Abstract
Objective
We aimed to explore cortical thickness abnormalities in a homogeneous group of patients with migraine without aura and to delineate possible relationships between cortical thickness changes and clinical variables.
Methods
Fifty-six female migraine patients without aura and T2-visible white matter hyperintensities and 34 female controls were scanned on a 3T magnetic resonance imager. Cortical thickness was estimated and compared between patients and controls using a whole-brain vertex-by-vertex analysis. Correlation analysis was conducted between cortical thickness of significant clusters and clinical variables.
Results
Compared to controls, migraine patients had cortical thickening in left rostral middle frontal gyrus and bilateral post-central gyri. Region-of-interest analysis revealed cortical thickening of bilateral post-central gyri in migraine patients relative to controls. The average thickness of bilateral post-central gyri positively correlated with disease duration as well as estimated lifetime headache frequency.
Conclusions
We have provided evidence for interictal cortical abnormalities of thickened prefrontal cortex and somatosensory cortex in female migraine patients without aura. Our findings of greater thickening of the somatosensory cortex in relation to increasing disease duration and increasing headache frequency suggest that repeated migraine attacks over time may lead to structural changes of the somatosensory cortex through increased noxious afferent input within the trigemino-thalamo-cortical pathway in migraine.
Introduction
Remarkable advances in computational analysis of multimodal neuroimagings have offered an opportunity to explore brain regions or networks that are likely to be involved in generation and mediation of migraine headache, thus contributing greatly to our understanding of both functional and structural changes of migraine (1). Recent morphometric magnetic resonance imaging (MRI) studies using voxel-based morphometry (VBM), the most frequently used tool to evaluate structural changes of the brain in a fully automated, objective, operator-independent manner (2), have repeatedly shown grey matter (GM) atrophy in brain regions that are involved in central pain networks (3–5). Given their findings of greater GM atrophy in relation to increasing disease duration or headache frequency, such GM alterations could be interpreted as progressive brain damage to the pain processing areas as a consequence of repeated migraine attacks.
An alternative approach to quantify morphometric changes of cortical GM is the measurement of cortical thickness, a relatively new and well-validated analytic procedure (6,7). It can provide complementary information to VBM in understanding the neuroanatomy of various brain disorders by allowing the regional distribution and quantification of cortical GM changes to be specifically examined in contrast to VBM, which often combine GM and white matter (WM) within regional volumes (6). Although VBM does permit precise assessment of GM changes, VBM is limited by the fact that it provides a mixed measurement of GM, including surface area, cortical folding and cortical thickness. In addition, VBM is considered to be less robust to noise and mis-segmentation, less accurate due to the limited resolution of the voxel grid and affected by partial volume effects at the boundaries of highly convoluted structures, such as deep sulci (8). In contrast, cortical thickness analysis allows a more direct index of cortical morphology that is less susceptible to positional variance given that the extraction of the cortex follows the GM surface despite local variations in its position, enabling a more sensitive measurement of cortical morphological changes (9). Recently, several studies have evaluated cortical GM changes using cortical thickness analysis in patients with migraine relative to healthy controls. However, the results showed considerable variance across the studies in which cortical structures are affected and in whether the cortical GM abnormality is associated with an increase or a reduction in cortical thickness (10–14). Moreover, another study failed to replicate such cortical abnormalities in either migraine with or without aura (15).
In the present study, we aimed to explore cortical thickness abnormalities in a large homogeneous group of migraine patients. To this end, we prospectively recruited only female patients with migraine without aura, who had no WM hyperintensities (WMH) on T2-weighted magnetic resonance (MR) images. We predicted that migraine patients had cortical thickness abnormalities in cortical regions involved in central pain network as compared to healthy controls and that these changes correlated with clinical variables representative of disease severity.
