Abstract
Background
Calcitonin gene-related peptide is an important target for migraine and other painful neurovascular conditions. Understanding the normal biological functions of calcitonin gene-related peptide is critical to understand the mechanisms of calcitonin gene-related peptide-blocking therapies as well as engineering improvements to these medications. Calcitonin gene-related peptide is closely related to other peptides in the calcitonin gene-related peptide family of peptides, including amylin. Relatedness in peptide sequence and in receptor biology makes it difficult to tease apart the contributions that each peptide and receptor makes to physiological processes and to disorders.
Summary
The focus of this review is the expression of calcitonin gene-related peptide, related peptides and their receptors in the central nervous system. Calcitonin gene-related peptide is expressed throughout the nervous system, whereas amylin and adrenomedullin have only limited expression at discrete sites in the brain. The components of two receptors that respond to calcitonin gene-related peptide, the calcitonin gene-related peptide receptor (calcitonin receptor-like receptor with receptor activity-modifying protein 1) and the AMY1 receptor (calcitonin receptor with receptor activity-modifying protein 1), are expressed throughout the nervous system. Understanding expression of the peptides and their receptors lays the foundation for more deeply understanding their physiology, pathophysiology and therapeutic use.
Introduction
Calcitonin gene-related peptide (CGRP) is an important sensory neuropeptide that is involved in pain modulation (1,2). CGRP has attracted particular interest in regard to its role in migraine and likely plays a role in other primary headache disorders and painful conditions (2,3). Presently, three investigational classes of drug aim to reduce CGRP activity to prevent and treat migraine (3). These are small molecule antagonists against a CGRP receptor and antibodies that either block receptor activity or bind directly to the CGRP peptide. This ‘first generation’ of CGRP-based treatments will likely lead to further drugs over time, which will result from a deeper knowledge of the CGRP system at a cellular and molecular level and a greater understanding of the molecular neuroanatomy of CGRP and its receptors. This information guides understanding of the relative role of central and peripheral processes in which CGRP participates in the pathophysiology of migraine, and other disorders.
Understanding CGRP biology is complex due to the existence of similar peptides in the same family, and shared receptor complexes. The aim of this review is to summarize information about the expression of CGRP and to compare this to its binding sites and molecularly defined receptors. We will detail what is known about the expression of CGRP and related peptides, where binding sites for labelled peptides are found, and what information is available for the molecular correlate(s) to that binding. We focus our attention on the central nervous system (CNS) and peripheral ganglia, with emphasis on brain regions that are of particular relevance to migraine. The sensory circumventricular organs (CVOs) will also be considered, as circulating factors can act directly on these sites within the brain.
Migraine and the nervous system
Historically, there has been considerable debate around the importance of central and peripheral mechanisms of migraine. This initially took the form of the competing vascular and neuronal hypotheses for migraine. It is now clear, based on recent clinical trials, that migraine can be treated through peripheral blockade of CGRP action (3,4). This does not preclude a central origin of migraine. Although the peripheral aspects of the trigeminovascular system are important in migraine, it is still worthwhile to consider central contributions (5). Numerous regions of the CNS are activated during a migraine attack and central phenomena are associated with migraine symptoms, such as the link between cortical spreading depression and migraine aura (3). It is unclear whether blocking central CGRP action may be beneficial or perhaps a hindrance in treating migraine. However, central CGRP receptors should be an important consideration for developing a ‘second generation’ of CGRP system-based treatments with potentially greater effectiveness or fewer side effects.
This review will focus on the craniofacial pain pathway and other brain regions associated with migraine. The craniofacial pain pathway begins with Aδ and C fibres of the trigeminal nerve, whose cell bodies are located peripherally in the trigeminal ganglia and project centrally primarily into the spinal trigeminal nucleus (STN) of the brainstem and into the C1/C2 levels of the spinal cord. The STN is proposed as a possible site of migraine initiation and displays increased activity immediately prior to a migraine attack (6). Higher order processing of painful signals primarily involves the thalamus, insular cortex and somatosensory cortex. Interconnected with these regions are other sites, including the amygdala, raphe nuclei, periaqueductal gray (PAG), parabrachial area, gracile nucleus and locus coeruleus, which play roles in pain processing, proprioception, stress or aversion. Interestingly, in migraine patients, altered connectivity between these regions is reported, such as from the thalamus to the insular cortex and somatosensory cortex or from the PAG to the cortex and amygdala (7,8). Other brain regions involved in migraine may include the hypothalamus, implicated in attack initiation; the hippocampus, which displays greater pain-induced activity in migraine patients; and the cortex, cerebellum and visual network, which may be involved in cortical spreading depression and symptoms of migraine aura (9–11).
