Abstract
Spatial changes in pressure pain hypersensitivity are present throughout the cephalic region (temporalis muscle) in both chronic tension-type headache (CTTH) and unilateral migraine. The aim of this study was to assess pressure pain sensitivity topographical maps on the trapezius muscle in 20 patients with CTTH and 20 with unilateral migraine in comparison with 20 healthy controls in a blind design. For this purpose, a pressure algometer was used to assess pressure pain thresholds (PPT) over 11 points of the trapezius muscle: four points in the upper part of the muscle, two over the levator scapulae muscle, two in the middle part, and the remaining three points in the lower part of the muscle. Pressure pain sensitivity maps of both sides (dominant/non-dominant; symptomatic/non-symptomatic) were depicted for patients and controls. CTTH patients showed generalized lower PPT levels compared with both migraine patients (P = 0.03) and controls (P < 0.001). The migraine group had also lower PPT than healthy controls (P < 0.001). The most sensitive location for the assessment of PPT was the neck portion of the upper trapezius muscle in both patient groups and healthy controls (P < 0.001). PPT was negatively related to some clinical pain features in both CTTH and unilateral migraine patients (all P < 0.05). Side-to-side differences were found in strictly unilateral migraine, but not in those subjects with bilateral pain, i.e. CTTH. These data support the influence of muscle hyperalgesia in both CTTH and unilateral migraine patients and point towards a general pressure pain hyperalgesia of neck-shoulder muscles in headache patients, particularly in CTTH.
Introduction
Headache is the most prevalent neurological disorder and experienced by almost everyone (1). Among headaches, tension-type headache represents the most common type with 1-year prevalence rates of 38.3% for the episodic form and 2.2% for the chronic form (2). In addition, the prevalence of tension-type headache has increased in recent years (3). The second most common headache, migraine, has a lifetime prevalence of 16% worldwide, and a last-year prevalence of 10% (4). Migraine and tension-type headache may cause substantial levels of disability not only to the individual patient and their family but also to the entire global society due to very high prevalence in the general population (5). Globally, the percentage of the adult population with a headache disorder is 47% for headache in general, 10% for migraine, 38% for tension-type headache and 3% for chronic headache that lasts for > 15 days/month (6).
Furthermore, there has been increasing interest in the pathogenic mechanisms of these disorders within recent decades, but knowledge is scant, despite some advances in aetiology. It seems clear that hyperexcitability of nociceptive pathways plays an important role in both headaches (7,8). The sensitization of these pathways causes increased muscle tenderness in tension-type headache (9,10) and cutaneous allodynia in migraine patients (11–13). Furthermore, these manifestations of sensitization are corroborated by increased mechanical pain sensitivity. Previous studies have found a decrease in pressure pain thresholds (PPTs) in chronic tension-type headache (CTTH) (14–16) However, the results on PPTs in migraine populations are contradictory, since some studies have reported lower values in patients compared with controls (17,18), whereas others have not (9,19,20).
These conflicting results in migraine may be related to the following aspects. First, previous studies have included patients with bilateral or side shifting migraine (9,17–20). A recent study from our group has demonstrated side-to-side differences in mechanical pain sensitivity in patients with strictly unilateral migraine (21), with significant variations between sides that reveal that the side of stimulation can be highly relevant. Muscle tenderness was mostly related to hyperaesthesia of the following specific muscles, i.e. suboccipital, temporalis, sternocleidomastoid, rather than generalized muscle tenderness akin to that present in both tension-type headache forms (21). These findings suggest that mechanical sensitivity in migraine can be a more complex clinical picture than expected, because of lateralization of pain sensitivity.
A second aspect to explore is that previous studies investigating pain sensitivity in patients with migraine or tension-type headache have used a single standardized point in the anterior part of the temporalis muscle (9,14–20). However, it is known that pressure pain sensitivity is not uniformly distributed over this muscle, since spatial changes in mechanical pain sensitivity are present throughout the cephalic region in both CTTH (22) and unilateral migraine patients (23). The anterior column of the temporalis muscle has been found to be the most sensitive part as depicted by pain sensitivity maps in strictly unilateral migraine and CTTH (22,23), but not in healthy matched controls.
