Abstract
Background
The present study aimed to describe the prevalence and evolution of depressive symptoms in a cohort of migraine patients treated with anti-CGRP monoclonal antibodies.
Methods
This is an exploratory, prospective, unicentric, one-year longitudinal study. We included migraine patients who started treatment with anti-CGRP monoclonal antibodies. Baseline demographic data, medical history, concomitant medication and migraine characteristics were collected. The presence of depressive symptoms was evaluated using the Beck Depression Inventory-II quarterly and treatment response was categorized according to the reduction in monthly headache days. A generalized mixed-effect regression model was used to model depression score over a one-year treatment taking into account frequency response rates.
Results
We included 577 patients: 84.2% females; median (range) age 47.0 (39.0–53.0) years, 46.1% (266/577) of them presented depressive symptoms at baseline (16.1% mild, 13.3% moderate and 16.6% severe). After six-month treatment, 47.4% (126/266) reduced headache frequency ≥50% after one year and 63.5% (169/266) achieved a clinically significant improvement in depression symptoms. We observed a 30.8% (−50.0%, −3.2%) main reduction in depression score during the first quarter. The improvement in depression symptoms was independently associated with headache frequency response: non-responders, −25.0% (−43.9%, −1.1%); partial responders, −30.2% (−51.3%, −7.6%); and good responders, −33.3% (−54.6%, −7.5%).
Conclusions
Anti-CGRP monoclonal antibodies targeting CGRP are effective in reducing depressive symptoms in patients with migraine. The main change of depression score happens during the first three months of treatment. The reduction in depressive symptoms is independent of migraine frequency improvement.
Keywords
Introduction
Migraine and depression frequently coexist in the same patient and are common brain diseases and leading causes of chronic burden (1). The possible mechanisms behind this association are still unknown and the direction of the relationship between migraine and depression is controversial (2). Genome-wide association meta-analysis of depression and migraine have demonstrated a polygenic complex architecture (3,4) even suggesting that migraine may share some of its genetic heritability with psychiatric disorders (5). Beyond genetic background, both disorders are strongly influenced by environmental factors such as educational level, reproductive status, obesity or traumatic life events (6,7). It is well known that the presence of depression contributes to migraine chronification (8,9) and increases migraine-related disability reducing quality of life (10). Furthermore, migraine patients often report emotional changes, including difficulty with concentration and irritability in the premonitory phase, the headache phase, and even the postdromal phase of the migraine attack (11). The presence and severity of depressive symptoms (DS) becomes more frequent when migraine frequency increases (12,13), which might point to the existence of emotional symptoms also as a migraine associated symptom.
Calcitonin gene-related peptide (CGRP) is a potent neuropeptide known for its vasodilator properties, playing a major role in the pathophysiology of pain and other migraine symptoms (14). Recently, elevated salivary CGRP levels have been associated with the presence of depression-like behavior and increased migraine frequency in animal models (15–17). These data suggest that CGRP may also be involved in the pathophysiology of depressive symptoms and could, in part, explain the high comorbidity of migraine and depression, especially when these conditions become chronic.
Monoclonal antibodies targeting CGRP (antiCGRP-MAb) are effective and safe preventive treatments for migraine patients (18). Post-hoc studies with fremanezumab and galcanezumab (19,20) have also demonstrated efficacy in patients with migraine and DS reducing headache frequency and headache-related impact, but the direct effect of these treatments on depressive symptoms has not been explored.
Our hypothesis was that DS in migraine patients may also be CGRP-related so that preventive treatment with antiCGRP-MAbs could also reduce DS. Hence, the aim of this exploratory prospective study was to model longitudinally the presence of DS in a cohort of migraine patients treated with antiCGRP-MAbs over a one-year period. Our objectives were (i) to describe the prevalence of DS in a cohort of resistant migraine patients; (ii) to study its evolution pattern during one-year treatment; and (iii) to determine whether there was an association between DS change and headache frequency-related treatment response.
