Abstract
Objective
The objective of this article is to investigate the neurological substrates associated with medication overuse (MO) in patients with chronic migraine (CM).
Methods
We recruited age- and sex-matched CM patients with MO (CMwMO), CM patients without MO (CMwoMO), and healthy controls (HCs). Magnetic resonance T1-weighted images were processed by voxel-based morphometry, and the findings were correlated with clinical variables and treatment responses.
Results
A total of 66 patients with CM (half with MO) and 33 HCs completed the study. Patients with CMwMO compared to the patients with CMwoMO showed gray matter volume (GMV) decrease in the orbitofrontal cortex and left middle occipital gyrus as well as GMV increase in the left temporal pole/parahippocampus. The GMV changes explained 31.1% variance of the analgesics use frequency. The patients who responded to treatment had greater GMV in the orbitofrontal cortex (
Conclusions
Our study showed GMV changes in CMwMO patients compared to the CMwoMO patients. These three cerebral regions accounted for significant variance in analgesics use frequency. Moreover, the GMV of the orbitofrontal cortex was predictive of the response to MO treatments.
Keywords
Introduction
Migraine is traditionally recognized as a chronic disorder with episodic attacks; however, migraine can evolve from episodic migraine (EM) to chronic migraine (CM), i.e. headache for ≥15 days per month for >3 months (1,2). Patients with CM have more severe disability, poorer quality of life, and higher direct and indirect economic losses compared with patients with EM (3). A longitudinal study reported that 2.5% of patients with EM developed CM annually (4). The mechanism underlying the evolution from EM to CM remains elusive; however, dysfunctions in endogenous pain modulation, cortical hyperexcitability, central sensitization and medication overuse (MO) have been proposed to affect this evolution (5).
MO is defined as the regular intake of analgesics for ≥10 or 15 days per month for >3 months according to the types of analgesics being overused (2). A recent review reported that approximately half (54%) of CM patients overuse medications (6). Psychological studies suggest that MO is, like drug addiction, a disorder of dependence and maladaptive decision making (7–10). Neuroimaging studies have reported functional and structural changes in multiple brain regions of MO patients, including areas participating in reward and motivation circuits such as the orbitofrontal cortex and also areas concerning pain perception and modulation (11–13). However, these studies compared patients with migraine and MO to healthy controls (HCs), which created ambiguity in the interpretation of the two effects and their interactions.
In this study, we aimed to explore the possible neurological substrate of MO. To focus on the effect of MO, we compared patients with and without MO who had the most similar CM backgrounds. The significance of the results was assessed to identify correlations between gray matter volume (GMV) and clinical parameters, including the response to treatments.
Methods
Participants
Age- and sex-matched, right-handed chronic migraineurs without aura were recruited from the Headache Clinic at Taipei-Veterans General Hospital (VGH), and HCs were recruited from the hospital staff. The diagnoses of CM with MO (CMwMO) and CM without MO (CMwoMO) were determined by the revised International Classification of Headache Disorders, second edition (ICHD-2) criteria (codes A1.5.1 and A8.2) (14). All these patients also fulfilled the latest criteria of the ICHD third edition beta (ICHD-3 beta) version (2). Demographics and clinical data, including headache characteristics, analgesics use, and Hospital Anxiety and Depression Scale (HADS) scores, were recorded in a structured questionnaire before the magnetic resonance imaging (MRI) scan (15). The durations between collection of clinical and imaging data were similar between CMwMO and CMwoMO patients (17.2 ± 12.0 versus 13.7 ± 9.5 days,
MRI data acquisition
Structural T1-weighted MRI scans were collected using a 1.5 Tesla GE Excite II MR system (General Electric Healthcare, Milwaukee, WI, USA) with an eight-channel phase array head coil at Taipei-VGH. A T1-weighted three dimensional inversion-recovery fluid-attenuated fast spoiled gradient-recalled echo pulse sequence with the following parameters was used to acquire the T1 scans: repetition time/echo time/inversion time = 8.548/1.836/400 ms, flip angle = 15 degrees, number of excitation = 1, field of view = 260 × 260 mm2, matrix size = 256 × 256, without inter-slice gap and voxel size = 1.02 × 1.02 × 1.5 mm3. In total, 124 contiguous axial slices that were aligned to the anterior and posterior commissure line were obtained to cover the entire brain. The total acquisition time was 8 minutes and 6 seconds. All T1-weighted scans were checked to identify any gross anatomical abnormalities and substantial image-related artifacts. No participants were excluded from this visual evaluation.