Work-related musculoskeletal disorders are often accompanied by pain in the neck-shoulder region (24) and headache (25). An overlap of trigeminal and upper cervical spinal afferent fibres in the caudal components (trigeminal nucleus caudalis) of the trigeminal brainstem complex and in the C1–C3 segments has been described in animals (26). This has also been corroborated by electrophysiological findings revealing convergence of trigeminal and cervical afferents onto both nociceptive and non-nociceptive neurons in trigeminal subnucleus caudalis in animals (27). Moreover, this is also supported by data reporting convergence between afferents from trapezius and temporalis muscles (28). Thus, there is a need for studies focusing on the relationship between headache and neck-shoulder muscles (29). Some studies have reported similarities between neck-shoulder pain and tension-type headache (30,31). Non-uniform distribution of pain sensitivity has also been reported in the shoulder region (32,33). Nevertheless, topographical distribution of pressure sensitivity maps over the trapezius muscle has not been previously investigated in primary headaches. The aim of this study was to investigate mechanical pain sensitivity distribution over the trapezius muscle in patients with CTTH and strictly unilateral migraine compared with matched controls.
Materials and methods
Subjects
Twenty women diagnosed with CTTH, aged 25–53 years (mean 39 ± 8 years), 20 women diagnosed with migraine, aged 23–55 years (mean 37 ± 9 years) and 20 healthy women (age matched) acting as controls, aged 27–54 years (mean 37 ± 8 years) with no history of head or neck-shoulder pain participated in this study. Participants with previous whiplash or neck trauma were excluded. No significant differences between groups for age were found (F = 0.27; P > 0.7). All subjects were right-handed (dominant side). Patients were recruited from the Neurology Department of the Fundación Hospital Alcorcón, an urban hospital, whereas controls were recruited from the general population by advertisement in local newspapers.
Patients were diagnosed following the International Headache Society (IHS) criteria (34). Headache features, temporal profile, family history and medications were ascertained from the clinical history. To be included within the CTTH group, patients had to describe all the characteristics typical of this headache condition: bilateral location, pressing and tightening pain, mild or moderate intensity (≤ 6 on 11 points of the numerical pain rate scale) and no aggravation of headache during physical activity. In addition, patients had to have headaches for at least 15 days/month. None of the patients reported photophobia, phonophobia, vomiting or nausea during attacks. Each patient fulfilled the criteria for CTTH, and there was no evidence of secondary headaches. Patients completed a headache diary for 4 weeks in order to complement the diagnosis (35). To be included in the migraine group, patients had to present the following features typical of migraine attacks: unilateral location, pulsating quality, moderate or severe pain intensity, photophobia, phonophobia, vomiting or nausea during headache and pain aggravation during physical activity. Furthermore, patients had to describe strictly unilateral headache attacks without side shift. Other primary headaches were excluded. A neuroimaging examination (magnetic resonance imaging or computed tomography) of the head was performed in all patients in order to exclude other disorders. Medication overuse headache as defined by the IHS (34) was ruled out. Patients whose headache was related to previous neck trauma were also excluded (34). All patients had received several prophylactic drugs, but none of them was taking any migraine preventive at the time the study was performed. Ethical approval was granted by the Local Ethics Committee (URJC/FHA 028). Informed consent was obtained from all participants, and procedures were conducted according to the Declaration of Helsinki.
Pressure pain thresholds
PPT was assessed from 11 locations in a blind design. For that purpose, an electronic pressure algometer (Somedic® Algometer type 2, Sollentuna, Sweden) with a 1-cm2 rubber tipped plunger mounted on a force transducer was used. PPT is defined as the minimal amount of pressure where a sense of pressure first changes to pain (36). The subject pushed a button to stop the pressure stimulation when the threshold was reached. The instrument was calibrated at the start of each session. Pressure was applied at a rate of 30 kPa/s. Three PPT measurements (intra-examiner reliability) were taken at each point with a 15-s interval in between two consecutive measurements over the same point, with randomization in the order of the points' assessment. The distance among adjacent points and the elapsed time between consecutive PPT recordings prevented spatial and temporal summation (37). Averaged values over the 11 locations were interpolated using an inverse distance weighted interpolation (38) for each side in each group.
