Abstract
Background
This study investigated the effects of medication overuse and withdrawal on modulation of pain processing in women with migraine. Temporal summation of laser-evoked thermal pain was used to measure the effects of conditioned pain modulation.
Methods
36 female participants (12 healthy volunteers, 12 with episodic migraine and 12 with medication overuse headache) were included in a two session protocol. Medication overuse headache subjects were also tested three weeks after medication overuse headache withdrawal. Mechanical and laser-evoked thermal pain thresholds were measured on the back of the non-dominant hand where, later, temporal summation of laser-evoked thermal pain to repetitive thermal stimuli was elicited for 30 min, at an intensity producing moderate pain. Between the 10th and 20th minutes, the contralateral foot was immersed into a water bath at a not painful (30℃) or painfully cold (8℃; conditioned pain modulation) temperature.
Results
Episodic migraine, medication overuse headache and medication overuse headache withdrawal were associated with an increase in extracephalic temporal summation of laser-evoked thermal pain as compared to healthy volunteer subjects, while there was no alteration of laser-evoked thermal and mechanical extracephalic pain thresholds in these subjects. Conditioned pain modulation was highly efficient in temporal summation of laser-evoked thermal pain in healthy volunteer subjects, with a solid post-effect (reduction of pain). Conditioned pain modulation was still present, but reduced, in episodic migraine. By contrast, conditioned pain modulation was normal in medication overuse headache and strongly reduced in medication overuse headache withdrawal. Furthermore, in medication overuse headache withdrawal, the post-effect was no longer a decrease, but a facilitation of pain.
Conclusions
These data show that a decrease in conditioned pain modulation does not underlie medication overuse headache in women. On the contrary, medication overuse reinstated conditioned pain modulation in female migraine patients. They also identify different phenotypes of pain modulation in migraine patients.
Introduction
Migraine is a common and disabling neurological disorder that manifests as a recurrent, episodic, often severe, headache. Among episodic migraineurs, a subset develops chronic migraine, a very invalidating condition characterized by headaches for more than 15 days a month. The most prominent risk factors that can promote chronic migraine include the frequency of attacks, psychiatric co-morbidities such as anxiety, depression, addictive behavior, and medication overuse (1). Indeed, the regular overuse for more than three months of one or more of the drugs normally used to relieve acute and/or symptomatic headache can lead to medication overuse headache (MOH). According to cross-sectional studies, chronic migraine and MOH are associated in about 65% of patients (2). The pathogenesis of medication overuse headache is unclear. It is still not known whether chronic medication use increases sensitivity to pain perception and thus leads to chronicity, or whether migraine progression on its own facilitates medication abuse. However, one important issue to understand the mechanisms underlying such progression, from episodic to chronic migraine associated with medication overuse, is to determine whether and how endogenous pain modulation systems are altered in medication overuse headache vs. episodic migraine.
One of the major endogenous pain inhibitory systems is conditioned pain modulation (CPM; formerly diffuse noxious inhibitory controls (DNIC)), a powerful general endogenous analgesic mechanism that can completely inhibit incoming nociceptor signals at the primary synapse (3). CPM is believed to play an important role in the development and exacerbation of chronic pain, because dysfunction of CPM is associated with a shift in balance between pain facilitation and pain inhibition. In many (but not all) patients with central sensitization, CPM is less efficacious (4). Migraine has been linked to an impairment of endogenous pain inhibitory systems, including subtle deficits in CPM (5). MOH could also result from a deficit in CPM (6,7). Chronic treatment with oral opioids is associated with altered CPM in patients with chronic pain (8). Accordingly, CPM was found to be reduced in MOH patients, while the situation improved after medication withdrawal (9). By contrast, a failure of CPM upon the R2 component of the blink-reflex was reported in another population of chronic migraineurs who had discontinued drug overuse for at least two months (10). There are thus possible conflicting results for the effect of medication overuse and withdrawal on modulation of pain processing in migraine patients. Here, we have sought to determine whether medication overuse and withdrawal could interfere with central modulation of pain processing in migraine and, more specifically, whether a decrease in CPM could play a part in medication overuse headache. To address this issue, we evaluated the effect of CPM on extracephalic pain perception in healthy volunteers (HV), episodic migraineurs (EM), and MOH patients, before and three weeks after withdrawal of medications. Since migraine is mostly a women’s disease and there are sex differences in CPM (11,12), we only included women in our study.
