Abstract
Background
Medication-overuse headache leads to high disability and decreased quality of life, and the best approach for withdrawal has been debated.
Aim
To compare change in disability and quality of life between two withdrawal programs.
Methods
We randomized medication-overuse headache patients to program A (two months without acute analgesics or migraine medications) or program B (two months with acute medications restricted to two days/week) in a prospective, outpatient study. At 6 and 12 months, we measured disability and headache burden by the Headache Under-Response to Treatment index (HURT). We estimated quality of life by EUROHIS-QOL 8-item at 2-, 6-, and 12-month follow-up. Primary endpoint was disability change at 12 months.
Results
We included 72 medication-overuse headache patients with primary migraine and/or tension-type headache. Fifty nine completed withdrawal and 54 completed 12-month follow-up. At 12-month follow-up, 41 patients completed HURT and 38 completed EUROHIS-QOL 8-item. Disability reduction was 25% in program-A and 7% in program-B (p = 0.027). Headache-burden reduction was 33% in program-A and 3% in program-B (p = 0.005). Quality of life was increased by 8% in both programs without significant difference between the programs (p = 0.30). At 2-month follow-up, quality of life increased significantly more in program-A than program-B (p = 0.006).
Conclusion
Both withdrawal programs reduced disability and increased quality of life. Withdrawal without acute medication was the most effective in reducing disability in medication-overuse headache patients.
Trial registration
Clinicaltrials.gov (NCT02903329).
Introduction
Medication-overuse headache (MOH) is a condition, where intake of symptomatic medication for headache may paradoxically lead to a worsening of headache (1). It affects 1–2% of the general population worldwide and 30–50% of patients in specialized headache centres. Medication overuse is associated with chronification of headache, especially migraine (2,3).
The recommended treatment of MOH is to cease the overuse (1,3), and withdrawal has demonstrated to revert at least half of patients to episodic headache (4,5). There is a lack of international consensus on withdrawal strategies. However, it is agreed that advice and patient education should be an essential first part of a treatment program (1,6,7). Most international treatment programs use restricted withdrawal, which means a certain decrease in frequency in monthly medication intake, often combined with medical preventives (5,6,8). Complete stop of acute medication combined with rescue medication, patient education, and close follow-up is recommended in current Danish guidelines, a procedure supported by several studies (4,9–11). Recently, Carlsen et al. demonstrated that withdrawal without any analgesics or acute migraine medication was more effective than restricted withdrawal with acute medications and/or analgesics up to two days/week regarding reduction of headache days and hereby number of patients reverting to episodic headache (4). Disability and quality of life, which also are very important end-points, were measured but not analyzed in that part of the study (4). It has been suggested that MOH has negative impact on disability and quality of life (12–15) which to some extent are improved when the overuse is treated (15–19).
The aim of the study was to examine whether change in disability and quality of life differed between a withdrawal program with complete stop of acute medications and a program that allowed restricted intake of analgesics. We hypothesized that the program with complete stop would reduce disability and headache burden more, but that the program with restricted intake had a better chance in improvement of quality of life, especially in the first two months, based on the more gradual approach with fewer withdrawal symptoms.
Methods
Study design
The present study was a predefined part of a larger study with the primary aim of comparing two withdrawal programs regarding percentage reduction in headache days/month (4). It was a randomized, prospective, longitudinal, open-label, controlled study. Patients with MOH were randomized to either treatment program A or B, both with a withdrawal period of 2 months with 12 months follow-up. In program A, patients underwent withdrawal without any analgesics or acute migraine medication. In program B, the intake was restricted, allowing patients to continue with their usual acute analgesics and/or migraine medication up to two days/week during the withdrawal. All patients were requested to continuously register headache and medication use in a standardized headache calendar. Both A and B were outpatient programs with the same patient education and follow-up visits, managed by a multidisciplinary team, and a more detailed description of the protocol has already been published (4). Patients were asked to complete the Headache Under Response to Treatment (HURT) and EUROHIS WHO Quality of Life 8-item (QOL-8) index at baseline, 6- and 12 months after withdrawal. In addition, the QOL-8 index was required at 2 months.
