Abstract
Previous studies on pressure pain sensitivity in patients with migraine have shown conflicting results. There is emerging evidence suggesting that pain sensitivity is not uniformly distributed over the muscles, indicating the existence of topographical changes in pressure pain sensitivity. The aim of this study was to calculate topographical pressure pain sensitivity maps of the temporalis muscle in a blind design in patients with strictly unilateral migraine compared with controls. For this purpose, an electronic pressure algometer was used to measure pressure pain thresholds (PPT) over nine points of the temporalis muscle: three points in the anterior, medial and posterior parts, respectively. Pressure pain sensitivity maps of both sides (dominant or non-dominant; symptomatic or non-symptomatic) were calculated. The analysis of variance showed significant differences in PPT values between both groups (F = 279.2; P < 0.001) and points (F = 4.033; P < 0.001). Patients showed lower PPT at all nine points than healthy controls (P < 0.001). We also found lower PPT in the centre of the muscle compared with the posterior part of the muscle within both groups (P < 0.01). Interaction between group and points (F = 1.9; P < 0.05) was also found. Within the migraine group, PPT levels were decreased bilaterally from the posterior to the anterior column of the temporalis muscle (Student-Newman-Keuls analysis; P < 0.05), with the most sensitive in the anterior part of the muscle. For controls, PPT did not follow such anatomical distribution, the most sensitive point being the centre of the mid-muscle belly. This study showed bilateral sensitization to pressure in unilateral migraine, suggesting the involvement of central components.
Introduction
Migraine is a common disorder with a lifetime prevalence of 16% worldwide, and a last-year prevalence of 10% (1, 2). Despite major advances in the understanding of migraine pathophysiology, including activation of the trigeminovascular system (3) and hyperexcitability of the nociceptive pathways within the central nervous system (4), a number of unresolved issues persist. The sensitization of central pathways induces increased muscle tenderness (5, 6) and cutaneous allodynia (7–10). This may be corroborated by a decrease in pressure pain thresholds, which have been demonstrated in tension-type headache (TTH) (11, 12), but not in migraine patients (13). Previous studies comparing pressure pain sensitivity in patients with migraine and healthy subjects have used a single standardized point in the anterior part of the temporalis muscle. However, it is not known whether pain sensitivity is uniformly distributed over the whole muscle. In fact, spatial changes in mechanical pain sensitivity may be present throughout the cephalic region in headache patients.
Nie et al. (14) found an increased mechanical pain sensitivity of muscle belly locations of the upper trapezius muscle compared with the sensitivity of musculo-tendinous junctions, indicating that mechanical pain sensitivity is not uniformly distributed over the muscle. We have recently investigated topographical pressure sensitivity maps of the temporalis muscle as a method to standardize mechanical pain sensitivity of cephalic muscles (15). In patients with chronic TTH, pain sensitivity maps showed a decrease of pain thresholds from the posterior to the anterior part of the muscle on both sides of the head, whereas among healthy controls pressure pain distribution maps were inhomogeneous within each muscle and between sides (15).
In the present study, we hypothesized that patients with strictly unilateral migraine might show unilateral hypersensitivity in pressure pain threshold maps of the temporalis muscles.
Material and methods
Subjects
Fifteen women diagnosed with migraine, aged 25–59 years (mean age ±
Pressure pain sensitivity mapping
The study procedure was established following previous guidelines (15). Patients were not allowed to take analgesics or muscle relaxants through the 24 h prior to the examination. The evaluation was held when all migraine patients were headache free, and when ≥ 1 week had elapsed since the last migraine attack to avoid migraine-related allodynia (17, 18).
