Abstract
Background
The diagnosis of transient ischemic attacks is fraught with problems. The inter-observer agreement has repeatedly been shown to be low even in a neurological setting, and the specificity of the diagnosis is modest to low, reflected in a poor separation of transient ischemic attacks and mimics, particularly migraine with aura with its varied symptomatology. In other disease areas, explicit diagnostic criteria have improved sensitivity and specificity of diagnoses. We therefore present novel explicit diagnostic criteria for transient ischemic attacks tested for sensitivity and for specificity against migraine with aura.
Methods
The proposed criteria were developed using the format of the international headache classification. We drew upon the existing literature about clinical characteristics and diagnosis of migraine with aura and transient ischemic attacks. We tested the criteria for sensitivity in a prospectively-collected material of 120 patients with transient ischemic attacks diagnosed before we developed the criteria using extensive semi-structured interview forms in the acute phase after admission. Eligible patients had focal brain or retinal ischemia with resolution of symptoms within 24 hours without presence of new infarction on magnetic resonance imaging with diffusion weighted imaging (n = 112) or computed tomography (n = 8). These criteria were also tested for specificity against a Danish (n = 1390) and a Russian (n = 152) material of patients with migraine with aura diagnosed according to the International Classification of Headache Disorders edition 3 (beta).
Results
The sensitivity of the proposed criteria was 99% in patients with transient ischemic attacks. The specificity was 95% in the Danish material of patients with migraine with aura and 96% in the Russian material.
Conclusions
Proposed explicit diagnostic criteria for transient ischemic attacks showed both high specificity and sensitivity. They are likely to improve the emergency room diagnosis of transient ischemic attacks. Further testing in unselected materials referred to transient ischemic attacks clinics was beyond the scope of the present study but is recommended for future study.
Introduction
The diagnosis of transient ischemic attacks (TIAs) is fraught with problems. The Inter-observer agreement has repeatedly been shown to be low even in a neurological setting (1,2,3) and the specificity of the diagnosis is modest to low (4,5,6,7), reflected in a poor separation of TIA from mimics. As expected, the separation is worst for general physicians and emergency room physicians but, even after consultation by a neurology resident, it is poor. The main reason for these difficulties is probably that there are no explicit diagnostic criteria for TIA to guide the diagnostic process, only a so-called definition (or in fact several definitions) (8,9). For many other neurological diseases such as headache diseases and for all psychiatric diseases, definitions have now been replaced by explicit (operational) diagnostic criteria (10). This has resulted in more reliable diagnoses and has improved comparisons of studies in different countries. To fulfill such diagnostic criteria, very specific clinical information that is part of the criteria must be obtained. In contrast, the definition of TIA contains almost no guidance about necessary clinical questioning, except that symptoms must be of acute onset and fully reversible. It is stated that they must be caused by cerebral ischemia but also that there must be no evidence of ischemic lesion on neuroimaging (preferably diffusion weighted MR imaging but varying between the different definitions). That makes it almost impossible to prove the ischemic nature of the problem.
In an attempt to improve the situation, we have developed explicit diagnostic criteria for TIA and have tested them for sensitivity in a prospectively collected material of TIA patients using extensive semi-structured interview forms in the acute phase after admission. We also test the criteria for specificity against two large materials of patients with migraine with aura which is the commonest TIA mimic (4,11).
Materials and methods
Development of explicit diagnostic criteria for TIA
We developed the criteria based on the explicit diagnostic criteria for migraine with aura in the International Classification of Headache Disorders 3rd edition beta version (ICHD-3beta) appendix. These will replace the criteria in the main body of ICHD-3beta in the forthcoming final version of the ICHD-3, which is expected to be published in early 2018. To some extent, our proposed criteria for TIA are the opposite of the migraine with aura criteria but there are also elements derived from the well-known clinical features of TIA (3,4,5,7). We applied the criteria to a number of theoretical cases of TIA and cases of migraine with aura that we thought might pose diagnostic problems, and made some minor modifications of the criteria. Thus we arrived at the intellectually most satisfactory set of diagnostic criteria, adopting the format used in the classification of headache disorders (10) and its more than 20 translations. The criteria are presented in Figure 1. These criteria were tested against a material of TIA patients for sensitivity and against a Danish and a Russian material of patients with migraine with aura for specificity in the present study.