Voxel-based morphometry (VBM) analysis
The T1-weighted scans were processed with a VBM approach using statistical parametric mapping (SPM8; http://www.fil.ion.ucl.ac.uk/spm; Wellcome Institute of Neurology, University College London, UK) and the VBM8 toolbox (http://dbm.neuro.uni-jena.de) (16). In our current study, the VBM processing pipeline was the same as in our previous studies (17,18). To achieve better registration performance, the original native space T1-weighted scans were manually set to the anterior commissure. These reoriented T1-weighted scans were then bias corrected and segmented into three distinct tissue components (gray matter (GM), white matter (WM) and cerebral spinal fluid (CSF)), in the native space using a tissue-prior free segmentation algorithm available in the VBM8 toolbox with default settings. These tissue segments were used to calculate the overall tissue volume (V) (including GMV, WMV and CSFV) and total intracranial volume (TIV) in the native space. Next, to account for differences in global brain size among participants, these native space tissue segments were registered to the standard Montreal Neurological Institute (MNI) template using the affine registration algorithm. Subsequently, to refine the inter-subject registration, the iterative high-dimensional registration algorithm provided by diffeomorphic anatomical registration through the exponentiated lie algebra (DARTEL) toolbox was simultaneously applied to the affine-aligned GM and WM segments of all the study participants (19). The GM segments were modulated with a non-linear deformation component of the DARTEL registration approach to enable a comparison of the relative GMV adjusted for individual brain size rather than GM tissue concentrations. Next, the covariance-based data homogeneity assessment module provided by the VBM8 toolbox was applied to confirm the quality of the processed tissue segments, and no images were identified as outliers in this step. The image resolution of the resultant GM tissue segments was resampled with an isotropic voxel resolution of 1.5 mm3. Finally, the MNI space-modulated GM tissue segments were smoothed with an isotropic Gaussian kernel of 8 mm3 full width at half maximum (FWHM) and entered as inputs for the following statistical analyses.