Study population
The study population was patients with MOH referred to the Danish Headache Center (DHC) included consecutively from October 2013 to March 2016. All participants signed informed consent. The inclusion criteria were confirmed MOH based on the ICHD-3 beta (which has same diagnostic criteria for MOH as the ICHD-3), previous diagnosis of a primary headache (chronic migraine (CM), episodic migraine (EM) and/or tension-type headache (TTH)) and being eligible for outpatient treatment (not needing in-patient care) (4). Patients had to register days with any headache for one month in a headache diary prior to the visit, and a headache specialist diagnosed MOH based on a detailed history of three months at minimum. In addition, patients had to complete the respective withdrawal strategy and fill out the questionnaires to be included in the present study.
Exclusion criteria were having a current substantial psychiatric disorder, such as schizophrenia or severe depression (the latter means a need for anti-depressive medication, treatment by a psychiatrist or treatment in a psychiatric department) or physical disorder (e.g. concomitant heart disease, severe co-morbid pain, or cancer), being previously diagnosed with another secondary headache other than MOH, being pregnant or breast feeding, or having inadequate language skills (requiring an interpreter). The study was approved by the Regional Ethical Committee in Denmark (H-1-2012-116).
Randomization
Patients were randomized to either program A or B in blocks of 10 by use of opaque, sealed envelopes as previously described (4).
Headache Under-Response to Treatment (HURT)
Headache Under-Response to Treatment Questionnaire (HURT) scoring system.

HURT-3: The sum of scores for the first three questions (min 0, max 9). HURT-8: The sum of scores for all 8 questions (min 0, max 24).
A Danish version translated according to an official translation protocol (made by Lifting The Burden) was accessible. The options for responses to questions 1 to 7 were either number of days or a “time aspect”. Question 8 was dichotomous, and non-response as well as lack of description of diagnosis, were considered a “no”. The options were later transformed to numerical data ranging from 0 to 3, which is in accordance with the official HURT Score guideline (Table 1) (24). The score for HURT-3 ranges from 0 to 9 and HURT-8 from 0 to 24. A high value indicates severe headache burden (HURT-3) or disability (HURT-8).
EUROHIS WHO Quality of Life 8-item (QOL-8)
QOL-8 is a systematic and validated shortened version of the 26-item The World Health Organization Quality of Life (WHOQOL)-BREF (25–27). The questionnaire comprises eight items: Two general items; overall quality of life rating and general health satisfaction, and six items that relate to the last 4 weeks. These are: Ability to perform daily life activities, patient's self-esteem, relationships, satisfaction with housing level, daily energy, and finances to handle daily necessities. Each question has 5 options of responses, for instance from “not at all” to “completely”. According to EUROHIS, the response of an item is transformed to a scale ranging from 1–5 points, where each subscale is scored positively. The eight items are then summed to a QOL-8 score (ranging from 8–40), with higher scores indicating increased quality of life (26,28).
Endpoints
The primary outcome of the present study was comparison of changes in HURT-8 score in the two programs from baseline to 12 months follow-up. Secondary outcomes regarding HURT were changes in HURT-8 from baseline to 6 months and changes in HURT-3 from baseline to 6 and 12 months. Last, we investigated change in QOL-8 from baseline to 2, 6, and 12 months' follow-up in total and for comparison between program A and B.
Statistics
Statistical calculations were performed using Rstudio for Mac, and the level of significance was set at p < 0.05. HURT and QOL-8 were transformed to numerical data according to the guidelines of each questionnaire. Continuous variables were presented as mean with SEM or 95% CI. Student's t-test was used for paired analysis on continuous variables following a normal distribution. When comparing program A and B, regarding continuous data, we used analysis of covariance (ANCOVA), which can adjust for baseline values. Categorical variables were presented as percentage and number and analysed by Fischer's exact test (for fewer than five samples).
Results
Study population
Seventy-two patients out of 553 screened patients were included (Figure 1). Thirty-five patients were randomized to program A and 37 to program B. In total, 59 patients (82%) completed 2 months withdrawal with a similar number of drop-outs in both programs at 2 months (Figure 1). Fifty-four patients (75%) attended the 12 months follow-up. Forty-one (69%) had followed the withdrawal approach and completed HURT at 12 months, and 38 patients (64%) completed QOL-8 at 12-month follow-up (Figure 1).
Flowchart for the study population.