The calcitonin gene-related peptide family of peptides
CGRP belongs to a small family of structurally related peptides (Figure 1). There are two forms of CGRP, α and β. The broad term “CGRP” is used for either α or β, unless specifically noted. The other major members of this family are amylin, adrenomedullin (AM) and adrenomedullin 2 (AM2). Another member of this family, the calcitonin receptor-stimulating peptide (CRSP) was reported in some mammals (12); however, CRSP is not expressed in primates or rodents. Each of these peptides contains a conserved pair of cysteine residues that form a disulfide bond, creating a loop. They also all contain a C-terminal amide. Amylin and CGRP are the most closely related peptides in terms of amino acid sequence, resulting in overlapping actions in pain modulation and nutrient balance (13,14). The similarities between the peptides within the CGRP family causes significant overlap in their ability to activate each other’s receptors. This “blurred” receptor pharmacology is particularly evident at rodent receptors, where there is noticeable cross-reactivity. This makes working with this peptide family challenging because it is often very difficult to determine which of the many possible molecularly defined receptors actually mediates an effect for a given peptide (15).
Amino acid sequences of calcitonin gene-related peptide family members. Alignment of the amino acid sequences for human αCGRP, βCGRP, calcitonin (CT), AM, AM2 and amylin using the single letter code. AM and AM2 have been truncated at the N-terminal. All the peptides have a C-terminal amide (-NH2). A conserved disulfide bond between the two N-terminal cysteine residues is indicated by a solid line.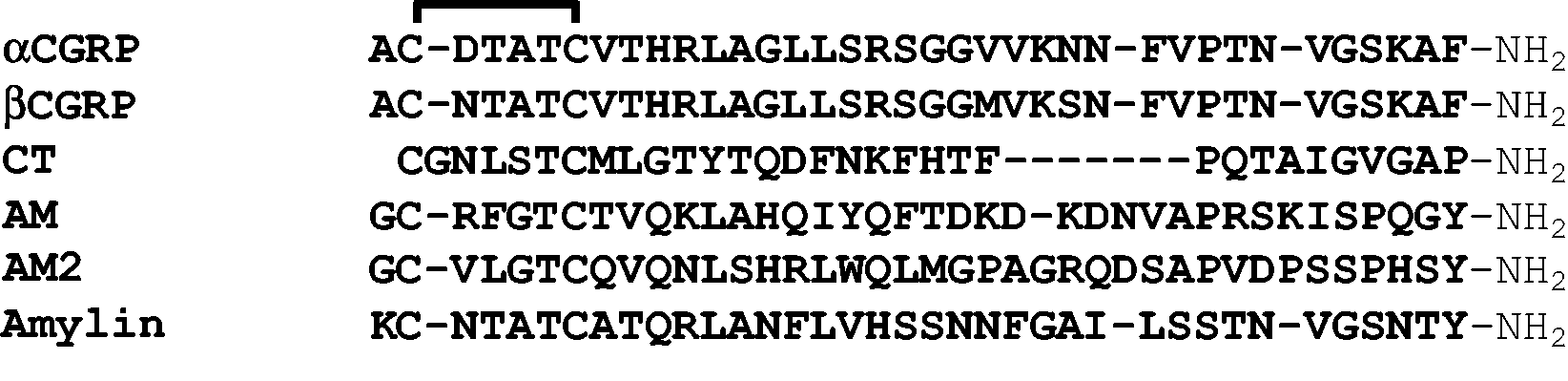
Receptor composition and pharmacology
The molecular composition of the CGRP family of receptors is illustrated in Figure 2, which highlights the overlapping activity of the peptides at the different receptors. Overlap occurs, not only because the peptides have shared features but also because the receptors themselves have shared and closely related components. All of the receptors within this family are G protein-coupled receptors (GPCRs). Two specific GPCRs form high affinity receptors for the different peptides due to their association with accessory proteins. There are three of these accessory proteins, receptor activity-modifying protein (RAMP) 1, 2 and 3. The CGRP, AM1 and AM2 receptors consist of the calcitonin receptor-like receptor (CLR) and RAMP1, RAMP2 or RAMP3, respectively (16,17). The AMY1, AMY2 and AMY3 receptors consist of the calcitonin receptor (CTR) with each RAMP (17,18). CLR alone does not appear to act as a functional receptor, whereas CTR is the receptor for calcitonin (16,17). Figure 2 shows that CGRP can activate both CLR/RAMP1 and CTR/RAMP1 equally. CGRP is weaker at other human receptor complexes, but there are clear deviations in other species (15). At rat CLR and CTR-based receptors, CGRP is potent at both RAMP3-based receptors (19). Amylin can potently activate CTR/RAMP1, CTR/RAMP2 and CTR/RAMP3 but not the CLR-based receptors. AM activates all three CLR/RAMP complexes but has a preference for CLR/RAMP2 and 3 (17).