Participants were asked to take a side-lying position on an examination bed with a suitable pillow under the head. Nine points over both temporalis muscles were marked with a wax pencil. The ear of each subject was taken as the anatomical reference point. The vertical line of the ear defined as the central column was considered as the centre of the muscle belly. In this way, three vertical points separated by 15 mm were marked. These three points (labelled 2, 5 and 8) were used to define the anterior and posterior columns. The points located in the anterior part of the muscle (labelled 3, 6 and 9) were located 10 mm anterior to each respective vertical point, whereas the points located in the posterior part (labelled 1, 4 and 7) were located 10 mm posterior to each respective vertical point (Fig. 1).

Schematic representation of the nine points for pressure pain threshold assessment in the temporalis muscle.
The pressure pain threshold (PPT) was assessed on each point. For that purpose, a pressure algometer (Somedic®, Hörby, Sweden) with a 1-cm2 rubber-tipped plunger mounted on a force transducer was used. PPT was defined as the minimal amount of pressure where a sense of pressure first changes to pain (19). The participant pushed a button to stop the pressure stimulation when the threshold was reached. Pressure was applied at a rate of 30 kPa/s. Three PPT measurements were taken at each point with a 15-s interval between two consecutive points and 2.5 min between two identical locations, with randomization in the order of the points' assessment, by an investigator blinded to the subject's condition. The mean of the three measurements was computed for each point and each subject. Group-averaged values over the locations were interpolated using an inverse distance weighted interpolation (20) for each side in both groups.
Statistical analysis
Results are expressed as means and standard deviations (
Results
Clinical characteristics of the migraine sample
Within the patient group, migraine was always strictly unilateral. Eight patients (55%) reported attacks on the right side, and the remaining seven (45%) on the left side. Headache attacks had typical features of migraine without aura, and were severe and invalidating. Ten patients recalled a positive family history of migraine in first-degree relatives. Migraine history ranged from 5 to 20 years (mean duration 12 ± 6 years). The frequency of headache attacks was one to four attacks per month (mean ±
Pressure pain sensitivity map of the temporalis muscle
The intra-examiner repeatability of PPT readings ranged from 0.93 to 0.96 in patients, and from 0.95 to 0.97 in controls, suggesting high repeatability of PPT data. The standard error of measurement (
The three-way

Pressure pain threshold (PPT) levels (average of both sides) for each point in either the patient or control group. Data are expressed as mean and standard deviation. ∗Significant lower PPT for the migraine group compared with the control group in all the points. #Significantly lower PPT in the centre of the muscle belly compared with points in the posterior part of the muscle in both groups (for location of points see Fig. 1).
A significant interaction between group and points (F = 1.9; P < 0.05) was also found. In migraine patients, PPT levels decreased from the posterior to the anterior column of the temporalis muscle (SNK; P < 0.05). The most sensitive points were those located in the anterior column (points 3, 6 and 9), followed by those located in the central (points 2, 5 and 8), and the posterior columns (points 1, 4 and 7) of the temporalis muscle. In contrast, PPT levels did not follow any anatomical distribution in healthy subjects: the most sensitive point was at the centre of the mid-muscle belly (point 5) on both sides, whereas the sensitivity of the remaining points was distributed differently on each side. Figure 3 shows pressure pain sensitivity maps on both sides (symptomatic or non-symptomatic; dominant or non-dominant) in each group, and Table 1 summarizes mean PPT values for each point and side in both groups.
Pressure pain thresholds (kPa) of each point over the temporalis muscle on each side in patients with unilateral migraine and controls

∗Significant differences between patients and controls.