Proposed tissue based diagnostic criteria for transient ischemic attacks*.
Study populations
Patients with TIA were eligible for enrollment if they had had a transient ischemic attack (TIA) before admission at the stroke unit of city hospital “New Hospital” in Yekaterinburg, Russia. Eligible patients had focal brain or retinal ischemia with resolution of symptoms without presence of new infarction on MRI with DWI or CT. All patients were evaluated within one day of admission, usually within a few hours by a neurologist. Sixty two patients had TIA < 6 hours before admission to the hospital, 50 patients had TIA in an interval from 6–12 hours, six had TIA in an interval from 13–24 hours, and two had TIA in an interval from 24–36 hours before admission.
The period of recruitment of patients was from 2014–2016, that is, before we developed the explicit diagnostic criteria. We consecutively included patients with TIA who agreed to be interviewed. The reasons for exclusion were: Previous stroke, subarachnoid haemorrhage, intracranial aneurysm, intracranial haemorrhage, brain tumor, any operation on the brain, multiple sclerosis, epilepsy, encephalitis, meningitis, dementia or memory problems, transient global amnesia, speech problems and other serious neurological or somatic disorder. Furthermore, patients with CT or DWI evidence of relevant infarction were excluded. A total of 131 patients were examined, 11 patients were excluded and 120 were included in the study. Among them, 112 patients had MRI with DWI and eight patients had CT. These examinations were done at the time of admission to the hospital. For the specificity analysis, we used a large, previously-published cohort of patients diagnosed with migraine with aura according to a validated physician conducted semi-structured interview (12). Furthermore, we also calculated specificity from a prospective Russian patient material with migraine with aura diagnosed in a specialist practice using the same semi-structured interview conducted by an experienced neurologist with a special interest in migraine.
Evaluation
One neurologist (NMG) collected patient data prospectively, using a standardized case-report form during face-to-face interviews at the time of evaluation of the TIA after MRI or CT. Sociodemographic characteristics of patients, medical history, use of drugs, clinical symptoms during TIA and risk factors were recorded. Past history of headache and headache around the time of TIA were recorded using extensive semi-structured interview forms that contained all necessary information to diagnose both previous headaches and headaches at the time of TIA. Furthermore, the information contained detailed recording of characteristics necessary to diagnose migraine aura. Results of imaging and laboratory tests were also recorded in the case-report form as well as previous and current treatment. All data about TIA were collected before the diagnostic criteria were developed and tested.
Definitions and diagnostic criteria
TIA was defined as new neurologic deterioration lasting less than 24 hours with no infarction in the relevant area on neuroimaging (9). Two neurologists (NMG and ERL) performed telephone interviews in difficult cases when we suspected that patients, originally diagnosed as TIA, could have migraine with aura. This interview was done 1–3 years after the first interview and focused on the possibility of repeated migraine with aura attacks. Patients were asked about recurrent similar episodes, their characteristics, and some other information about risk factors and headache.
Ethical considerations
The Medical Ethics Committee of the Urals State Medical University approved this study. All respondents were informed of the purpose of the survey. Written informed consent was obtained from all participants.
Statistical analysis
We tested the proposed explicit diagnostic criteria for TIA for sensitivity in our 120 TIA patients and for specificity on 1390 Danish migraine with aura patients and 152 Russian migraine with aura patients. The number of patients fulfilling each sub-criterion was recorded. The sensitivity was calculated as the number with true positive diagnosis (fulfilling our explicit diagnostic criteria for TIA) divided by 120. Specificity of the TIA criteria was calculated as the number of MA patients minus the number fulfilling TIA criteria divided by the number of MA patients in both the Danish and the Russian material.
Results
Distribution of patients with TIA by age and sex.

Duration and type of symptoms in TIA patients.

