Abstract
Background
Medication-overuse headache is a costly disease for individuals and society.
Objective
To estimate the impact of medication-overuse headache treatment on direct and indirect headache-related health care costs.
Methods
This prospective longitudinal study was part of the COMOESTAS project (COntinuous MOnitoring of Medication Overuse Headache in Europe and Latin America: development and STAndardization of an Alert and decision support System). Patients with medication-overuse headache were included from four European and two Latin American headache centers. Costs of acute medication, costs of health care services, and measurements of productivity were calculated at baseline and at 6-month follow-up Treatment consisted of overused drug withdrawal with optional preventive medication.
Results
A total of 475 patients (71%) completed treatment and were followed up for 6 months. Direct health care costs were on average reduced significantly by 52% (p < 0.001) for the total study population. Significant reductions were seen in both number of consumed tablets (−71%, p < 0.001) and number of visits to physicians (−43%, p < 0.001). Fifty percent of patients reduced their number of consumed tablets ≥ 80%. Headache-related productivity loss, calculated either as absence from work or ≥ 50% reduction of productivity during the workday, were reduced by 21% and 34%, respectively (p < 0.001).
Conclusion
Standardized treatment of medication-overuse headache in six countries significantly reduced direct health care costs and increased productivity. This emphasizes the importance of increasing awareness of the value of treating medication-overuse headache.
Trial registration
The trial was registered at ClinicalTrials.gov (no. NCT02435056)
Keywords
Introduction
Medication-overuse headache (MOH) is a common disease (1,2) affecting 58 million individuals worldwide (3) with a prevalence ranging from 0.5% to 7.2% in the general population (4,5).
MOH is one of the most expensive neurological diseases (6). The estimated annual cost/person with MOH is €3561 on average – three times the cost of migraine and more than 10 times the cost of tension-type headache (1,7). MOH accounts for a higher proportion of headache-attributed lost work-days on a monthly basis (14 days/month) compared to other chronic headaches (3 days/month) (8–10). From a patient’s perspective, MOH is a disabling disease associated with decreased quality of life and a heavy burden of comorbidities (11–13).
To a large extent, these costs are unnecessary because MOH is preventable and, for the most part, a treatable condition (14). The recommended treatment according to European Guidelines is discontinuation of the overused drugs, which can be done either abruptly or by tapering use of analgesics and acute migraine medication, possibly supplemented by pharmacological preventive treatment (15–18).
Improved treatment is likely to contribute to a considerable reduction in socio-economic and health care costs. A study based in a highly specialized clinic reports an overall reduction of 24% in medication expenses after detoxification (19). However, there is still lack of evidence on the potential economic benefits of MOH treatment from a multi-national perspective.
The COMOESTAS project was primarily designed to develop a detoxification program for global implementation. The project was implemented in six countries from two continents – Europe: Denmark, Germany, Italy, Spain; and Latin-America: Argentina and Chile – from 2008 to 2010. The main results have already been published (2,20,21). The studies showed that 67% of subjects no longer overused acute pain medications, and 46.5% of patients reverted to episodic headache at the 6-month follow-up. Half of patients (50.7%) reported reduced symptoms of depression and 27.1% reported reduced anxiety after detoxification (2). An extensive analysis of the baseline data has also been published (22,23).
The COMOESTAS project also aimed to estimate the economic benefits of the detoxification program. In this paper, we report on the economic impact of MOH treatment on direct and indirect headache-related health care costs in a large population of MOH patients enrolled, treated, and followed up for 6 months in six countries. Our aim is to estimate the economic impact of MOH and the reduction in costs that is associated with treating it.
Methods
Study design
This is a prospective longitudinal study and a part of the COMOESTAS project (20). All patients underwent a detoxification program with a 6-month follow-up according to a consensus protocol (21). The detoxification was performed as out-patient treatment, except in Italy where all patients were treated as in-patients. On day one, all analgesics and acute migraine medications were abruptly stopped, and patients were given rescue therapy until day 7 to relieve withdrawal symptoms. Personalized preventive treatment was initiated after the first seven days if the physician and patient found this relevant. The preventive treatment was selected among documented first-line options: Beta-blockers, valproic acid, topiramate, flunarizine, amitriptyline or candesartan. OnabotulimumtoxinA was not yet approved for the prophylaxis of migraine and was therefore not included in this study. A more detailed description of the detoxification program has already been published (21).