The calcitonin gene-related peptide (CGRP) receptor family. The relative potency of CGRP, calcitonin (CT), AM, AM2 or amylin is shown below a schematic representation of the appropriate receptor complex (17). The potential overlap between the CGRP and amylin receptors is highlighted in purple. The receptor components are described in the legend.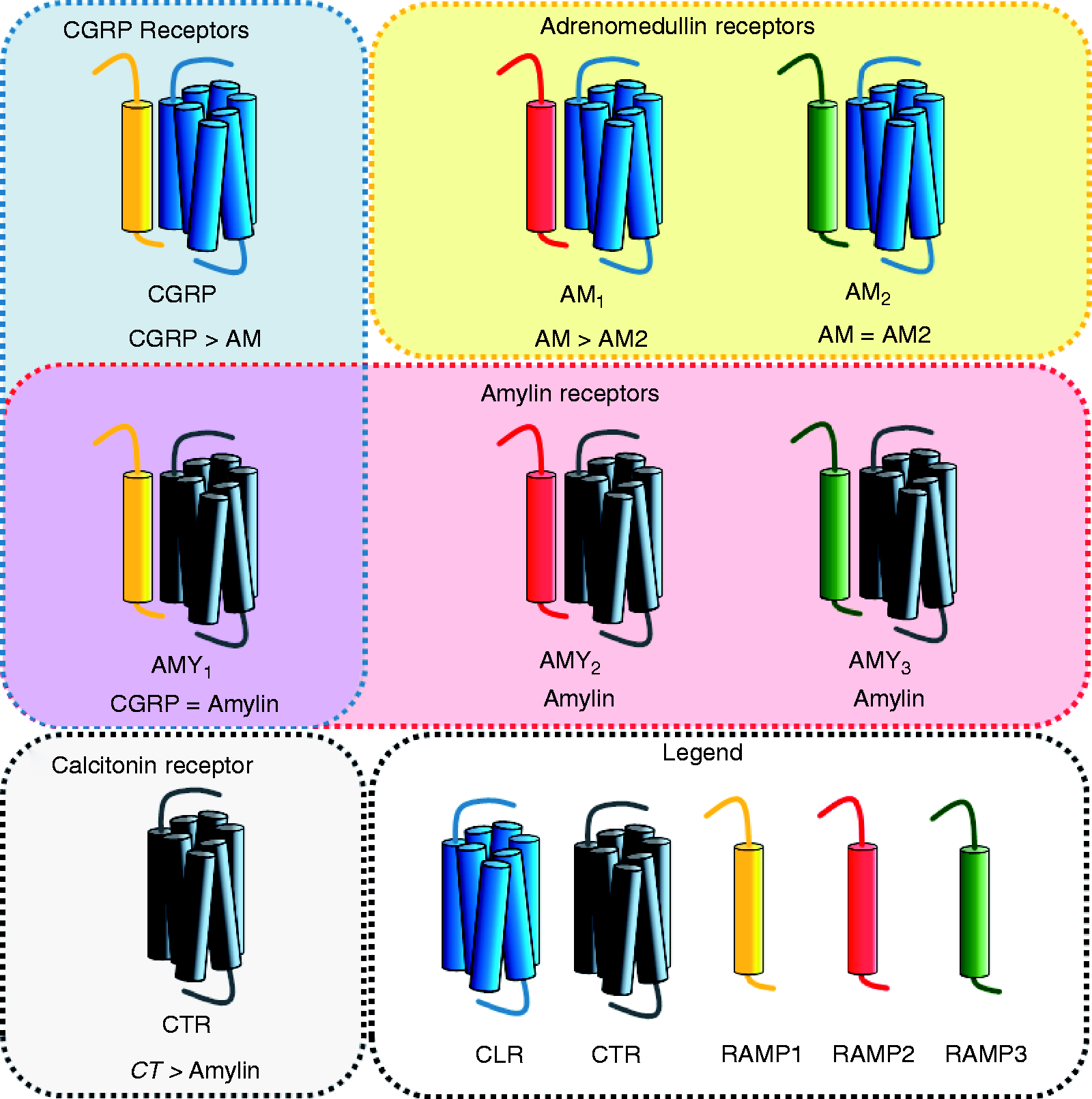
Summary of small molecule CGRP receptor antagonists (gepants) for migraine treatment.