Methods
Data were collected from a prospective clinical cohort that has been previously described (21–24). We have updated our clinical series including all migraine patients who initiated preventive treatment with antiCGRP-MAb from February 2019 to May 2023. According to Spanish regulations, all treated patients fulfilled International Classification of Headache Disorders 3rd edition (ICHD-3) criteria for migraine (25) with a headache frequency ≥8 days/month who had previously failed at least three preventive medications, including onabotulinumtoxinA in chronic migraine patients (26,27). We did not exclude patients with concomitant stable dose of migraine preventive or antidepressant medications.
Erenumab 140 mg monthly, galcanezumab 240 mg first dose and 120 mg monthly or fremanezumab 675 mg quarterly or 225 mg monthly were administered following the manufacturer’s recommendations. Treatment response was assessed quarterly with the data collected from a daily electronic headache diary completed by the patient where they recorded the presence of headache, pain intensity using a 0–3 numerical scale (0, no pain; 1, mild; 2, moderate; 3, severe pain) and use of acute medication to treat headache. We included in the analysis all patients who had completed at least three months of treatment with antiCGRP-MAb at the moment of the analysis (Figure 1).
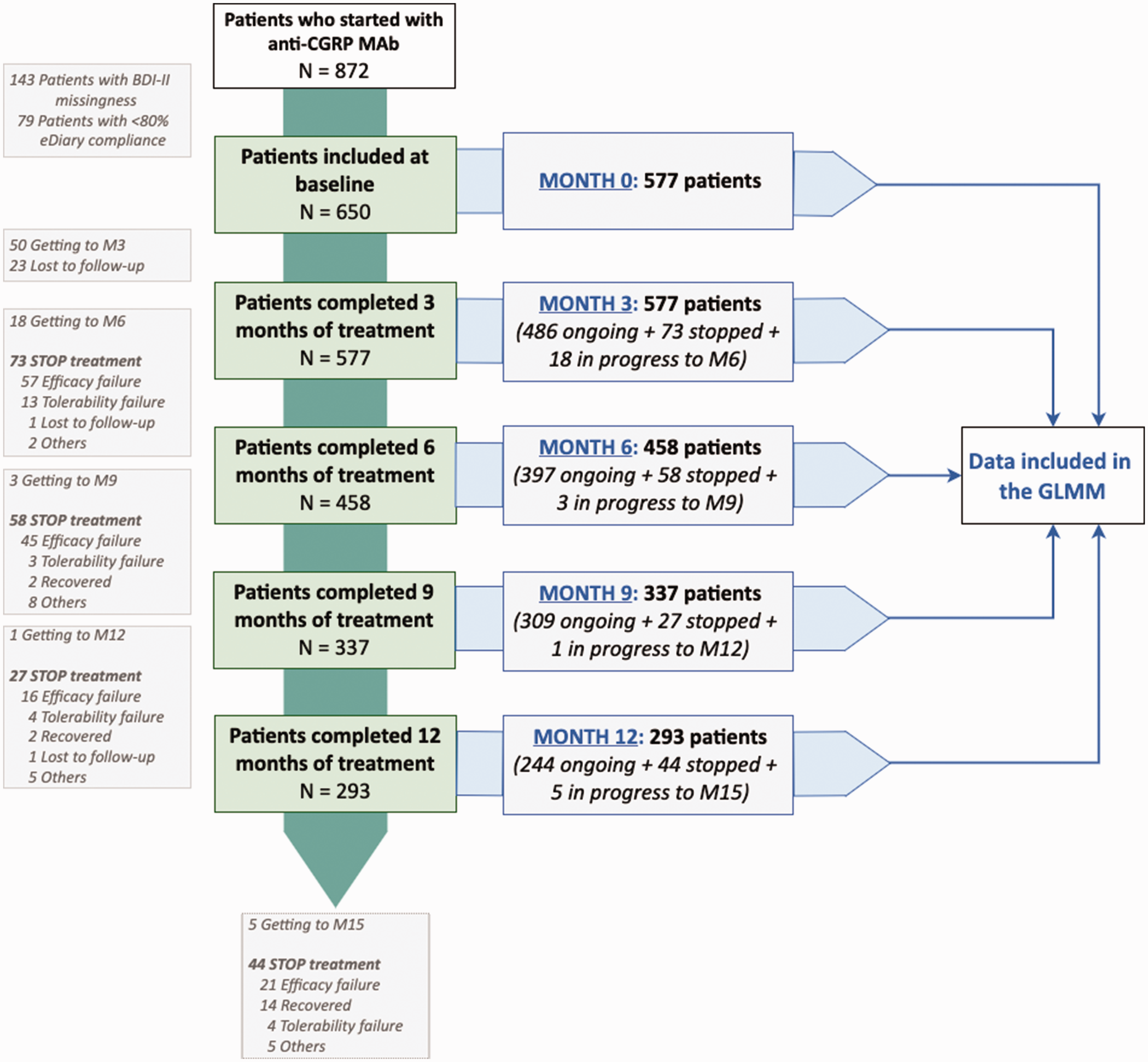
Flow-chart of patient selectionWe included patients who started anti-CGRP MAbs (erenumab, galcanezumab or fremanezumab) from February 2019 to June 2023 who had 12 months of treatment. GLMM, generalized linear mixed models.
Relevant to this study, baseline demographic data, medical history and migraine characteristics were collected. Presence of depressive symptoms were evaluated at baseline and every three months using the Beck Depression Inventory II (BDI-II), a widely used patient reported outcome questionnaire in the evaluation of interventions for depression (28,29): none DS (score 0–13), mild DS (score 14–19), moderate DS (score 20–28) and severe DS (score 29–63). According to previous published data (30), a mean reduction of a ≥17.5% in BDI-II score was considered as the minimal clinically important difference for considering a clinical improvement in DS (DS improvement). Anti CGRP-MAb response was categorized according to the mean reduction in monthly headache days (MHD) from baseline: non-responders (MHD reduction <30%), partial responders (MHD reduction 30–49%) and good responders (MHD reduction ≥50%).