1
Need for interpretation assistance (n = 17); severe psychiatric or physical co-morbidities (n = 55); secondary headache from other than MOH (n = 9); unclear headache diagnosis (n = 9). 2Protocol A: One patient had surgery, two continued analgesics once or twice a week. Protocol B: One patient had surgery, and two needed daily analgesics without any specific reason. 3Patients did not show up for the 2 months' control visit and thereby were lost to follow-up. 4Protocol A: All patients with successful detoxification did not take any migraine medication or analgesics for two months, except two patients who respectively had 1 and 4 days with acute medication. 5Patients in brackets are those who answered QOL question 1 (Q1) at the respective month. 6Protocol B: One patient dropped out because of pregnancy. 7Protocol A: One patient dropped out because of pregnancy and one was lost to follow-up. Protocol B: One patient emigrated from Denmark, and two were lost to follow-up.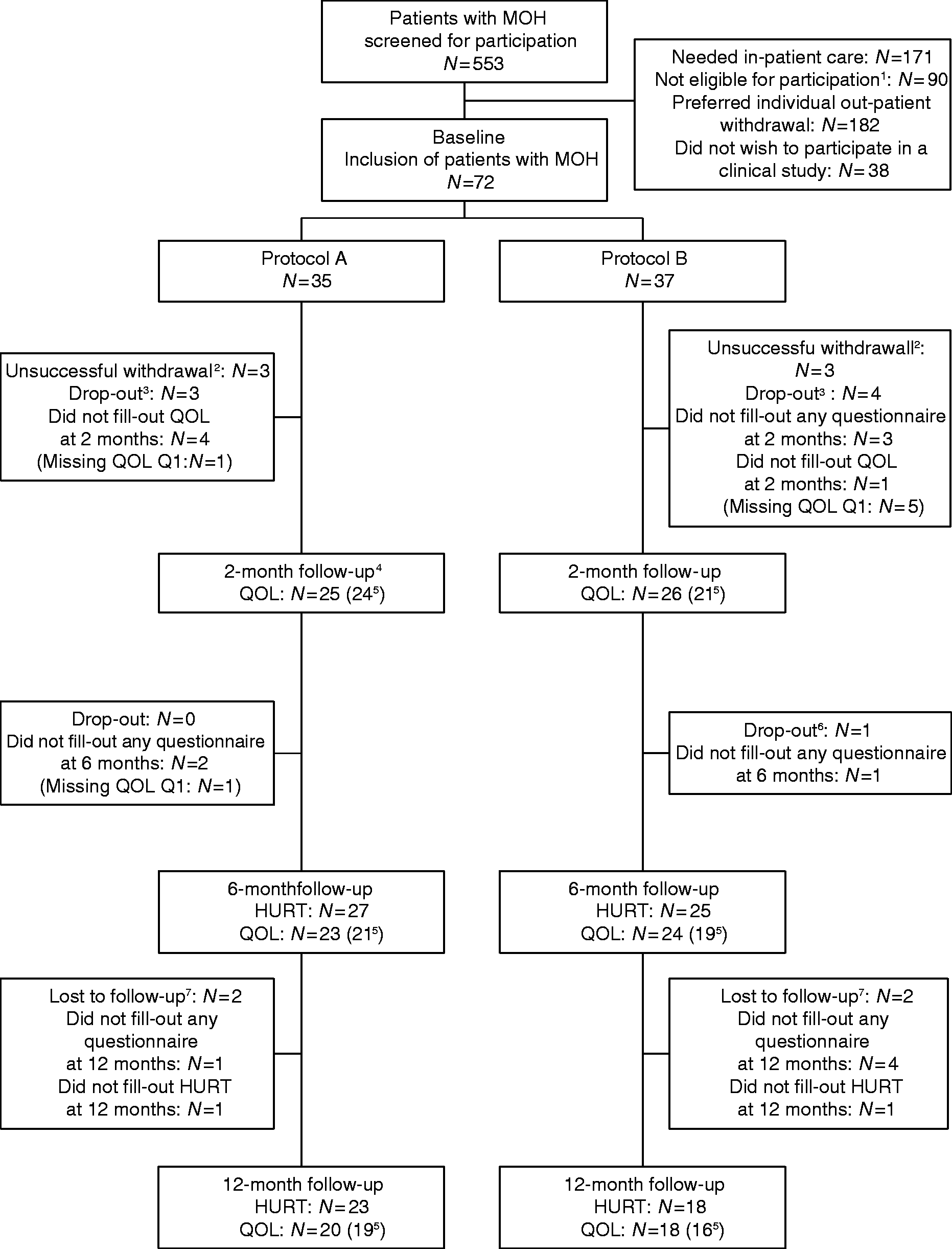
Baseline characteristics.