Study population
The project was implemented in four European and two Latin American headache centers: Denmark (Danish Headache Center, DHC, Copenhagen, Denmark); Germany (University Hospital, UKE, Essen, Germany); Italy (C. Mondino National Neurological Institute, FMPV, Pavia, Italy); Spain (University Clinical Hospital, FIHCUV, Valencia, Spain); Argentina (Foundation for Combating Neurological Diseases of Childhood, FLENI, Buenos Aires, Argentina); and Chile (Pontificia Universidad Católica de Chile, UC, Santiago, Chile). The European centers are public tertiary academic headache centers; the two Latin American centers are private institutes serving insured patients. All patients were recruited consecutively from January 2008 to February 2009 if they matched the inclusion and exclusion criteria.
The inclusion criteria were: a) a diagnosis of MOH according to the revised ICHD-II criteria (24), and subsequently confirmed with ICHD-III beta criteria (25) and b) the ability to fill in a paper or electronic diary. Exclusion criteria were a) parallel diagnosis of complicating, significant physical or psychiatric diseases; b) known overuse of pure opioids, benzodiazepines or alcohol; c) actual treatment with migraine preventive medication; d) inefficacy of previous detoxification programs; e) inability to provide reliable medical history; f ) pregnancy or breast feeding (20).
Standard protocol approvals, registrations, and patient consents
The COMOESTAS project was approved by local ethics committees in all participating headache centers except the Danish Headache Center, where the local ethics committee did not find application for approval necessary, since the study did not contain use of different/new pharmacological treatment. All patients signed an informed consent form prior to entering the study.
Definition of terms
“Direct health care costs” refer to the costs of three variables: a) Amount of acute medication: ergotamines, triptans, opioids, simple analgesics, and combination-analgesics (e.g. combinations of analgesics with codeine, caffeine and/or antiemetics); b) visits to emergency rooms (ERs), general practitioners (GPs), and headache specialists (SPs) (mainly neurologists but also neurosurgeons and pain specialists); c) laboratory investigations (X-rays, EEGs, and CT and MR scans).
“Productivity loss” was defined as a decrease in work capability due to headache. Productivity was quantified according to the Migraine Disability Assessment (MIDAS) questionnaire (26): Question 1 – “On how many days in the last 3 months did you miss work or school because of your headaches?” and Question 2 – “How many days in the last 3 months was your productivity at work or school reduced by half or more because of your headaches? (Do not include days you counted in question 1 where you missed work or school)”.
Collection of data
Baseline data: We retrospectively collected information about the types and number of clinic visits, and types and number of laboratory investigations performed during the previous year. Productivity loss in the previous 3 months was assessed using the two MIDAS questions. Use of acute medication was calculated with a prospective one-month recording of data in an ad hoc headache diary at baseline.
6-month follow-up: All the data above was captured prospectively using the headache diary (paper or electronic version) and an electronic patient’s report form.
For the calculation of costs associated with visits, investigations, and medications, we applied local reference prices derived from a questionnaire that was filled in by participating centers. Data were obtained from their official pricing registries.
Indicators and their calculation: The following direct health care costs were estimated at baseline and at the 6-month follow-up after detoxification:
Costs of acute medications was calculated at baseline and at month 6 by multiplying the number of doses with the cost of each drug type in the patient’s home country. Utilization of health care services was presented as the number of consultations with GPs and SPs; the number of admissions to ERs; the number of diagnostic investigations (EEG/MR/CT/X-rays), and the accompanying estimated costs. For the baseline calculation, we collected the total number of events (visits and investigations) that occurred during the previous year, multiplied them by the respective price in the patient’s home country and subsequently normalized the price to one month (total number of events times the individual national price, divided by 12 months).
For the 6-month follow-up, the number of visits and investigations were calculated as a mean value over the last four months, multiplied by respective national price and then normalized to one month (total number of events times the individual national price, divided by 4 months).