Methods
This is an experimental study conducted in the Neurology Department of Clermont-Ferrand University Hospital (France).
Standard protocol approvals, registrations, and subject consents
The study was performed after approval was obtained from the regional Ethics Committee “CCP Sud-Est” in 2012. Standard written informed consent was obtained from all participants, according to the 1964 declaration of Helsinki. The study was registered as N° 2008-A00471-54.
Participants
In a population of female subjects aged 18–65 years having an appointment for migraine at the Neurology Department of Clermont-Ferrand University Hospital, two groups of subjects were successively recruited: Episodic migraineurs without aura (EM), and subjects suffering from medication overuse headache (MOH). After the first phone contact for appointment, a three-month diary was sent to patients and they were asked to indicate headache episodes and treatment intake on a daily basis. A third group of age-matched volunteer females was later recruited from a database of healthy people. Episodic migraineurs met the diagnosis criteria of the International Headache Society (IHS), had two to five attacks per month without aura, and had not used preventive medications for at least two months. Subjects suffering from MOH met the diagnosis criteria of the revised International Classification of headache Disorders that was available at the time of the study (second edition, ICHD-II (13)) and had migraine as the primary headache. MOH subjects with tension-type headache were not considered. Only women were included in the study, since migraine is mostly a women’s pathology and CPMs display sex-specific features (11,12,14).
All subjects underwent a standardized interview, as well as a clinical neurological examination, and all were free of any other neurological dysfunction or chronic pain disease. All experiments were performed during the menstrual cycle follicular phase. Oral contraception was not an exclusion criterion. In patients with no menstrual cycle as a consequence of hormonal treatment, the treatment was maintained during experiments. In all groups, exclusion criteria were as follows: Treatment with tricyclic antidepressants or serotonin reuptake inhibitors, alcohol or drug addiction, pregnancy. All experiments in EM and MOH subjects were performed at least 24 hours after any acute migraine attack. If an attack happened within the 24 hours after a test, patients were asked to notify the investigator and the experimental results were discarded. In the MOH group, CPM experiments were performed again three weeks after treatment withdrawal (MOHw).
Questionnaires
Standard demographic information, duration of migraine, prophylactic and attack headache medications used before were collected on the day of inclusion. At the same time, all subjects also completed a series of self-report questionnaires including the Beck Depression Inventory short form (BDI-SF), the State trait inventory anxiety (STAI YA) form, the Pain Catastrophizing Scale (PCS), the Headache Impact Test (HIT-6) and the Medication Dependence Questionnaire in Headache patients (MDQ-H).
Pain threshold measurements
Extracephalic mechanical and thermal pain thresholds were assessed in all subjects, while they sat quietly in a room maintained at a constant temperature (23℃).
Mechanical stimuli were applied on the back of the non-dominant hand using an electronic algometer (electronic von Frey, Bioseb, France). The strain gauge was connected to a plastic sterile cone (Eppendorf, Hamburg, Germany), the tip of which was applied perpendicularly to the studied skin area. The punctate pressure was gradually increased with a constant slope under visual control of the pressure value up to the detection of the mechanical pain threshold. The threshold was defined as the lowest pressure that produced a sensation of pain. The results of three separate consecutive measurements at different points in the testing area were averaged to establish the mechanical pain threshold value.
Thermal pain was evoked by laser stimulation of the back of the non-dominant hand using a YAP laser stimulator (fiber–optic guidance, diameter 5 mm, duration 2 ms). The stimulator, with optic-fiber guidance, was placed on a skin area of about 6 cm2 at the dorsal aspect of the hand. Cutaneous heat stimuli were applied in incremental intensities in steps of 0.25 J, starting at a minimum of 0.75 J and reaching a maximum of 2.00 J. The threshold was defined as the lowest intensity that produced a sensation of pain.