No data, no published data is available.
Challenges associated with determining the molecular target for a given peptide
To understand the biology of the CGRP peptide family, it is crucial to define the molecular identity of the peptide and receptor responsible for a biological effect in physiological systems. The overlapping pharmacology described above represents a major challenge associated with studying this important family of receptors. Therefore, pharmacological approaches should be complemented with other methods. The measurement of mRNA can be a useful guide. However, the detection of mRNA does not necessarily mean that protein is present, especially in neurons where proteins can be transported to projections distant from the cell body (27). Stable membrane proteins including CLR, CTR and RAMPs may not need high levels of mRNA expression to maintain protein expression in steady-state cells. This has been illustrated where moderate to high levels of immunoreactivity were observed in regions where the corresponding mRNA was low or not detected (28,29).
The development of techniques including high-resolution confocal imaging has allowed researchers to detect the precise cellular localization of a receptor using antibodies. However, when used to directly detect protein expression, antibodies also have limitations. They require comprehensive validation to confirm specificity and selectivity for their targets (30). In many cases, antibodies are used without sufficient evidence of validation and it may not be possible to draw the conclusions that the authors suggest. For example, studies may simply lack the required information regarding the antibodies used to allow evaluation of the data (31) or the antibodies used have been shown to recognize multiple proteins. This prevents observed immunoreactivity from being conclusively associated with a single protein (30). The latter situation has been a significant issue for antibodies designed to detect RAMPs (32).
Even if a receptor subunit has been identified with a well-validated antibody, this alone is not meaningful for this receptor family, except when considering calcitonin activity at CTR alone. Expression of CLR or CTR by themselves says little about the receptor phenotype without further examining potential co-localization with a RAMP. RAMPs are also known to modify the signaling of other receptors, forming alternative complexes that complicate interpretation (33). However, valuable information has been obtained from studies showing co-localization between CLR and receptor-component protein or between CLR and RAMP1, indicating the likely presence of the CGRP receptor (34–36). The ideal solution maybe to develop antibodies that specifically target the heteromeric complex. This strategy was employed to detect the CLR/RAMP1 complex in the trigeminal ganglia, dura mater and the spinal trigeminal nucleus (37). However, antibodies that recognize heteromeric complexes are difficult to generate and characterize. Thus, protein expression data should be used alongside other experimental observations. For example, mRNA and peptide binding correlations were informative in early studies validating CLR/RAMP complexes as receptors for CGRP and AM (38). Another consideration relates to the level of expression reported. For many receptors, there is considerable amplification in signal following ligand-receptor binding. Hence, relatively low expression does not automatically translate into little function.
Regardless of method, much research relies on qualitative description of the biological target within anatomical locations. This is a highly subjective process, and is open to individual interpretation of intensity and location. Collating data, improving its accessibility, and standardisation all help to address these issues. For example, the Human Protein Atlas, the Allen Brain Atlas and other web-based tools provide excellent resources for expression data, assisting identification of patterns (39–41).
Peptide accessibility to the CNS and relevance of receptors in different locations
The blood-brain barrier (BBB) is an important consideration when looking at the origin, expression and activity of neuropeptides in the CNS. Peptides present in the blood may cross the BBB either through active transport or passive diffusion (42,43). Alternatively, circulating peptides can interact directly with CVOs. CVOs are highly vascularized, with fenestrated capillaries, allowing peptides access to these discrete parts of the CNS. The CGRP family of peptides do not freely cross the BBB (44). The proportion of peripheral amylin that can cross the BBB is low and is unlikely to be sufficient to activate receptors inside the BBB at physiological concentrations (45,46). Interestingly, the highest amount of labelled amylin that was reported to penetrate the brain was observed in the hypothalamus and medulla (46), which are associated with sensory CVOs and thus are permeable to circulating hormones. Hence, relatively high levels of amylin experimentally reaching these regions is not surprising. The reported actions of exogenous amylin at sites inside the BBB, such as the ventral tegmental area, are unlikely to be explained by amylin crossing the BBB (47–49). Alternative explanations could include local production of amylin or another member of the CGRP peptide family triggering receptor activation in these regions. The BBB penetrance of CGRP and AM have been examined primarily in vascular models, which suggest that neither peptide can cross the BBB when the vascular endothelium is intact (50–52). A more definitive study has been performed for AM, which, under normal conditions, does not appear to cross the BBB and penetrate the brain (53). Curiously, definitive experiments have not been performed for CGRP. However, given the lack of brain penetrance displayed by calcitonin, amylin and AM and data from vascular models, significant crossing of the BBB by CGRP seems unlikely. Overall, the low BBB permeability for CGRP family peptides suggests that CNS expression of these peptides is required for them to have activity inside the BBB.
