Abstract
Background
There is lack of evidence on how to detoxify medication-overuse headache.
Aim
To compare the effect of complete stop of acute medication with restricted intake.
Methods
Medication-overuse headache patients were included in a prospective, outpatient study and randomized to two months’ detoxification with either a) no analgesics or acute migraine-medication (program A), or b) acute medication restricted to two days/week (program B). Detoxification was followed by preventives if indicated. Patients were followed up at 2, 6 and 12 months. Percentage reduction in headache days/month after 6 months was the primary outcome.
Results
We included 72 medication-overuse headache patients with a primary migraine and/or tension-type headache diagnosis. Fifty-nine completed detoxification, 58 (81%) were followed up at month 6 and 53 (74%) at month 12. At month 6, program A reduced headache days/month by 46% (95% CI 34–58) compared with 22% (95% CI 11–34) in program-B (p = 0.005), and 70% in program A versus 42% in program B were reverted to episodic headache (p = 0.04). Migraine-days/month were reduced by 7.2 in program A (p < 0.001) and 3.6 in program B (p = 0.002) after 6 months.
Conclusion
Both detoxification programs were very effective. Detoxification without analgesics or acute migraine-medication was the most effective program.
Trial registration
Clinicaltrials.gov (NCT02903329).
Introduction
Medication-overuse headache (MOH) is common, costly, and disabling, and affects approximately 60 million people worldwide (1). Medication overuse is an important risk factor for chronification of headache, especially migraine. MOH is treatable with detoxification as recommended by the European guidelines (2), but there is lack of consensus on how to perform the detoxification. Different approaches to detoxification programs have been suggested: Simultaneous preventive medication, prednisone treatment, rehydration and in- or out-patient programs (3–10). Most programs allowed restricted intake of acute headache medication during detoxification to relieve withdrawal symptoms (4–6). The standard procedure in current Danish guidelines is abrupt and complete stop of all acute analgesics and migraine medications in two months accompanied by rescue medication, patient education and close follow-up (11,12). Initiation of preventive medication is postponed to after detoxification, as a previous study has reported that subsequent medical preventives were indicated in only 47–61% of patients (12). This approach was reported to be effective in treating MOH, even in patients considered intractable (12,13). However, patients may be less compliant to follow this kind of detoxification, since withdrawal symptoms may be aggravated when all acute headache medication is stopped. Therefore, a less strict approach with some doses of acute headache medication could be more feasible. To compare the effect of restricted intake of acute medication to maximally two days/week versus complete stopping of all medication during the detoxification period, we conducted a randomized, controlled study. We hypothesized that there was a significant difference between the two detoxification programs in favor of the restricted medication intake.
Methods
Study design
The study was a prospective, longitudinal, open-label, randomized, controlled study comparing two detoxification programs. The study was conducted in a tertiary headache care center. Patients with MOH were randomized to treatment in either program A or program B. In program A, patients underwent detoxification without any analgesics or acute migraine medication during two months (standard care at the Danish Headache Center (DHC) with complete stop of acute medication intake). In program B, patients were allowed to continue with their usual analgesics and migraine medication up to two days/week during the detoxification (restricted acute medication intake). A two-month detoxification is recommended in the Danish treatment guidelines (11). Both A and B were out-patient programs, and all patients received exactly the same education, consisting of eight lectures managed by headache nurses in collaboration with specialized psychologists and physiotherapists (Figure 1). All patients were offered maximally 10 tablets of rescue medication (levomepromazine max. 75 mg per day or promethazine max. 75 mg per day) during the detoxification period.