The study was approved by the Vall d’Hebron Ethics Committee (PR(AG)53/2017/2022). All patients provided their written informed consent.
Statistical analysis
This exploratory analysis was a secondary pre-planned analysis of previously collected data (22). Data were prospectively collected to specifically evaluate treatment response with clinical as well as research purposes. We reported nominal (categorical) variables as frequencies (percentages) and continuous variables as the median and interquartile range (IQR). We checked normality assumption of continuous variables through visual methods (Q-Q plots).
To fulfil the objectives of the present study, two different multivariate generalized mixed-effect regression models (GLMMs) were estimated for a count outcome variable (BDI-II score). The aim was to investigate whether alterations in the dependent variable (BDI-II) over time were associated with DS at baseline, as well as the minimal clinically important difference in BDI-II outcomes among individuals, and also whether this change was associated with the MHD-treatment response.
GLMM are powerful and flexible statistical models that are particularly well-suited for analyzing longitudinal data, especially when there is no independence in data (different BDI-II measurements from the same patient over one year of treatment) and to account for both patient-specific variability and treatment-specific effects that need to be considered as random effects. A zero-inflated link function was employed because of the BDI-II error distribution (overdispersed count outcome variable with an excess of zeros). Both models were adjusted by fixed-effect covariates (concomitant antidepressant treatment and migraine diagnosis). For the time variable, both linear and quadratic terms were also included in the model after data visual inspection. Different combinations of two-way and three-way interactions were tested. Only random intercepts per patient were implemented as random effects. An autocorrelation structure of order 1 was considered for the variance–covariance matrix of the residuals of the model to take into account the time correlation for data clustered by patient.