AM and AM2
AM and AM2 expression in the CNS
AM is best known as a regulator of the cardiovascular and lymphatic systems. It is a potent vasodilator and is highly expressed in the vascular endothelium (54,55). Vascular AM may be involved in maintaining the BBB, cerebral circulation and the volume of cerebrovascular fluid (56–58). Expression in the vasculature may complicate expression analysis that relies on homogenized tissue, including mRNA analysis and membrane binding. mRNA and immunoreactivity for AM has been reported in the human and rat brain, localized to the vasculature, the choroid plexus and in neurons and glia of the hypothalamus, cerebellum and medulla (59–63). However, the cerebellum, which has been implicated in AM-regulated blood pressure control, was the only brain region where mature AM peptide was detected (59,64). Interestingly, in a genetic mouse model that was modified to lack AM expression in the CNS, altered responses to pain, anxiety and stress were observed (65,66). Similarly, AM has been reported to induce pain, and AM-like immunoreactivity was detected in dorsal root ganglia neurons and axon terminals in the dorsal horn from rats (67,68). These studies suggest that AM may play a physiological role in the CNS despite limited expression.
The physiology of AM2 is poorly understood; however, central or peripheral administration can modulate blood pressure (69). AM2 mRNA and immunoreactivity has been found in the pituitary and hypothalamus of humans and rats as well as the spinal cord and dorsal root ganglia of rats (70–74). AM2 may be involved in the hypothalamo-pituitary axis and the central control of blood pressure in the hypothalamus (69,75–77).
AM and AM2 binding sites in the CNS
AM binding sites have been measured in rat and human brain homogenates, with high levels of 125I-AM binding in the hypothalamus, thalamus and spinal cord (72,78–80). AM2 binding sites in the CNS have not been specifically studied. More work is required to determine the prevalence and location of AM and AM2 binding sites within the brain.
Molecular composition of AM and AM2 binding sites in the CNS
Expression of CGRP family receptor components and peptides and their binding sites in rodents.

M: mRNA detected by polymerase chain reaction or in situ hybridisation; P: protein/immunoreactivity in tissues; Y: specific radioligand binding detected; N: no binding detected; Empty space: no expression or data not available; CVOs: circumventricular organs; OVLT: organum vasculosum of lamina terminalis.
Very low expression.
Expression of CGRP family receptor components and peptides and their binding sites in primates including humans.

M: mRNA detected by polymerase chain reaction or in situ hybridisation; P: protein/immunoreactivity in tissues; Y: specific radioligand binding detected; N: no binding detected; Blank space: no expression or data not available; NS: non-specific binding; CVOs: circumventricular organs; OVLT: organum vasculosum of lamina terminalis.
Very low expression or binding.
Calcitonin
Calcitonin expression in the CNS
Canonically, calcitonin is expressed by thyroid C cells and regulates calcium homeostasis (88–90). The CALCA gene encodes calcitonin and CGRP mRNA (91). Tissue-specific alternative splicing of CALCA mRNA results in the preferential expression of CGRP mRNA in the nervous system; calcitonin mRNA is not detectable in these regions (1,88,92,93).
Calcitonin binding sites in the CNS
Although calcitonin does not appear to be expressed in the nervous system, binding sites for calcitonin are found in many brain structures (94–96). Salmon calcitonin has frequently been used as a tool in calcitonin studies because it has higher affinity than human or rat calcitonin and can bind CTR alone, AMY1, AMY2 and AMY3 receptors with high affinity (18). Thus, depending upon the presence or lack of a RAMP, salmon calcitonin binding may indicate the presence of a receptor for amylin, CGRP or calcitonin. Salmon calcitonin binding sites have been examined in rat and primate nervous systems using autoradiography (Tables 2 and 3). The data are generally consistent between these studies with only minor differences. Overall, autoradiography shows high levels of salmon calcitonin binding in the hypothalamus, sensory CVOs and the nucleus accumbens, along with moderate binding in the bed nucleus of the stria terminalis (BNST), PAG and locus coeruelus (95,97–100). The spinal cord has minimal salmon calcitonin binding in the dorsal horn (95,97). There also seems to be very little binding in the cerebral cortex and in the cerebellum. Data from the human brain is more limited. In a single study, salmon calcitonin binding was examined by autoradiography in the human medulla (Table 3). Discrete binding sites were observed in several nuclei, including high densities of binding in the NTS and raphe nuclei. Very little binding was observed in the fibre tracts (96). In another study, human hypothalamic membranes showed high levels of salmon calcitonin binding (101). The variability in displacement of salmon calcitonin by human calcitonin between brain regions suggests that AMY receptors account for a significant proportion of salmon calcitonin binding sites (102,103). However, more work is required to determine the prevalence and location of calcitonin binding sites within the human brain, and their direct relevance to calcitonin, amylin and/or CGRP activity.
