Abstract
Introduction
Migraine and cluster headache are challenging to manage, with no tailored preventive medications available. Targeting the calcitonin gene-related peptide (CGRP) pathway to treat these headaches may be the first focused therapeutic option to date, with the potential for promising efficacy.
Methods
We systematically searched PubMed and clinicaltrials.gov for randomized controlled trials investigating the preventive potential of monoclonal antibodies against the CGRP pathway in the treatment of migraine and cluster headache.
Results
The literature search returned a total of 136 records, of which 32 were eligible for review.
Discussion
Clinical data from phase II and III trials of the four monoclonal antibodies targeting the CGRP pathway: Eptinezumab, erenumab, fremanezumab, and galcanezumab, collectively show a positive effect in the preventive treatment of episodic and chronic migraine. Multiple phase II and III trials are under way to further determine the efficacy and safety of this new drug class. It may be particularly important to assess the cardiovascular effects of long-term CGRP blockade. Phase III trials are also currently in progress for the preventive treatment of cluster headache.
Conclusion
Efficacy of anti-CGRP monoclonal antibodies spells a promising future for the many patients suffering from migraine, and possibly also for the smaller but severely-affected population with cluster headache.
Introduction
Migraine and cluster headache are recurring painful primary headaches, often treated with a combination of acute and preventive medication. Preventive therapy may help reduce attack frequency and severity as well as alleviate accompanying disability. While clinicians have a broad armamentarium of preventive medications to choose from, none of these remedies were developed specifically to target the molecular pathway of either migraine or cluster headache. This lack of specificity poses a challenge due to the unfavorable adverse events associated with these therapies, inadvertently affecting the treatment outcome. A systematic review of observational studies and randomized controlled trials (RCTs) of three migraine preventive drugs (propranolol, amitriptyline, and topiramate) demonstrated poor adherence and persistence to oral preventive therapies for migraine. Both parameters declined over the treatment duration, with adherence dropping below the acceptable threshold of 80% as early as 6 months after treatment initiation (1). Adverse events were the most common cited reason for discontinuation. In chronic migraine, preventive therapy with antidepressants, beta blockers, or anticonvulsants has shown particularly low adherence, ranging from 26% to 29% at 6 months and 17% to 20% at 12 months (2). While data on adherence to preventive treatment in cluster headache has not been investigated systematically, we know that treatment options are limited and that 10–20% of patients are either refractory to treatment or proceed to develop drug resistance (3). In addition, the brief duration of cluster headache attacks can make abortive treatment a challenge (4), further stressing the importance of effective preventive therapy options.
Due to the suboptimal adherence and tolerability of existing treatments, there is an obvious unmet need for preventive medication specifically targeting the pathophysiological mechanisms underpinning migraine and cluster headache. Neuropeptides that are central in the pathophysiology of these debilitating headaches, including calcitonin gene-related peptide (CGRP), may be the optimal target for such specifically-tailored treatment.
CGRP-targeted treatment was first introduced with the small molecule CGRP receptor antagonists (the –gepants); olcegepant for intravenous administration (5) and telcagepant for oral administration (6). A number of CGRP receptor antagonists were later tried for both acute and preventive treatment of migraine (7–10), and despite beneficial effects on reducing migraine headache days, development was halted largely due to concerns about hepatotoxicity (6–8).
Currently the focus is on the latest advent in migraine preventive therapy: Monoclonal antibodies (mAbs) against either the CGRP ligand or the CGRP receptor. The initial reports from phase II studies on both episodic and chronic migraine have shown promising results (11–15), with more phase II and III trials under way to further elucidate the efficacy and tolerability of this targeted treatment option. In addition, anti-CGRP mAbs are currently also being tested for the prevention of cluster headache.
Here, we perform a systematic review of the current clinical data on anti-CGRP mAbs in the preventive treatment in migraine. We will discuss the rationale for treating cluster headache with anti-CGRP mAbs, and finally we will present ongoing trials testing anti-CGRP mAb treatment of both migraine and cluster headache.