Methods
Subjects
Patients were prospectively recruited from the migraine population who were followed in the outpatient clinic of the Department of Neurology at Korea University Guro Hospital from January 2012 to December 2012. During the study period, only four male patients who met the following inclusion criteria were recruited. Given a highly disproportionate number of patients according to sex (56 females vs. four males) and the well-known sex differences in cortical thickness in healthy subjects (16,17) as well as migraineurs (13), only female subjects were included to avoid possible sex-related bias. Patients were eligible for inclusion if they met the following criteria: (1) episodic migraine without aura based on the criteria established by the International Headache Society (ICHD-II) (18); (2) 18–55 years old; (3) normal neurological examination; (4) no WMH on T2-weighted and fluid attenuated inversion recovery (FLAIR) images. Exclusion criteria that we used were the following: (1) pregnant or breast-feeding women; (2) claustrophobia or any MRI incompatibility; (3) hypertension, diabetes mellitus, ischaemic heart disease, chronic liver disease or other chronic systemic disorders; (4) cerebrovascular disease, epilepsy, dementia, mental retardation or other proven neurological or psychiatric disorders; (5) chronic alcohol consumption or substance abuse. Since cortical volume or excitability can be affected by valproate (19) or topiramate (20), the widely used medications for migraine prevention, patients who were on or had a history of taking any prophylactic medications were further excluded. Demographic data and the following clinical characteristics were obtained from the patients: age of onset; disease duration; maximal pain intensity measured by a 4-point scale (0, none; 1, mild; 2, moderate; 3, severe); dominant side of headache attack (left, right or alternating); frequency of migraine attacks recorded in a 3-month headache diary before the MRI examination; estimated lifetime headache frequency (frequency of migraine attacks during 3 months × 4 × disease duration in years).
Thirty-four age-matched, right-handed healthy volunteers (all females, mean age 34.2 years, age range 18–51 years) were prospectively recruited during the study period. All control subjects had extremely few (less than a few days per year) spontaneous non-throbbing headaches, no family history of migraine, no history of alcohol and substance abuse, chronic pain disorders, systemic, psychiatric or neurological disorders. Neurological examination was normal in all participants. This study was approved by the local ethics committee and written informed consent was obtained from all subjects prior to study inclusion.
MRI acquisition
MRI data were acquired on a Siemens Trio 3T scanner (Erlangen, Germany) with a 12-channel phased array head coil. All patients had been free from a typical migraine attack for at least 72 h prior to and 24 h after the MRI scanning. For volumetric analysis, a high-resolution 3D MP-RAGE sequence was acquired with the following parameters: TR = 1780 ms, TE = 2.34 ms, matrix = 256 × 256, FOV = 256 × 256 mm, voxel size = 1 mm3. For identification of structural abnormalities, the following MR images were acquired: axial T2-weighted and FLAIR images (4 mm thickness); oblique coronal T2-weighted and FLAIR images perpendicular to the long axis of the hippocampus (3 mm thickness). The MR images of all participants were reviewed by a board-certified neuroradiologist (S.I.S.) for any structural abnormalities and reported as normal without any WMH. Diffusion tensor imaging and resting-state functional MRI data were acquired simultaneously but not included in the current analysis.
MRI data pre-processing and cortical thickness analysis
Data pre-processing and subsequent cortical thickness analysis were performed using FreeSurfer image analysis suite (version 5.1, http://surfer.nmr.mgh.harvard.edu/). Automatic segmentation enables labelling cortical and subcortical tissue classes using an atlas-based Bayesian segmentation procedure (21). Briefly, the automated procedures included removal of non-brain tissue, automated Talairach transformation, segmentation of the subcortical WM and deep GM volumetric structures, intensity normalization, tessellation of the GM–WM boundary, automated topology correction and surface deformation. The last procedure is accomplished by following intensity gradients to optimally place the GM/WM and GM/cerebrospinal fluid borders at the location where the greatest shift in intensity defines the transition to the other tissue class (6). All images were visually inspected for accuracy by an experienced neuroradiologist (S.I.S.), who was blinded to the subject’s diagnosis. A first visual inspection was done after the skull stripping to detect skull tissues not successfully removed and a second inspection was necessary after the GM/WM segmentation to visualize possible tissue misclassification. In six patients and three control subjects, minor manual corrections were performed by editing out the skull tissue and adding control points to correct topological defects, according to FreeSurfer guidelines (http://surfer.nmr.mgh.harvard.edu/fswiki). The distance between the WM surface and pial surface yields an estimate of cortical thickness at each vertex. Automated cortical parcellation was conducted using the Desikan–Killiany parcellation atlas as provided in FreeSurfer (22). The reconstructed cortical surfaces for each participant were then aligned to produce an average cortical surface. A mapping was obtained between each vertex on the average surface and the corresponding vertex on the surface of each subject’s cortical reconstruction. The cortical thickness maps for each subject were then resampled onto the average surface and smoothed with a 15-mm full width at half maximum Gaussian kernel to improve inter-subject averaging.