Full models were fitted using R package glmmTMB, version 1.1.7. Variance inflation factors were computed for all the parameters in order to estimate how much the variance of an estimated regression coefficient is inflated as a result of correlated variables, preventing potential overfitting issues in the final models. Model diagnostic plots, including residual QQ plot, residuals vs. fitted quantile plot and overdispersion test, were performed using R package DHARMa, version 0.4.6. The analysis of deviance table of model’s main effect was performed and main effect plots were plotted using the R package sjPlot, version 2.8.14. Lastly, the estimated marginal means were computing using the R package emmeans, version 1.8.6, to perform post-hoc comparisons between study visits after fitting the GLMMs.
We did not conduct a statistical power calculation prior to the study because sample size was determined by available data of our cohort. All participants included had a ≥80% compliance rate of the headache diary. In our data, we detected a missingness rate <9% in some continuous variables at baseline. In those cases, we used random forest imputations to estimate these values according to their other variables using the R package MICE (Multivariate Imputation via Chained Equations), version 3.8.0 (31).
The p-values presented are for a two-tailed test and we considered p < 0.05 statistically significant. As a result of the exploratory nature of the present study, all p-values were adjusted applying the false discovery rate (Benjamini–Hochberg procedure). All analysis were performed using R, version 4.3.0 (R Foundation, Vienna, Austria) and the corresponding packages.
Results
Since 2019, 872 patients with migraine had started on antiCGRP-MAb therapies in our clinic. At the time of the analysis, 577 patients had received treatment for at least three months and a 50.8% (293/577) had completed 12 months of treatment. We excluded patients with a headache eDiary compliance <80% (9.1%, 79/872) and patients who did not complete the BDI-II questionnaire during treatment period (16.4%, 143/872). Some 35.0% (202/577) of patients discontinued treatment during the one-year follow up (lack of efficacy (68.8%, 139/202) and non-severe adverse events 11.9% (24/202). Patients who discontinued treatment were also included in the GLMM (Figure 1).
Therefore, we included at baseline in our analysis 577 patients: 84.2% females; median (range) age 47.0 (39.0–53.0) years. Chronic migraine criteria were fulfilled by 67.8% (391/577), the median (range) headache frequency at baseline was 18.0 (13.0–27.0) MHD and the median (range) frequency of monthly acute medication use was 11.0 (8.0–15.0) days/months. A 50.4% (291/577) received erenumab, 31.0% (179/577) received galcanezumab and 18.5% (107/577) received fremanezumab. At baseline, 47.1% (272/577) were using antidepressant drugs indicated for either migraine, anxiety or depression treatment. Regarding depressive symptoms, the median (range) BDI-II score at baseline was 12.0 (5.0–22.0) and 46.1% (266/577) of patients presented DS: 16.1% (93/577) mild DS, 13.3% (77/577) moderate DS and 16.6% (96/577) severe DS. The main demographic and baseline characteristics are shown in Table 1.
Demographics, comorbidities and migraine characteristics at baseline.

Continuous data are represented as the median (IQR) and categorical data as % (n). IQR, interquartile range; anti-CGRP MAb, anti-CGRP monoclonal antibodies; TCA, tricylic antidepressant; SSRI, selective serotonin reuptake inhibitor; SNRI, serotonin-norepinephrine reuptake inhibitor; SARI, serotonin antagonist and reuptake inhibitor; MIDAS, migraine disability assessment; HIT-6, headache impact test; BDI-II, Beck Depression Inventory – Second Edition.
Evolution on depressive symptoms during one-year treatment
In this analysis, we included 266 migraine patients with DS at baseline (BDI-II score ≥14). After 12 months of treatment, we found that a 63.5% (169/266) of patients presented a DS improvement (defined as ≥17.5% BDI-II reduction). A lower headache frequency at baseline was associated with DS improvement: 19.0 (14.0, 28.0) MHD vs. non-DS improvement 24.0 (16.0, 28.0) MHD; adjusted P = 0.026. However, no statistically significant differences were found for other baseline characteristics, including the use of concomitant antidepressant medications or the specific antiCGRP-MAb treatment used (see supplementary Table 1).