CPM measurements
CPM was assessed on the temporal summation of pain evoked by thermal stimuli. Blocks of four thermal test stimuli (Nd:YAP laser stimulator, diameter 5 mm; duration 2 ms) were delivered at 0.2 Hz on the back of the non-dominant hand. First, the lowest laser energy able to produce a moderate pain (3–6 on a 0–10 VAS scale) was determined (∼10 min before CPM assessment) by applying one block of four thermal test stimuli (spaced 5 sec apart) at an intensity just below the pain threshold. Thereafter, the intensity of the stimulation was adjusted according to the subject’s response, and further blocks were repeated every 40 sec for 30 min, at an intensity producing a moderate pain (3–6 on a 0–10 VAS scale). After each block of stimulation, subjects were asked to score pain on a 0–10 VAS scale. The laser beam was moved (∼1 cm) between each block to avoid skin lesions.
To produce CPM, the contralateral foot was immersed into a water bath (a conditioning stimulus) at a noxious cold (8℃) temperature, between the 10th and 20th minute of the experiment. In control experiments, performed on a different day in the same subjects, the water bath temperature was neutral (30℃). In all cases, water was constantly re-circulated to prevent laminar warming around the immersed foot. All subjects were asked to rate the cold pain on a 0–10 VAS scale immediately at the end of immersion. The order of CPM vs. control experiments was assigned randomly.
The responses for each block in each condition were plotted over time. The first 15 responses were used to study the unconditioned temporal summation of pain, the next 15 to study CPM, and the 15 final responses to study the post-effects of CPM.
Treatment withdrawal management
One week after the initial CPM experiments, MOH subjects were instructed to abruptly stop headache medication intake and immediately hospitalized for five days. During this period, headache was managed using ice packs applied on the forehead, and when necessary, sedative medication (hydroxyzine, 25 mg per day). At discharge from hospital, patients were asked to continue withdrawal at home, until they could wake up without headache. Afterwards, they were allowed to take a triptan at the beginning of the next attack. Preventive therapy was begun three weeks after treatment withdrawal, just after the last CPM experiment was performed.
Data analysis
Statistics were computed with STATA V12 (Stata Corp, College Station, TX, USA). Results were expressed as mean ± standard error of the mean (SEM) or median (interquartile range) and as frequencies (percentage). Groups were compared using the Chi square test (or Fisher’s exact test when appropriate) for categorical data, followed by the Marascuillo procedure when signification was reached comparing groups, and using Student t-test (for comparing two groups) or ANOVA (for comparing three or more groups), or the Mann-Whitney/Kruskal-Wallis test (for comparing two or more groups), depending on the data distribution for continuous data. Those tests were followed by a Dunn’s post-hoc test when significance was reached between groups. Relationships between the number of headache days and BDI, STAI, PCS and MDQ-H scores were assessed by Spearman’s correlation coefficient. The evolution of the number of headache days and tablet intake from baseline to three weeks after withdrawal was analyzed using the Wilcoxon matched-pairs signed rank test.
Pain perception was analyzed using linear mixed models with random subject intercept. Groups, time, and foot water immersion were tested as fixed effects. Those methods were developed for temporal subdivision (control, water immersion and post-water immersion periods) in order to test: a) the session effect, b) the effect of the repeated painful stimulation, and c) the water immersion effect and the post-water immersion effect. Those analyses were repeated intra and inter groups. All tests were two-sided and a p-value < 5% was considered statistically significant.
Results
Study population
Thirty-six female subjects were included in the present study, out of 42 who were screened. Six subjects were excluded (four in the HV group, two in the MOH group) because their pain tests were not sufficiently painful (<3 on VAS). There were finally 12 subjects in each group.