Statistical analyses of VBM data
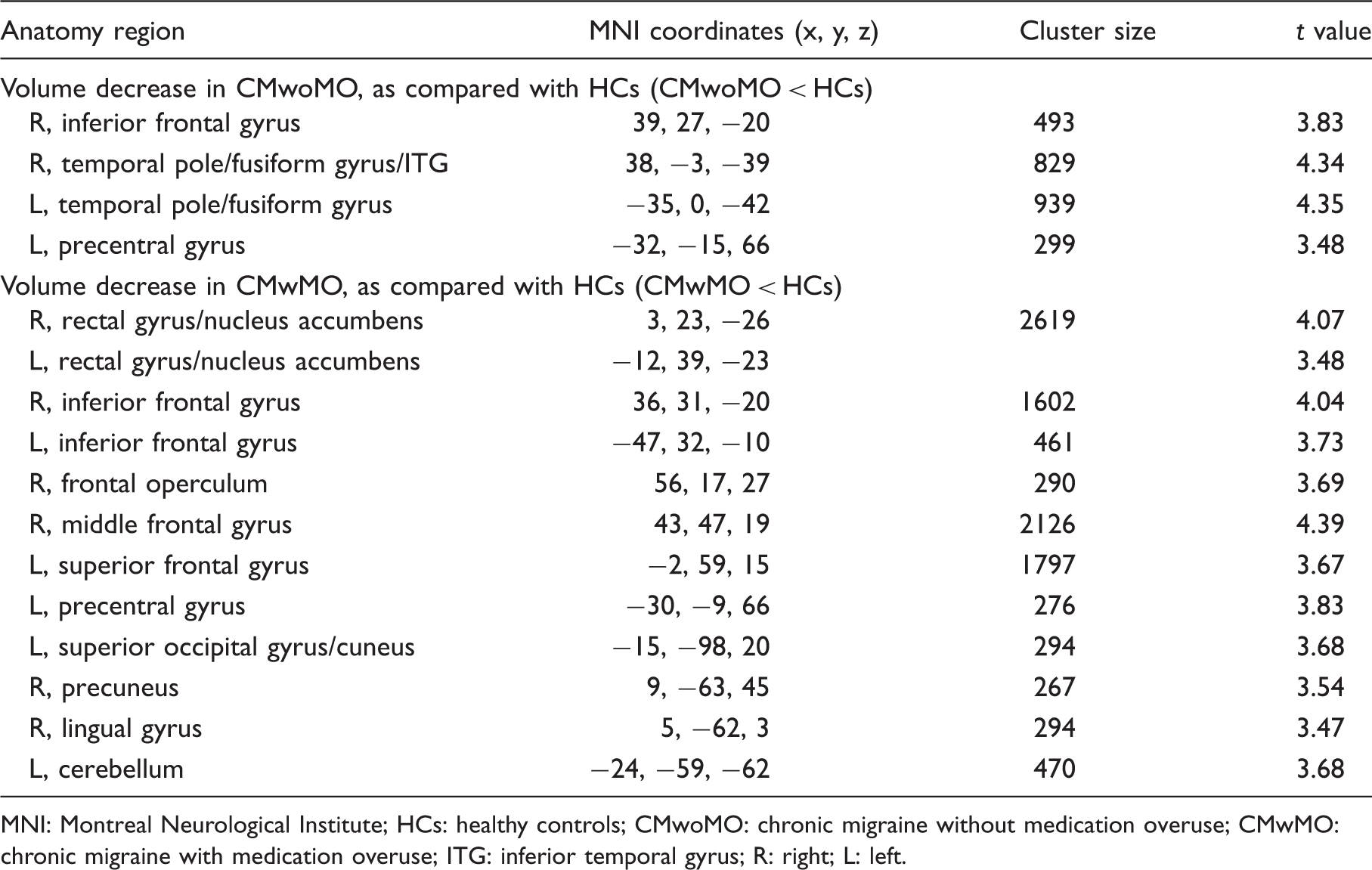
Voxel-wise statistical analyses of GMV data were conducted using the GLM Flex toolbox (http://mrtools.mgh.harvard.edu/index.php/GLM_Flex). We used a one-factor three-level (HCs, CMwoMO and CMwMO) analysis of covariance (ANCOVA) design with appropriate statistical contrast to identify the regional GMV differences between the study groups. The tested group comparison contrasts included two tiers. First, we explored the effect of MO and performed an analysis between CMwMO and CMwoMO patients. Second, we aimed to reveal the effects of migraine by conducting an analysis between CM (both with and without MO) patients and HCs. Potential confounding factors, including age and migraine ictal status, served as nuisance covariates. To ensure that statistical inferences were restricted in the GM area, we used the explicit masking procedure with the GM probability of 0.2 to exclude the GM voxels with lower GM probability. To correct for multiple comparisons, the significance level of whole-brain inferences was set at the cluster-level family-wise error (FWE) corrected with Voxels with significant differences of gray matter volumes between patients with chronic migraine with medication overuse (CMwMO) and chronic migraine without medication overuse (CMwoMO). The Montreal Neurological Institute coordinates and sizes of the clusters are shown in Table 1. Voxels with significant decrease of gray matter volumes in patients with chronic migraine (without (A) and with (B) medication overuse), as compared with healthy controls. The Montreal Neurological Institute coordinates and sizes of the clusters are shown in Table 2. L: left; R: right. Regions of gray matter volume change in chronic migraine with or without medication overuse. MNI: Montreal Neurological Institute; CMwMO: chronic migraine with medication overuse; CMwoMO: chronic migraine without medication overuse. Regions of gray matter volume decrease in patients with chronic migraine, without or with medication overuse. MNI: Montreal Neurological Institute; HCs: healthy controls; CMwoMO: chronic migraine without medication overuse; CMwMO: chronic migraine with medication overuse; ITG: inferior temporal gyrus; R: right; L: left.