We observed a reduction of the BDI-II score during the use of antiCGRP-MAb treatment over a one-year period (Figure 2a). According to the overall effect of the fitted GLMM 1 (Table 2), we found statistically significant main effects on: (i) Severity of DS at baseline (Wald χ2 = 140.14; p < 0.001), showing higher values of BDI-II in moderate and severe DS than mild DS during all the longitudinal assessment; (ii) DS improvement (Wald χ2 = 71.31; p < 0.001), presenting lower BDI-II score, in those patients who showed ≥17.5% BDI-II reduction; and (iii) the evolution time (Wald χ2 = 6.48; p = 0.048), showing a main BDI-II score change (reduction) from baseline to month 12. From all the interaction terms, only the two-way interaction between evolution time and DS improvement (Wald χ2 = 71.43; p < 0.001) remained statistically significant, meaning that the change of the BDI-II scores during all the active treatment period depended on the improvement achieved in DS. Nevertheless, no statistically significant interaction was found between the presence of DS at baseline and DS improvement, suggesting that a clinically significant improvement in depression impairment is independent of its severity at the start of the treatment (Table 2).

(a) Effect plots and (b–d) post-hoc comparisons from the fitted generalized linear mixed models (GLMM) according to the presence of depressive symptoms at baseline (mild, moderate and severe) and the clinically BDI-II improvement between each quarterly time pointIn (a) is represented the change of BDI-II score over a 12-month period in migraine patients with anti-CGRP MAb treatment according to their presence of depressive symptoms at baseline. (b) to (d) represent the post-hoc comparison of BDI-II scores change (estimated marginal means derived from the GLMM) according to the presence of depressive symptoms at baseline between each quarterly time point. Statistically significances were found between baseline (M0) and follow-up visits (M3, M6, M9 and M12) but no differences were found between follow-up visits. The red dashed line represents the absence of depressive symptoms (BDI-II ≤14) threshold. BDI-II, Beck Depression Inventory – Second Edition; MCID, minimal clinically important difference; M0, baseline; M3, month 3, M6, month 6; M9, month 9; M12, month 12. p-values are adjusted by false discovery rate correction in the post-hoc analysis (between study visits). Adjusted p-value significance codes: ***0 to 0.001 ; **0.001 to 0.01; *0.01 to 0.05.
Analysis of deviance table (Type III Wald χ2 tests) of the main effects for the analysis of the BDI-II change after 12 months with anti-CGRP MAb treatment (GLMM 1) and the influence of patients’ treatment MHD-related response rate (GLMM 2).

df, degrees of freedom; BDI-II, Beck depression inventory-second edition; MCID, minimal clinically important difference; FUP, last follow-up visit; DS, depressive symptoms; ×, interaction between variables.
Bold indicates statistically significant variables.
Continuous independent variables were rescaled to a z-score metric (mean = 0, SD = 1) in the mixed model.
In a post-hoc analysis, we also saw this effect. All patients groups (mild, moderate or severe DS) statistically significantly reduced their BDI-II score with a global reduction of −30.8% (−50.0%, −3.2%). This reduction mainly occurred during the first three months of treatment and remained stable during all the follow-up (Figure 2b-d). After three months of treatment, the BDI-II reduction in the mild DS group was −25.0% (−46.7%, −3.0%), −30.2% (−50.0%, −7.8%) in patients with moderate DS and −35.2% (−52.1%, −6.9%) in patients with severe DS at baseline.
Relationship between depressive symptoms improvement and treatment response
From patients with DS at baseline, 47.4% (126/266) had a good response (≥50% MHD reduction), 19.9% (53/266) were partial responders (30-49% MHD reduction) and 32.7% (87/266) did not achieve 30% MHD reduction. We recomputed previous GLMM adding the MHD response groups to investigate the relationship between MHD improvement and DS improvement. If MHD and DS were strictly dependently associated, one would expect that BDI-II score would not change at the end of the treatment period in those patients without a MHD improvement (either non-responders or partial responders), as we saw in the GLMM 1 (patients without DS did not change their BDI-II score).