Methods
Completed phase II and III randomized placebo-controlled trials of anti calcitonin gene-related peptide monoclonal antibodies in the prophylactic treatment of migraine.

AE: adverse event.
★Significantly different from placebo.
*Preliminary results have been presented, complete results are pending publishing.
N/A: not applicable.
Ongoing phase II and III randomized placebo-controlled trials of anti calcitonin gene-related peptide monoclonal antibodies in the prophylactic treatment of migraine.

Finally, we searched the website cgrpforum.org to include the latest, unpublished presentations of clinical data from ongoing anti-CGRP monoclonal antibody trials. This search was also conducted on 7 August 2017.
Prior to publication, two press releases from 2017 on the preliminary results of two phase III trials investigating TEV-48125 were identified and added to the results.
Results
The PubMed search returned 92 records, of which seven were eligible for review. The clinicaltrials.gov search returned 44 records, of which 25 were eligible for review (Figure 1). Thus, a total of 32 articles form the basis of this review, consisting of published or preliminary clinical data of anti-CGRP monoclonal antibody treatment in migraine (n = 14, Table 1), and ongoing trials testing anti-CGRP monoclonal antibody treatment in migraine (n = 12, Table 2) and cluster headache (n = 6, Table 3).
Flowchart of search protocol and results. Ongoing phase III randomized placebo-controlled trials of anti calcitonin gene-related peptide monoclonal antibodies in the prophylactic treatment of cluster headache. N/A: not applicable.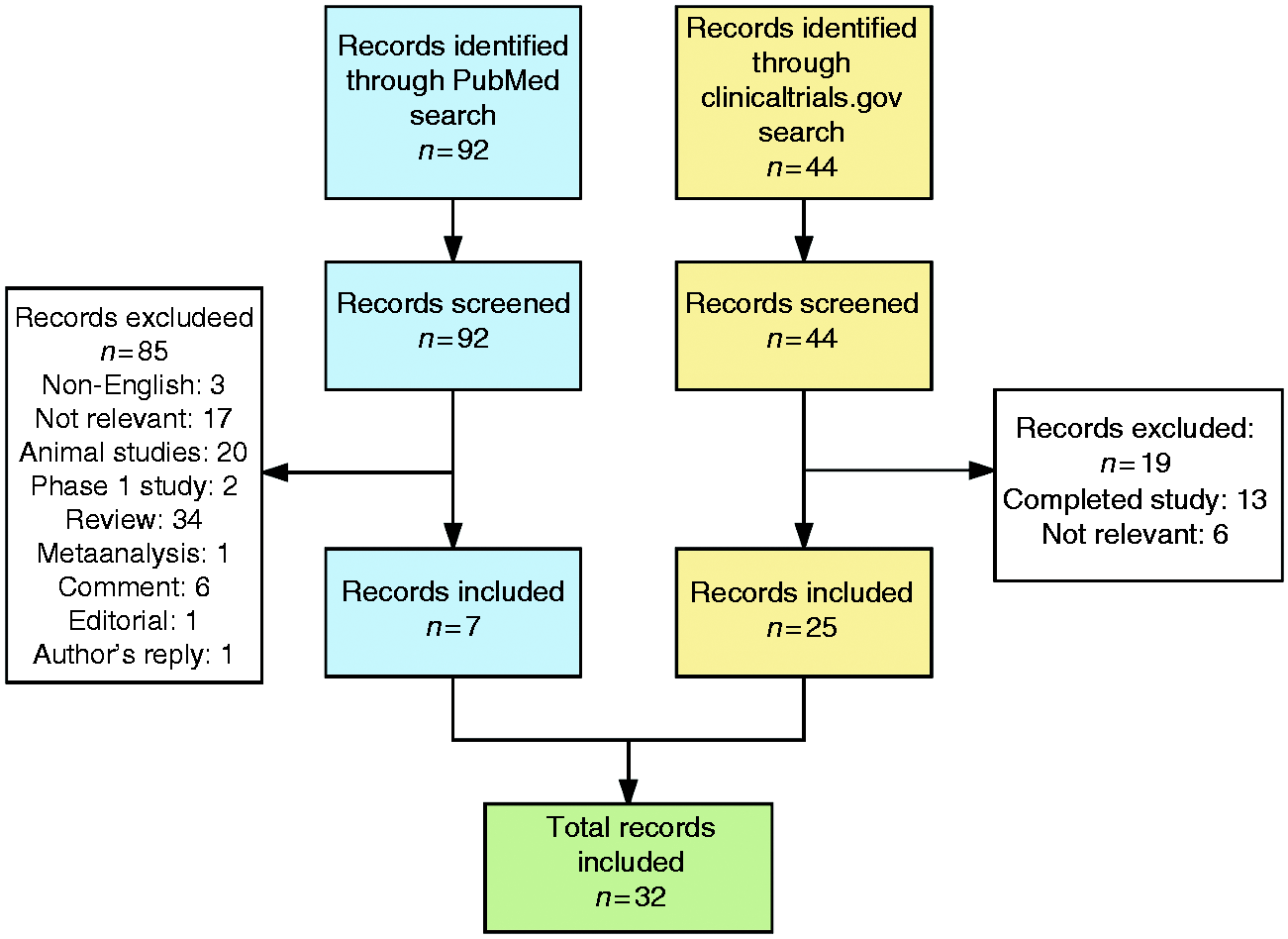

The search on cgrpforum.org (16) returned 24 records, of which five reported on preliminary results of ongoing trials. These preliminary results are also presented in Table 2.
Discussion
The first line of evidence: CGRP receptor antagonists (the –gepants)