In two patients (both females) symptoms spread gradually. A 19-year-old woman had gradual development of hypoesthesia of the left arm during 15 minutes, vertigo and confusion during 2 hours. A 23-year-old woman had gradual development of hypoesthesia of the right arm, vertigo and amnesic aphasia during 3 hours. Both patients had no headache and experienced these episodes for the first time in their life. MRI did not reveal any abnormalities. At follow up after 2 years, there had been no further attacks and no kind of migraine attacks.
In three other patients, symptoms occurred in succession. A 22-year-old woman had right-sided hemianopsia during 30 minutes; after that, right-sided hemihypoesthesia during 60 minutes, and at the end paresis of her right arm and motor aphasia during 6 hours. A 23-year-old female had gradual development of hypoesthesia of the right arm during 2 hours, then amnestic aphasia during 2 hours, and at the end vertigo during 2 hours. A 37-year-old man had gradual development of left sided paresthesias, which started from his leg and then to the arm and right side of the face. These symptoms were present from 11 am to 2 pm. After one hour, he felt paresis of his left arm and leg lasting 3 hours. MRI did not reveal any abnormalities in any of these patients. All these patients had no further attacks and no kind of migraine attacks during two years of follow-up.
Sensitivity of the proposed diagnostic criteria and specificity versus migraine with aura
Testing of TIA diagnostic criteria for sensitivity in patients with TIA.

Testing of TIA diagnostic criteria for specificity in patients with migraine with aura in the Danish material.
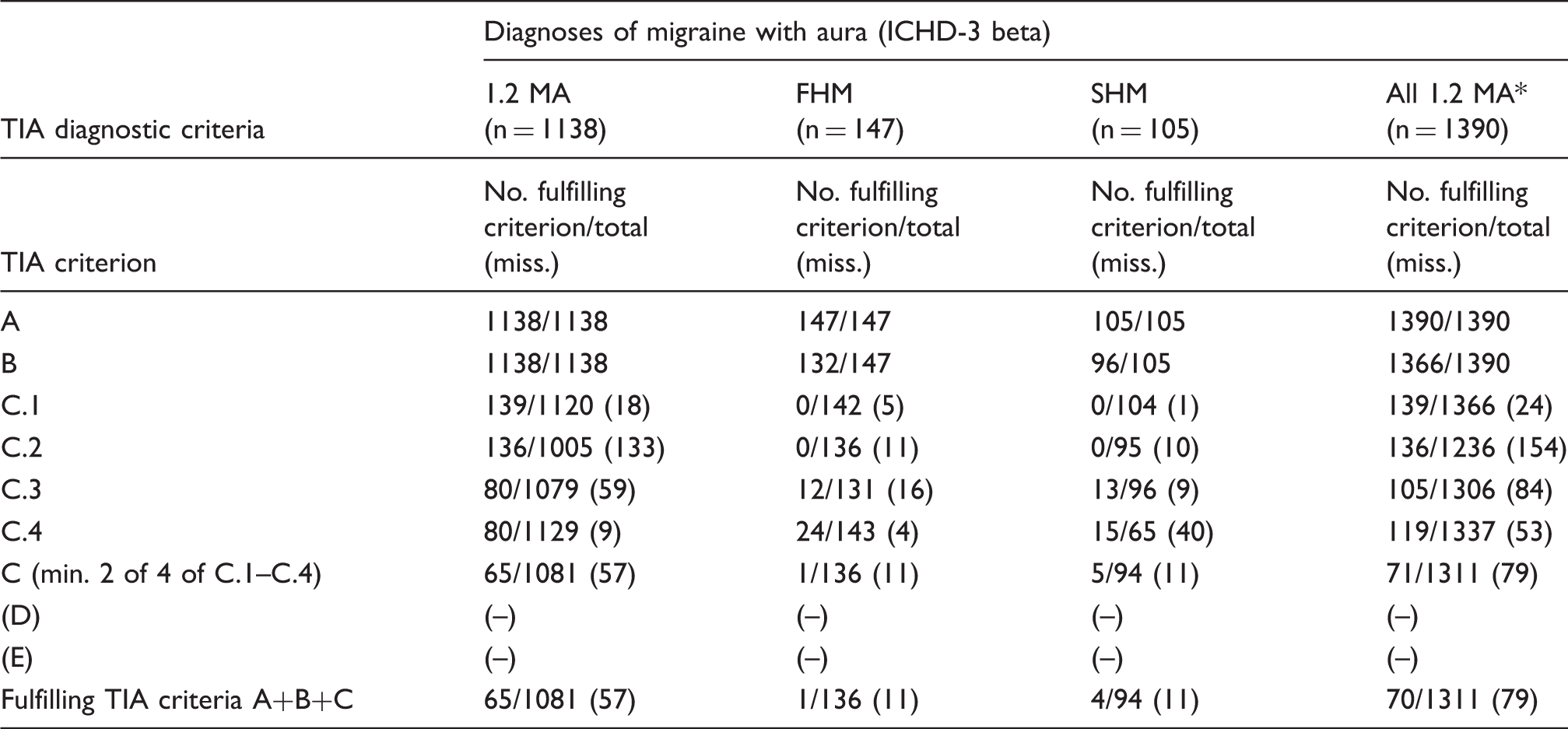
MA: Migraine with aura; FHM: Familial hemiplegic migraine; SHM: sporadic hemiplegic migraine. *Diagnosis made in all cases according to International Classification of headache disorders (ICHD) 3 beta, appendix.
Testing of TIA diagnostic criteria for specificity in patients with migraine with aura in the Russian material.
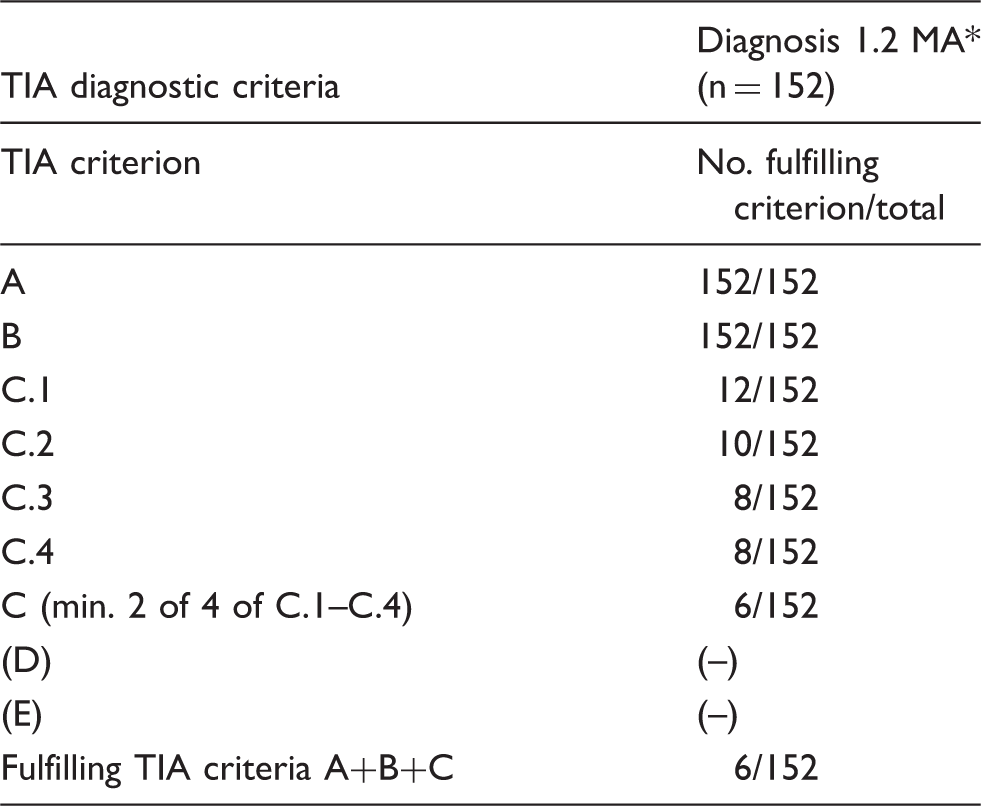
MA: Migraine with aura, diagnosis made in all cases according to International Classification of headache disorders (ICHD) 3 beta, appendix.