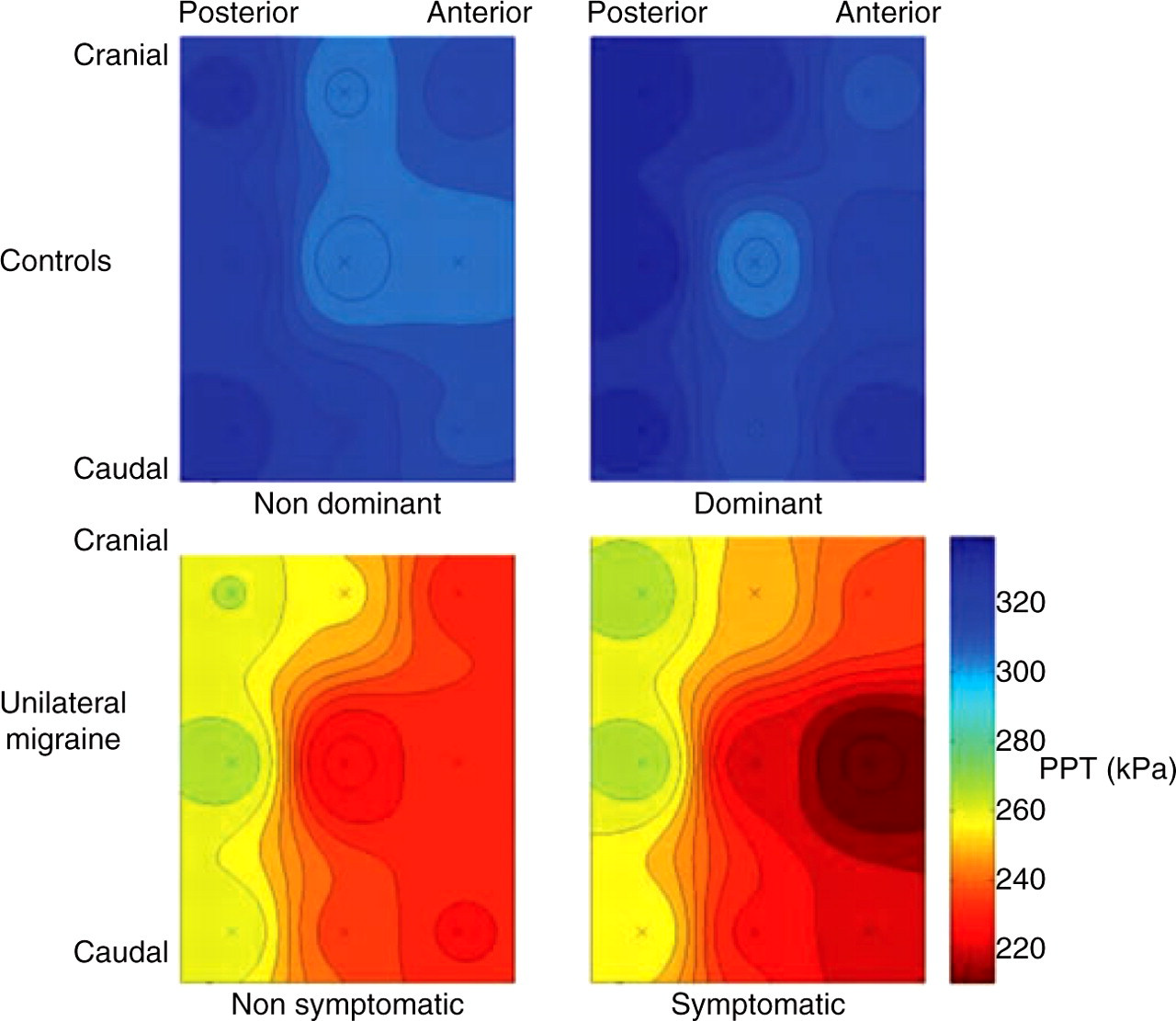
Average pressure pain threshold (PPT) maps for migraine patients and controls for both sides. ‘X’ represents the location of the measured points (see Fig. 1).
Discussion
The present study applied topographical pressure sensitivity maps to assess the distribution of pain sensitivity in the temporalis muscle in patients with strictly unilateral migraine. We found that patients with unilateral migraine had lower pressure pain thresholds compared with healthy subjects in all the analysed points. Within the patient group, pain sensitivity maps were characterized by an anterior–posterior pressure pain threshold gradient on both symptomatic and non-symptomatic sides, whereas in healthy controls pain sensitivity maps did not follow any spatial distribution, with the mid-muscle belly being the most sensitive point to pressure.