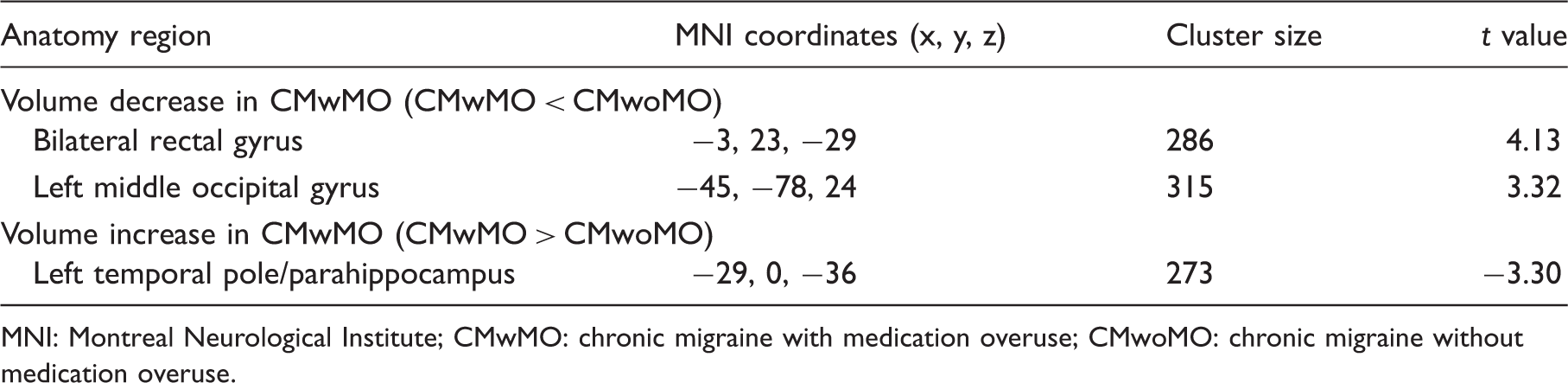
Clinical correlations
To test the clinical significance of the VBM findings, we correlated clinical parameters with the GMV comparison data between the CMwMO and CMwoMO patients. First, the GMV of these brain areas in combination were used to predict the frequency of analgesics use in CMwMO patients. Second, we retrospectively reviewed the medical records within one year after the MRI scans and divided the patients into two groups according to their responses to treatments. A good response was defined as a decrease >50% of either headache or analgesics use frequency at 12 months of treatment that included verbal advice regarding medication withdrawal and preventive and abortive drugs at our clinics (13).
Statistical analyses of clinical data
Statistical analyses were performed using SPSS software, version 17 for Windows (SPSS, Chicago, IL, USA). The demographics and headache profiles were compared among the study groups using one-way analysis of variance (ANOVA) with post-hoc analysis of the least-significant difference (LSD) or a two-sample
Results
Demographics
Demographics and headache profiles of the participants.

CMwMO: chronic migraine with medication overuse; CMwoMO: chronic migraine without medication overuse; HCs: healthy controls; HADS: Hospital Anxiety and Depression Scale;
VBM results on MO and clinical correlations
The CMwMO patients displayed GMV decrease in the bilateral rectal gyrus of the orbitofrontal cortex and left middle occipital gyrus compared with the CMwoMO patients. In contrast, GMV increase in the left temporal pole/parahippocampus was also observed (Figure 1, Table 1). Multiple linear regression showed that GMV of these three areas accounts for 31.1% variance (adjusted
Baseline demographics and headache profiles of patients with chronic migraine and medication overuse, with or without good treatment responses.

HADS: Hospital Anxiety and Depression Scale. Standard deviation in parentheses.
VBM results on CM compared with HCs
The VBM analysis between patients with CM (with and without MO) and HCs showed decreased GMV of multiple brain areas including the frontal, temporal and occipital lobes, precuneus and cerebellum (Table 2, Figure 2).