Demographic, clinical characteristics of migraine attacks and psychiatric comorbidities in healthy voluntary (HV) subjects, episodic migraine (EM) patients and medication overuse headache (MOH) patients.

p-values indicated in bold are the only ones that were significantly different. *(median, interquartile range).
Psychiatric symptoms were more often associated with MOH than with EM (Table 1). MOH patients had significantly higher BDI, PCS and STAI scores than HV subjects and EM patients. The MOH patient MDQ-H score was twice as important as the EM patient MDQ-H score. Spearman's rank correlation revealed a highly significant correlation between the number of headache days and BDI score (r = 0.46; p = 0.02), STAI score (r = 0.51; p = 0.01), PCS score (r = 0.55; p = 0.006), MDQ-H score (r = 0.78; p < 0.001).
Acute headache treatments were classified as non-opioids (triptans, NSAIDs, acetaminophen, alone or in combination) or opioids (tramadol, codeine) even when associated with any non-opioid acute headache treatment. Most patients used triptans or simple analgesics, alone or in combination. Opioid consumption was the same in the two groups [(EM group (n = 3) and MOH group (n = 4)] and always associated with non-opioid medication. No EM patient took preventive treatment. Only three MOH patients were taking prophylactic drugs [indoramine (n = 1), beta blockers (n = 1) and carbamazepine (n = 1)] at inclusion. Treatments were kept during the study and until discharge, without changing doses.
All MOH patients completely stopped taking analgesics during the withdrawal period, while four of them needed hydroxyzine infusion especially for sleeping assistance. Ice packs were used sporadically during hospitalization by all MOH patients. Three weeks after withdrawal, the mean number of headache days/month and tablet-intake had dropped dramatically, from 27.5 ± 1.4 to 8.5 ± 2.6 days/month (Wilcoxon signed rank test, p = 0.002) and from 70.0 ± 14.8 to 9.0 ± 3.0 tablets/month, Wilcoxon signed rank test, p = 0.004) respectively. One patient found no significant improvement of headache after withdrawal.
All MOHw patients initiated a preventive treatment after the last experiment, including tricyclic antidepressants (4), valproate (3), topiramate (1), oxerotone (2) beta-blockers (1) or nadolol (1). Six months after withdrawal, the mean number of headache days/month and tablet-intake in these patients were 10.5 ± 3.8 and 11.5 ± 4.1, respectively.
Extracephalic pain thresholds and temporal summation of pain
Mechanical and thermal pain thresholds.

Mechanical and thermal pain thresholds evoked, respectively, by application of an electronic algometer and Nd:YAP laser stimulator on the back of the non-dominant hand in healthy voluntary (HV) subjects, episodic migraine (EM) patients and medication overuse headache (MOH) patients.
Following repetitive laser-mediated thermal stimuli applied on the hand, pain intensity progressively increased in all tested groups: HV, EM, MOH and MOHw, showing temporal summation in all cases (Figure 1). Pain intensity increased significantly from the first to the 15th block of stimulation in HV subjects (p = 0.001), from 4.0 ± 0.4 to 5.1 ± 0.4, as well as in EM subjects (p < 0.001), from 4.7 ± 0.4 to 6.3 ± 0.4, in MOH subjects (p < 0.001), from 4.8 ± 0.4 to 6.4 ± 0.4 and after withdrawal (MOHw, p < 0.001), from 5.4 ± 0.5 to 6.1 ± 0.3. EM, MOH and MOHw subjects had significantly (p < 0.001) greater scores than HV subjects (Figure 1). The differences were significant mainly between the 10th and the 15th blocks of stimuli. There was no difference between EM, MOH and MOHw subjects.