Previous studies investigating pressure pain sensitivity in patients with migraine and TTH have reported conflicting results. Some studies have found lower PPT in TTH (11, 12, 15), whereas others have found no significant differences between patients with migraine or TTH and healthy subjects (13, 21). The discrepancies between the former results and our findings may be due to differences in the assessment procedure. For example, most previous studies used a standardized point in the anterior part of the temporalis muscle for PPT measurements. According to our results, the anterior column of the temporalis muscle appears to be the most sensitive part in patients with unilateral migraine or TTH (15), but not in healthy controls. Future studies on muscle hyperalgesia should not be limited to a particular point, since the spatial distribution of pressure pain thresholds seems to be inhomogeneous between headache patients and healthy subjects.
Contrary to our hypothesis, patients with strictly unilateral migraine showed a similar distribution on both sides of the head. This was unexpected, since our patients had a unilateral distribution of their symptoms. Bilateral pressure pain sensitivity maps in unilateral migraine patients were similar to those maps previously found in patients with chronic TTH (15) and are consistent with bilateral pressure hyperalgesia in the cephalic region in both types of headache. A different distribution of nociceptors between headache patients and controls in both temporalis muscles could be responsible for these findings, but this is unlikely. Central sensitization most probably accounts for bilateral hyperalgesia in unilateral pain syndromes, as previously suggested (22, 23). Burstein has proposed that hyperalgesic and allodynic responses in migraine were due to the development of central sensitization involving second-order neurons in the trigeminal nucleus (that receive convergent information from the meninges, the scalp and the face) and at least third-order neurons in the thalamus (that process information from the whole body) (4, 24). In this process, it seems that the hyperexcitability of the central nervous system is a dynamic condition, which may be influenced by the activity of peripheral nociceptive inputs (25). However, further studies investigating the relationship between pain symptoms and pain sensitivity maps are needed.
This study has some limitations in terms of sample size, but the low deviation of the PPT in both study groups supports that a greater sample size would not significantly change our findings, with the exception of possible side-to-side differences in controls. In fact, our former study on pressure pain sensitivity maps in TTH found lower PPT on the non-dominant side within the control group (15). This could be due to an effect of age (26), as on average the age of controls was higher in the present study. Despite the low deviation of our data, extrapolation of the present results to the general migraine population should be done with caution. Population-based epidemiological studies with larger samples are needed with a view to permitting a more generalized interpretation of these results. Otherwise, muscle trigger points were not searched for. Muscle trigger points are hypersensitive spots within a taut muscle band that elicit a referred pain upon examination (27), and the decrease in PPT could be potentially related to the presence of trigger points in the temporalis. In addition, we only investigated pressure pain sensitivity maps of the temporalis muscle. It would be interesting to describe pressure pain sensitivity maps of other cranio-cervical muscles, particularly those with non-trigeminal nociception whose nerve inputs may be relevant for migraine, e.g. upper trapezius or sternocleidomastoid muscles (28). Finally, we have described the spatial distribution of pressure pain sensitivity only in patients with unilateral migraine or TTH. An assessment of pressure pain sensitivity maps in other unilateral headaches, e.g. trigeminal autonomic cephalalgias, may expand current knowledge about the state of nociception in headache disorders.
Conclusions
Unilateral migraine patients had bilateral pressure hyperalgesia in the temporalis muscle compared with controls. Headache patients showed a gradual decrease of PPT from the posterior to the anterior part of the temporalis muscle, whereas PPT maps in healthy subjects showed no such gradient. The most sensitive locations were the anterior part of the temporalis muscle in the patient group and the centre of the mid-muscle belly in healthy subjects. Further studies on the spatial distribution of muscle hypersensitivity may provide relevant information about pain perception in headache disorders.
Footnotes
Acknowledgements
This study received funds from a grant conceded by Universidad Rey Juan Carlos and Comunidad de Madrid (reference CCG07-URJC / BIO-1420).