Trapezius muscle mapping
The procedure was the same for patients and controls. Patients were not allowed
to take analgesics or muscle relaxants through the 24 h prior to the
examination. Within the migraine group, evaluation was held when all patients
were headache free and when at least 1 week had elapsed since the last migraine
attack to avoid migraine-related allodynia (11,12). CTTH patients were
examined on days in which headache intensity was < 4
points on a 10-cm horizontal visual analogue scale. Subjects were asked to take
a prone position on an examination bed. The following 11 points according to Nie
et al. (32) over the upper, middle and lower trapezius muscle, in both
dominant and non-dominant sides, were marked with a wax pencil: (1) occiput: at
the suboccipital muscle insertions; (2) cervical muscle: transverse process of
C5; (3) cervical myotendinous spot: transverse process of C7; (4) upper
trapezius: middle point between the spinous process of C7 and the acromion; (5)
levator scapulae: 2 cm superior to the superior angle of the
scapulae; (6) superior angle of the scapulae; (7) 1 cm medial to the
acromion-clavicular joint; (8) supraspinatus: 3 cm superior to the
middle of spina of the scapulae; (9) supraspinatus: 2 cm distal to
the middle of spina of the scapulae; (10) middle trapezius: middle point of
spinous process of T4 and medial border of spina scapulae; and (11) lower
trapezius: middle point of spinous process of T6 and medial border of spina
scapulae (Fig. 1). Schematic representation of the 11 points for pressure pain threshold
assessment.
The examiner located the C7 spinous process by the following procedure. First, he palpated the most prominent spinous process (C6 or C7) at the base of the cervical spine. After it was identified, he passively flexed and extended the lower cervical spine to verify which one moved first: C6 vertebra should be more mobile, whereas C7 should demonstrate less motion.
Sample size determination
Sample size determination and power calculations were performed (Tamaño de la Muestra, 1.1©, Madrid, Spain). Calculations were based on detecting at least clinically significant differences of 20% on PPT levels between the groups (39) with an α level of 0.05 and a desired power of 80%. This generated a sample size of at least 16 participants per group.
Statistical analysis
Mean ± standard deviations
(
Results
Clinical characteristics of the sample
Within the CTTH group, headache history was 10.2 years (95% CI 6.9, 13.4). The mean headache period per day was 5.5 h (95% CI 3.9, 7.1), the mean intensity per episode was 5.4 (95% CI 5.1, 5.6) and the number of days per week with headache was 5.2 (95% CI 4.9, 5.6). Mean headache intensity during the examination was 2.5 (95% CI 2.1, 2.8). Headache intensity was positively associated with the frequency of the headaches (r = 0.47; P = 0.04).
Within the migraine group, nine patients (45%) reported attacks on the right side and the remaining 11 (55%) on the left. Headache attacks had typical features of migraine without aura, and were severe and incapacitating. Sixteen patients recalled a positive family history of migraine in first-degree relatives. Migraine history was 12 years (95% CI 8.8, 15.3). All patients were classified as having episodic migriaine, since they reported two to three attacks per month (2.6 ± 0.7 attacks/month).
Pressure pain sensitivity map of the trapezius muscle
The intra-examiner repeatability of PPT readings ranged from 0.89 to 0.94 in migraine patients, from 0.90 to 0.93 in CTTH patients, and from 0.9 to 0.95 in controls, suggesting high repeatability of the PPT data. The standard error of measurement ranged from 5.6 to 6.7 kPa, depending on the assessed point.
The analysis of variance detected significant differences in mean PPT between groups (F = 56.2; P < 0.001) and between the measurement points (F = 61.3; P < 0.001). Post hoc comparisons revealed: (i) lower PPT in CTTH subjects compared with both migraine (SNK; P = 0.03) and controls (SNK; P < 0.001); (ii) lower PPT levels within the migraine group compared with controls (SNK; P < 0.001); (iii) PPT in the cervical part of the upper trapezius muscle (points 1, 2) was lower than in all remaining points (SNK; P < 0.001); (iv) lower PPT in the mid-belly of the upper trapezius muscle (point 4) compared with the lower (points 6, 7, 9, 10, 11) trapezius muscle (SNK; P < 0.001); (v) PPTs in the cervico-thoracic part of the trapezius muscle (point 3) were higher than in the upper trapezius muscle (points 1, 2, 4; P < 0.001), but lower than in the lower part of the muscle (points 9, 11; P < 0.001).
In addition, significant interactions between group and point
(F = 14.6;
P < 0.001) and between
group and side (F = 3.6;
P = 0.03) were found. Post
hoc revealed that the cervical portion of the upper trapezius muscle (points 1,
2, 4) within the CTTH group had lower PPT than both migraine and healthy
controls (P < 0.001). PPT
levels of the remaining points of the trapezius muscle were not significantly
different between CTTH and migraine patients
(P > 0.2). Figure 2
summarizes mean ± Pressure pain thresholds (PPT) (average of both sides) for each point
in both patient groups and healthy controls (data are expressed as
mean and standard deviation). *Significantly lower PPT
levels in both headache groups compared with healthy controls
(P < 0.001).