Time course of pain perception evoked by repetitive laser painful stimuli of the hand in healthy voluntary (HV) subjects, episodic migraine (EM) patients and medication overuse headache patients before (MOH) and (MOHw) after withdrawal. Testing sessions consisting of a series of 15 blocks of four thermal test stimuli (Nd:YAP laser stimulator) were delivered at 0.2 Hz on the back of the non-dominant hand, repeated every 40 sec for 10 min, at an intensity producing a moderate pain (3–6 on a 0–10 VAS scale). Mean pain ratings (± SEM) are shown from the first to the 15th stimulation. EM, MOH and MOHw patients showed significantly greater summation of pain than HV subjects.
CPM measurements
All participants experienced the conditioning stimulation (foot immersion in water at 8℃) as painful (VAS: HV = 8.3 ± 0.4, EM = 7.1 ± 0.7, MOH = 8.4 ± 0.4) and achieved the experimental procedures. There was no difference between the groups (p = 0.27). In all participants, the application of the non-painful conditioning stimulation (foot immersion in water at 30℃) did not induce pain.
In HV subjects, laser-evoked pain was depressed during (p < 0.001) and after (p < 0.001) the application of the painful conditioning stimulation of the contralateral foot, whereas non-painful stimulation had no effect (Figure 2(a)). The inhibitory effects appeared immediately after foot immersion in cold water, reached their maximum within a few minutes, and outlasted the conditioning period for several minutes.
Time course of pain perception evoked by repetitive painful stimuli of the hand before (first to 15th), during (16th to 30th), and after (31st to 45th), application of non-painful (in yellow) or painful (in green) conditioning thermal stimulation on the contralateral foot in (a) healthy voluntary (HV) subjects, (b) episodic migraine (EM) patients, and medication overuse headache (MOH) patients before (c) and after (d) medication withdrawal (MOHw). To assess CPM, blocks of four thermal test stimuli (Nd:YAP laser stimulator) were delivered at 0.2 Hz on the back of the non-dominant hand, repeated every 40 sec for 30 min, at an intensity producing a moderate pain (3–6 on a 0–10 VAS scale). Between the 10th and 20th minute, the contralateral foot was immersed into a water bath at a non-painful (30℃) or painful cold (8℃) temperature. A mixed linear model was used to explore the effects of CPM. The p-values indicate the differences between the non-painful and painful conditioning thermal stimulation.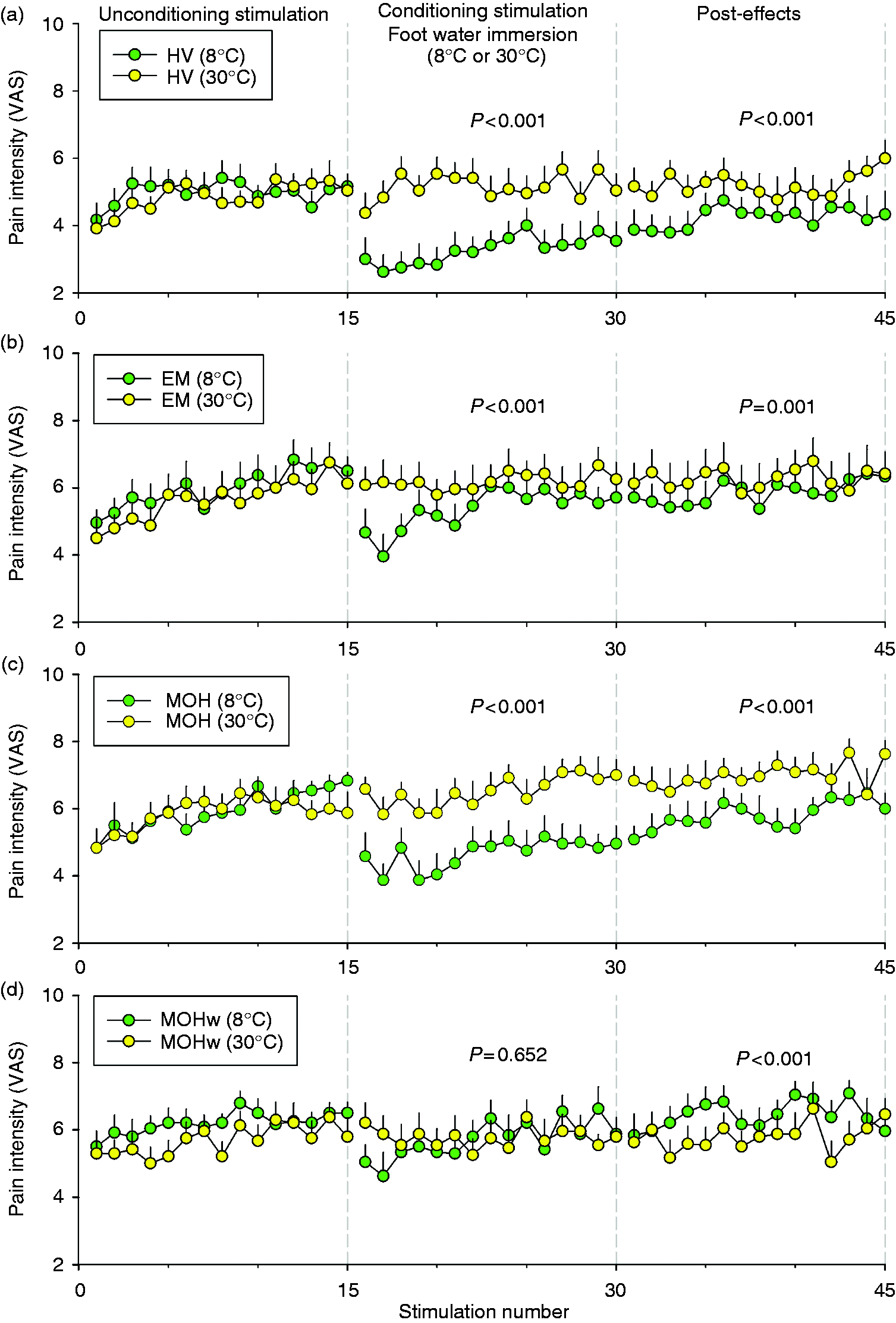