Flowchart for patient education in the two detoxification programs.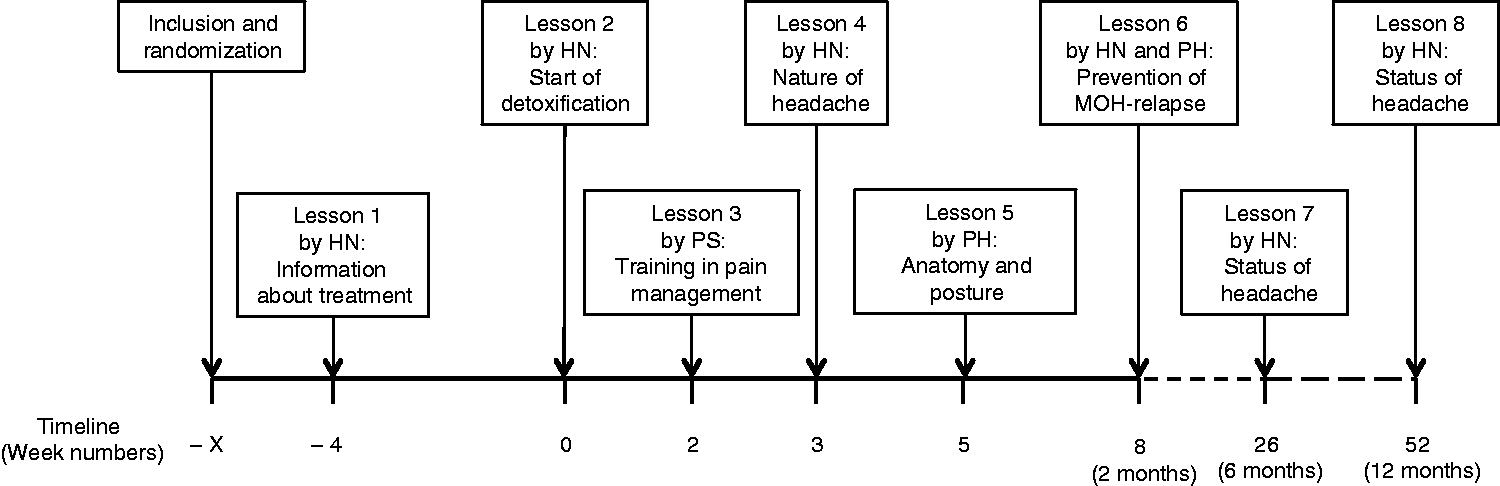
Pharmacological preventive treatment was initiated after detoxification ended, if indicated according to Danish guidelines and patients’ history of previous preventive treatment (11). None of the patients received preventive treatment during detoxification. Patients were seen by headache specialists and headache nurses during the education program (Figure 1). Data were collected at baseline, 2, 6 and 12 months after detoxification. All patients were asked to continuously register headache and medication use in a standardized headache calendar.
Study population
Patients with MOH referred to DHC were included consecutively from October 2013 to March 2016. The inclusion criteria were: Confirmed MOH based on the ICHD3-beta, previous diagnosis of a primary headache (chronic migraine (CM), episodic migraine (EM) and/or tension-type headache (TTH)), and being eligible for outpatient treatment (14). Headache diagnoses were given by headache specialists at DHC based on a minimum three months’ detailed history and the patient having completed a headache calendar for one month prior to the visit.
Patients with a current substantial psychiatric (e.g. schizophrenia or severe depression) or physical disorder (e.g. concomitant heart disease, severe co-morbid pain, or cancer), previously diagnosed with another secondary headache, pregnant or breast feeding, or with inadequate language skills (requiring an interpreter) were excluded.
The study was approved by the Regional Ethical Committee in Denmark (H-1-2012-116). All participants signed informed consent. The study was registered at Clinicaltrials.gov (NCT02903329).
Randomization
Patients were allocated to either program A or B by a concealed randomization process and randomized in blocks of 10 by use of opaque, sealed envelopes.
Endpoints
The primary outcome was comparison of the percentage reduction in headache days/month from baseline to 6-month follow-up between program A and B. The secondary outcomes were:
Comparison of the proportion of patients reverted to episodic headache (defined as headache days/month ≤ 15) at 2, 6, and 12 months’ follow-up; Reduction in migraine-days/month from baseline to 2, 6, 12 months for each program and comparison between the programs; Reduction in headache days/month from baseline to 2, 6 and 12 months for each program; Comparison of distribution of headache diagnoses (CM, EM, combination of EM and TTH, and TTH) defined according to the ICHD3-beta between groups at 2, 6 and 12 months (14); Comparison of percentage reduction in days/month with acute medication intake from baseline to 6- and 12-month follow-up; Comparison of proportion of patients with cured MOH at 2-, 6- and 12-month follow-up. Cured MOH was defined as not fulfilling the MOH criteria anymore, which is present in three scenarios: i) No acute medication overuse and remission to episodic headache, ii) no acute medication overuse despite remaining chronic headache (≥15 headache days/month), or iii) episodic headache despite an ongoing medication overuse. Comparison of headache intensity at 2-, 6- and 12-month follow-up. The daily headache intensity was recorded on a 0–3 scale (0 = no pain, 1 = mild pain, 2 = moderate pain, 3 = severe pain), and the headache intensity was calculated as the sum of daily headache intensity for one month; Dropout rates in percentage from baseline to 2, 6 and 12 months.