Statistical analysis
A general linear model was used to test for cortical thickness differences between migraine patients and controls at each vertex while controlling for the effect of age. The statistical surface maps were cluster-wise corrected for multiple comparisons using Monte Carlo simulation with 10,000 permutations and the p value for each cluster was determined. Only clusters that survived this correction were considered statistically significant (p < 0.05). Average cortical thickness was then measured for each cluster that survived the correction for multiple comparisons and plotted for correlation analysis. Linear correlation analysis was performed between average cortical thickness of each cluster and clinical variables such as disease duration, 3-month headache frequency and estimated lifetime headache frequency (partial correlation accounting for the effect of age, p < 0.05).
In order to confirm the results of whole-brain cortical thickness analysis, we further performed region-of-interest (ROI)-based analysis. The average cortical thickness of 34 cortical regions for each hemisphere was computed according to the Desikan-Killiany’s parcellation atlas (22). This parcellation method was shown to be valid and reliable, with high intraclass correlation coefficients between the manual and automated procedures for volume measurement of 34 cortical regions. Data were first tested for normality of distribution and homogeneity of variance assumption using Kolmogorov–Smirnov test and Levene’s test, respectively. In variables where the assumption of normality was not met, a natural log transformation was applied to conform the data to a normal distribution. Prior to final analysis, interactions between the dependent variables and the covariate were checked by using a custom model, which included group and age as main effects and the group × age interaction. When there was no significant age × group interaction (p > 0.05), a full factorial model was chosen. Group comparisons between migraine patients and controls were made using analysis of covariance, adjusting for the effect of age. Bonferroni’s correction was further applied and statistical significance was set at p < 0.0015 (0.05/34). Statistical analyses were performed with the Statistical Package for Social Sciences (Version 19.0; IBM, Armonk, New York).
Results
Clinical characteristics
Clinical characteristics and cortical thickness measurement in migraine patients and controls.
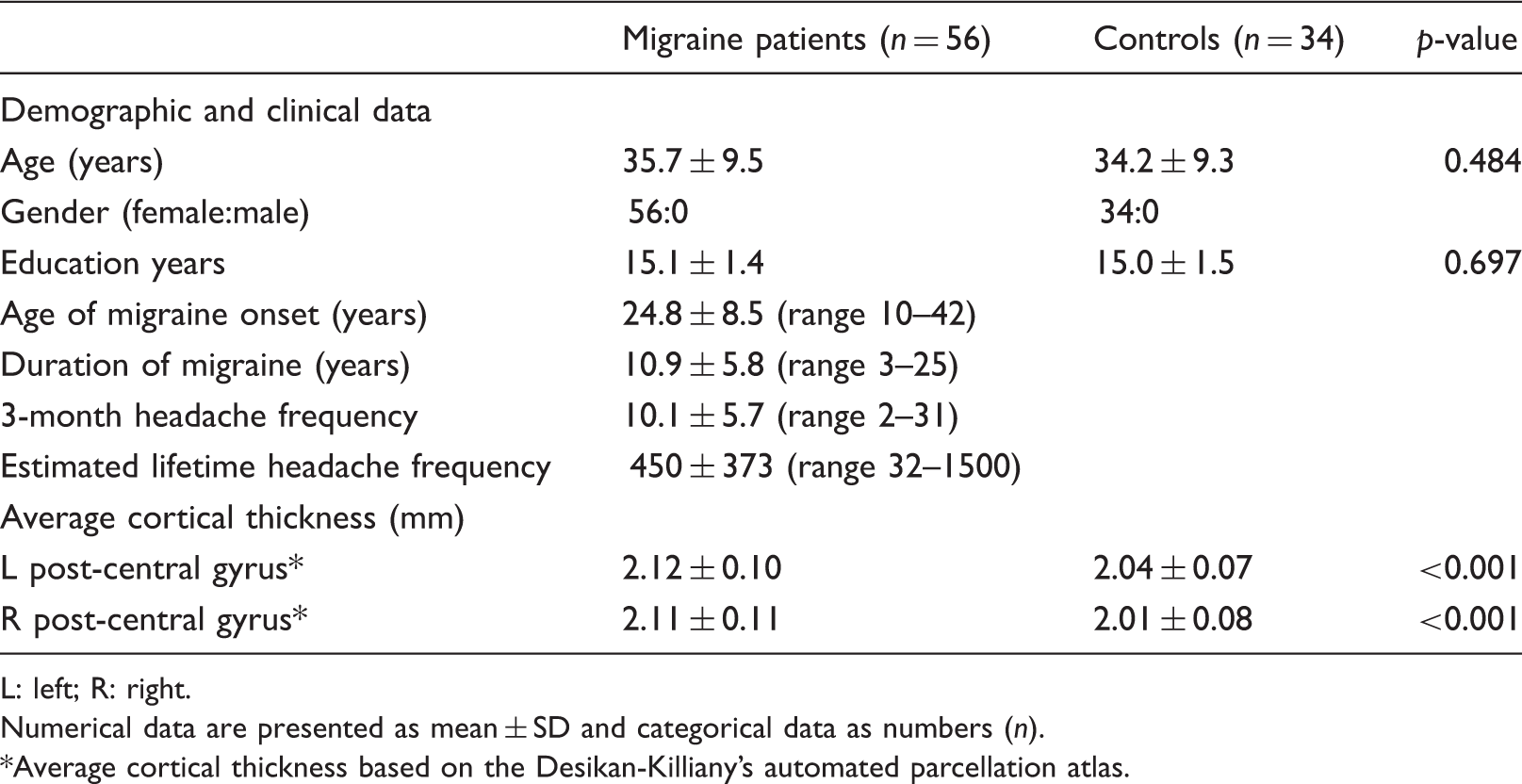
L: left; R: right.
Numerical data are presented as mean ± SD and categorical data as numbers (n).
Average cortical thickness based on the Desikan-Killiany’s automated parcellation atlas.
Whole-brain cortical thickness analysis