Discussion
Our study showed that CMwMO patients, compared to CMwoMO patients, had lower GMV in the orbitofrontal cortex and occipital lobe and higher GMV in the temporal pole/parahippocampus. These regions explained the 31.3% variance in analgesics use frequency after controlling for the demographic and clinical variables. The GMV at orbitofrontal cortex was positively correlated with better treatment response at one year.
The strength of this study involves the direct comparisons between the CMwMO and CMwoMO patients. This experimental design highlights the effect of MO by minimizing the effect of migraine on patients with similar clinical conditions. The specific involvement of these brain areas provides a possible pathomechanism basis of MO as a separate headache disorder and not an epiphenomenon of migraine pain (21). Our study is also unique in that the imaging findings were correlated with clinical parameters and prognosis, enabling a more practical interpretation of the data and the possibility of clinical applications.
In an early study involving patients with MO and chronic tension-type headache, instead of migraine, only nonsignificant GMV decreases in the left orbitofrontal cortex and right midbrain were noted (22). Another study including medication-overuse headache (MOH) patients with migraine and HCs showed GMV decreases in the orbitofrontal cortex, anterior cingulate cortex, insula and precuneus and also GMV increases in the periaqueductal gray, thalamus and ventral striatum (11). A recent study explored both functional connectivity and morphology changes in MOH patients with migraine, while EM patients and healthy individuals were recruited as controls (23). In this study, no structural difference was noted in group comparisons; however, negative correlations were identified between the migraine duration and the GMV of the frontal regions, precuneus and hippocampus. Functional studies using either position emission tomography (PET) or functional MRI (fMRI) reported hypofunction of multiple brain areas concerning pain perception and modulation (pain-matrix) in patients with MOH (12,24,25). These changes were often reversible after withdrawal of analgesics (24) though persistent dysfunction has been noted as well (12,25). To the best of our knowledge, no VBM study has made direct comparisons between CMwMO and CMwoMO patients. Further research is needed to confirm the GMV changes specific to MO in migraine and other pain disorders.
In our study, GMV decrease was noted in the bilateral rectal gyrus of the orbitofrontal cortex. Together with the nucleus accumbens and other brain regions, the orbitofrontal cortex is the core of the mesocorticolimbic dopaminergic circuit (also known as the reward system), which has been considered the neurological substrate of drug addiction (7). Previous studies have provided neuropsychological evidence of orbitofrontal cortex dysfunction in MOH such as dependence and impaired decision making (7–10). A PET study in patients with MOH showed hypometabolism of multiple brain areas but only hypometabolism of the orbitofrontal cortex persisted after analgesics withdrawal (12). Another study that followed MOH patients before and after treatment revealed reversible changes of the GMV in the midbrain but not the orbitofrontal cortex (13). Patients without a treatment response showed lower orbitofrontal cortex volumes compared with responders, which supports our results (13). These imaging studies underlie the importance of the orbitofrontal cortex in MO, which is consistent with animal and clinical studies of drug addiction and MOH (7–10). The location of orbitofrontal cortex involvement in our patients is similar to these neuroimaging studies (12,13).
In addition to the orbitofrontal cortex, our results also showed a decrease in GMV in the left middle occipital gyrus and an increase in GMV in the left temporal pole/parahippocampus. The occipital lobe is known to be associated with cortical spreading depression and photophobia in migraine (26,27). Notably, the occipital lobe has been found to participate in the aforementioned dopaminergic reward system (28,29). Changes in functional activity and the decrease in GMV of the occipital lobe have been reported in patients with dependence problems such as cocaine addiction and gambling disorder (30,31). The temporal pole and parahippocampus have been considered parts of the limbic system and pain-matrix (32). Structural involvement of these regions has been noted in patients with EM or CM (without MO) (26,33,34). In patients with MOH, functional connectivity between the hippocampal temporal region and precuneus is higher, and the strength of this functional connectivity is positively correlated with number of pills ingested per month (23). Whether this increased functional connectivity predisposes the GMV increase associated with MO is not currently known. An alternative explanation for our results is that these three brain regions might belong to the same network. Further studies are needed to test this emerging network hypothesis in MO and migraine (35).