In addition, the proportion of patients with medical preventives and type of preventive medication was registered.
Intention-to-treat (ITT) analyses were conducted for the above defined outcomes, except analysis of preventives. All data used for the endpoints were patient reported and were assessed from the previous 30 days (e.g. headache days at 6 months’ follow-up were counted from month 5 to 6). The ITT analyses were conducted based on follow-up data from the patients who failed detoxification, achieved in the same way as described above. If patients were lost-to-follow-up, the “last value carried forward” principle was used.
Statistics
SAS Enterprise Guide 7.1 was used for statistical calculations. Continuous variables were presented as mean with standard error of the mean (SEM) or 95%CI. For continuous variables, we used paired and unpaired Student’s t-test. Categorical variables were presented as percentage and number, and analyzed by χ2-test or Fischer’s exact test (if expected counts were ≤ 5). The p-value was two-sided and p < 0.05 was considered significant. The primary outcome was percentage reduction in headache days/month. Clinically-relevant difference was estimated at 25% between the groups. The standard deviation based on previously published literature was estimated at 35%. Accepting an alfa-error on 5% and 80% power, approximately 64 patients were needed. Based on previous studies, the dropout-rate was estimated at 15–20%. Therefore we aimed to include 76 patients, corresponding to 38 in each group.
Results
Study population
Out of 553 screened patients, 72 with confirmed MOH were included (Figure 2). Thirty-five were randomized to program A, and 37 to program B. Fifty-nine (82%) completed the detoxification and attended 2-month follow-up; 58 (81%) patients were followed up after 6 months, while 14 patients dropped out (Figure 2). At 12-month follow-up, 53 patients completed the protocol (77% in program A and 70% in program B). Baseline characteristics for dropped-out patients are presented in supplementary Table 3.
Flowchart for the study population.
Baseline characteristics.

Note: Age, headache days/month, migraine days/month, duration of medication overuse and days with acute medication/month are presented as mean (SEM). Gender, MOH-subtype, previous history of MOH and pharmacological preventive treatment are presented as N (%).
For the variable migraine days/month, n = 23 (program A) and n = 23 (program B), corresponding to the number of patients diagnosed with migraine.
Other preventive treatments cover valproate, venlafaxine, riboflavin and magnesium.

Distribution in percentage of headache diagnosis at baseline, 2, 6 and 12 months. N = 27 in program A; n = 26 in program B.
Outcomes for 6-month follow-up
Headache days/month were reduced significantly more in program A, by 46% (95% CI 34–58), compared to 22% (95% CI 11–34) in program B (p = 0.005). In program A, 70% reverted to episodic headache compared to 42% in program B (p = 0.04). Migraine-days/month were reduced by 7.2 in program A (p < 0.001) and by 3.6 in program B (p = 0.002) (Figure 5). There was no significant difference between the groups (p = 0.08). Both programs proved effective in reducing headache days/month (p < 0.001 for both programs) (Figure 4). The number of patients with CM was reduced to two (7%) in program A, and to 5 (19%) in program B (p = 0.60) (Figure 3).
Mean changes in number of headache days from baseline to 2, 6 and 12 months for the two programs. N = 27 (program A) and n = 26 (program B). The mean values and corresponding 95% CI are shown. *p < 0.05 for changes from baseline to each time point. Comparisons between the two programs were not calculated in terms of reduction in headache days/month. Mean changes in number of migraine days from baseline to 2, 6 and 12 months. N = 23 (program A) and n = 23 (program B). The mean values and corresponding 95% CI are shown. *p < 0.05 for changes from baseline to each time point. There was no significant difference between the two groups in comparison of reduction in migraine days/month.

Headache characteristics.
