Abstract
Background
Identifying specific subsets of patients within the clinical spectrum of migraine could help in personalizing migraine treatment. Profiling patients by combining clinical characteristics and neurophysiological biomarkers is largely unexplored. We studied the association between migraine attack triggers and habituation of visual evoked potentials.
Methods
We personally interviewed 25 patients about their migraine triggers following a structured list, and measured the N1-P1 habituation slope over six blocks of 100 averaged pattern-reversal VEP afterwards.
Results
The mean number of triggers per patient was 4.52 ± 1.42. Habituation slopes differed significantly between subjects who reported stress as a migraine trigger (deficient VEP habituation) and subjects who did not (preserved VEP habituation). For the remaining categories, the mean amplitude slope was always positive, indicating deficient habituation, and was not significantly different between subgroups.
Conclusions
Migraine patients not reporting perceived stress as a trigger for their attacks might constitute a distinct clinic-physiological subset within the migraine spectrum.
Introduction
The quest towards a personalized migraine treatment follows many pathways. Most – if not all – approaches, namely clinical, genetic, imaging, and neurophysiological, convey the assumption that specific traits can be found in some, though not all, migraineurs. Identifying specific subsets of patients within the widespread clinical spectrum of migraine could help in developing such a beneficial tailored migraine treatment approach. While no single para-clinical investigation can by itself reliably identify or categorize a given migraine patient, the possibility of such categorization combining clinical features with accessible para-clinical tests remains quasi unexplored.
Migraine attacks can be triggered by a number of factors, among which stress, hormonal changes, missing a meal, and lack of sleep are the most frequently referenced (1). Migraine triggers are not part of the diagnostic criteria for migraine, but they may characterize subgroups of patients (2). On the other hand, deficient habituation to repeated stimulations is a neurophysiological feature commonly found in migraine cohorts (3). It cannot, however, be demonstrated in all patients diagnosed according to ICHD2-3 criteria and has not been reproduced in all studies (3). One may thus assume that patients with different habituation profiles might differ by phenotypic features, such as attack triggers, that are not used to diagnose migraine.
We therefore analysed the VEP habituation profile in relation to the report of migraine triggers during a head-to-head, semi-structured interview, which preceded the neurophysiological recordings.
Subjects and methods
Twenty-five migraine without aura patients (ICHD-3 beta 1.1, mean age 26.3 ± 6 years, 80% females) who underwent visual evoked potential (VEP) recordings (4) were interviewed face to face by two of the investigators (ML and ERR) about their migraine triggers. They were instructed to provide yes or no answers to a list of potential triggers drawn according to the results of a large epidemiological study (1). The list included stress, not eating, sleeping less, sleeping more, hormonal triggers (for females), weather changes, food, alcohol, perfumes/strong odours, bright lights, smoke, physical exercise and sexual activity. Contrary to Kelman (1), neck pain was not included because of its high prevalence in the premonitory phase (5), heat was enclosed within weather, and sleeping less was specifically addressed instead of sleep disturbance, which was considered to be less specific. Both interviewing investigators emphasized the importance of distinguishing and excluding symptoms occurring during the headache phase.
Pattern reversal VEPs were registered immediately after the interview. Recordings were performed within a headache-free interval of at least 72 hours before and after a migraine attack. None of the patients had prophylactic migraine treatment, or any other pharmacological treatment other than the contraceptive pill. A detailed description of the methodology employed for VEP acquisition and processing can be found elsewhere (4). In brief, 600 responses were recorded at Oz (reference: Fz) at temporal and spatial frequencies of 3.1 Hz and 14’ respectively. The responses were averaged on-line and partitioned into six sequential blocks of 100 epochs. The mean N1-P1 habituation slope over the six blocks was compared between subjects responding positively or negatively to each trigger using the Mann-Whitney U Test. Two-tailed p values were calculated with the significance level set at p < 0.05. We used SPSS for Windows (Version 20.0, IBM Corp, Armonk, NY) for statistics and Prism version 6.00 for Windows (GraphPad Software, La Jolla, California, USA) for designing figures. All participants provided written informed consent, and the study was approved by the Institution’s Ethics in Health Research Committee (Clínica Universitaria Reina Fabiola, Córdoba, Argentina).