Molecular composition of calcitonin binding sites in the CNS
Consistent with widespread calcitonin binding sites, CTR mRNA and protein expression has been observed throughout the nervous system of rodents (Table 2). Several splice variants of the CTR have been reported. These variants are not necessarily conserved across species and can display altered functions (104). This review does not distinguish between these, because there are no tools to determine the expression profile of specific splice variants at the protein level. We discuss CTR only in general due to limited data, even at the mRNA level. In the peripheral nervous system, CTR staining was observed in neuronal cell bodies in the trigeminal and dorsal root ganglia (20,29). Centrally, dense protein expression was reported in fibres and cell bodies of brain regions including the nucleus accumbens, the substantia nigra, the NTS and the area postrema (105). Moderate staining of cell bodies and fibres was observed in the spinal cord, amygdala, PAG and dorsal raphe nucleus (105). Limited but discrete staining has also been observed in the thalamus, BNST and regions of the somatosensory cortex (29,105). Interestingly, CTR protein has also been reported in several regions involved in the perception and higher-order processing of craniofacial pain (Table 2). The pattern of CTR expression in human neural tissues is similar to that of rodents, although data are limited to the trigeminal ganglia and the medulla (Table 3; (20,106). Widespread expression of CTR was observed in the human medulla, including the NTS, STN, cuneate, gracile and hypoglossal nuclei (106). Considering the lack of calcitonin expression in central and peripheral nervous tissues and its limited ability to cross the BBB, it is surprising that CTR is so widely expressed within the nervous system. This suggests that the CTR probably functions in concert with one or more RAMPs inside the nervous system to bind amylin, CGRP, or another peptide.
Amylin
Amylin expression in the CNS
The pancreatic β-cell is a major site of amylin expression and releases amylin into the circulation to induce meal-ending satiation (13). Early reports show a lack of amylin expression in the nervous system (107,108). However, later work reported limited amylin expression within the brain and in sensory neurons (Table 2; (109–111). It is likely that some amylin antibodies can also detect CGRP and some CGRP antibodies can detect amylin. Potential cross-reactivity between amylin and CGRP antibodies may account for some discrepancies between studies (112). Hence, care should be taken to confirm that antibodies detect only the desired target. More recently, amylin mRNA expression was reported in the preoptic area and BNST neurons of lactating dams (113). Amylin mRNA and immunoreactivity has also been reported in hypothalamic neurons in mice (114). Although amylin expression has not been examined in the human brain (Table 3), the limited amylin expression in the CNS suggests that its central actions likely occur predominantly outside the BBB through the CVOs, although discrete expression in a limited number of CNS regions is possible.
Amylin binding sites in the CNS
Despite the reportedly low expression of amylin in the CNS, 125I-amylin binds with widespread distribution in rat brain when measured using autoradiography (Table 2; (115)). This included moderate to high levels of binding in the nucleus accumbens, BNST, amygdala, hypothalamus, locus coeruleus, the NTS and the sensory CVOs – the area postrema and subfornical organ. These CVOs can access circulating amylin, consistent with the physiology of amylin following its release from the pancreas (13,116). A similar pattern of binding was observed in primates, with the hypothalamus described as having the densest binding of 125I-rat amylin (Table 3; (95)). If amylin is produced in the hypothalamus, then hypothalamic binding sites for amylin imply a paracrine action of the peptide. However, the physiological relevance of other amylin binding sites inside the BBB is not clear.