Compared to control, migraine patients had significant cortical thickening in left post-central gyrus (Montreal Neurological Institute (MNI) 305 coordinates = −32.2/−34.4/63.9, cluster size = 1045 mm2, cluster-wise p (CWP) = 0.0007), left rostral middle frontal gyrus (MNI coordinates = −21.8/54.0/7.6, cluster size = 593 mm2, CWP = 0.0315) and right post-central gyrus (MNI coordinates = 51.5/−15.0/39.5, cluster size = 743 mm2, CWP = 0.0098) (Figure 1). There was no cortical region of significant interaction between age and diagnostic group (p > 0.05). No region of significant cortical thinning was found in patients relative to controls at the same statistical threshold.
Statistical maps rendered on the standard pial cortical surface showing cortical areas of significant thickening (red) in left rostral middle frontal gyrus and post-central gyrus (a) and right post-central gyrus (b) in migraine patients compared to controls. Only clusters surviving a cluster-wise probability of p < 0.05, corrected for multiple comparisons using Monte Carlo simulation (10,000 permutations), are displayed. The colour bar scale is logarithmic and represents –log10(p): 1.3 corresponds to p value of 0.05 and 5 to p value of 0.00001.
Average cortical thickness of 34 cortical regions for each hemisphere was computed based on the Desikan-Killiany’s automated parcellation atlas and compared between patients and controls. Compared to controls, patients had significant increases in average cortical thickness of left post-central gyrus (F[2,87] = 13.850, mean square error = 0.007, p < 0.001) and right post-central gyrus (F[2,87] = 19.724, mean square error = 0.008, p < 0.001) (Table 1). There was no significant interaction term of age × group in average cortical thickness of the left post-central gyrus (p = 0.755) and right post-central gyrus (p = 0.350). Average thickness of the remaining 33 cortical regions for each hemisphere did not differ between the groups (data not shown).
Correlation analysis
Partial correlation controlling for the effect of age showed that disease duration in years of migraine patients positively correlated with average thickness extracted from the left post-central cluster (r = 0.465, p < 0.001) and right post-central cluster (r = 0.369, p = 0.006), but not with that of left rostral middle frontal cluster (p > 0.05) (Figure 2). Headache frequency recorded in a 3-month headache diary did not correlate with average cortical thickness of left rostral middle frontal cluster and both post-central clusters (all p > 0.05). Estimated lifetime headache frequency positively correlated with average thickness of left post-central cluster (r = 0.310, p = 0.021) and right post-central cluster (r = 0.298, p = 0.027) (Figure 2).