Results
The mean number of triggers per patient was 4.52 ± 1.42, the most common being stress, not eating, and sleeping less (76%, 72%, and 64% respectively) (Figure 1, Table 1). The average VEP habituation slope of the sample was positive, indicating a deficient habituation (0.030 ± 0.47). Regarding individual triggers, the mean habituation slope differed significantly between subjects who reported stress as a precipitating factor (n = 19) (0.042 ± 0.05) and those who did not (n = 6) (−0.007 ± 0.02) (p = 0.009) (Figure 2). The inverse relation between stress as a trigger and lack of habituation was independent of the total number of triggers, which did not differ between the stress “yes” (4.789 ± 1.134) and stress “no” groups (3.667 ± 1.966, p = 0.22). We found no significant difference in mean VEP habituation slope for the remaining trigger categories.
Percentage of patients referencing each of the individual factors as migraine triggers. The hormonal category is restricted to females. Triggers of each participant. Dots (•) indicate a positive response to an item from the list of potential triggers. Negative responses are represented by empty cells. *The hormonal category is restricted to females (cells of male participants are filled with a dash (–). Mean VEP habituation slope in relation to the positive (rhombus) or negative (circles) report of each trigger. The hormonal category is restricted to females. Error bars correspond to the standard error of the mean. The dashed line at y = 0.03 represents the mean habituation slope of the sample. The dotted line at y = 0 corresponds to the upper limit of habituation (i.e. values above 0 indicate deficient habituation). The asterisk (*) denotes a p value < 0.01.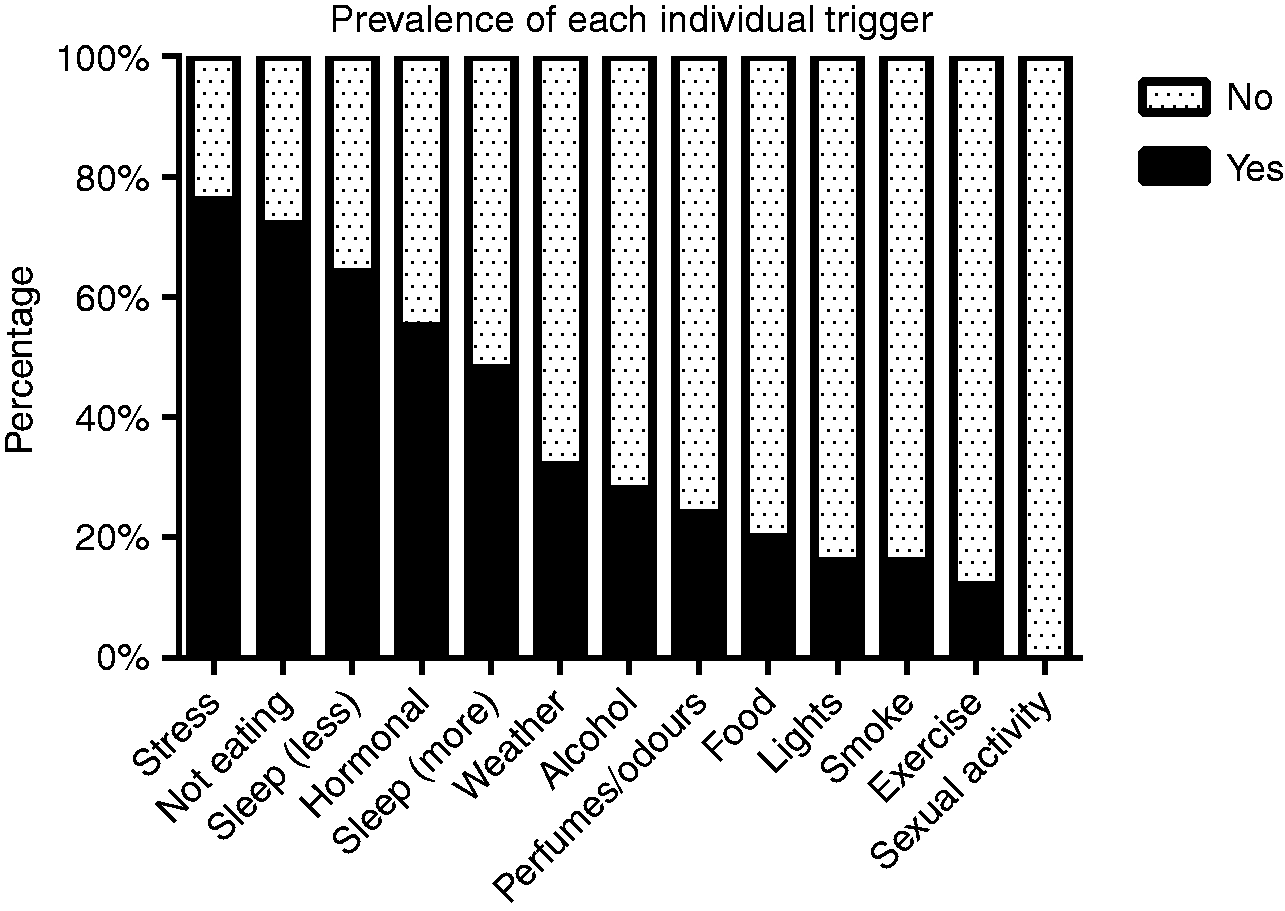


Discussion
Although sometimes questioned because evidence from retrospective and cross-sectional studies is criticisable, the role of stress as a trigger of migraine attacks was corroborated in two prospective studies that are by nature not affected by recall bias (6,7). In our sample, just as in large epidemiological studies (1), stress was the most common trigger referenced by migraine patients. Nevertheless, there is a small subgroup of patients who do not subjectively perceive stress as a potential attack-precipitating factor. From an electrophysiological perspective, this subgroup differs from the other patients and from published cohorts (3) by a normal habituation of visual evoked potentials, although not quite reaching that found in a sample of healthy volunteers recorded concurrently with the same methodology (4). This finding is noteworthy for the long-standing debate about the variability of VEP habituation in migraine. While fluctuations over the migraine cycle likely account for intra-subject variability, there is no unanimously accepted explanation for the between-subject differences (8,9).