Loss of productivity was measured as the number of days in three months where the patient missed a whole work/school day (MIDAS1) or number of days in three months with more than 50% reduced productivity at work/school (MIDAS2).
Management of missing data: We adopted a conservative approach whenever data were missing. If the precise number of doses/day was missing, we assumed that the patient had taken one dose in each day they indicated use of acute medication. If no change of acute drug was reported, we assumed that the patient used the same acute medication prescribed after the detoxification for the entire 6-month follow-up.
At the 6-month follow-up, the diagnostic investigations were not specified in groups (EEG/MR/CT/X-rays) but given as a total. To estimate the costs, we used the average price per investigation at baseline in the patient’s country and multiplied it by the total number of investigations.
As regards utilization of health care services, one of the countries had variable prices depending on the type of visit (initial or follow-up). This variability was considered in the calculations. If the expense for a health care service varied regionally within a country, the lowest price was used. The costs of health care services in each country were estimated during the time of the COMOESTAS project (2008–2010).
Statistics
Statistical Package for Social Sciences (SPSS) version 24 was used for statistical analysis. Data were shown as number per month with Standard Deviation (SD) or Standard Error of Mean (SEM). Costs were presented in Euros reported as Euros per month per patient.
Percentage change in acute medication, visits, direct health care costs and productivity loss from baseline to 6-month follow-up were calculated for each patient, and then an average percent reduction was calculated for each country and for the total study population. Shapiro-Wilk test was used to determine if the percent differences were normally distributed. Paired t-test or Wilcoxon signed-rank test (its non-parametric equivalent) were used to test the significance of differences in costs and productivity loss. All p-values were two-tailed and p < 0.05 was considered significant.
Results
Out of 918 screened patients, 669 patients were included in the COMOESTAS project at baseline; 475 patients (71%) were followed-up after 6 months and included in the analysis (Figure 1). The baseline characteristics of the included patients are presented in Table 1. The cumulative percentage in the primary headache types is higher than 100% because some patients suffered from more than one type headache.
Flowchart showing patient inclusion and exclusion in six participating headache centers. Baseline characteristics of included patients examined at six headache centers. Note: Episodic Migraine includes patients diagnosed with migraine with aura and/or without aura. Tension-type headache (TTH) includes patients diagnosed with either chronic or episodic forms. Age, age of onset of primary headache, duration of chronic headache, duration of overuse, and headache frequency are presented as mean and standard error of the mean in brackets. Gender and primary headache types are presented in number and percentage in brackets.

Direct health care costs
Total number of doses of acute headache medication consumed per month before and after detoxification among patients treated at six headache centers.

Note: Data are shown with standard error of the mean in brackets. p-values were calculated with the Wilcoxon Signed Rank test.
Similarly, the number of consumed medications were reduced significantly (p < 0.001) in each headache center. The average reduction ranged from 63% to 85%. The highest reduction was observed in Chile (85%), where patients on average reduced their intake by approximately 62 doses/month (from 70 to 8). At 6-month follow-up, patients in Argentina consumed the lowest number of tablets (seven tablets/month) compared with the other centers.
Number of visits per month at emergency rooms, general practitioners’ clinics and specialist clinics before detoxification and at the 6-month follow up among patients treated at six headache centers.