In EM patients, laser-evoked pain was also depressed during (p < 0.001) and slightly after (p = 0.001) the application of the painful conditioning stimulation on the contralateral foot (Figure 2(b)). Non-painful stimulation had no effect. However, the magnitude of CPM was significantly lower in EM vs. HV subjects during (p < 0.001) and after (p = 0.002) the conditioning stimulation.
In MOH patients, laser-evoked pain was significantly depressed during (p < 0.001) and after (p < 0.001) the application of the painful conditioning stimulation on the contralateral foot (Figure 2(c)). Non-painful stimulation had no effect. The magnitude of CPM was significantly higher in MOH patients compared to EM patients during (p < 0.001) and after (p < 0.001) conditioning stimulation. CPM in MOH patients had a similar magnitude compared to HV subjects.
In MOHw patients (after medication withdrawal), laser-evoked pain was not significantly depressed during (p = 0.65) the application of the painful conditioning stimulation on the contralateral foot (Figure 2(d)). Non-painful stimulation had no effect. The magnitude of CPM was significantly lower in MOHw patients compared to HV subjects (p < 0.001), EM patients (p < 0.001) and MOH patients (p < 0.001). By contrast, during the period following the conditioning stimulation (31st to 45th block of stimulation, Figure 2(d)), MOHw patients showed facilitation of pain compared to the control condition (p < 0.001) and higher pain scores than HV subjects (p < 0.001), EM patients (p < 0.001) and MOH patients (p < 0.001).
Discussion
In this study, we investigated whether a decrease in CPM could be one of the mechanisms leading from EM to MOH in women with migraine. We first showed that EM, MOH and MOHw were associated with an increase in temporal summation of extracephalic pain compared to HV subjects, while there was no alteration of laser-evoked thermal and mechanical extracephalic pain thresholds in these patients. We then found out that CPM was highly efficient at temporal pain summation in HV subjects, with a solid post-effect (reduction of pain). CPM was still present, but reduced in EM. By contrast, CPM was normal in MOH and strongly reduced in MOHw. Furthermore, in MOHw, the post-effect was no longer a decrease, but a facilitation of pain. Altogether, these data show that a decrease in CPM does not underlie medication overuse headache in women. On the contrary, medication overuse reinstated CPM in female migraine patients.
