive therapy.
Migraine patients included in the study were chosen from a group of 62 patients with migraine with aura who visited the Headache Center, Neurology clinic, Clinical Center of Serbia, from the beginning of 2009 to the end of 2015. From this group, 23 patients met the inclusion/exclusion criteria.
Selected participants were instructed to complete a specific questionnaire developed for this study after each attack of migraine with aura. The questionnaire was filled out one day after the attack to minimize a possible bias of failing to recall symptoms during attacks.
Migraine Aura Complexity Score (MACS)
Study questionnaire.

As there is no definition of complex migraine aura, we decided to use K-means cluster analysis of derived data in order to obtain two groups of patients, the first having complex auras and the second having non-complex auras. Further, regression analysis was used as the regression method to determine significant predictors chosen from the aura characteristics data. We considered dichotomous variable (complex and non-complex aura), derived from K-means cluster analysis, as a dependent variable and all investigated characteristics of the aura as separate predictors. Because of the low frequencies of visual HCDs, we decided to join micropsia, macropsia, dysmorphia, fractured vision and prosopagnosia into one variable (HCDs-V). The same thing has been done with somatosensory HCDs, which included astereognosis, dyspraxia, and unawareness of one’s own body parts (HCDs-S). We also gather all symptoms of dysphasia (Broca’s dysphasia, Wernicke’s dysphasia, and dysnomia) and memory difficulties (difficulties in remembering or recalling events, recalling names, and calculating and/or memorizing numbers) into a single variable (HCDs-D). Items that were shown to be significant predictors were further evaluated with exploratory factor analysis in order to determine a dimension of factors. Also, from the estimated regression coefficients of the positive predictors for the aura complexity, we derived a risk score for each predictive variable as the approximated proportion of the regression coefficients. Next, according to the determined separate factors, a theoretical model for confirmatory factor analysis was constructed and tested in order to test the reliability and validity of this model for scoring the MACS.
MRI data acquisition
MR examinations of patients were performed using a 1.5 T MR scanner with an eight-channel head coil (Signa, General Electric Healthcare, Milwaukee, WI, USA). The imaging protocol consisted of T2 weighted spin echo (T2W) in an axial plane (Echo time (TE) = 105.8 ms, repetition time (TR) = 5700 ms, flip angle (FA) = 90 degrees, 24 slices with 0.47 × 1 × 5 mm2 voxels, slice thickness = 5 mm, acquisition matrix 512 × 512) and three-dimensional T1 weighted fast spoiled gradient-echo (T1-3D-FSPGR) series (TE = 3.60 ms, TR = 8.12 ms, FA = 15 degrees, 248 continuous slices with 0.47 × 0.47 × 1.4 mm voxels, slice thickness = 1.4 mm, acquisition matrix 512 × 512, FOV = 256 × 256 mm2). T2W images were only used to exclude the presence of brain lesions.
Images were transferred to a PC workstation and converted to NifTI-1 (Neuroimaging Informatics Technology Initiative) using the dcm2nii software. Only T1W FFE volumetric images were included in the analysis. In the first step, images were checked for orientation using a fslreorient2std routine from the FSL 5.0 package. Freesurfer (version 5.3.0) analysis was performed on an HP 350 server (Intel Xeon 1800 Mhz, eight cores, 16 GB RAM) using a recon-all script for automatic cortical reconstruction and segmentation of brain structures. Average run time (with the parallelization option used) was 6 h. Average run time (with the parallelization option used) was 6 h. Details about Freesurfer and its routines can be found elsewhere (14,15). After finalization of the script, segmentation of the brain structures was checked using the tkmedit routine and final surfaces were built using the reconall2 script. After these 3D surfaces were reconstructed, cortical thickness was estimated as the shortest distance from the white matter surface to the pial surface at each surface vertex. Cortical parcellation was done according to the Desikan-Killiany Atlas (16).
Statistical analysis
Subject demographics and MA characteristics were reported using descriptive statistics. The K-means cluster analysis was applied in order to obtain clusters that distinguish participants according to their symptoms during the aura. Median values of the scored items were analyzed in the AMOS (version 21) in order to perform confirmatory factor analysis to test model reliability and validity. Median values were used because the scale is not necessarily linear in all participants. Also, we used ROC curve analysis (95% Confidential Interval) to find a cut-off value of the MACS score that can distinguish between patients who have a complex aura and those without complex manifestation of migraine aura.