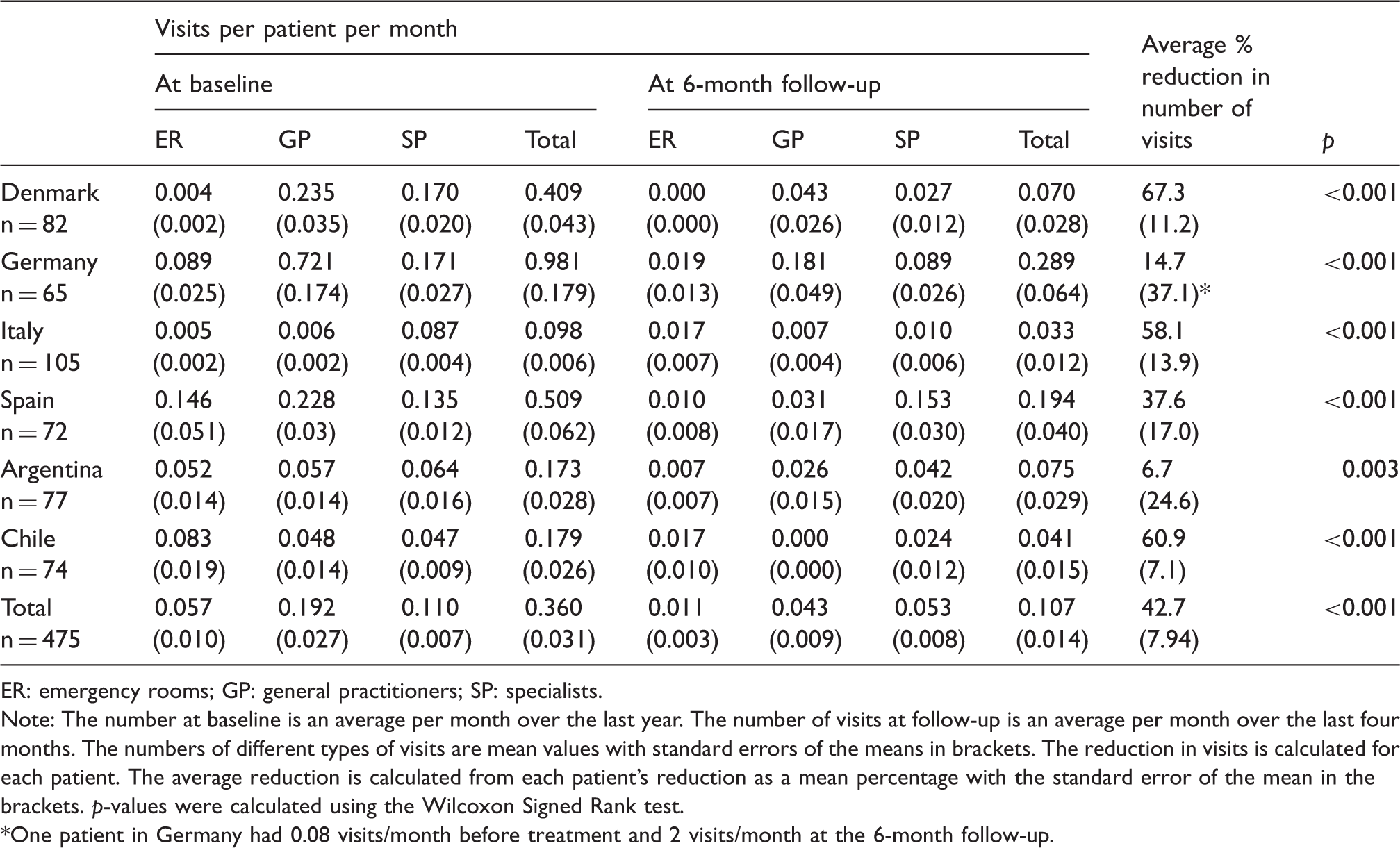
ER: emergency rooms; GP: general practitioners; SP: specialists.
Note: The number at baseline is an average per month over the last year. The number of visits at follow-up is an average per month over the last four months. The numbers of different types of visits are mean values with standard errors of the means in brackets. The reduction in visits is calculated for each patient. The average reduction is calculated from each patient’s reduction as a mean percentage with the standard error of the mean in the brackets. p-values were calculated using the Wilcoxon Signed Rank test.
One patient in Germany had 0.08 visits/month before treatment and 2 visits/month at the 6-month follow-up.
At baseline, Latin American centers, Spain and Germany had a least 10 times more visits to the ER per patient (0.05–0.15 visits/month) compared with Denmark (0.004 visits/month) and Italy (0.005 visits/month) (Table 3). Before detoxification, Italy had the lowest number of monthly visits per patient (0.098 visits/month) while Germany had the highest (0.980 visits/month). After 6 months, Germany reduced the number of visits per patient by 15%, but still had the highest number of total visits per patient (0.289 visits/month).