Molecular composition of amylin binding sites in the CNS
Amylin potently activates CTR in complex with RAMP1, RAMP2 or RAMP3. As described above, CTR is present in many regions of the brain. In a similar manner to the AM receptors, the presence of CTR alone is not sufficient evidence for the existence of a high affinity amylin receptor. RAMP must be co-localized. For RAMP2 and RAMP3 there are only mRNA studies in rodents (Table 2; (86)). Interestingly, CTR, RAMP1, RAMP2 and RAMP3 mRNA were all identified in single area postrema neurons (117). For RAMP1, there is protein data in the context of the CGRP receptor, which is discussed below (Tables 2 and 3). There has been very little investigation of CTR alongside RAMP1. To date, only a single study has examined the co-expression of CTR and RAMP1 proteins using validated antibodies. This study suggested that these proteins are co-expressed in neuron cell bodies from human or rat trigeminal ganglia and in fibres from the human spinal trigeminal tract (20). As noted above, given the lack of amylin in the brain, it is unclear why CTR and RAMP proteins are co-expressed or why is there so much CTR in the brain. One possibility is that the CTR acts as a receptor component for another endogenous ligand. It has been suggested that the AMY3 receptor could be activated by amyloid β42 and may contribute to Alzheimer’s disease (118). However, this is difficult to rationalize based on the known structure-function relationships between the receptors and their ligands for the CGRP peptide family (119–121), and contradicts the generally positive benefits amylin is reported to have in Alzheimer’s disease models (122). Furthermore, amyloid β42 was unable to activate amylin receptors in a separate study (123). A more likely explanation is that CTR complexes with RAMP1 to form receptors for CGRP in the CNS. Perhaps both the CLR/RAMP1 CGRP receptor and AMY1 are needed to exert the full repertoire of effects of CGRP.
Calcitonin gene-related peptide (CGRP)
CGRP expression in the CNS
CGRP is implicated in a wide range of physiological effects including vasodilation, nociception and energy metabolism (2,124–126). In most studies, αCGRP and βCGRP cannot be distinguished, partly due to a lack of specific antibodies. However, αCGRP and βCGRP mRNA are both expressed in the central and peripheral nervous systems and can coexist in a single neuron (127–130). CGRP immunoreactivity was reported throughout the nervous system of αCGRP knockout mice, suggesting that βCGRP is present throughout the nervous system (131).
CGRP expression has been primarily studied in rats; despite its value, primate and human data are more limited (Tables 2 and 3). Unsurprisingly, CGRP has widespread expression. Peripherally, CGRP is expressed in Aδ and C fibres of the trigeminal nerve and neuronal cell bodies in the trigeminal ganglia and in dorsal root ganglia neurons (1,2,70,82,93,132–138).
In the spinal cord, CGRP is expressed in fibres in both the ventral and dorsal horns of rats, predominantly in the outer laminae (70,82,92,139,140). Some cell bodies containing CGRP were also detected in the ventral horn (110). In human tissue, CGRP immunoreactivity was present in laminae I and II, consistent with rat studies (141).
In the brain, CGRP expression is abundant and there are discrete patterns of expression in regions studied in the rat (2,81). Areas positive for CGRP mRNA or protein in rats include the brainstem, cerebellum, midbrain, pituitary, hypothalamus, hippocampus, thalamus, amygdala and the BNST (Table 2). In most brain regions, CGRP immunoreactivity was present in the soma of neurons (81). CGRP-expressing fibres are less common, with the exception of the septal nucleus (81,142). The brainstem appears to have particularly high expression of CGRP, with staining in neuronal cell bodies of all brainstem nuclei, including the locus coeruleus, raphe nuclei, gracile nucleus and STN, consistent with a central role in migraine (81,92,139,141). In contrast, the hippocampus has generally weak CGRP staining in neurons (81,92). The Purkinje cells of the cerebellum stain strongly for CGRP in rat (143). Data describing the expression of CGRP in peripheral and central glial cells is somewhat conflicting, with both positive and negative reports (35,132,144). Of all the CGRP family peptides, CGRP appears to be the most abundant and most widespread in the CNS.
CGRP binding sites in the CNS
Consistent with its widespread expression, autoradiography has detected CGRP binding sites throughout the brain and spinal cord (Tables 2 and 3; (145–147)), with some discrepancies between studies. For example, contrary results were reported for 125I-CGRP binding in the nucleus accumbens (147,148). Species differences may also exist – for example, in rat brain, 125I-CGRP binding sites in the nucleus accumbens were more sensitive to competition with salmon calcitonin than corresponding sites in monkey brain. Experimental differences make these results difficult to compare, but this may suggest differences in the underlying molecular nature of the CGRP receptors present (148,149). The reported CGRP binding sites could be a mixture of CGRP and AMY1, with contributions from other receptors such as AM2 and AMY3 receptors, depending on the species.
Molecular composition of CGRP binding sites in the CNS
CGRP and AMY1 receptors comprise either CLR or CTR and RAMP1 proteins (17). The majority of research has focused on examining the canonical CGRP receptor: CLR and RAMP1 (Table 2; (70,86,150–152). The availability of specific CGRP receptor antibodies has been relatively recent and their usage limited; however, several studies have mapped the expression of these receptor proteins in the nervous system (Tables 2 and 3; (28,35,70,81).