The mean cortical thickness of mapped cortical regions (such as occipital lobe – cuneus, pericalcarine cortex, lingual cortex, lateral occipital cortex; parietal lobe – precuneus, superior parietal cortex, inferior parietal cortex, postcentral cortex, supramarginal cortex; temporal lobe – superior, middle and inferior temporal cortex, fusiform cortex, parahippocampal cortex; and frontal lobe – pars triangularis and opercularis) were extracted from surface-based morphometry results and exported into the R statistics program. The regions were chosen as potentially linked, from a functional point of view, to the symptoms that develop during the aura. Mean cortical thickness of these regions in migraineurs was correlated with the MACS. We used a partial correlation test, controlled for the effect of age and sex to avoid spurious results. The results were corrected for multiple comparisons using a Bonferroni correction with a significance level of p < 0.0016 (p < 0.05/32).
Results
The study included 16 females and 7 males suffering from a migraine with typical aura, aged 38.65 ± 11.4 years. The average disease duration was 19.57 ± 9.9 (range: 5–37) years. All patients had visual aura; 15 (65%) patients had visual and somatosensory auras; and 13 (57%) patients had visual, somatosensory and dysphasic auras.
Visual, somatosensory and higher cortical dysfunctional symptoms during the aura.
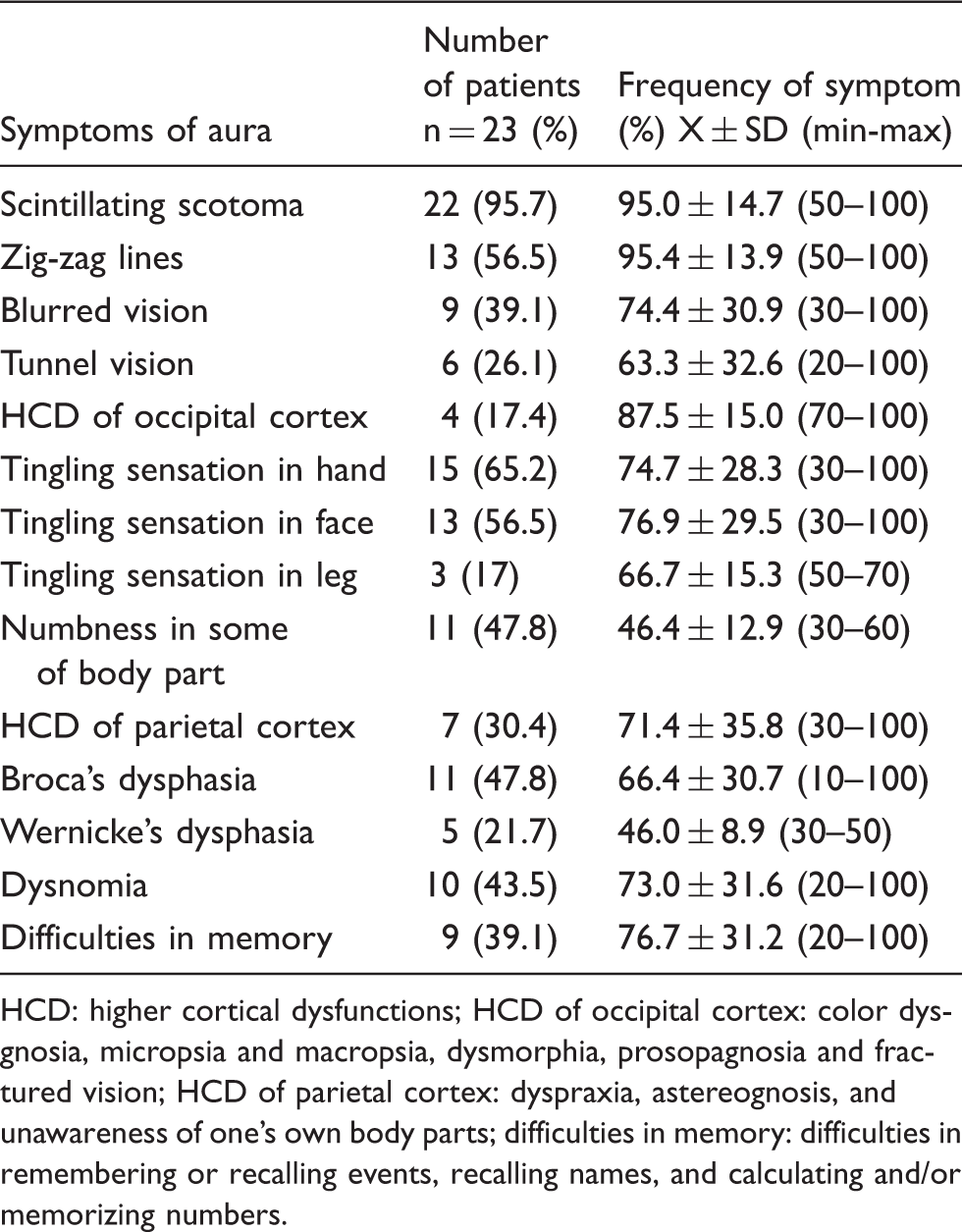
HCD: higher cortical dysfunctions; HCD of occipital cortex: color dysgnosia, micropsia and macropsia, dysmorphia, prosopagnosia and fractured vision; HCD of parietal cortex: dyspraxia, astereognosis, and unawareness of one’s own body parts; difficulties in memory: difficulties in remembering or recalling events, recalling names, and calculating and/or memorizing numbers.
The average duration of the aura was 41.96 ± 28.9 (range: 15–120) minutes. One patient had prolonged visual symptoms (lasting for more than 60 minutes), while another patient had both somatosensory and dysphasic prolonged symptoms.
Migraine Aura Complexity Score
Significant predictors for complex aura.

Visual field significantly affected: more than half visual field is affected by visual aura; all body regions significantly affected: whole surface of primary somatosensory cortex is affected by cortical spreading depression; HCDs-V: micropsia, macropsia, dysmorphia, fractured vision and prosopagnosia; HCDs-S: astereognosis, dyspraxia, and unawareness of one’s own body parts; HCDs-D: dysphasic symptoms (Broca’s dysphasia, Wernicke’s dysphasia, and dysnomia) and memory disturbances (difficulties in remembering or recalling events, recalling names, and calculating and/or memorizing numbers).