Discussion
The main result of this study was that it is possible, using only data from the patient history, to develop explicit diagnostic criteria that have excellent sensitivity when tested against conventionally-diagnosed TIA and specificity tested against migraine with aura.
Previous attempts to improve the TIA diagnosis
Previous attempts to improve TIA diagnosis include stroke diagnostic tools such as FAST and ROSIER. They have been developed for use by pre-hospital assessors and emergency room clinicians (13,14). The ABCD2 score has been used as a crude diagnostic aid for TIA (15). More recently, the ability of the ABCD2 score to reliably discriminate between those at high or low risk after a TIA has been called into question because a third of mimics had ABCD2 scores ≥4 (16). The score of Dawson and colleagues was not designed for retinal and some posterior circulation events and is not widely used (17). It has shown limited accuracy when used in a primary care setting (18). Recently the “Diagnosis of TIA Score (DOTS)”, which is a new tool to help non-specialists make the diagnosis of TIA with greater accuracy, was presented, but it does not convincingly solve the problem (19). The sensitivity and specificity of the DOT score were 89% (CI: 84–93%) and 76% (70–81%) respectively. This is lower than in the explicit criteria that we propose. The DOT score uses the terminology “visual aura”, and it is difficult to be sure about the presence of other aura symptoms because it was not indicated that some other symptoms, for example hemihypoesthesia, can have a gradual spread too if it is aura, but not if it is TIA. We specify this better: “At least one symptom is maximal in < 1 minute (no gradual spread)”. The DOT score also contains “Headache” but no description of headache: Is it previous history of headache, is it new headache at the time of TIA? We describe it better: “No headache accompanies or follows the neurological symptoms within one hour”. Finally, and most importantly, rating scales are almost never used by non-experts because they are too abstract. Diagnostic criteria have a better chance because they provide a diagnosis in a more direct way. A study validating TIA diagnoses in the Swedish stroke registry operated with probable and possible diagnoses, which makes it difficult to interpret in relation to the present study (20). Furthermore, the diagnoses were discharge diagnoses after admission and full investigation. The aim of the diagnostic criteria proposed by us is that they should be useful in the emergency room or acute stroke admission unit when applied by the younger physicians on duty.
Development of the proposed explicit diagnostic criteria
The basis for the development of explicit diagnostic criteria for TIA was the criteria for migraine with aura, which have been extremely successful (ICHD-3). It was obvious, therefore, to use the same format, which allows the use of all important clinical information that is readily at hand on the first patient encounter. For TIA, it must of course be supplemented by neuroimaging to rule out stroke. Ideally, this should be diffusion weighted MR imaging, but the WHO has tried to avoid this degree of specificity because DWI is not available in large parts of the world. For that reason, we have chosen neuroimaging rather than DWI. From the known clinical facts in the literature, we next constructed criteria so that they would optimally distinguish TIA from the migraine aura, which is probably the commonest TIA mimic (4,11). There are excellent studies showing the most typical symptoms of TIA and, although not strictly part of the criteria, we have mentioned them in parenthesis for clinical guidance (3,4). More importantly, it has been shown which symptoms most often cause non-stroke specialists to make an erroneous diagnosis of TIA (3,4,5,6,7). These symptoms have been included as exclusionary in the criteria for TIA because as isolated symptoms they very rarely represent TIA. Thus, isolated shaking spells, diplopia, dizziness, vertigo, syncope, decreased level of consciousness, confusion, hyperventilation-associated paresthesias, unexplained falls and amnesia rule out TIA according to criterion D. There may, however, be rare patients with isolated vertigo who actually have TIA in the brain stem. But criteria can never be totally sensitive. If they were, they would lose specificity. Diagnostic criteria must always represent a compromise between sensitivity and specificity. A big advantage of the proposed diagnostic criteria is that they are explicit, also called operational. They can therefore easily be tested in several different clinical settings and they can easily be modified according to future results of field testing. This kind of process has led to gradual improvement of the criteria for the different headache disorders (ICHD-3).