#Significantly lower PPT levels in points 1, 2 and 4 within the
chronic tension-type headache (CTTH) group compared with both
migraine and healthy controls
(P < 0.001).
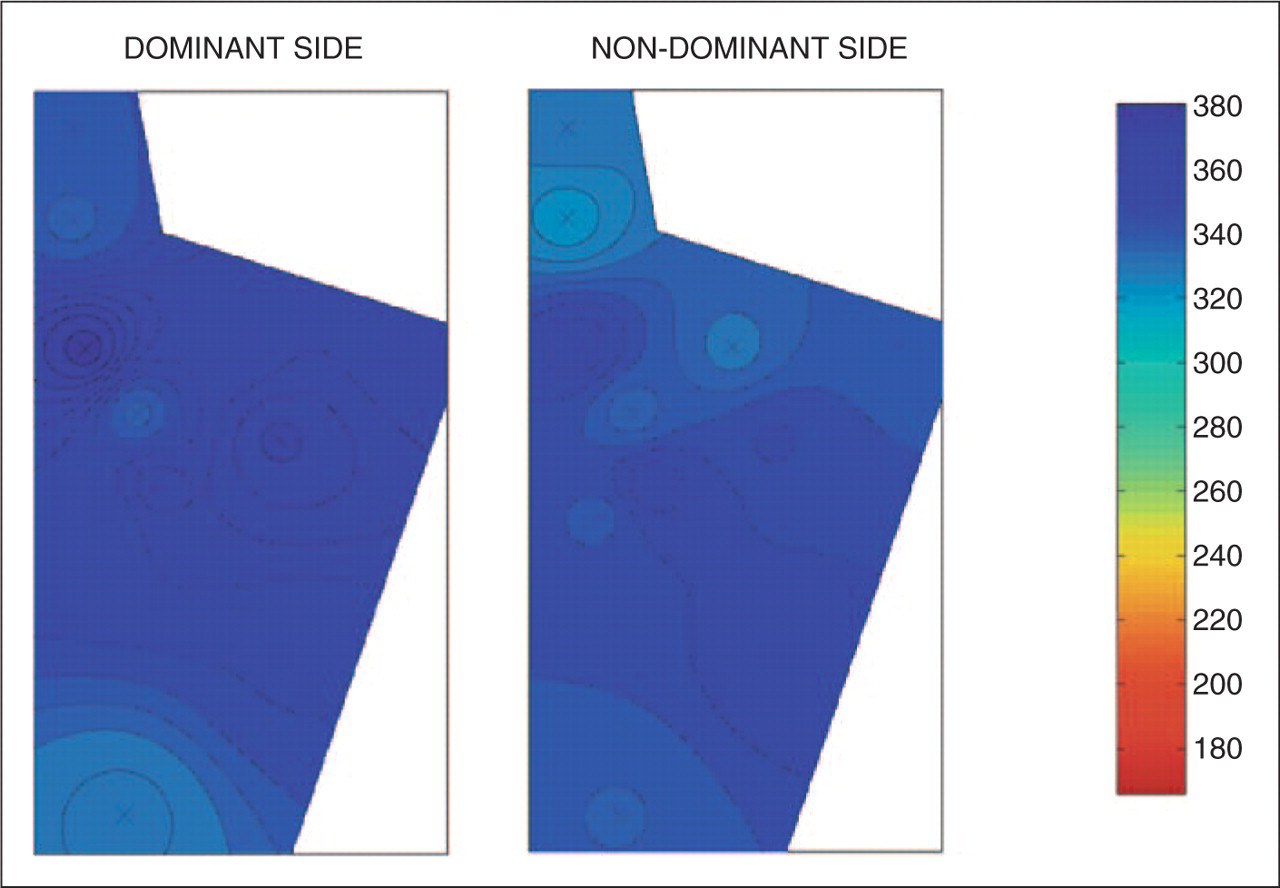
Finally, symptomatic/non-symptomatic and dominant/non-dominant pressure pain
sensitivity maps were quasi-symmetric in, respectively, CTTH patients (Fig. 3) and healthy controls (Fig. 4), but not in
unilateral migraine (Fig. 5). Within the migraine group, PPT of upper and
middle trapezius (points 1–5, 7, 8) was lower on the symptomatic
side compared with the non-symptomatic side. In contrast, PPT levels over the
lower part of the trapezius muscle (points 6, 9–11) were higher on
the symptomatic side. Table 1 summarizes PPT values
of each point for both sides (dominant or non-dominant, symptomatic or
non-symptomatic) in all groups. Average pressure pain threshold maps for chronic tension-type
headache patients. ‘X’ represents the
measured points. Average pressure pain threshold maps for healthy controls.