It has been shown that stress is able to increase event-related potentials by decreasing habituation to a greater degree in migraine patients than in healthy subjects (10), and just before a migraine attack (11). In theory, there could be multiple connections between stress and abnormality of cortical evoked potentials. Even though the precise pathophysiological substrate of the deficient habituation of evoked potentials in migraine remains to be determined, there is evidence indicating that it could be related to decreased pre-activation levels of sensory cortices allowing an increased range of cortical activation, dysfunction of cortical inhibitory interneurons, and neuronal hyperexcitability (see Coppola et al. 2007 for a review) (12). All the latter can be favoured by low serotoninergic neurotransmission, thought to play a role in migraine pathogenesis but also in stress sensitivity. Another possible link between several triggers such as fasting, sleeping less, and stress could be the metabolic facet of migraine pathogenesis, based on evidence that the mitochondrial phosphorylation potential and ATP synthesis are reduced in the migrainous brain (13). Last, but not least, there may be a common genetic denominator. Variants in the CNR1 gene coding for the endocannabinoid receptor 1 (CB1), for instance, were recently found to predispose to migraine with nausea in the presence of life stress (14), which could be explained by the critical role of the endocannabinoid system in the control of the serotonin system and stress.
Whatever the neurobiological underpinning may be, the subgroup of migraine patients in whom stress is not an attack trigger may differ from the majority of patients by their pathophysiological and, possibly, genetic profile. In isolation, having a preserved or deficient habituation has not been shown to be useful for therapeutic decisions. However, if the findings of this small pilot study can be replicated in a larger sample and some insight can be gained about the underlying mechanisms, novel, better targeted therapeutic approaches based on pathophysiology might be developed in future.
Clinical implications
VEP habituation is preserved in migraine patients who do not recognize stress as a trigger. The subgroup of migraine patients in whom stress is not an attack trigger may differ from the majority of patients by their pathophysiological and, possibly, genetic profile.
Footnotes
Acknowledgements
This project forms part of the EUROHEADPAIN project - FP7 n° 602633.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