Various small molecule CGRP receptor antagonists have been studied for both preventive and acute migraine treatment (5,7,10,17–21), providing support for targeting the CGRP pathway as a plausible treatment option for migraine. Early trials demonstrated the efficacy of CGRP receptor antagonists olcegepant (administered intravenously) and telcagepant (administered orally) comparable to the triptans (5,19,21). Further development of olcegepant was hindered due to difficulty in obtaining an oral formulation. Later, telcagepant was also tested for its migraine preventive properties, with participants randomized to 140 mg (n = 265) or 280 mg (n = 264) twice daily for 12 weeks. However, the trial was discontinued prematurely due to elevated levels of serum alanine aminotransferase (ALT) and aspartate aminotransferase (AST) in a subset of patients (n = 13 for ALT, n = 7 for AST), raising concerns about hepatotoxicity. Interestingly though, interim analysis up to termination showed that telcagepant was effective in reducing both mean monthly headache days and migraine days compared to placebo (18). Telcagepant was also investigated in women suffering from perimenstrual migraine (8). Participants were randomized to telcagepant (n = 3018) or placebo (n = 1502) for seven consecutive days perimenstrually. The study showed no statistical effect of telcagepant versus placebo in reducing mean monthly headache days; however, the authors reported a nominally-significant reduction in on-drug headache days of 0.4 headache days per week. This trial also revealed elevated transaminase levels (n = 13 for ALT, n = 7 for AST) associated with telcagepant treatment.
MK-3207 was the second orally bioavailable CGRP receptor antagonist investigated for the acute treatment of migraine. Despite a positive dose-response trend across all doses for 2-hour pain freedom, further development of MK-3207 was also halted due to elevated liver enzymes in a subset of patients (7). Select other CGRP receptor antagonists investigated include BI 44370 TA (17) and BMS-927711 (rimegepant) (10).