After we identified significant predictors, we then used explorative factor analysis to investigate their structure. The Kaiser-Meyer-Olkin measure of sampling adequacy was 0.729, which satisfied the criteria for factor analysis. Three factors were derived with cumulative squared loadings of 91.05%. Factors were derived using Oblimin with the Kaiser Normalization rotation method because of positive correlations between components. The first factor (Cronbach’s alpha = 0.859) contains the following items: HCDs-V, HCDs-S, and the presence of dysphasic and/or memory disturbance symptoms (HCDs-D). The second factor (Cronbach’s alpha = 0.977) contains the following items: “visual field significantly affected” and “all body regions significantly affected”. The third factor (Cronbach’s alpha = 0.908) contains the following items: “somatosensory symptoms affecting hand” and “somatosensory symptoms affecting head”. In synthesis, the first factor is linked to dysfunctions of higher cortical functions during the aura, while the second factor is associated with significant involvement of the visual and somatosensory primary cortex and the third factor is associated with body regions that are usually affected by somatosensory aura.
The second-order latent factor model considering the MACS was tested. According to the fit indices employed (GFI: Goodness-of-fit index; NFI: Normed Fit Index; CFI: Comparative fit index; RMSEA: root mean square error of approximation; PCLOSE: p-value for testing the null hypothesis that the population RMSEA is no greater than 0.05), CFA indicated that the second order proposed model (Figure 1) was an acceptable fit on any of the fit indices employed (CMIN (df) = 7.171 (11), p = 0.785, GFI = 0.924, NFI = 0.950, CFI = 1.000, RMSEA = 0.000, PCLOSE = 0.816). As shown in Figure 2, all factor loadings were in the expected direction and had significant regression weights with their related latent variables (critical ratio > 1.96), indicating convergent validity. The inter-factor correlation (correlation between the three latent variables, not shown in Figure 2) between Higher cortical symptoms and Whole primary cortical surface affected by aura (0.428), Higher cortical symptoms and Basic aura characteristics (0.581), and Whole primary cortical surface affected by aura and Basic aura characteristics (0.344) indicated acceptable discriminant validity (<0.85).