Scatter plot graphs of correlation analysis showing significant positive correlations between disease duration and average cortical thickness of left (a) and right (b) post-central clusters (p < 0.05, partial correlation controlling for the effect of age). Estimated lifetime headache frequency also positively correlated with average cortical thickness of left (c) and right (d) post-central clusters (p < 0.05, partial correlation controlling for the effect of age).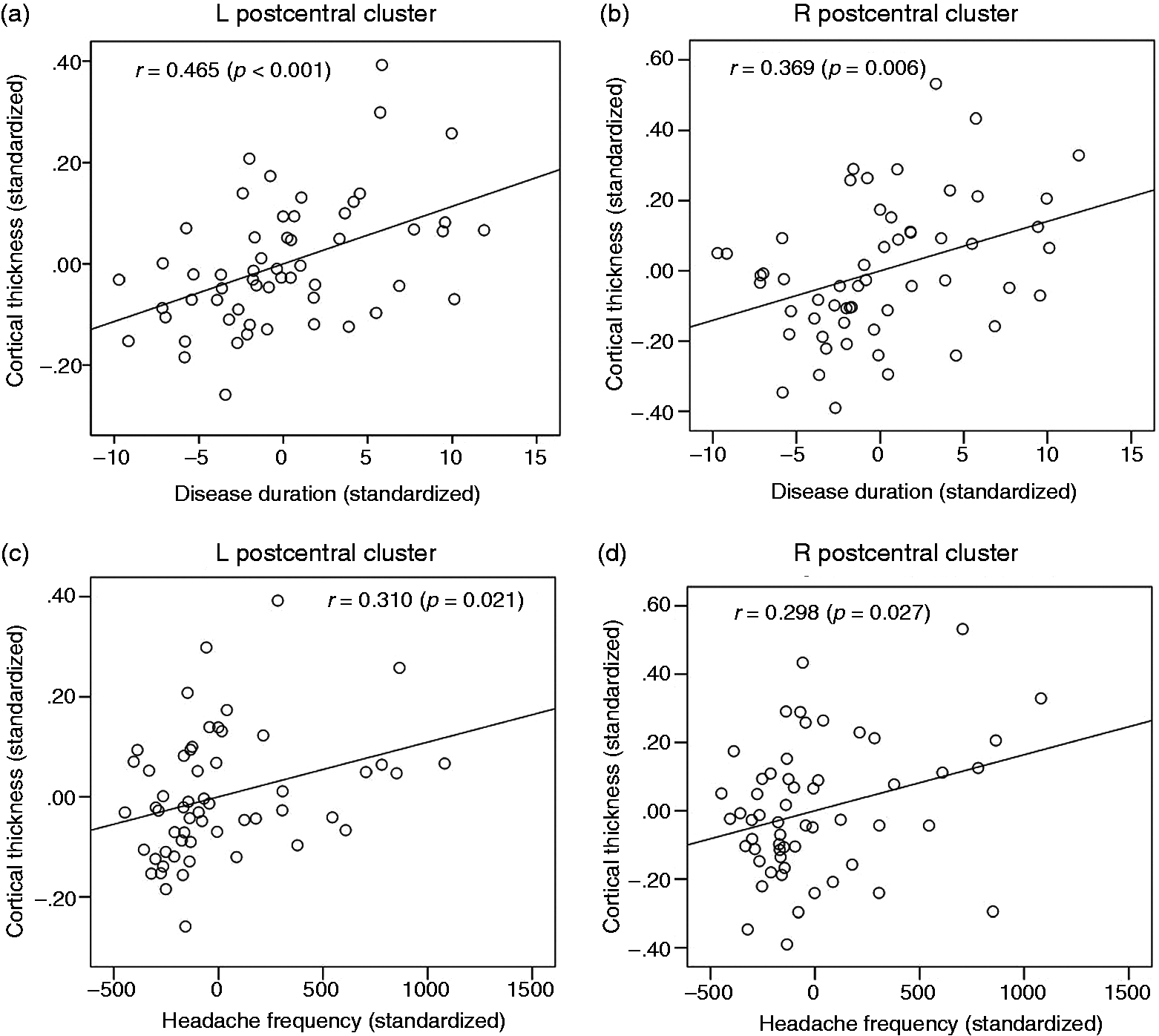
Discussion
We attempted to explore cortical thickness changes in a homogeneous group of patients with migraine without aura and to delineate relationships between anatomical changes and clinical variables. We found significant thickening of left middle frontal cortex and bilateral somatosensory cortex (SSC) in migraine patients compared to controls. Cortical thickness of bilateral SSC positively correlated with disease duration and estimated lifetime headache frequency.