Anti-CGRP monoclonal antibodies in migraine treatment
Anti-CGRP mAbs are target-specific, a feature that may diminish or eliminate adverse events relating to other organ systems. Furthermore, the metabolism of anti-CGRP mAbs differs from that of CGRP small molecules, as the antibodies are renally excreted or excreted through the liver without undergoing hepatic processing and thus do not cause hepatotoxicity. This aspect is of particular interest when considering the elevated liver enzymes observed with CGRP receptor antagonists. Also, the long half-life of anti-CGRP mAbs allows for less frequent administration, making this drug class optimal for preventive treatment (22).
Currently, four anti-CGRP mAbs are in development after showing promising results in phase II trials. Additional phase II and III RCTs are in progress to further clarify the efficacy and safety of these new drugs. Of the four currently known anti-CGRP mAbs, one binds to the CGRP receptor (erenumab; AMG334) (11), and the remaining three bind to various locations on the CGRP molecule itself (eptinezumab; ALD 403, fremanezumab; TEV-48125, and galcanezumab; LY2951742) (12–14). The main routes of administration are intravenous and subcutaneous. Table 1 summarizes published and presented clinical data from phase II and III trials testing the efficacy and safety of the four anti-CGRP mAbs in the preventive treatment of episodic and chronic migraine. In the following, we will present an overview of these clinical findings in alphabetical order of the mAb.
Eptinezumab was tested for prevention of frequent episodic migraine, defined as 5–14 migraine days per month. Patients were randomized to receive a one-time dose of 1000 mg intravenous eptinezumab or placebo. In this RCT, weeks 1–4 were used as a stabilization period and weeks 5–8 were the primary efficacy endpoint, versus week 9–12 as applied by the remaining studies. At weeks 5–8, the net reduction in monthly migraine days was 1.0 days in the treatment group compared to placebo (13). The most common adverse events included upper respiratory tract infection and urinary tract infection. Three patients reported six serious adverse events, all deemed unrelated to the investigational drug. In anti-drug antibody assays, 14% in the active group had analytical results suggesting the potential for developing anti-eptinezumab antibodies during the study. However, the corresponding anti-drug antibody titres were low, with no obvious effects of immunogenicity on pharmacokinetic parameters of efficacy.
Erenumab was tested for the prevention of episodic migraine, defined as 4–14 migraine days per month. Participants were randomized to subcutaneous erenumab 7 mg, 21 mg, 70 mg or placebo, every 4 weeks for 12 weeks. Results from weeks 9–12 showed that only the highest dose, 70 mg, resulted in a significant reduction of migraine days after treatment compared to placebo, with a net reduction of 1.1 days in favor of erenumab (11). At week 12, the 50% responder rate was significantly greater for patients who received erenumab 70 mg compared to placebo, but not for lower doses of erenumab. There was no significant difference in monthly migraine attacks at week 12 in the erenumab 70 mg group compared to placebo.
The most common adverse events reported included nasopharyngitis, fatigue and headache, with similar prevalence across the treatment groups. Serious adverse events were reported for two patients across treatment groups; none of these were considered related to erenumab treatment. Nine patients had anti-erenumab neutralizing antibodies.
An open-label extension trial continued after the initial trial, testing the long-term effect of 70 mg erenumab in episodic migraine (23). Interim analysis showed that at week 64, compared to baseline, 65% experienced ≥50% reduction in migraine days, 42% experienced ≥75% reduction in migraine days, and 26% experienced a 100% reduction in migraine days. No new safety findings were identified in the open-label extension.
Erenumab has also been tested for the prevention of chronic migraine (15), defined as 15 or more headache days per month, of which eight or more were migraine days. Participants were randomized to subcutaneous erenumab 70 mg, 140 mg or placebo, administered every 4 weeks for 12 weeks. Results from weeks 9–12 showed a net reduction in monthly migraine days of 2.4 at both tried doses. The most common adverse events were injection site pain, upper respiratory tract infection, and nausea, and these were similarly prevalent in treatment and placebo groups. Serious adverse events were reported in a total of 15 patients across treatment groups; none led to discontinuation. Eleven patients receiving 70 mg and three patients receiving 140 mg had anti-erenumab binding antibodies; none had anti-erenumab neutralizing antibodies.