Nevertheless, the statistically significant factors associated with the BDI-II score change in the GLMM 2 (Table 2) were again (i) the severity of DS at baseline (Wald χ2 = 99.32; p < 0.001); (ii) the evolution time (Wald χ2 = 6.25; p = 0.046); and (iii) we also found a main effect of the MHD response groups (Wald χ2 = 8.37; p = 0.015). We observed that partial responders obtained a main greater reduction in BDI-II score than non-responders [GLMM est. (95% confidence interval): 0.765 (0.638–0.917); p = 0.004] but no differences were found between non-responders and good responders [0.912 (0.793–1.05); p = 0.196; p = 0.339) (see supplementary Table 2). When we inspected the interaction terms, we also found a statistically significant two-way interaction between the evolution time and MHD response groups (Wald χ2 = 15.21; p = 0.004), showing that BDI-II score reduction was different between non-responders, partial responders and good responders. However, in the post-hoc analysis, no statistically significant differences were found in the BDI-II reduction achieved at month 12 between these MHD groups: −25.0% (−43.9%, −1.1%) in non-responders, −30.2% (−51.3%, −7.6%) in partial responders and −33.3% (−54.6%, −7.5%) in good responders.
Hence, BDI-II scores were reduced for all MHD response groups but the trend of this reduction differed between them. We found a more pronounced reduction for individuals who have shown a partial or good response to the treatment (following a statistically significant quadratic trend) in contrast to the non-response group (adjusted by a linear trend) (Figure 3; see also supplementary Table 2).

Effect plots from the generalized linear mixed models (GLMM) according to the presence of depressive symptoms at baseline (mild, moderate and severe) and patient’s treatment response (non-responder, partial responder and good responder) between each quarterly time pointIn (a) is represented the two-way interaction of BDI-II change over time according to patient’s MHD-related RR and in (b) is represented the three-way interaction between the BDI-II change over time, the presence of DS at baseline (mild/moderate/severe) and patient’s MHD-related RR (non-responder, partial responder and good responder). BDI-II, Beck Depression Inventory – Second Edition; MHD, monthly headache days; RR, response rate; NR, non-responder; PR, partial responder; GR, good responder; DS, depressive symptoms; M0, baseline; M3, month 3, M6, month 6; M9, month 9; M12, month 12. The red dashed line represents the absence of depressive symptoms (BDI-II ≤14) threshold.
Finally, as most of the reduction in the BDI-II score occurred during the first three months of treatment, we adjusted the previous model (Table 2) for all cohort of patients who had both month 0 and month 3 data (n = 577). This adjustment was made to assess the robustness of the previously reported effects. GLMM was fitted considering only linearly evolution time (baseline to month 3) and we reproduced the same results of BDI-II reduction after three months of treatment for all response group.
Discussion
Patients with resistant migraine frequently have comorbid mood disorders that have sometimes been attributed to a poor migraine control (12,13). Since antiCGRP-MAb have become available for migraine preventive treatment, patients have reported improvement in mood-related symptoms together with migraine frequency or intensity of pain reduction, correlating the DS changes to an improvement in their migraine.
The present study explores the change in DS, measured by BDI-II score, in a longitudinal way, in a large cohort of resistant migraine patients treated with antiCGRP-MAbs. In this clinical cohort, 46.1% of migraine patients had DS. Our study demonstrates that, in patients with migraine and DS, preventive antiCGRP-MAb treatment is associated with a median reduction in BDI-II score in the range 25.0–35.2% for all groups (mild, moderate or severe DS at baseline), which occurs during the first three months of treatment and stabilizes during the 12-month follow-up. The most surprising result was the fact that the reduction in BDI-II score did not appear to be directly related to the reduction of migraine frequency because all response groups statistically significantly reduced DS. We found that the relationship between MHD improvement and BDI-II improvement was not equally linear when we compared groups with good, partial or no MHD response, showing a slower reduction in the BDI-II score over time in those patients with <30% headache frequency reduction. However, at the end of the follow-up, no differences were found in the BDI-II reduction rate between MHD response groups. Accordingly, the improvement of DS is independent to the improvement in headache frequency in patients with resistant migraine treated with antiCGRP-MAbs for one year, with a significant change in the first three months of treatment.