Schema of the Migraine Aura Complexity Score (MACS). The second-order latent factor model contains the standardized factor loadings that were derived from estimating the study model for the Migraine Aura Complexity Score (MACS).

Average Migraine Aura Complexity Scores and subscores from 10 consecutive migraine with aura attacks.

patients who were assigned into the group that had complex aura.
N: patient number; MACS: migraine aura complexity scores from 10 consecutive migraines with aura attacks displayed as median and mean values with standard deviations; HCDs: subscores of higher cortical dysfunctional features from 10 consecutive migraines with aura attacks displayed as median (Med) and mean values with standard deviations (SD); primary cortex: subscores of significantly affected primary cortical areas from 10 consecutive migraines with aura attacks displayed as median and mean values with standard deviations; body regions: subscores of involvement of hand and head by somatosensory aura from 10 consecutive migraines with aura attacks displayed as median and mean values with standard deviations.
Relationships between average thickness of cortical regions and the MACS.

Significant after Bonferroni correction.
L: left hemisphere; R: right hemisphere; MACS: Migraine Aura Complexity Score.
Discussion
In the present study, we describe the characteristics of typical MA and proposed a Migraine Aura Complexity Score system (MACS). The MACS is a simple rating scale that could serve as a tool for a stratification of patients affected by migraine with aura into subgroups. Moreover, our findings revealed that MACS has a positive correlation with cortical thickness in the multiple regions of the left and right hemispheres.
MA presents a very heterogeneous phenomenology, not just among patients but also in the same patient (4,6). Because of that, it is difficult to investigate the pathophysiology of MA in a group of migraineurs. Our previous investigations demonstrated that there are several differences among patients with MA, in terms of duration, quality, degree of involvement of aura symptoms and neuroradiological findings (8,17,18). In order to stratify patients with MA, and to explore connections between the complexity of MA and structural changes in the cortex or other neuroradiological investigations, we decided to develop the MACS. The MACS can be applied to one attack or can be averaged using multiple scores of one patient, with the aim of getting an impression of the overall complexity of the MA of that patient. We think that development of this type of scoring system is a useful tool for further neuroradiological studies, in order to improve investigations of the pathophysiology of MA.
Previous studies showed a wide range of MA features, from short-lasting auras with only visual symptoms to long-lasting auras with visual and somatosensory symptoms and HCDs (complex aura) (6,8). However, there was no attempt to develop a scoring system that measures the complexity of migraine aura. Thus, there is no “gold standard” for measuring the complexity of the aura. The MACS was developed on the basis of the clinical data reported by patients who experienced a migraine with typical aura in different forms and levels of complexity. Cluster analysis was used to define homogeneous groups of individuals based on given parameters (19). In this way, we identified patients who had complex aura and then investigated characteristics of migraine aura as potential predictors of aura complexity. Our findings showed that the qualities of visual disturbances were not significant predictors of the complexity of an aura. On the other hand, the characteristics that were shown to be significant predictors of complex aura were used to develop the MACS. After we applied explorative and confirmatory factory analysis, the model for the MACS was developed and validated. Indeed, this model should be cross-validated in future independent studies with a larger number of participants. The strength of the MACS is that it is derived from the dataset of 230 carefully investigated attacks of MA in 23 patients, who were free from other comorbidities and without a migraine preventive therapy that could have influenced investigation.
The MACS is influenced by three factors. The first factor (HCDs) is derived from the presence of visual, somatosensory, dysphasic or memory disturbances during the aura, which is an important part of the MACS because it evaluates an aura’s influence on higher cortical functions in migraine patients. The second factor (related to involvement of primary cortices) is made by the degree of involvement of the visual field and body areas, and therefore to the degree of involvement of the surface of primary visual and somatosensory areas, which is another important aspect when evaluating migraine aura complexity. The third factor (involvement of hand and head by somatosensory aura) is made by summing somatosensory symptoms that involved hand and/or head/face, which indeed are the most common ones, after visual symptoms, in the MA (3,4). Combing these three factors into the MACS score gives a model that better assesses the complexity of the aura than just one of these domains. Moreover, all three domains give strong predictions of the complexity of migraine aura, only in different, but complementary ways. Thus, the MACS can range from 0 points (i.e. a patient who has a visual aura that involves a quarter of the visual field) to 9 points (i.e. a patient who has a visual aura that involves more than half of the visual field accompanied by hemiparesthesia, dysmorphia, dyspraxia, dysphasic and memory disturbance symptoms). The cut-off value in the MACS for distinguishing those patients who have complex aura is 4.50 points, which can be useful in creating groups for investigation of the complexity of migraine aura. However, there are some patients with notable variability across attacks and for those patients average scores could lead to possible bias, which is not the case in our study when we look at average scores and predictions of complex aura. In further studies, what number of attacks is sufficient to estimate someone’s aura should be evaluated. Also, it should be considered that those with significant variability across attacks could be stratified into a separate group, or MACS could be applied to them to investigate the influence of some treatment on the manifestation of their auras.