The phase 3 trials STRIVE (NCT02456740) and ARISE (NCT02483585) in episodic migraine have completed within the first half of 2017, with no published results as of yet. However, preliminary trial results were presented at the 2017 American Academy of Neurology Annual Meeting, 25 April 2017, Boston (24).
For STRIVE, results showed a net reduction in monthly migraine days at weeks 13–24 by 1.4 days for 70 mg erenumab and 1.9 days for 140 mg erenumab, both significantly different from placebo. Regarding ≥50% reduction in monthly migraine days, this endpoint was achieved for 43.4% in the 70 mg group and 50% in the 140 mg group versus 27% in the placebo group (odds ratios 2.13 and 2.81). Furthermore, the study showed greater reduction in the number of days involving use of migraine-specific medications in the active treatment groups (1.1 and 1.6 days versus 0.2 days with placebo, p = <0.001).
For ARISE, results showed a net reduction in monthly migraine days at weeks 9–12 by 1.1 for 70 mg erenumab, significantly different from placebo. It was found that 39.7% in the 70 mg group versus 29.5% in the placebo group experienced at least 50% reduction in monthly migraine days (odds ratio 1.6, p = 0.01). Also, there was a significant reduction in acute migraine-specific medication days between the 70 mg group (1.2 days’ reduction) and the placebo group (0.6 days’ reduction, p = 0.002).
Fremanezumab was tested as a preventive treatment of high-frequency episodic migraine, defined as 8–14 migraine days per month (25). Participants were randomized to subcutaneous fremanezumab 225 mg, 675 mg, or placebo, every 4 weeks for 12 weeks. Results from weeks 9–12 showed a net reduction in monthly migraine days of 2.81 days for the 225 mg group, and 2.64 days for the 675 mg group, both significantly different from placebo. Furthermore, at weeks 9–12, both active treatment groups showed a significant decrease in monthly headache hours, days with photo- and phonophobia, and days of acute migraine treatment compared to placebo. Also, there was a consistent reduction in migraine disability assessment (MIDAS) for both doses compared to placebo. The most common adverse events reported were injection site pain and erythema, equally distributed between the three treatment groups. No serious treatment-related adverse events were reported. Two patients tested positive for antibodies against fremanezumab before taking any study drug, and also tested positive after drug administration.
Like erenumab, fremanezumab has also been tested for chronic migraine (12). Patients were randomized to receive subcutaneous fremanezumab 675/225 mg (675 mg in the first treatment cycle and 225 mg in the second and third treatment cycles), 900 mg, or placebo.
At weeks 9–12, the net reduction in mean monthly headache hours was 22.74 hours in the 675/225 mg group and 30.41 hours in the 900 mg group, both significantly different from placebo. After the third treatment cycle, there was also a significant reduction in the least square mean of moderate or severe headache days compared to baseline and compared to placebo, with a net reduction of 1.84 days in the 675/225 mg group, and 1.96 days in the 900 mg group.
A post-hoc analysis revealed that the beneficial effect of fremanezumab set in within the first week of treatment, with a net reduction in headache hours of 6.23 hours for the 675/225 mg group, and 8.52 hours for the 900 mg group, compared to placebo (26).
The most common adverse events were mild injection site pain and pruritus. No treatment-related adverse events were serious. Two patients showed antibodies against fremanezumab before receiving study medication, with no changes to the concentration after drug administration, for which reason the antibody response was considered not related to treatment.
Most recently, two phase III studies have been completed with fremanezumab, aimed at assessing the mean change in monthly migraine days and the percentage of patients with adverse events in episodic (NCT02629861) and chronic (NCT02621931) migraine. Unique for these two studies, the efficacy and safety of monthly versus quarterly dose administration was also tested. Preliminary results were announced in press releases in May and June 2017 (27,28), showing that fremanezumab met all primary and secondary endpoints across both the monthly and quarterly dosing regimens for both episodic and chronic migraine. In episodic migraine, there was a monthly decrease in migraine days of −3.7 for fremanezumab versus −2.2 days for placebo for the monthly dose regimen, and −3.4 days for the quarterly dose regimen. In chronic migraine, patients experienced a decrease in monthly headache days of −4.6 for the monthly dose regimen and −4.3 for the quarterly dose regimen, compared to −2.5 days after placebo.