TIA definitions in relation to present results
The so-called definitions of TIA in several versions are unhelpful when junior doctors in the emergency room or in an acute TIA or stroke clinic have to make the diagnosis. All they say is that symptoms must occur acutely and be fully reversible within 24 hours. But patients and doctors use the word “acute” with a variety of meanings spanning from seconds to hours. Migraine aura usually develops within 20 minutes but may do so as fast as in 5 minutes (ICHD-3) (10). That is acute. It is necessary to know exactly how many seconds or minutes it took for the symptoms to fully develop. The definition also requests that the neurological deficits must be caused by cerebral ischemia, but that can only be determined on the basis of a very acute onset, because there must not be a relevant lesion on neuroimaging. Thus, the migraine aura actually fulfils the definitions of TIA. Put in another way, the definitions of TIA have zero specificity tested against migraine with aura. To distinguish between TIA and migraine aura, it is necessary to use more clinical characteristics. This can be done using the format of the diagnostic criteria of the ICHD-3. It allows the use of characteristics that are not always present because of the so-called polythetic criteria, that is, criteria that require only, for example, two out of four clinical features. Thus, it became possible to use the fact that TIA symptoms are almost always negative, while migraine aura often has positive symptoms such as pins and needles or flickering light. Also, absence of headache immediately after the neurological deficits can be used. Furthermore, our criteria rule out mistaking TIA for most mimics, which are specifically mentioned as exclusionary. Current definitions do nothing to avoid mimics. Thus, our criteria direct systematic and specific questioning. Despite their relative simplicity, the criteria can diagnose TIA with 99% sensitivity. The criteria can also distinguish TIA from migraine with aura with 95% specificity. These criteria will therefore be a great help for the physician in the emergency room or in migraine, TIA, or stroke clinics. It should be mentioned that women, in particular, may be difficult to diagnose. They have migraine twice as frequently as men, so many women with TIA already have a history of migraine. Therefore, a TIA may be missed in the belief that it is just another migraine attack.
Suggestions for further testing of the proposed diagnostic criteria
It would be important to perform further testing of the proposed diagnostic criteria for TIA. It is preferable to make this in specialized stroke or TIA clinics, and also in the emergency rooms of hospitals with a large intake of patients with cerebrovascular diseases. All patients should be interviewed at the time of admission by the junior physician on duty, using the proposed diagnostic criteria. Patients should then have extensive cerebrovascular work-up including DWI and a final diagnosis made by an experienced stroke physician.
Strengths and weaknesses of the present study
One limitation of this study is the quick disappearance of clinical symptoms in TIA patients before admission to the hospital. Some patients could not remember details that are important in the differential diagnosis of TIA and MA. For example, some patients could have missed the gradual spread of symptoms or the presence of a succession of symptoms. Some patients could not describe the characteristics of headache during TIA very well. Therefore, some cases of MA could have been missed. However, we performed follow-up of 118 out of 120 patients with TIA. The period of follow-up varied from 6 months to 4 years. We found only one case of migraine with aura that was missed during the first interview. This patient experienced three more similar attacks of migraine with aura during the two following years.
One might argue that 120 TIA patients is not a large material. It is, however, very demanding to prospectively interview patients in the acute phase after TIA using a neurologist-conducted semi-structured interview about both the previous and actual headache history and about the characteristics of the cerebrovascular event. We believe that this study has made great strides towards valid explicit diagnostic criteria for TIA. As mentioned above, it would, however, be good to further test the criteria in other countries and other settings to further analyze their efficacy in distinguishing TIA from mimics other than migraine with aura.
Conclusions
Prompted by previous demonstration of poor validity and repeatability of the TIA diagnosis, we have developed explicit diagnostic criteria. Tested for sensitivity in a TIA material and for specificity in two materials of migraine with aura they perform excellently.
Clinical implications
Because the proposed explicit diagnostic criteria for TIA specifies precisely what questions to ask, and what answers that must be obtained, they are very likely to improve the diagnosis of TIA versus mimics. We give proof that this is so versus migraine with aura. The criteria are expected to be useful for all physicians, not only neurologists.
Footnotes
Acknowledgement
We gratefully acknowledge the following doctors who participated at this study: TS Tsypushkina and PA Filimonova.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