Furthermore, MACS was used to explore the connection with the cortical thickness of regions, derived from the Desikan-Killiany Atlas, which have been linked to MA. The analysis identified several cortical regions of the occipital and parietal lobes within both hemispheres, where the cortex was thicker in those patients who had higher MACS. This result could be explained by better-developed connections between occipital and parietal lobes, which allows spreading of cortical spreading depression from the occipital lobe to the parietal lobe (20,21). Also, changes to the cortical thickness in the left and right superior parietal cortices suggest a role for these areas in manifestations of the somatosensory disturbances and HCDs during the aura. Moreover, averaged cortical thicknesses of the left and the right hemispheres show a positive correlation with the MACS. These findings are in line with the results of previous studies that demonstrated higher cortical thickness in migraineurs who have aura (9,22).
We did not find any significant correlation between cortical thickness in the frontal regions and the MACS, which implies a less significant role for the frontal lobe in the pathophysiology of migraine aura. This finding is opposite to expectations that changes in the pars opercularis could be responsible for language impairment during an aura and question the role of Broca’s region in dysphasic aura (23–25).
Limitations
In our study, patients had to answer the questionnaire the day after an MA attack. Indeed, a questionnaire to be fulfilled during the aura would have provided more reliable data, avoiding the recall bias. Yet, answering the questionnaire just one day after the MA attack reduced recall bias with respect to answering the same questions after many days – that is, at the follow-up visit. Moreover, answering the questionnaire after each MA attack (out of 10) has guaranteed that patients had described every single attack instead of reporting general information regarding several MAs.
Conclusion
Our results show that the newly developed Migraine Aura Complexity Score could be a valuable tool for assessing the complexity of the aura. MACS could be used for stratification of patients with MA in order to better investigate changes in migraineurs’ brains, both structurally and functionally, as well as for prognostic purposes in the evaluation of a treatment response.
Clinical implications
Clinical investigations of migraine aura require stratification of patients according to their aura phenomenology and, so far, a scoring system for this purpose is not available. We here propose to evaluate the complexity of migraine aura with a Migraine Aura Complexity Score (MACS) that takes into account several aspects of aura such as the presence and quality of visual, somatosensory, dysphasic symptoms and other higher cortical disturbances. The MACS total score exhibited positive correlations with neuroimaging findings of cortical thickness in patients, among which the left and right lateral occipital, right cuneus, right precuneus, left postcentral, and left and right superior parietal cortices were the most significant. MACS could be a valuable tool for assessing the complexity of the aura, for stratification of migraineurs into subgroups, and potentially to serve as a prognostic tool of therapy outcome.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: IP, MV, MD and JTZ have no conflict of interest. PJG reports, unrelated to this study, grants and personal fees from Allergan, Amgen, Eli-Lilly and Company, eNeura Inc and personal fees from Alder Biopharmaceuticals, Avanir Pharma, Cipla Ltd, Dr Reddy’s Laboratories, Electrocore LLC, Novartis, Pfizer Inc, Quest Diagnostics, Scion, Teva Pharmaceuticals, and Trigemina Inc., personal fees from MedicoLegal work, Journal Watch, Up-to-Date, Oxford University Press, Massachusetts Medical Society, and Wolters Kluwer; and a patent Magnetic stimulation for headache assigned to eNeura without fee.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: IP and MD received research grant support from the Ministry of Education and Science, Republic of Serbia (project no. III 41005), and JZT received research grant support from the Ministry of Education and Science, Republic of Serbia (project no. 175022).