Previous studies and a recent meta-analysis have also shown that other migraine preventive treatments, specifically onabotulinumtoxinA, lead to a significant reduction of disease severity of both chronic migraine and depressive disorder (32–35). One potential interpretation for this finding would be to associate improvement in DS to the reduction in headache days. As patients experience fewer headaches, they generally feel better, resulting in improvements in both their depression and anxiety levels. However, and similar to our data, Blumenfeld et al. (32) found that patients with chronic migraine and baseline depressive symptoms treated with onabotulinumtoxinA showed reduced depression and anxiety symptoms even with limited improvements in headache days, also suggesting that the effect may be independent of the effect on headache day frequency. Accordingly, both this previous study and the present study indicate that an improvement DS is not directly linked to a reduction in headache frequency.
Despite headache frequency being used as the main efficacy endpoint in clinical trials, in the real world, an improvement in headache severity, analgesic use or emergency visits can have a key impact in headache-related disability and quality of life of our patients (36). One hypothesis could be that the improvement of DS seen in our patients with poor frequency response could be an indirect consequence of CGRP-MAbs improvement in headache day severity (presence of accompanying symptoms, intensity of pain, duration of attack) or acute treatment response, leading to a better control of the disease. Alternatively, DS in migraine patients could also be understood as a reversible (or not) accompanying symptom of the disease. Mood changes had been described as a premonitory symptom as well as during the attack phase of migraine (11,37). In a diary-based study, migraine patients reported more acute DS as depressive mood, loss of energy or interest or feelings of worthlessness during their migraine headache day than on all other days (38). Hence, treatment with antiCGRP-MAbs could directly act to reduce DS similarly to how they act for other migraine characteristics such as attack frequency or severity.
The relationship between DS and CGRP has been supported by animal models that demonstrated increased brain levels of CGRP in a rat model of depression (39) and enhancement of depressive-like behavior after the central administration of CGRP (40,41). In humans, an increase in CGRP was observed in cerebral spinal fluid, sweat and plasma of patients with major depression (42,43). In a recent study (16), we described higher salivary CGRP levels in patients with migraine and DS compared to patients without DS. These differences become more significant as migraine frequency increases, especially for CM patients. After erenumab treatment, salivary CGRP levels decrease both in patients with and without DS. According to these results, we can hypothesize a positive linear relationship between CGRP levels and the presence of DS in migraine patients.
Although the exact pathophysiology behind DS and CGRP in migraine and non-migraine patients needs further investigation, neuroinflammation has received increasing attention as a pathophysiological mechanism in depressive disorders, based on the existence of a strong association between depression and peripheral markers of inflammation in both blood and cerebral spinal fluid, as well as the antidepressant effects of numerous anti-inflammatory agents (44). However, we are still not able to clarify whether depression is a primary brain inflammatory disorder or consequence of other chronic inflammatory conditions involving increased circulating markers of inflammation that are able to cross the blood–brain barrier and sustain neuroinflammation. Translated to the migraine field, elevated CGRP levels in migraine patients could reinforce depression behavior. Accordingly, CGRP-pathway blockade by monoclonal antibodies could decrease CGRP levels and directly reduce DS in migraine patients, even in those that do not experience a great effect on migraine frequency.
The strict prospective data collection method and the use of an eDiary and electronic PROs, together with the precise statistical modelling, are one of the main strengths of the present study. Moreover, the use of mixed effect models allows us to include all the variability of patients who discontinue treatment before the end of the follow-up period (one year), and the use of different antiCGRP-MAb treatments allows us to capture patient’s inter- and intra-variability in the model. The main limitation to consider is the lack of a control group as well as the high percentage of use of antidepressants as migraine preventive or antidepressant indication. Considering the exploratory nature of the present study, change in concomitant medications (including antidepressants) might not been captured. Regarding DS evaluation, patients included were not evaluated by a psychiatrist and presence of DS was considered using the BDI-II scale that has demonstrated to be a reliable and sensitive tool to measure depressive disorders in other diseases (45,46). Although patients with depression diagnosis or antidepressant treatment were not excluded from the study, basal evaluation and the BDI-II scale were completed after CGRP prescription, such that treating physicians were blind with respect to BDI-II score and concomitant antidepressant treatment was not systematically offered. Further studies with better phenotyping of the affective domain can contribute to advancing our understanding about the relationship between migraine, depressive symptoms and their treatment.