The mechanisms underlying the GMV differences associated with MO in our CM patients are not determined. One prominent argument is whether these imaging findings are causes or consequences of the diseases being investigated. As discussed above, the hypometabolism and GMV decrease of the orbitofrontal cortex are persistent while changes in other brain regions are reversible after successful MO treatment (12,13). We hypothesize that the low GMV of the orbitofrontal cortex may be the cause rather than the consequence of MO in our study. The low GMV of the orbitofrontal cortex may indicate atrophy and dysfunction of neurons that lead to impaired decision making and dependence behavior related to MO (7–10). The changes of the middle occipital gyrus and temporal pole/parahippocampus, like many other regions reported in previous studies, may be more likely consequences (12,13). Further studies, particularly longitudinal follow-up ones, are needed to confirm the hypothesis.
Our study also demonstrated decreased GMV in patients with CM (with and without MO), as compared with HCs (Table 2, Figure 2). Similar results have been noted in several studies enrolling mainly EM patients, as well as MOH patients with migraine history (11,13,36). Although the involved areas were not the same among the studies, many of them are considered to take part in different aspects of pain perception or modulation. Reversal of GMV decrease has been reported after successful treatment or spontaneous recovery, suggesting the GMV changes are likely consequences (37–40).
Our study has potential clinical implications. First, our results highlight the importance of the orbitofrontal cortex in CMwMO patients, which is consistent with previous studies (7–10). Intervention targeting the orbitofrontal cortex such as repetitive transcranial magnetic stimulation has been successfully applied in patients with obsessive-compulsive disorder (41). Similar approaches may be tested in patients with MOH. Further studies combining imaging and interventions may help us understand the neuroplasticity and pathogenesis of related headache and pain disorders.
Our study has limitations. First, this study did not provide direct evidence of a volume change along with the transition from without MO to with MO. The evidence could be provided only in a cohort study with longitudinal follow-up. Second, migraine patients with aura were not included; therefore, our results cannot be generalized to this patient group; however, a recent study reported no detectable differences in the brain structure regarding the aura (42). We recruited only migraine patients without aura because most CM patients begin experiencing migraine without aura (43). Third, in evaluation of the treatment responses, the headache and analgesics use frequencies were not separated. This combined outcome may not be ideal since these two factors might not have the same weight. The treatment responses could also be biased because of the variation of preventive treatments. Fourth, we did not collect HADS scores from HCs. Since anxiety and depression have been reported to be associated with structural changes of the brain (44,45), the comparisons between CM patients and HCs (Table 2 and Figure 2) may thus be biased.
Key findings
Structural changes related to medication overuse in patients with chronic migraine included the orbitofrontal cortex, middle occipital gyrus and temporal pole/parahippocampus. These structural changes explained significant variance in analgesics use frequency. The treatment response in patients with medication overuse was associated with gray matter volume in the orbitofrontal cortex.
Footnotes
Acknowledgments
Author contributions are as follows: TH Lai: manuscript preparation, collection of clinical data, statistical analysis; KH Chou: neuroimage analysis and interpretation, statistical analysis; Fuh JL: initiation of the study, recruitment of patients and healthy controls; PL Lee: assistance in neuroimage analysis, preparation of figures; YC Kung: assistance in neuroimage analysis; CP Lin: initiation of the study, monitoring of neuroimage analysis; and SJ Wang: initiation of the study, recruitment of patients and healthy controls, monitoring of manuscript preparation. CP Lin and SJ Wang contributed equally to this work.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported, in part, by grants from the Ministry of Science and Technology (MST; 100-2314-B-010-019-MY2, 100-2314-B-010-018-MY3, 103-2314-B-418-009), Taipei-Veterans General Hospital (V103C-080, VGHUST103-G7-4-1, V103E9-006), MST support for the Center for Dynamical Biomarkers and Translational Medicine, National Central University, Taiwan (NSC 102-2911-I-008-001), Brain Research Center, National Yang-Ming University, and a grant from the Ministry of Education, Aim for the Top University Plan.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