In some countries, a few patients reported a high increase in visits over the follow-up period leading to the wide distribution of data. For example, one patient in Germany had 0.08 visits/month before treatment and two visits/month at the 6-month follow-up. One patient in Italy had an increase in number of visits/month from 0.08 to 1 at the 6-month follow-up.
Diagnostic investigations: The number of investigations/month was significantly reduced by 26% in the total study population (p = 0.001). In all centers except Italy (p = 0.1), we detected a significant reduction in number of investigations compared to baseline (p < 0.002–0.040).
Reduction in health care costs: The direct health care costs for the whole study population were on average reduced by 52% (p < 0.001) (Figure 2 and Appendix 1).
Distribution of direct healthcare costs at baseline and at 6-month follow-up in six headache centers and in the total study population. Direct healthcare costs include acute medication, clinic visits, and laboratory investigations at baseline and at 6-month follow-up. The total column shows average health care cost per patient per month at baseline and at 6-month follow-up for the study population. The reductions are significant. Summary of total health care costs in six headache centers at baseline and at the 6-month follow-up. Note: Health care costs at baseline and at the 6-month follow-up are calculated per patient per month. Acute medication covers the costs of analgesics and migraine medication (ergotamines, triptans, opioids, simple analgesics and combination analgesics (e.g. combinations of codeine, caffeine and/or antiemetics). Visits include expenses incurred at emergency rooms, or clinics of specialists or general practitioners. Investigations include expenses for EEG, X-ray, CT or MR. p-values were calculated using Wilcoxon Signed Rank test.

At baseline, Denmark had the highest total cost/patient (184 Euros/month) and Argentina had the lowest (40 Euros/month). Also, distribution of direct health care costs varied among the centers: Chile spent 90% of total costs on medications (134 Euros/month/patient), six times more than Spain (22 Euros/month/patient, corresponding to 45% of total costs). Denmark spent 68% of direct cost/month on visits (126 Euros/patient), while Argentina used only 5% (2 Euros/month out of 41 Euros/month). Details are summarized in Appendix 1.
At 6-month follow-up, on average the total study population significantly reduced their costs from 87 to 20 Euros/month/patient (p < 0.0001). A significant reduction in total direct costs/month was recorded in all countries (Figure 2). Average reduction percentages ranged from 64% (Denmark) to 20% (Germany). Denmark had the highest monthly reduction of approximately 150 Euros/patient, equivalent to 1800 Euros/year/patient.
Overall, 60% of cost reduction in the total study population was due to reduction in medication costs, especially seen in Italy, Argentina and Chile with 80%, 84%, and 88%, respectively (Figure 3). The main reduction in Denmark originated from expenses on visits, equivalent to 105 Euros/patient/month (70% of total reduction). Only in Chile, a minor increase in costs of investigations (approximately 9%) was observed after 6 months follow-up.
Distribution of reduction of direct healthcare costs in six headache centers and total study population. The values used for this figure are detailed in Appendix 1. The negative value in Chile is due to an increase in the costs of investigations comparing baseline costs to cost at the 6-month follow-up.
Out of 475 patients, 292 (62%) reduced their direct health care costs by ≥ 80%.
Loss of productivity
Loss of productivity in terms of missed days and reduced productivity by 50% or more among patients treated in six headache centers.

MIDAS 1: Number of missed work- or school days during the last 3 months due to headache.
MIDAS 2: Number of days with 50% or more reduced productivity at work or school more because of headache during the last 3 months.
Note: The days are indicated as a mean value with the standard error of mean in brackets. Average percentage reduction is a mean calculated from individual reductions per patient with standard error of the mean in brackets. If the patient had 0 days lost before and 0 days lost after, a reduction of 0% was recorded. p-values were calculated with the Wilcoxon Signed Rank test.
Number of patients who had more than 50% reduction in MIDAS1 or MIDAS2:
MIDAS1: A1n = 28. B1n = 52. C1n = 19. D1n = 31. E1n = 31.F1n = 22. Total = 183.
MIDAS2: A2n = 35. B2n = 55. C2n = 28. D2n = 41. E2n = 51.F2n = 46. Total = 256.