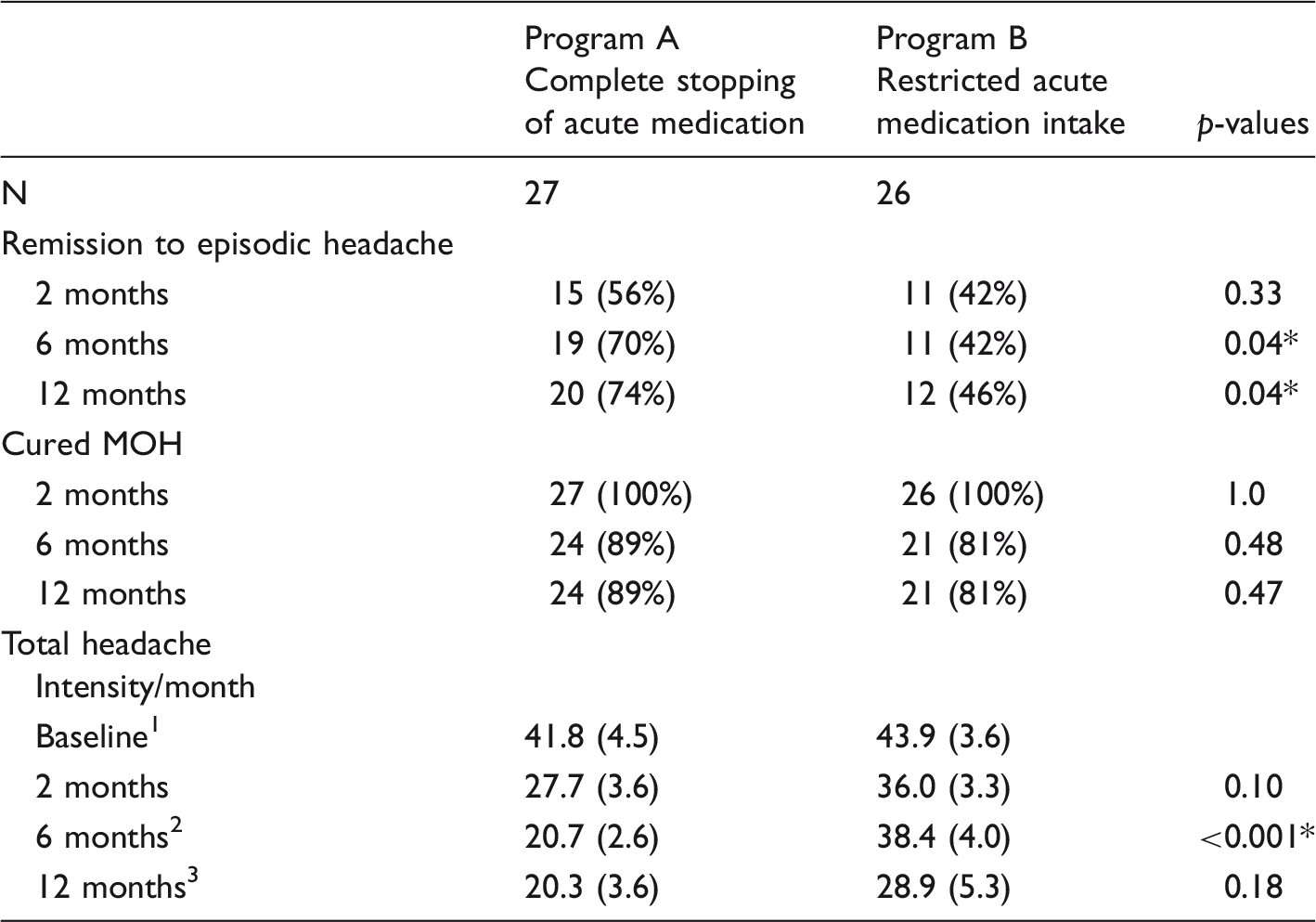
Remission to episodic headache, chronic migraine and cured MOH are presented as N (%). Days with acute medication/month and total headache intensity/month are presented by mean (SEM).
N = 23 (program A) and n = 20 (program B).
N = 25 (program A) and n = 19 (program B).
N = 22 (program A) and n = 12 (program B).
p < 0.05, indicating significant difference between program A and program B.