Galcanezumab also underwent a phase II trial for the prevention of episodic migraine. Patients were randomized to subcutaneous galcanezumab 150 mg or placebo every 2 weeks for 12 weeks. The net reduction in monthly migraine days between galcanezumab and placebo was 1.2 days in favor of galcanezumab (14). Also, the galcanezumab group showed greater reduction in number of headache days (least squares mean difference −1.3, 90% CI −2.1 – −0.5), number of migraines plus probable migraine headache days (least squares mean −1.3, 90% CI −2.2 – −0.5), and the number of migraine attacks (least squares mean −0.8, 90% CI −1.3 – −0.3). Finally, there was a higher degree of ≥50% responders in the galcanezumab group compared to the placebo group (odds ratio 2.88, 90% CI 1.78 – 4.69). Adverse events occurred more frequently with galcanezumab than with placebo, the most common being injection site pain, erythema, upper respiratory tract infections, and abdominal pain. A total of six serious adverse events were recorded, none of which were considered related to the investigational drug.
Most recently, three phase III studies have been completed using galcanezumab, and preliminary results were presented at the 2017 Annual Scientific Meeting of the American Headache Society in June 2017. Results for episodic migraine in the EVOLVE-1 (NCT02614183) study show a net reduction in monthly migraine days of 1.9 days for 120 mg and 1.8 days for 240 mg, significantly different from placebo (29). The EVOLVE-2 study, also in episodic migraine (NCT02614196), showed a net reduction in monthly migraine days of 2 days for 120 mg and 1.9 days for 240 mg, both significantly different from placebo (29). For chronic migraine, the REGAIN study (NCT02614196) showed a net reduction in monthly migraine days of 2.1 days for 120 mg and 1.9 days for 240 mg, both significantly different from placebo (29).
Collectively, none of the four tested anti-CGRP mAbs showed indications of serious substance-specific adverse events in the phase II RCTs. Importantly, none of the tested anti-CGRP mAbs showed signs of altered laboratory results, including elevated liver enzymes. In the fremanezumab trial for chronic migraine, four patients had a transient increase in liver enzyme concentrations during the treatment phase, which was considered non-treatment related. Liver enzymes returned to normal during the study period.
Hypertension was seen in a few patients (n = 5) in the galcanezumab trial, but it is unknown whether these patients had hypertension at enrollment. The other anti-CGRP mAbs showed no signs of hypertension (30).
Ongoing studies of anti-CGRP monoclonal antibodies in migraine
Many large-scale RCTs investigating anti-CGRP mAbs for the prevention of episodic and chronic migraine are in progress, as summarized in Table 2. In the following, we will review these ongoing trials in alphabetical order of the mAb.
Eptinezumab is currently undergoing a phase II trial in chronic migraine to evaluate efficacy in reducing mean monthly migraine days (clinicaltrial.gov identifier NCT02275117). The study concluded in November 2016 and results are pending. Phase III trials are also in progress to evaluate laboratory values, electrocardiogram, and adverse events in chronic migraine (NCT02985398), and to evaluate efficacy in reducing mean monthly migraine days in episodic (NCT02559895) and chronic (NCT02974153) migraine.
Erenumab is being tested for treatment of episodic migraine in a phase II trial (NCT02630459) in Japanese patients. For chronic migraine, an open-label extension of the concluded phase II trial (NCT02174861) will assess the long-term safety and tolerability of the drug in a 13-month treatment period. This open-label trial concluded in May 2017 and results are pending. Furthermore, erenumab is being tested for prevention of episodic migraine in a phase III trial (LIBERTY, NCT03096834) aimed at patients who have failed previous preventive treatment.