‘X’ represents the measured points. Average pressure pain threshold maps for unilateral migraine
patients. ‘X’ represents the measured
points. Pressure pain thresholds of each point in either dominant (right) or
non-dominant (left) sides within chronic tension-type headache,
migraine and healthy controls Pressure pain thresholds are expressed as
means ± standard deviation
(kPa). Significant differences between patients and controls.


Pressure sensitivity and pain features in CTTH and unilateral migraine patients
Finally, significant negative correlations between PPT data and several pain features were found: (i) within the migraine group between the frequency of attacks and PPT over point 1 (r = −0.7; P < 0.001), point 5 (r = −0.5; P = 0.02) and point 8 (r = −0.47; P = 0.03) on the affected side; (ii) within the CTTH group between the duration of the headache and PPT levels over point 3 (r = −0.6; P = 0.01), point 6 (r = −0.46; P = 0.04), point 7 (r = −0.48; P = 0.04), point 9 (r = −0.8; P < 0.001), point 10 (r = −0.59; P = 0.01), point 11 (r = −0.72; P < 0.001) in both sides, and between headache intensity and PPT over point 2 (r = −0.5; P = 0.02). Thus, the greater the frequency of headache or the longer the headache duration, the lower was the PPT.
Discussion
Our study has shown that the upper part of the trapezius muscle is the most pressure pain sensitive part in both headache patients and controls. Generalized lower PPT was found over the whole trapezius muscle in CTTH patients compared with both migraine and healthy controls. Furthermore, patients with strictly unilateral migraine had lower pain thresholds in the trapezius muscle than healthy controls. Finally, side-to-side differences were also found in patients with strictly unilateral symptoms, but not in patients with bilateral pain symptoms (CTTH).
PPT recordings are recommended by the IHS classification (34) However, the classification of headache patients into subgroups is still not clearly related to possible pathophysiological mechanisms. PPT levels are considered a valuable clinical test for the evaluation of pericranial muscle sensitivity. Furthermore, we have demonstrated the utility of multiple site recordings for PPT mapping, leading to a new imaging modality of muscle sensitivity to pain (22,23,33). The technique enables the detection and visualization of non-uniformity in muscle pain sensitivity as reported in headache and musculoskeletal pain (22,23,40).
Our results support the relevance of assessing pressure pain sensitivity at multiple sites in the trapezius muscle in both CTTH and migraine patients. The existing neurophysiological and morphological evidence of convergence from cervical sensory and muscle afferent inputs onto trigeminal sub-nucleus caudalis nociceptive and non-nociceptive neurons is considered an explanation for the phenomenon of cervical-to-trigeminal and trigeminal-to-cervical pain referral commonly seen clinically. Therefore, our data support the relevance of cervical afference in both CTTH and unilateral migraine patients and point towards a general hyperalgesia of neck-shoulder muscles in headache patients, particularly in CTTH. The fact that work-related neck-shoulder pain is accompanied by headache (25,41) underlines these convergence mechanisms, making muscle pain one possible cause or effect of headache (42). Nevertheless, our results can also be explained in the opposite way, i.e. trigeminal afference may be also relevant for the sensitization of cervical muscles. Nie et al. found increased mechanical pain sensitivity of muscle belly locations of the trapezius muscle compared with the pressure pain sensitivity of musculo-tendinous junctions after the induction of delayed onset muscle soreness (32), indicating that changes of mechanical pain sensitivity are not uniform over this trapezius region, in line with our findings in headache patients. This could be due to a different distribution of muscle nociceptors between the different subdivisions of the trapezius muscle.