We used temporal summation of laser-evoked thermal pain to explore the effects of CPM, because temporal summation of pain relates to the hyperexcitability of nociceptive neurons that has been identified in preclinical models of chronic pain conditions. This, therefore, offers an opportunity to link preclinical and clinical results. Temporal summation of extracephalic laser-evoked thermal pain was significantly higher in EM, MOH and MOHw vs. HV subjects. Similar facilitation of extracephalic temporal summation of pain after electrical stimulation has been reported in both episodic (15, but see 9,16) and chronic (9,16, but see 17) migraine patients and enhanced temporal summation of extracephalic pain has been also reported in other cephalic (18,19,20) and extracephalic (9,21) pain conditions. Temporal summation of pain thus appears to be a general feature of chronic or recurrent clinical pain conditions.
Temporal summation of pain was similar in EM, MOH and MOHw patients. This suggests that medication overuse and withdrawal did not affect the hyperexcitability of nociceptive spinal cord neurons directly.
Increases in temporal summation of extracephalic pain in patients were not associated to any changes in laser-evoked and mechanical extracephalic pain thresholds. Consistently, EM patients have been shown to exhibit normal laser-evoked (10,22,23,24) and mechanical (25,26,27, but see 20,28) pain thresholds. In chronic migraineurs, some authors have reported that interictal chronic migraineurs have lower mechanical pain thresholds (17,28), compared with healthy controls, while others, consistent with our findings, reported no differences in mechanical (27,29,30,31,32) and laser-evoked (23,33) pain thresholds.
Finally, the temporal summation of pain was not associated to extracephalic pain symptoms in this study, as in our previous preclinical work using a rat model of chronic migraine (34,35,36). In the patients, hyperexcitability may have remained under the threshold required for clinical expression of pain symptoms (34,35,36). The threshold might only be reached during (37) or close to (38,39,40,41) migraine attacks or when migraine is associated with other painful conditions such as temporomandibular disorders (39) or fibromyalgia (42). Indeed, in these conditions, most patients exhibit extracephalic cutaneous hypersensitivity.
Migraine, medication overuse and withdrawal could affect the hyperexcitability of nociceptive spinal cord neurons through CPM. Indeed, CPM is a powerful endogenous analgesic mechanism that works in animals (43) as in humans (3), where nociceptive stimulation applied to a given body location reduces the perception and brain responses elicited by noxious test stimuli delivered at a remote body location. The CPM denomination replaces the previous term “diffuse noxious inhibitory controls (DNICs)”, which provide one of the main supraspinal pain inhibitory pathways. Studies in animals have shown that DNICs involve the brainstem subnucleus reticularis dorsalis (44) and a loop between lamina I/III spinoparabrachial neurons that express the NK1 receptor (45,46) and hypothalamic dopaminergic descending neurons (47,48). Dysfunction of CPM is believed to shift the balance between pain facilitation and pain inhibition and facilitate the development of chronic pain, including various kinds of cephalalgia from tension headache to cluster headache (22,49,50,51,52), temporomandibular disorders (53) and fibromyalgia (54). In our study, CPM was highly efficient on temporal pain summation in female HV subjects, with a solid post-effect (reduction of pain). Consistent with previous studies in migraine patients (52) EM patients showed reduced CPM. However, other studies found no CPM impairment in patients suffering from EM (9,55). This difference could result from the different experimental paradigms used by the different authors, including the type of conditioning stimuli, test stimuli, locations, and individual factors (reviewed in 56,57). For instance, individual factors such as young age, male gender, ovulatory phase, positive expectations, attention to the conditioning stimulus, and being a carrier of the 5-HTTLPR long allele result in better CPM (58). Interestingly, although we found a similar decrease in scores for the first responses in EM and HV subjects, EM patients demonstrated reduced CPM with repetition, suggesting a subtle pronociceptive pain modulation pattern, as also shown by others (5).