Age, headache days/month, duration of medication overuse, and days with acute medications/month are presented as mean (SEM). Gender, comorbidities, work status and social status are presented as N (%). A: Program A complete withdrawal for 2 months. B: Program B restricted intake of acute medication for 2 months. 1Number corresponds to the number of patients answering either HURT or QOL until 12 months (column 2 and 3 respectively) and 6 months for HURT (column 1). 2Severe physical and/ or psychological comorbidities were exclusion criteria.
Baseline characteristics for patients not completing the study and/or questionnaires and for all included patients are presented in Supplemental Table 1. The number of non-completers was, for HURT, 12 in program A and 19 in program B and, for QOL-8, 16 in program A and 21 in program B. Non-completers were comparable with completers in headache frequency and number of days with acute medication intake.
Disability and headache burden
Outcomes 12 months follow-up
The reduction in HURT-8 was significantly higher in program A (from 16.7 to 12.6 corresponding to 25% reduction) than program B (from 17.1 to 15.9 corresponding to 7 % reduction) (p = 0.027) (Figure 2). Compared to baseline, HURT-8 was also reduced significantly more in program A (p < 0.001), but not in program B (p = 0.25) (Figure 2, Table 3).
Scores for HURT-8 at 12 months presented as mean and SEM. n = 23 in program A with complete withdrawal vs. n = 18 in program B with restricted intake. Paired comparisons from baseline to 12 months' follow-up were made for program A and program B, as well as unpaired comparison between program A and B after 12 months. HURT scores and QOL score. All data are presented as mean (SEM). 1For HURT: n = 27 (program A), n = 25 (program B) at 6 months and n = 23 (program A), n = 18 (program B) at 12 months, which corresponds to the number of patients answering HURT at the respective month. 2For QOL-8: n = 21 (program A), n = 19 (program B) at 6 months and n = 19 (program A), n = 16 (program B) at 12 months, which corresponds to the number of patients answering QOL-8 at the respective month. Figures in bold: p<0.05 is considered significant and indicates significant difference from baseline to 6 or 12 months for the respective program.

In program A, HURT-3, reflecting headache burden, was reduced by 33% from baseline to 12 months and there was a trend towards reduction by 3% in program B (Table 3). The reduction in HURT-3 was significantly higher in program A than B (p = 0.005).
Outcomes 6 months follow-up
In program A, HURT-8 was reduced from 16.9 to 13.4 (p < 0.001, (95% CI 2–5)), corresponding to 21%, which was significantly more than the 8% reduction in program B (p = 0.019) (Table 3).
HURT-3 was reduced by 23% and 10% for program A and B respectively (Table 3). When comparing HURT-3 in program A and B, there was no significant difference (p = 0.12).
Quality of life
Outcomes 12 months follow-up
In program A, QOL-8 increased 2 points from 28 at baseline to 30 (p = 0.037, (95% CI −4.4 – −0.1)) and in program B, QOL-8 increased 2 points from 24 to 26 (p = 0.096), corresponding to an increase of 8% in both program A and B, with no difference between the programs (p = 0.30) (Table 3) (Figure 3).
Development in QOL-8. Presented in mean score difference from baseline. Only includes patients completing all QOL-8-questionnaires (i.e. baseline, 2 months, 6 months and 12 months).
Outcomes 6 months follow-up
In total, QOL-8 increased by 2% from baseline to 6 months. This was based on an increase in program A by 7% and a reduction by 4% in program B in favour of program A at 6 months (p = 0.023) (Table 3).