Outcomes for 2-month follow-up
Headache days/month were reduced by 41% (95%CI 28–55) in program A and by 26% (95% CI 16–36) in program B (p = 0.06). Both groups improved significantly in headache days/month, number with episodic headache, and migraine days/month (Table 2, Figures 4 and 5). The number of patients with CM was reduced from 15 (55%) at baseline to five (18%) in program A, and from 17 (65%) to eight (31%) in program B (Figure 3), with no difference between the programs in distribution of headache diagnoses (p = 0.60). All included patients completed detoxification, and were thus cured of MOH after 2 months (Table 2).
Outcomes for 12-month follow-up
Headache days/month were reduced by 45% (95% CI 33–57) in program A, and by 31% (95% CI 16–46) in program B (p = 0.14). Headache days/month and migraine days/month were significantly reduced in both programs (Figures 4 and 5), without any difference between the groups. More patients in program A (74%) than in program B (46%) reverted to episodic headache (p = 0.04). There was significant difference between the programs in the distribution of headache diagnoses (p = 0.03): Only one (4%) patient in program A and eight (31%) in program B fulfilled the CM criteria (Figure 3).
There was no significant difference between percentage reduction in days with acute medication, with 65% (95% CI 55–75) in program A and 56% (95% CI 44–67) in program B (p = 0.22). In program A 89% were cured of MOH, and 81% were cured in Program B (p = 0.47) (Table 2). The corresponding relapse rates were 11% and 19%, respectively.
Pharmacological preventive treatment
Preventive treatment.

All data are presented as N (%).
Intention-to-treat analysis
In ITT analyses, all 72 patients were included. Baseline characteristics are presented in Supplementary Table 1.
The reduction in headache days/month was 33% (95% CI 21–45) in program A and 21% (95% CI 13–29) in program B (p = 0.09) after 2 months, 34% (95% CI 21–48) in program A and 19% (95% CI 10–28) in program B (p = 0.06) after 6 months. After 12 months, headache days/month were reduced by 37% (95% CI 26–48) in program A, and by 24% (95% CI 13–35) in program B (p = 0.10). Both programs significantly reduced headache days/month after 2, 6 and 12 months (p < 0.001 in all cases). Numbers of patients with remission to episodic headache at 2, 6 and 12 months are presented in Supplementary Table 2.
Migraine days/month were reduced by 6.0 (95% CI 3.0–8.8) in program A and 3.1 (95% CI 1.0–5.2) in program B after 2 months (p = 0.09), by 5.1 (95% CI 2.0–8.2) in program A and 3.7 (95% CI 2.0–8.2) in program B after 6 months (p = 0.44) and by 6.1 (95% CI 3.1–9.1) in program A and 4.8 (95% CI 1.9–7.7) in program B (p = 0.52) after 12 months. Distribution of headache diagnoses at 6 and 12 months are presented in Supplementary Figure 1, and there was no significant difference between the programs (p = 0.87 at 6 months and p = 0.15 at 12 months).
There was no significant difference in the reduction of days with acute medication/month at 6-month follow-up (51% [95% CI 40–61] vs. 43% [95% CI 30–57]; p = 0.40) or at 12-month follow-up (52% [95% CI 41–63] vs. 49% [95% CI 38–61]; p = 0.74). Patients cured of MOH at 6 and 12 months, and headache intensity at 2, 6 and 12 months are presented in Supplementary Table 2.
Discussion
In this randomized, controlled study with carefully classified patients, we found that complete stop of acute headache medication during the two-month detoxification was the most effective treatment in terms of percentage reduction in headache days/month after 6 months. There was no difference in the dropout rate. Thereby, our results reject our hypothesis that restricted medication intake during detoxification would be the most effective and feasible treatment for MOH.
There was no significant difference in percentage reduction of headache days/month between the groups after 2 and 12 months. However, at month 12, more patients had reverted to episodic headache and fewer patients had CM in program A than in program B. The mean value for headache days/month in program A remained under 15 days/moth, the limit for chronic headache, at 2, 6 and 12 months. Contrarily, headache days/month in program B did not decrease below 15 days/month. Of note, the largest decrease in headache days/month and migraine-days/month had already occurred at month two.