There are, to our knowledge, six published studies that investigated cortical thickness alterations in a migraine population (10–15). A pioneering study using a ROI-based approach reported significant cortical thickening in the motion processing areas, namely, V3A and MT+, in migraine patients with and without aura compared to controls (10). This cortical abnormality could account for the exaggerated excitability of neurons in these motion processing areas and establish neuroanatomical correlates of classical visual aura and motion perception deficits that have frequently been observed in migraine patients (23,24). A subsequent study performed by the same group found that migraineurs had a thicker SSC, especially in the caudal regions representing head and face, which may be related to increased noxious stimulation within the somatosensory system (11). This could be either the consequence or cause of repetitive migraine attacks, although no correlation was found between the SSC thickness and disease duration or headache frequency (11). A recent study recruiting a larger patient cohort, however, failed to replicate such cortical thickness changes in both migraine patients with and without aura relative to controls (15). More recently, Maleki and collaborators found that patients with frequent migraine attacks had cortical thickening in the area representing the face in the post-central gyrus as compared to those with less frequent attacks and controls, which correlated well with the stronger functional activation of the SSC in response to noxious thermal stimulation, suggesting an adaptive response to repeated migraine attacks (12). The same group also reported that, compared to controls, migraineurs had cortical thickening in the posterior insula and precuneus that are known to be involved in pain perception and sensorimotor or associative/cognitive processing, respectively (13). It is particularly intriguing that female migraineurs had thicker cortex in the posterior insula and precuneus compared with both male migraineurs and controls, implicating sex-related differences in cortical morphology in a migraine population (13). In a recent study, migraineurs were found to have both increases and decreases in cortical thickness in multiple areas that are thought to be involved in the pain processing network (superior frontal gyrus, precentral gyrus) and visual motion processing (temporo-occipital cortex) as well as executive functions (middle frontal gyrus) (14). The inconsistent findings between the above-mentioned studies could not be properly explained but might, in part, be attributed to genetic heterogeneity, different sample size, differences in MRI parameters (e.g. pulse sequences, manufacturer, upgrade) (7), differences in analytic approach, statistical threshold and selection of covariates in the statistical model adopted in each study. Clinical complexities and confounders within the patient population, such as disease duration (short vs. long duration of migraine), different migraine subgroups (patients with aura vs. those without aura, high frequency vs. low frequency attacks) and sex difference (female vs. male) may also account for these inconsistencies between the studies.
Our results from the whole-brain level analysis accord well with those of the previous study using a ROI-based measurement (11), corroborating that migraine patients do have a thickening of the SSC, specifically the caudal part where the trigeminal area, including head and face, is somatotopically represented. The primary SSC is known to play a prominent and highly modulated role in the sensory aspects of pain, including localization and discrimination of pain intensity (25). Involvement of primary SSC in migraine has been suggested in magnetoencephalographic and somatosensory evoked high-frequency oscillations studies, indicating hyperexcitable primary SSC in migraine patients with frequent attacks (26,27). Furthermore, our findings of greater SSC thickening in relation to increasing disease duration and estimated lifetime headache frequency further support the notion that repetitive noxious stimuli involving the face and head in the form of migraine headache may lead to structural changes of the SSC by means of increased afferent activity within the trigemino-thalamo-cortical pathway in migraine. Our view could be supported by the recent study that found thicker SSC in migraine patients with high attack frequency than those with low attack frequency (12), implicating SSC thickness as a potential biomarker for the disease progression or severity in migraine.