Currently there is one ongoing phase III study using fremanezumab, (NCT02638103) aimed at assessing the safety, efficacy, and tolerability of fremanezumab in episodic and chronic migraine patients, with the percentage of patients with adverse events as the primary endpoint.
For galcanezumab, two phase II studies are in progress to evaluate the decrease in mean monthly headache days in episodic migraine (NCT02163993 and NCT02959177). Also ongoing are two phase III open-label extension studies in episodic and chronic (NCT02959190 and NCT02614287) migraine to evaluate the percentage of patients who discontinue treatment.
Ongoing studies of anti-CGRP monoclonal antibodies in cluster headache
Experimental data suggest a central role of CGRP in the pathophysiology of cluster headache. It has been shown that plasma levels of CGRP are elevated in the external jugular vein ipsilateral to the pain side during a cluster headache attack, and decrease back to normal after sumatriptan or oxygen treatment (31,32). The prospect of treating cluster headache with anti-CGRP mAbs is being tested in six phase III trials, at the current time limited to fremanezumab and galcanezumab.
Two phase III studies are in progress for preventive treatment of episodic (NCT02945046) and chronic (NCT02964338) cluster headache with fremanezumab. The primary endpoint is reduction in weekly or monthly average cluster headache attacks. Both studies are projected to be completed in 2018. Patients who complete either of the two trials may be included in the phase III study ENFORCE, with both episodic and chronic cluster headache patients (NCT03107052), to evaluate adverse events, injection site reactions, and the use of concomitant medication throughout the study.
Two phase III studies are being conducted, investigating the preventive effect of galcanezumab for episodic (NCT02397473) and chronic (NCT02797951) cluster headache in reducing weekly attacks. Patients from either of these two trials may continue in the open-label extension trial (NCT02797951) to evaluate the number of patients with treatment emergent adverse events and the number of patients with suicidal ideation.
CGRP as a target for preventive therapy in migraine and cluster headache
Upon reviewing published clinical data of anti-CGRP mAbs in the treatment of migraine, we can summarize that these targeted monoclonal antibodies are effective in the prevention of migraine, with acceptable safety and tolerability profiles across the completed studies. Phase III RCTs are currently in progress to investigate whether this effect may also apply to cluster headache.
The mechanism of action of the anti-CGRP mAbs is of continued ample interest. Early preclinical data reported that anti-CGRP mAbs inhibited CGRP-induced dilation of rat cranial arteries in vitro and thus suggested a possible efficacy in migraine (33,34). To what extent this vascular effect contributes to migraine prevention is unknown. A recent study on the modulatory effect of fremanezumab on meningeal sensory pathways (35) has shown that fremanezumab inhibits the activation and sensitization of high threshold neurons in the intracranial dura mater by cortical spreading depression, but not their activation from the facial skin or cornea. The authors suggest that this finding could explain the selectivity of anti-CGRP mAbs for migraine headache but not for other non-headache pain conditions, as well as indicating a peripheral site of action (35). At present, the mechanism and site of action are not fully clarified and more studies are needed to elucidate these fundamental characteristics.
Other aspects of anti-CGRP mAb treatment in migraine to receive much attention are efficacy and safety at the prospect of long-term treatment, which would apply if these drugs were introduced as preventatives. While we currently do not have results from long-term trials of all four tested mAbs, we do have data from the interim analysis of the open-label study of erenumab in episodic migraine (23). At data cut-off (median exposure 34.1 weeks), results showed that further reductions from baseline in mean monthly migraine days were sustained for at least 52 weeks. Reports after yet longer treatment periods will be exciting to follow.