Out of 475 patients, 183 patients (39%) had a reduction of more than 50% of missed workdays due to headache. The number of days with reduced productivity due to headache (MIDAS2) was on average significantly reduced by 34% for the total study population (p < 0.001) (Table 4). All centers had a significant reduction after 6 months (p < 0.001–0.030).
In the German sample, both variables (MIDAS1 and MIDAS2) showed a negative average in reduction percentage, which indicated the center overall had a decrease in productivity. The average was skewed by a few patients with a large increase in number of missed workdays. In total, nine patients in Germany had an increase in days with lost productivity at the 6-month follow-up, where one patient increased from 3 days/three months at baseline to 60 days/three months at follow-up. These outliers were not excluded.
Discussion
In this large prospective multi-national multi-center study, we report a significant reduction in total number of doses of acute medications, visits, and diagnostics, corresponding to a 52% reduction in these healthcare costs. Due to lack of precise data, the costs of preventive treatments before and after the withdrawal procedure were not included. OnabotulinumtoxinA was not available at the time of the study. We feel that the impact of the cost of preventive drugs may not have been relevant in this population. Future cost-effectiveness studies are needed to properly address this issue. We applied a conservative, societal perspective and did not discriminate between costs financed by individuals, governments or health insurances. This, and the differences in the different health care systems, have previously been described (22). Considering the prevalence of MOH (0.5% to 7%) (5), direct health care costs globally can be significantly lowered by an increased focus on MOH prevention and treatment (27). These investigations are important in order to help policymakers foster the adoption of appropriate intervention programs, allocate resources for headache services, and promote evaluations of new treatments.
The mean annual price for direct costs in our total MOH population before treatment was 1044 Euros/patient, and in the most expensive country (Denmark) 2208 Euros/patient. The Eurolight project estimated the mean annual cost/patient to 3561 Euros before treatment (1). This is more than three times our estimate, but the Eurolight project also included an extensive analysis of the important indirect costs of productivity loss and absenteeism from work (92% of total costs), which were not included in our study.
Shah et al. estimated costs of medication expenses per patient to 971 USD in Denmark – equivalent to 900 Euros (19), which is more than our estimate of 600 Euros. Furthermore, they found a cost decrease of 24% per year, while we observed 41% reduction per month. The difference might be due to Shah et al. evaluating long-term savings on medication costs over a 2-year period, while we had a shorter 6-month follow-up period, or it could be due to our conservative approach. Shah et al. collected data at roughly the same time as our study, so the change cannot be due to a time difference between estimations. Nonetheless, our findings agree with their overall conclusion that there is an impressive cost reduction after treatment. The results should be interpreted with some caution, due to the relatively low number of patients included in each country and the dropout rates.
In our study, distribution of direct health care costs and reduction in visits to ER, GPs, and SPs varied considerably among the centers. This could be due to differences in the health care structures, referral strategies on a national basis, and costs of services across countries, which makes the services in some of the countries more accessible and affordable than in others. The results reflect how health care services for headache were very different among the countries. The MOH treatment protocol was standardized and implemented in all participating centers despite differences in the health care systems. In this study, we focus on comparing individual costs before and after treatment, rather than comparing costs across countries. ER visits in most participating countries are free of costs for the patient, whereas visits to a GP or specialist are not. Differences in health care utilization between the Latin American and European centers have previously been described in the COMOESTAS project (22). One example here is Denmark, which had fewer visits than Germany but still had higher costs for these visits. The same national difference is also seen in the costs of analgesics, where the consumption levels were similar in two countries despite different costs. Our results thereby suggest the importance of calculating the impact of adequate care for MOH at the country level.
A significant part of the burden of MOH is attributed to reduced effectiveness at work (1). In our study, effectiveness at work increased significantly. Increased effectiveness at work could be due to marked reduction in number of days with headache and the disability this causes (2). An important point to remember is that significant improvement (such as reduction in total MIDAS score after treatment) likely leads to higher productivity. However, data are influenced by differences in national regulations regarding paid leave. In the Latin American countries, the lower number of missed workdays may be due to economic pressure and not necessarily better treatment or better outcome. In a 5-year follow-up study, Andrasik et al. reported a 76% reduction in total MIDAS score (28), and significant differences were seen in the MIDAS scores of successfully and non-successfully treated patients (29).