For complete detoxification, we found a 41% reduction in headache days/month after 2 months, 46% after 6 months and 45% after 12 months, which is comparable to similar studies (4,13). In the ComoEstas-study, with 6-month follow-up, it was reported that headache days/month were rapidly reduced within the first month and further reduced from 25 days/month at baseline to 10 days/month after 6 months (approximately 60% reduction) (4). Munksgaard et al. reported a headache reduction of approximately 40% 12 months after initiation of a multidisciplinary detoxification program. Importantly, that study also included patients with post-traumatic headache who previously were reported to have lower headache reduction than in TTH and migraine patients (15). In program B, treatment with restricted intake of acute medication did not reduce headache days/month as much as previously reported in the mentioned studies. However, Hagen and colleagues reported a 5.1-day reduction from 24.1 days 12 months after detoxification (approximately 20% reduction). This is more similar to the reduction we report in program B (31%). This could be because patients in Hagen’s study were allowed to use NSAIDs up to two days/week during detoxification (16).
The ComoEstas-study reported that approximately 68% of patients who completed the protocol reverted to episodic headache, comparable to our results with 70% (program A) and 42% (program B) (4). The ITT analysis at 6-month follow-up was comparable with our study too (46.5% reverted to episodic headache in ComoEstas compared to 57% in program A and 32% in program B) (4). Munksgaard and colleagues also found that 61% reverted to episodic headache after 12 months, similar to the 74% in complete detoxification and 42% with restricted intake (13) in our study. Of note, program A cured 14 patients out of 15 for CM, whereas program B cured only 9 of 17. However, several of the above-mentioned results were not significant in the intention-to-treat analysis.
Despite a significantly higher reduction in headache days/month in program A, both programs proved effective in reducing headache days/month and migraine days/month. A restricted intake of acute medication compared with continuous medication overuse is still a step forward. According to clinical experience, some patients find a restricted intake more feasible compared to a complete stop. Some situations (e.g. difficulties with sick leave, social aspects) do not allow weeks with potentially severe rebound headache and withdrawal symptoms. Some MOH patients have a complicated form; for example, based on secondary headaches, with opioid- or benzodiazepine overuse, or severe co-morbidities (17,18). Patients with complicated MOH were offered inpatient treatment and were not included in this study. This may be a limitation, making the results less applicable to the more heterogeneous group of patients who may experience severe withdrawal symptoms in an outpatient setting. A high number of patients preferred an individual outpatient detoxification rather than the more time-demanding study set-up with patient education, and were thus not included. Nevertheless, our results can strongly support clinicians in the importance of detoxification in all patients with overuse and advising patients’ choice of treatment.
We also found a very high proportion of cured MOH (not fulfilling the MOH criteria) with no difference between the groups probably due to there being no significant difference in acute medication intake between the groups. Both at 6- and 12-month follow-up, 89% in program A and 81% in program B were cured, and this is comparable to the previously-reported 91% cured of MOH (for completers) in the ComoEstas study (4) and to 83% reported by Munksgaard et al. (13).
It may be discussed whether a primary outcome at 6-month follow-up was appropriate. We chose this time point to permit comparison of our results with the ComoEstas study – the largest, and a very central study in the detoxification field (4,19–22). Moreover, we expected a higher dropout rate from 6 to 12 months, which is why a primary outcome at 6-month follow-up was more feasible for the study design. However, it was still interesting to follow the patients for one year, since 94% of relapses occur within the first 12 months from detoxification (23).
There were no major adverse events related to either of the treatment programs. During the initial detoxification period, only normal withdrawal symptoms were reported. These minor adverse events were not systematically registered.
Patients with EM (±TTH), TTH (±EM) or CM are all abundant in DHC and present often with MOH. Therefore, it was of clinical importance to include all three groups in this study. Being equally distributed in the two programs, it is unlikely that different headache diagnoses had a major impact on the results.
The remaining question is why complete stopping had the highest impact on headache days/month. It could be due to limitations of the study, for example, there were 10% more patients with CM and MOH in program B than A, or to natural fluctuations in headache days/month, which we cannot control for due to lack of a control group (24,25). Also, the use of rescue medication was not systematically collected, which to a minor extent could have influenced the result. However, it could be hypothesized that the effect of complete detoxification has an earlier onset due to complete stopping of input to pro-nociceptive mechanisms caused by the medication overuse. It has been reported that patients with MOH present with central sensitization, which is normalized up to one year after detoxification with complete stopping of acute medication (26).