Outcomes 2 months follow-up
In total, there was no change from baseline to 2 months (1%). In program A, QOL-8 increased 2 points from 27 at baseline to 29 (p = 0.018, (95% CI −3.6 – −0.4)) corresponding to an increase of 7%, while QOL-8 was reduced by 2 points from 25 to 23 (−7%) in program B (p = 0.36) (Figure 3). Between the groups, there was significant difference in favour of program A (p = 0.006). Supplemental Table 2 and 3 illustrate the results of HURT and QOL-8 without use of ANCOVA, but with Student's t-test instead.
Discussion
Main results
The main results of the present study were that disability was reduced significantly more in the program that used a complete stop of acute medications compared with the program that allowed a restricted intake of medications. The trend in the program with restricted intake was also a reduction of disability, though this was not significant (Figure 2). In addition, patients rated their quality of life equally higher at 12 months in the two programs. However, patients with a complete stop had a much faster turn-over to improved quality of life. They reported a higher score already after two months, leading to rejection of our hypothesis in favour of restricted withdrawal.
Complete withdrawal versus restricted intake
In line with the current results in this study, complete withdrawal has previously been shown to result in a significantly greater reduction of headache days/month at 6 months (46% vs. 22%) with a similar trend at 12 months (45% vs. 31%) compared to restricted intake (4). More patients also reverted to episodic headache after complete withdrawal compared with restricted intake (74 % vs. 46 % at 12 months) and migraine days/month were significantly reduced (7.8 vs. 4.6 at 12 months) (4). Regarding completion in the study by Carlsen et al., 77% in program A and 70% in program B finished the protocol at 12 months (4).
Our findings of a greater reduction in disability when initiating complete withdrawal relates to the findings of reduction in headache days/months in this study (4). This is to be expected, but it is interesting that the results only showed significance at 6 months (4) but were significant at both 6 and 12 months in our study. It can be suggested that the reduction in disability is not dependent on improvement of headache frequency alone. Reduced disability can also be due to how a headache attack is managed; for example, if the patients feel they have control over the headache, have proper treatment and can perform daily activities.
The question is why complete withdrawal seems to be the most effective treatment program in terms of both reducing headache days and disability. Concerning the reduction in disability, we suggest that it partly could be explained by an extra benefit of complete withdrawal in the form of a reinforced personal feeling of control when the medication-overuse is ceased.
And why does quality of life increase faster at a complete stop compared to restricted use? We suggest that it is a combination of faster improvement in headache frequency, the previously mentioned feeling of control, and that patients initiating a complete stop subsequently may feel better about themselves (4). At a complete stop, patients may be positively surprised that they can manage their headache without any acute medication, which may contribute to development of belief in self-efficacy and thereby higher quality of life. Self-efficacy is suggested to affect the success of MOH therapeutic strategies and high expectations of functionality in MOH patients have been addressed (12,29). On the contrary, patients with restricted intake may have had higher expectations compared to the actual profit of their treatment, reflected in decreased quality of life.
It has been addressed that MOH can cause a great individual loss for the patient, influencing education, work and private economy, but also in terms of psychological and social aspects (2). It can further be questioned how great an impact all these aspects of life have on coping opportunities and on how well the patient can adjust their life to the withdrawal program. In future studies, it could be interesting to investigate whether a patient's way of running their life can predict whether complete withdrawal or restricted intake would have the best long-term results for a specific patient.
In the study by Westergaard et al., patients with MOH at the initial visit had a mean HURT-8 score of 17.0 (24), consistent with our findings, and a HURT-8 score at the final visit of 12.5, which is almost identical to program A, while the reduction was less convincing in program B.
There are to our knowledge few studies assessing QOL in patients with MOH during a treatment intervention, and those that do exist use different questionnaires. A Russian cross-sectional study found a QOL-8 score of 24.7 (30). This is in line with program B and slightly lower than program A (Table 3), possibly explained by some national differences in aspects related to quality of life.
Strengths and drawbacks
A major strength of this randomized controlled study is the prospective long-term evaluation of the treatment programs at 12 months. The results add important new knowledge on outcome of medication withdrawal not only for headache parameters but also for disability and quality of life.
The screening of 553 patients indicates that patients with the diagnosis of MOH with migraine and/or TTH are thoroughly selected and confirmed, because patients with other secondary headaches are deselected. Yet, the number of screened patients also address the risk of selection bias as some patients did not wish to participate. In addition, some patients chose an in-patient program outside this study, though it was assessed by clinicians that the grade of severity of their condition was consistent with an outpatient approach.