In rat and human trigeminal ganglia, CLR and CTR mRNA and protein have been detected (Tables 2 and 3). CLR and CTR appeared to have limited co-localization, whereas both co-localize with RAMP1 in neuron cell bodies (20,35,132). This suggests that both AMY1 and CGRP receptors are present. Interestingly, CGRP and CLR/RAMP1 are rarely localized within the same neurons, indicating that CGRP released from neuron cell bodies may act locally upon a distinct population of neurons (35,132,133). It is not known whether CTR/RAMP1 and CGRP are co-localized in the trigeminal ganglia, but it is possible that CGRP acts in an autocrine manner via this receptor, and a paracrine manner via CLR/RAMP1.
In the rat, CNS, CLR and RAMP1 appear to be widely expressed (Table 2; (81,82)). CLR and RAMP1 immunoreactivity was reported primarily in neuronal processes or fibres. In the brainstem, CLR and RAMP1 staining was observed in fibres from several nuclei including the STN, gracile and hypoglossal nuclei. Staining was also observed in cell bodies and some fibres in the locus coeruleus and raphe nuclei (81). Fibres that span the cerebral cortex of rats were reported to express CLR and RAMP1 (81). Staining was also reported in the Purkinje cells and neuronal processes in the molecular layer of the cerebellum (81,143). Some staining for CLR and RAMP1 was observed in fibres from thalamic nuclei and the cell bodies of neurons in the PAG (81,153). Data from human and primate CNS are more limited (Table 3). In the human brain, CLR/RAMP1 protein has been co-localized in the STN and cerebellum (34,82). The pattern of expression is similar to that reported in rats. It has been suggested that there may be more RAMP1 present than CLR in some regions, and that CGRP binding does not always coincide with CLR mRNA expression (28,86,152). Attempts have been made to describe the ratio of CLR to RAMP1, but this has not been conclusively quantified (81,86). The presence of AMY1 receptors, which could act as a receptor for CGRP, could account for any discrepancies.
The widespread expression of CTR mRNA and immunoreactivity throughout the nervous system is described above. CTR is reported at many sites that express RAMP1 throughout the brain (29,105,106). Additionally, CTR and RAMP1 protein were co-localized in the human spinal trigeminal tract (20). Further studies are required to determine the extent of AMY1 receptor expression within the nervous system and the importance of this receptor for the physiological actions of CGRP. Tables 2 and 3 clearly identify the knowledge gaps that need to be filled in order to understand the contribution of this receptor to CGRP (or amylin) biology.
Concluding remarks
The expression of CGRP and related peptides has been extensively studied in the CNS. Coupled with receptor binding and expression studies, this research provides some insight into how these peptides may contribute to various biological functions. However, the overlapping expression and receptor pharmacology of these peptides, coupled with the lack of specific antibodies and effective pharmacological tools, has made determining the specific receptors involved in pharmacological and physiological responses difficult. There is still more that needs to be learned. The physiological contributions of the two receptors for CGRP remains poorly described. The co-expression of the subunits for the AMY1 receptor (subtype) has only been detailed in the trigeminal ganglia and spinal trigeminal tract. Although the molecular composition of receptors for CGRP in the CNS is not always clear, CGRP and its receptor components are widespread throughout the CNS. In particular, CGRP and components for both CGRP and AMY1 receptors are present at several central sites involved in processing of pain and other migraine symptoms. The potential role of the CVOs in responding to circulating signals, such as increased peripheral CGRP during a migraine attack, should also be considered. In the future, both peripheral and central sites could be targeted to more effectively treat migraine.
Article highlights
CGRP is expressed throughout the nervous system. Amylin and adrenomedullin display limited expression at discrete sites in the brain. CGRP peptide family members do not cross the blood-brain barrier in a physiologically relevant manner. The components of two receptors that respond to CGRP, the CGRP receptor (CLR and RAMP1) and the AMY1 receptor (CTR and RAMP1), are expressed throughout the nervous system.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Christopher S Walker and Debbie L Hay have received research support from Alder Biopharmaceuticals.
Funding
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Christopher S Walker is supported by a Sir Charles Hercus Health Research Fellowship from the Health Research Council of New Zealand. Debbie L Hay is supported by a James Cook Research Fellowship of the Royal Society of New Zealand. Erica R Hendrikse is supported by the Barbara Basham Doctoral Scholarship administered by the Auckland Medical Research Foundation and Perpetual Guardian.