In terms of the safety profile of long-term anti-CGRP mAb treatment, it has been stressed that CGRP is abundantly present and active not only in the peripheral and central nervous systems, but also in the cardiovascular and gastrointestinal systems (22). Due to the potent vasodilator effect of this neuropeptide and its ubiquitous presence, it has been proposed that blocking the CGRP system in the long run is potentially risky (36) as CGRP acts as a vasodilatory safeguard in hypertension (37), focal cerebral ischemia (38), and myocardial infarction (39). In the phase II RCTs reviewed above, no serious cardiovascular adverse events were reported (11–15). While one should keep in mind that these trials were relatively short lasting, the open-label extension of erenumab also did not identify any new safety concerns after 64 weeks of treatment (23). Importantly, the anti-CGRP mAbs have not been tested in patients with known gastric, renal, or cardiac disease, and therefore it cannot be assessed how patients with ischemic vasculopathies or genetic susceptibility to cardiac disease might react to this treatment. Also, it should be noted that the mean age of participants across the trials was approximately 40 years, most likely rendering the participants too young to have developed vascular pathologies. Moving forward, experimental animal studies would elucidate the true effect of anti-CGRP mAbs in cardiovascular disease in particular, and potentially exclude adverse events related to CGRP blockade (36). Long-term RCTs in humans with anti-CGRP mAbs should also be conducted to further estimate the cardiovascular risk profile of preventive treatment with this emergent drug.
While concerns about hepatotoxicity caused a halt in the development of CGRP receptor antagonists as preventive treatment for migraine, these small molecules are far from having been vanquished from potential therapeutic breakthroughs. Currently, the agents ubrogepant, atogepant, and rimegepant are under trial. In a phase IIb study, ubrogepant showed efficacy as an acute drug in two-hour pain freedom compared to placebo (25.5% versus 8.9%) (40), and is currently being tested in a phase III study (NCT02867709). Based on these positive preliminary findings, it would be interesting to consider whether ubrogepant may also exert a preventive effect. Atogepant (NCT02848326) and rimegepant (NCT03237845) are currently under trial as preventive and acute treatment for migraine, respectively.
Conclusion
At present, available treatment options for migraine and cluster headache are unsatisfactory in terms of efficacy, tolerability, and patient adherence. Clinical data indicates that monoclonal anti-CGRP antibodies have a beneficial effect in the treatment of episodic and chronic migraine, introducing the first-ever targeted treatment option for this agonizing headache. Extensive data is underway with a multitude of phase II and III trials to further elaborate on this positive effect. The prospect of using anti-CGRP mAbs for preventive treatment of cluster headache is also currently being examined, and results are highly anticipated.
Introducing target-specific preventive treatment for migraine and cluster headache with proven efficacy and a favorable safety profile would not only revolutionize the management of headache patients in clinical practice, but more importantly it would dramatically improve the quality of life for the millions of affected individuals globally.
Clinical implications
Migraine and cluster headache are challenging to manage, with no targeted preventive medications available. Clinical data from phase II and III trials of four anti-CGRP monoclonal antibodies show a positive effect in the preventive treatment of episodic and chronic migraine. Phase III trials are also being conducted for preventive treatment of cluster headache with anti-CGRP monoclonal antibodies. Efficacy of anti-CGRP monoclonal antibodies predicts a promising future for patients suffering from migraine, and possibly also for the smaller but severely affected population with cluster headache.
Footnotes
Acknowledgements
The authors would like to thank Casper Emil Christensen, MD, for his valuable input on the literature search.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: S Khan has acted as invited speaker for Novartis. M Ashina is a consultant or scientific advisor for Allergan, Amgen, Alder, ATI, Eli Lilly, Novartis and Teva, primary investigator for Alder ALD403-CLIN-011 (Phase 3b), Amgen 20120178 (Phase 2), 20120295 (Phase 2), 20130255 (OLE), 20120297 (Phase 3), GM-11 gamma-Core-R trials, Novartis CAMG334a2301 (Phase 3b), and PAC1 20150308 (Phase 2a).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: M Ashina reports grants from Lundbeck Foundation (R155-2014-171) during the conduct of the study.