A tendency toward expressing “MOH” as “CM with medication overuse” has during the last decade become more widespread, although not described in the current ICHD3-beta (14). According to the classification, both diagnoses should be given: CM and MOH. It is definitely a clinical challenge to distinguish between CM and MOH when patients overuse acute medication, but the ICHD-3 beta also recommends detoxification in CM and MOH (14). In this study, 32 patients fulfilled the criteria for CM and had simultaneous medication overuse at baseline. Already, after a two-month detoxification without preventives, only 13 patients still fulfilled the CM criteria, suggesting that the rest were unlikely to have suffered from CM, but simply from MOH and underlying EM. Therefore, we suggest that it is highly important to add both diagnoses (CM and MOH) to these patients and initiate detoxification as the first step in the treatment plan.
There was no significant difference between the groups in the number of patients treated with preventives, and they were equally distributed in the programs. All patients were treated based on current Danish guidelines (11), and the scope of the study was not to investigate the role of preventives in detoxification. Change in preventives could still influence the results, but patients changing preventives were equally distributed between the two groups (30% vs. 38% from 2–6 months and 33% vs. 31% from 6–12 months; Table 3). In future studies, the role of pharmacological preventives in treatment of MOH must be addressed. Zeeberg et al. reported that only 47% of patients actually needed preventive treatment at discharge after detoxification (12). In our study, preventive treatment was not indicated for 26% and 15% of patients, respectively, after successful detoxification. In other MOH studies, preventives were initiated before and/or during the detoxification period to relieve headache symptoms (3–5). It has also been questioned whether detoxification is absolutely necessary, and whether MOH can be treated with preventives alone (27), which has only partly been addressed in the study by Hagen et al. (16). In the PREEMPT study (n = 904 with CM and medication overuse) Onabotulinumtoxin A reduced headache days/month by 8 (approximately 40% from baseline) after 6 months (28). Topiramate reduced migraine-days/month by 5.5 days (38%) in patients with CM with simultaneous medication overuse (29). The CGRP antibody Erenumab reduced monthly migraine days by 6.6 after 12 weeks in patients with CM (41% with medication overuse) (30). However, a recent study reported low long-term adherence to preventive treatment for patients with CM, where only 14% of patients continued the first initiated preventive treatment after 12 months (31). We found similar effects after complete detoxification, and recommend that detoxification should be considered as a treatment for patients with CM and MOH before preventive medication is considered.
Conclusion
Detoxification with a two-month complete stop of acute medication is highly successful and more effective for relative reduction of headache days/month and in reverting chronic to episodic headaches compared with reduced medication intake. Both detoxification programs reduced headache days/month, migraine days/month, and cured more than 80% of the patients of MOH.
We suggest that it should be considered in a standard care program for headache before initiation of a pharmacological prevention.
Clinical implications
Complete stop of medication overuse is the most effective detoxification program compared with restricted medication intake. Detoxification is a very effective treatment for converting chronic headache with medication overuse to episodic headache. The proportion of patients with chronic migraine was reduced from 55% to 18% by complete detoxification. Over 80% were cured of medication-overuse after 6 months. According to the Classification of Evidence by the American Academy of Neurology, this randomized controlled trial can be classified as a Class II study.
Footnotes
Acknowledgement
The authors thank the headache nurses Malene Kjærgaard Danø, Annette Vangaa Rasmussen, Hjørdis Rasmussen and Annette Fjeldborg Jonasson for planning and management of the patient education program, as well as collecting data. Also, we would like to thank project nurse Mette Frank Fisker for data management and data typing.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SB Munksgaard has undertaken lectures for Berlin-Chemie and UCB. RH Jensen has participated in clinical trials for ATI, Electrocore, Eli-Lilly and Linde Gas Ltd., and is a director in LTB, trustee in IHS and past-president in EHF, and has the following additional disclosures: Lectures for Pfizer, Berlin-Chemie, Allergan, Merck, and ATI. L Bendtsen has the following disclosures: Advisory board member for Allergan, Novartis and Biogen.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: LN Carlsen has received funding from the Tryg-Foundation. SB Munksgaard has received travel grants from Allergan and Merck.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