We also observed cortical thickening of left rostral middle frontal gyrus, part of the medial prefrontal cortex, in patients relative to controls. Our findings are in line with those of previous studies showing both anatomical and functional changes of the prefrontal cortex (5,14,28–30), further suggesting the involvement of the prefrontal cortex in migraine. The prefrontal cortex is generally accepted to be involved in cognitive evaluation and attention modulation of pain as well as mediating attenuation of pain perception through cognitive control mechanisms (31). Moreover, several lines of evidence suggest hemispheric lateralization of pain modulation by the prefrontal cortex. The right prefrontal cortex is known to be related to attentional aspects of pain and involved in cognitive modulation of pain based on reappraisal strategy (31,32), while the left prefrontal cortex is hypothesized to exert antinociceptive effects through top-down influences on the supraspinal opioidergic circuit (33). Taken together, our finding of a thickened left prefrontal cortex might be interpreted as dysfunction of inhibitory control over nociceptive transmission. However, this implication about the observed lateralization in prefrontal morphological change remains only speculative. Further investigation is required to determine the specific contribution of left prefrontal cortical thickening to migraine pathophysiology.
One central question that remains to be answered is whether such cortical changes are the cause or the consequence of migraine headache. We assume that thickening of the SSC observed in our patients may occur as a consequence of repeated migraine attacks, which could be supported by the following observations. First, a number of activation studies using positron emission tomography and functional MRI have highlighted the role of brainstem structure, not the SSC, in the generation of migraine headache, indicating the brainstem as the key region specific to migraine (1). Second, it is noteworthy that such thickening of the SSC has also been observed in other forms of chronic pain disorders, including trigeminal neuralgia (34), temporomandibular disorder (35) and chronic low back pain (36). Moreover, a significant positive correlation was found between the SSC thickness and disease duration in patients with cluster headache, a distinct headache syndrome from migraine (37). In a recent study on healthy subjects, Erpelding and collaborators found a strong correlation between the SSC thickness and thermal and pain sensitivity, indicating that individuals who are highly sensitive to pain have thicker SSC (38). Third, human brains may have activation-dependent cortical plasticity on a structural level. Indeed, previous elegant studies have shown that juggling training (39,40) and painful stimulation (41) induced transient increases of GM in the visual area and SSC, respectively. Furthermore, recent studies infer that morphological GM changes are the consequence and not the cause of chronic pain, as structural changes reverse after cessation of pain with effective treatment (42,43). Taken together, we speculate that thickening of the SSC observed in our study is not the primary cause of generating migraine headache, but might reflect adaptive or secondary changes of the cortical GM as a consequence of repeated migraine attacks. Future longitudinal studies may help disentangling this issue.
Several limitations of our study should be addressed. First, the sample size of the control group was smaller than that of the patient group. Second, our patients were recruited from an outpatient clinic of a university-affiliated hospital rather than from the community-based patient population. It is therefore possible that they were not representative of the general migraine population. Moreover, only female migraine patients without aura and T2-visible WMH were included in our study. Given the possible differences in cortical thickness between male and female patients (13), between migraine patients with aura and those without aura (11,14) and between patients with WMH and those without (14), our findings cannot be generalized to the entire migraine population and should be restricted to female migraine patients without aura and WMH. Third, the possible influence of rescue medications for acute migraine attacks (triptans, ergotamine-caffeine preparations) could not be entirely discounted, although any study evaluating the effects of these drugs on brain structures is currently lacking.
In conclusion, we have provided evidence for interictal GM abnormalities of thickened prefrontal cortex and SSC in female migraine patients without aura. Our findings of greater thickening of the SSC in relation to increasing disease duration and increasing migraine attack frequency suggest that repeated migraine attacks over time may lead to structural changes of the SSC through increased noxious afferent input within the trigemino-thalamo-cortical pathway. Given the structural plasticity in response to chronic pain, future study incorporating a longitudinal design is required to establish the issue of whether the observed SSC thickening could be reversed with effective treatment in migraine.
Clinical implications
Our data indicate that female migraine patients without aura have interictal cortical grey matter changes of thickened prefrontal cortex and somatosensory cortex as compared to control subjects. Our findings of greater thickening of the somatosensory cortex in relation to increasing disease duration and increasing headache frequency suggest that repeated migraine attacks over time may lead to structural changes of the somatosensory cortex through increased noxious afferent input within the trigemino-thalamo-cortical pathway in migraine.
Footnotes
Acknowledgements
The authors are very grateful to the participants for taking part in the present study.
Funding
This work was supported by the National Research Foundation of Korea Grant funded by the Korean Government (grant number 2012007757).
Conflict of interest
None declared.