In addition, we also found bilateral topographical maps of the trapezius muscle in both CTTH and healthy controls, but only side-to-side differences in unilateral migraine. These results agree with those previously reported for the temporalis muscle in CTTH (22), but not in strictly unilateral migraine (23). In our previous study we found bilateral pressure hyperalgesia in the temporalis muscle in patients with strictly unilateral pain symptoms (23). However, pressure maps in the trapezius muscle were not symmetrical in unilateral migraine. It may be that sensitization of central pathways (7) accounts for bilateral hyperalgesia (43) in the trigeminal-related region (temporalis muscle), but not in the cervical-related region (upper trapezius) in unilateral pain headache. In agreement with that, side-to-side differences in pain sensitivity and tenderness over the upper trapezius, but not the temporalis, muscle were found in a cohort of 25 patients with unilateral migraine (21). Further studies investigating different pressure pain sensitivity topographical maps between muscles with trigeminal or cervical innervations should be conducted. Moreover, these investigations on muscle pain hyperalgesia should not be limited to a particular point, since the topographical distribution of pressure maps differs throughout the same muscle in headache and musculoskeletal pain (22,23,32,40).
Olesen has proposed that perceived headache intensity could be the sum of the nociceptive barrage from cranial and extracranial peripheral tissues converging on the neurons of the trigeminal nucleus caudalis (44). Burstein suggested that hyperalgesic and allodynic responses in migraine are due to the development of central sensitization involving second-order neurons in the trigeminal nucleus and at least third-order neurons in the thalamus (45). The existence of central sensitization mechanisms in local pain syndromes may suggest that sustained peripheral noxious input to the central nervous system plays a role in the initiation and maintenance of central sensitization. In the current study, PPT levels over the trapezius muscle were negatively associated with some clinical pain features. These results support a role of muscle peripheral sensitization mechanisms in the initiation and maintenance of central sensitization (8). In fact, increased recruitment of central neurons by peripheral nociceptive stimulus (46), enhanced spatial summation (47) and spatial referral (i.e. tonic nociceptive input from local tissue can result in pain in remote areas and increase pain intensity (48)) have been suggested to be the potential peripheral mechanisms. Furthermore, it seems that hyperexcitability of the central nervous system is a dynamic condition and influenced by the activity and location of peripheral nociceptive inputs (49). Therefore, prolonged nociceptive inputs from non-trigeminal, but cervical, innervated tissues (upper trapezius muscle) may result in spatial summation (50), increasing pain perception, mechanical pain hypersensitivity and the intensity of headache. In the present study we found a generalized increased hypersensitivity expressed by lower PPT in the trapezius muscle (including the asymptomatic lower part of the muscle) in both CTTH and migraine. Our findings support the role of central sensitization mechanism in CTTH. Nevertheless, our patients with strictly unilateral migraine were classified as episodic, so current findings in episodic migraine may result from a general putative dysfunction in sensory processing and pain control system, i.e. abnormal habituation in multimodal cortical evoked potential and cutaneous allodynia during attacks (51). Moreover, the observed spatial differences in PPT levels provide some evidence of different pathogenic factors underlying CTTH and unilateral migraine. Nevertheless, CTTH and migraine patients reported neck-shoulder pain, but the pain intensity and spreading was not assessed in the present study. Thus, pain in the neck-shoulder region could be the sole factor responsible for the general hyperalgesia in the trapezius muscle. However, it is most likely that convergence mechanisms from trigeminal and cervical afferents play a role in hyperalgesia observed in the temporalis muscle and trapezius muscles in CTTH and migraine patients (22,23).
Population-based epidemiological studies with larger samples are needed for a more generalized interpretation of these results. Furthermore, during face-to-face interview none of the included patients seemed to have psychiatric or depression symptoms; however, since pressure pain sensitivity can be influenced by depression or anxiety, specific questionnaires could have been used. Finally, we have described the spatial distribution of pressure pain sensitivity only in patients with unilateral migraine or CTTH. Assessment of pressure pain sensitivity maps in other headache disorders, e.g. trigeminal autonomic cephalalgias, and in both genders, may expand current knowledge about the state of nociception in headaches.
Conclusions
CTTH and migraine patients have generalized lower PPT levels in the trapezius muscle region compared with controls. CTTH patients also have lower PPTs in the trapezius muscle than migraine subjects. The topographical pressure pain map revealed that the upper part of the trapezius muscle is the most sensitive part of the muscle in both patients and controls. Finally, side-to-side differences were also found in patients with strictly unilateral migraine, but not in patients with CTTH. The present study supports the implication of cervical afferences in headache conditions.
Footnotes
Acknowledgements
This study received funds from a grand conceded by the Universidad Rey Juan Carlos and Comunidad de Madrid (reference CCG07-URJC/BIO-1420).