It is important to notice that as it only included patients suitable for outpatient treatment based on CM, EM and/or TTH, the present study did not deal with most complicated patients needing in-patient treatment; for example, with severe comorbidities including need of psychiatric expertise, use of anti-depressant medication, overuse of opioids failing previous outpatient treatments or based on a secondary headache other than MOH. A similar study of the severely affected MOH patients on an in-patient basis could be of interest in the future.
Another limitation is the number of patients not completing all questionnaires, causing the total completion rate to decrease from approximately 70–77% (caused by dropout alone) to 64–69% when combining dropout and non-completed questionnaires. However, completers and non-completers had similar baseline characteristics with only minor unimportant differences.
Our results can support clinicians in advising MOH patients with their selection of withdrawal approach. The potential advantage of a complete stop is emphasized, and in addition patients can be reassured that this stop is not unbearable and does not lower the quality of life compared to restricted withdrawal. The outcome measure is not only based on headache frequency, and thereby the assessment of efficiency of the two programs becomes more patient-oriented and clinically relevant.
Conclusion
At 12 months, disability was improved significantly in the program with complete withdrawal compared to the program with restricted withdrawal. Both withdrawal programs improved quality of life equally, yet complete withdrawal let to a faster increase.
This study establishes the importance of withdrawal and suggests an advantage and the obtainable gain in the patient's life when the withdrawal is two months of complete stop of analgesics and acute migraine medication, together with support and close follow-up.
Supplemental Material
Supplemental material for Complete withdrawal is the most effective approach to reduce disability in patients with medication-overuse headache: A randomized controlled open-label trial
Supplemental Material1 for Complete withdrawal is the most effective approach to reduce disability in patients with medication-overuse headache: A randomized controlled open-label trial by Mia Nielsen, Louise Ninett Carlsen, Signe Bruun Munksgaard, Ida Maria Storm Engelstoft, Rigmor Højland Jensen and Lars Bendtsen in Cephalalgia
Supplemental Material
Supplemental material for Complete withdrawal is the most effective approach to reduce disability in patients with medication-overuse headache: A randomized controlled open-label trial
Supplemental Material2 for Complete withdrawal is the most effective approach to reduce disability in patients with medication-overuse headache: A randomized controlled open-label trial by Mia Nielsen, Louise Ninett Carlsen, Signe Bruun Munksgaard, Ida Maria Storm Engelstoft, Rigmor Højland Jensen and Lars Bendtsen in Cephalalgia
Supplemental Material
Supplemental material for Complete withdrawal is the most effective approach to reduce disability in patients with medication-overuse headache: A randomized controlled open-label trial
Supplemental Material3 for Complete withdrawal is the most effective approach to reduce disability in patients with medication-overuse headache: A randomized controlled open-label trial by Mia Nielsen, Louise Ninett Carlsen, Signe Bruun Munksgaard, Ida Maria Storm Engelstoft, Rigmor Højland Jensen and Lars Bendtsen in Cephalalgia
Footnotes
Clinical implications
Complete stop of acute medication is the most effective method to reduce headache burden and disability.
Withdrawal improves quality of life in MOH patients and a complete stop of medication shortens the time to improvement.
Acknowledgements
The authors thank the headache nurses Malene Kjærgaard Danø, Annette Vangaa Rasmussen, Hjørdis Rasmussen and Annette Fjeldborg Jonasson for planning and management of the patient education program and collecting of data. Additionally, we thank project nurse Mette Frank Fisker for data management and data typing.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SB Munksgaard has undertaken lectures for Berlin-Chemie and UCB. RH Jensen has participated in clinical trials for ATI, Electrocore, Eli-Lilly and Linde Gas Ltd., and is a director of LTB, trustee of IHS and past president of EHF, and has the following additional disclosures: Lectures for Pfizer, Berlin-Chemie, Allergan, Merck, and ATI. L Bendtsen has the following disclosures: Advisory board member for Allergan, Novartis and Biogen. M Nielsen, LN Carlsen and IMS Engelstoft have nothing to declare.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: M Nielsen has received a 6-month scholarship from the Danish Medical Society Copenhagen (DMSK). LN Carlsen, SB Munksgaard, and R Jensen have received funding from the TrygFoundation.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