We expected a supplementary decrease in CPM to underlie MOH, and that CPM would normalize after withdrawal, as previously reported by Perrotta et al (9). To our surprise, CPM was normal in MOH, at the same level as HV subjects, and strongly reduced in MOHw. Several factors could contribute to the differences between the two works. First, we used thermal stimuli applied to the hand to evoke pain, rated pain using a VAS, and CPM was produced by a cold (8℃) stimulus applied to the lower limb for 10 minutes, while in their work, Perrotta et al. (9) used electrical stimuli applied to the leg, measured nociceptive withdrawal reflex area, used a temporal summation threshold of the nociceptive withdrawal reflex, used a numerical rating scale, and CPM was produced by a cold stimulus (3–4℃) applied to the hand for four to five minutes. These technical differences could influence the results (59). Furthermore, in our study, MOH patients were younger than those in Perrotta’s work (35 vs. 43 years), were women alone and not a population of men and women, suffered from more severe migraine (27.5 vs. 21.6 days of headache/month) and consumed more analgesics (70.0 vs. 24.3 tablets/month). Accordingly, chronic use of opioid pain-killers has already been shown to reduce CPM in patients suffering from chronic pain (8). We also measured CPM in MOHw three weeks after the start of the withdrawal treatment, while Perrotta et al. (9) measured CPM 8–10 days after withdrawal (9). This, combined with the differences in test stimulus modalities, age, gender, pain severity, and amount of medication, could explain the divergent results of the two studies. In line with our results, a failure of CPM upon the R2 component of the blink-reflex was reported in a population of chronic migraineurs who had discontinued drug overuse for at least two months (10). To sum up, our data show that a decrease in CPM does not underlie medication overuse headache. On the contrary, medication overuse reinstated CPM in migraine patients, the mechanisms of which are unknown. Furthermore, after withdrawal, CPM was strongly reduced and the post-effect of CPM was no longer a decrease, but a facilitation of pain, at a time when our patients had a significantly reduced number of headache days and analgesic consumption. This confirms that medication overuse influences CPM, but CPM reduction does not underlie headache in such conditions. To determine whether CPM can later normalize, it would be interesting to study the dynamics of CPM after medication withdrawal long term, with or without preventive drugs.
Although clear-cut, the results have limitations. First, the size of the population was limited; second, only women were included; and third, MOH participants were recruited from a tertiary hospital, which provides a potential bias for recruitment. The present results may not thus fully apply to all MOH patients. In line with what is known about chronic migraine (1), though, our population study shows that MOH was associated with psychiatric symptoms, and that the number of headache days was correlated to the psychiatric disorder scores.
In conclusion, our study provides a new experimental paradigm to study CPM in migraine patients, using temporal summation of pain as the measurement target. This offers an opportunity to link preclinical and clinical results. We show that in a specific population of women migraineurs, temporal summation of pain was increased in episodic migraine as well as in medication overuse headache, before and after withdrawal. Furthermore, CPM was reduced in episodic migraineurs and medication overuse reinstated CPM, while withdrawal strongly decreased CPM. This demonstrates that a decrease in CPM does not underlie medication overuse headache in women. Altogether, our results are in line with the general idea that there are differences in central pain modulation between patients with chronic pain, according to pain conditions (60). They thus allow the identification of different phenotypes of pain modulation in migraine patients, as described in patients suffering from other chronic pain conditions (60,61).
Clinical implications
Conditioned pain modulation (CPM) was reduced in episodic migraine. CPM was normal in medication overuse headache and reduced after medication withdrawal. A decrease in CPM does not underlie medication overuse headache. There are different phenotypes of pain modulation in migraine patients.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by funding from Institut National de la Santé et de la Recherche Médicale (Inserm), Université Clermont Auvergne (France), and CHU Clermont-Ferrand (France).