A full cost-benefit analysis was not included in our study. Costs of in-patient treatment, productivity/absenteeism from work, and pharmacological preventive therapy were not included due to lack of precise data. A future study on cost-effectiveness of withdrawal should include these calculations. In this frame, we would expect that the treatment costs might be even higher than the direct savings if we limited our evaluations to the first month or two after detoxification. However, our data demonstrate that as we extend the observation period by only a few more months, we obtain a marked reduction in the use of health care services. This study is limited to a 6-month follow-up period after treatment, and in future studies it could be interesting to expand this follow-up period to at least 1 year, especially considering that MOH is a chronic disorder that requires evaluation over longer periods.
Eighty-six percent of patients (407) received preventive treatment at the time of detoxification, mainly anti-epileptics and tricyclic antidepressants. The costs of preventive medication were not included in the analysis due to lack of data at follow-up, which is a limitation when estimating the direct healthcare costs. We do not have data on compliance to preventive therapy, but a recent study reports that patients in general have low compliance (30). Patients were instructed to record in the diary the number of acute medications they used, but they were not asked to do the same for preventive medications, which could be an important area to map for future studies.
Only patients who finished the treatment protocol were included in this analysis. Patients who dropped out from baseline to the 6-month follow-up (194 patients) were excluded. The reason for drop out is of relevance, as a drop out could be due to a lack of effectiveness, a relapse, lack of important data or, conversely, a significant improvement in headache. An intention-to-treat analysis was not conducted and could be regarded as a limitation of the study. It will be of relevance in future studies.
The COMOESTAS project included in-patient and out-patient treatment programs, both of which proved equally effective in a previous sub-analysis (31), in agreement with other data from the literature (32,33). The center in Italy used in-patient treatment exclusively. Health care system costs in Italy were approximately 2500 Euros per patient per one-week detoxification program. It is estimated that out-patient care is less costly, but it was outside the scope of the present study to compare the cost-effectiveness of in- and out-patient care. In this study we did not compute analytically the costs of withdrawal, because the majority of patients (78%) were treated with the quite inexpensive out-patient protocol and because we wanted to focus attention on the observation that addressing MOH properly is a cost saving process. Since data regarding long-term success of these two methods are similar, an out-patient MOH treatment would be ideal from the financial point of view since in-patient treatment is more expensive. However, there are certainly groups of patients who benefit most from in-patient treatment, particularly those with opioid overuse, complex pain syndromes and/or significant comorbidities.
A small number of enrolled patients lacked precise data on number of doses of acute medications per month, both at baseline and at follow-up. In these cases, we assumed the number of doses as one per drug-day. We are aware that this is a very conservative approach, since the daily intake often exceeds more than one tablet a day. On the other hand, these patients were aware of the importance of reducing intake of acute drugs, and we felt that this conservative assumption was adequate for avoiding overestimation.
Results from one center are not necessarily generalizable to the whole population of that country. For this purpose, larger studies are needed. Nonetheless, this study shows the benefits of an evidence-based approach in the treatment of MOH.
Conclusion
Treatment of MOH considerably reduces direct and indirect costs associated with the disorder. These reductions were observed in all six countries studied, despite differences in health care systems. Our results demonstrate the utility of globally adopted treatment protocols implemented in centers that facilitate access to adequate treatment.
With a high worldwide MOH prevalence, both increased awareness and effective treatment of MOH are of immense public health importance and can lead to a significant reduction in economic and personal disease burden.
Clinical implications
Treating patients with MOH using a detoxification strategy markedly reduces direct health care costs, including costs of acute medication, visits to health care professionals, and laboratory investigations. The majority of patients treated for MOH reported reduced work absence rates and higher productivity at work, thereby decreasing indirect costs. With a worldwide MOH prevalence of 0.5 to 7.2%, reduction in economic burden after effective treatment is of immense public health importance.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by the COMOESTAS Project – EC contract number 215366 (COMOESTAS) FP7 – Thematic priority ICT.