Conclusions
AntiCGRP-MAbs are effective in reducing comorbid depressive symptoms (BDI-II score) in patients with resistant migraine who start preventive treatment. The main change of BDI-II score happens during the first three months of treatment and we estimated a median reduction of 30.8% in patients with the presence comorbid depressive symptoms at baseline. We did not find a clear relationship between migraine improvement and a reduction of comorbid depressive symptoms showing that, although in some patients there is no improvement in headache frequency, patients with migraine and depressive symptoms can benefit from antiCGRP-MAb treatment.
Clinical implications
Anti-CGRP monoclonal antibodies have demonstrated effectiveness in reducing comorbid depressive symptoms. Improvement in depressive symptoms typically occurs within the first three months of treatment. A clear correlation between the reduction in headache frequency and the decrease in comorbid depressive symptoms was not observed. Patients with migraine and depressive symptoms can benefit from anti-CGRP MAb treatment, even in cases where there is no apparent improvement in headache frequency.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024231222923 - Supplemental material for Improvement of migraine depressive symptoms is not related to headache frequency: exploring the impact of anti-CGRP therapies
Supplemental material, sj-pdf-1-cep-10.1177_03331024231222923 for Improvement of migraine depressive symptoms is not related to headache frequency: exploring the impact of anti-CGRP therapies by Marta Torres-Ferrús, Victor J. Gallardo, Alicia Alpuente, Edoardo Caronna, Eulalia Giné-Ciprés and Patricia Pozo-Rosich in Cephalalgia
Footnotes
Author contributions
All authors made substantial contributions to conception and study design and data collection. MTF and VJG wrote the first draft. AA, EC, EGC and PPR critically revised for important intellectual content. All authors approved the version submitted for publication and agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Data availability
Data generated during this study are included in the published article and its supplementary information are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
MT-F has received honoraria as a consultant or speaker for Allergan-Abbvie, Almirall, Chiesi, Eli Lilly, Novartis, Teva and Medlink. VJG has received honoraria as speaker for Eli Lilly and Novartis. AA has received honoraria as a consultant from Allergan-Abbvie, Novartis, Chiesi and Medlink. EC-G has received honoraria from Eli Lilly, Novartis and Teva. EC has received honoraria as consultant or speaker for Novartis, Chiesi and Medlink. In the last five years, PP-R has received honoraria as a consultant and speaker for: Abbvie, Amgen, Biohaven, Chiesi, Eli Lilly, Lundbeck, Medscape, Novartis and Teva. Her research group has received research grants from AbbVie, Novartis and Teva; and has received funding for clinical trials from Alder, Abbvie, Amgen, Electrocore, Eli Lilly, Lundbeck, Novartis and Teva. She is the Honorary Secretary of the International Headache Society. She is in the editorial board of Neurologia, and Revista de Neurologia, associate editor for Cephalalgia, Headache and The Journal of Headache and Pain. She is a member of the Clinical Trials Guidelines Committee and Scientific Committee of the International Headache Society. She has edited the Guidelines for the Diagnosis and Treatment of Headache of the Spanish Neurological Society. She is the founder of ![]() . PP-R does not own stocks from any pharmaceutical company.
. PP-R does not own stocks from any pharmaceutical company.
Ethical statement
The study was approved by the Vall d’Hebron Ethics Committee. All patients provided their written informed consent for the analysis of patients’ data, which was collected according to Spanish regulation on clinical trials.
Funding
No funding was received.
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
