Abstract
Aims
The aim of this cross-sectional population-based study was to investigate the associations between migraine and type 1 and type 2 diabetes mellitus (DM).
Methods
We used data from the second (1995–1997) and third survey (2006–2008) in the Nord-Trøndelag Health Study. Analyses were made for the 26,121 participants (30–97 years of age, median 58.3 years) with known headache and DM status in both surveys, and for the 39,584 participants in the third survey (20–97 years, median 54.1 years). The diagnosis of migraine was given to those who fulfilled the questionnaire-based migraine diagnosis in the second and/or third survey. Associations were assessed using multiple logistic regression, estimating prevalence odds ratio (OR) with 95% confidence intervals (CIs).
Results
In the multivariate analysis of the 26,121 participants in both surveys, adjusting for age, gender, years of education, and smoking, classical type 1 DM (n = 81) was associated with a lower prevalence of any headache (OR = 0.55, 95% CI 0.34–0.88),and migraine (OR = 0.47, 95% CI 0.26–0.96) compared to those without DM (n = 24,779). Correspondingly, the merged group of classical type 1 DM and latent autoimmune diabetes of adults (LADA) (n = 153) were less likely to have migraine (OR = 0.53, 95% CI 0.31–0.91). Similarly, an inverse relationship between type 1 DM and migraine was found in analyses of 39,584 participants in the third survey. No clear association was found between headache and type 2 DM.
Conclusions
In this cross-sectional population-based study of mainly middle-aged participants, type 1 DM was inversely associated with headache, in particular migraine.
Background
A large number of disorders are comorbid with migraine, but very few conditions are associated with lower migraine prevalence (1). However, an inverse relationship between migraine and blood pressure has been reported (2–5). This finding is probably a consequence of hypertension-related hypalgesia (2,5), illustrating that identification of factors that seem to protect against migraine may suggest clues to the pathophysiology.
An inverse relationship between diabetes mellitus (DM) and migraine has been reported in several studies (6–10). This was the case for overall diabetes mellitus (DM) in some previous studies (6,7), although one of these did not include a control group (6). Later, several population-based studies reported no association between migraine and overall DM (11–13). More recently, studies have reported an inverse association between use of anti-diabetic medications and migraine (7,8). On the other hand, a positive association with migraine was found for non-insulin dependent DM (14). Many studies have investigated risk factors and consequences of type 1 DM and type 2 DM (15,16), but very few previous studies have evaluated the relationship between migraine and specific DM subtypes. Two studies found no association between type 2 DM and migraine (17,18). To our knowledge, only one previous cross-sectional study, which was based on the second wave of the Nord-Trøndelag Health Study (HUNT), has evaluated type 1 DM, reporting an inverse relationship with migraine (10). This is interesting, because it may lead to the identification of protective factors for migraine, and throw light on the pathophysiology.
The present population-based cross-sectional study evaluated the association between migraine and subtypes of DM. The primary aim was to analyze data for those who participated in both the second and third survey in the HUNT study, and the secondary aim was to analyze data for those who participated in the third survey.
Methods
The HUNT study
The HUNT study is a longitudinal cohort study in which all inhabitants ≥ 20 years old in Nord-Trøndelag county, Norway, were invited to participate. Subjects were examined three times; 1984 to 1986 (HUNT1), 1995 to 1997 (HUNT2) (19) and 2006 to 2008 (HUNT3) (20). The two last surveys covered a large number of health-related items in two different questionnaires (Q1 and Q2), and the participants were also invited to clinical consultations, which included non-fasting blood samples and measurements of blood pressure, height and weight.
Headache classification based on HUNT2 and HUNT3
Both HUNT2 and HUNT3 questionnaires included the screening question “Have you suffered from headache during the last 12 months?” Individuals who answered “yes” in HUNT2 and/or HUNT3 were classified as having “any headache”, whereas those who answered “no” in both HUNT2 and HUNT3 were classified as “headache-free”. More details of the headache questionnaire in HUNT2 and HUNT3 are published elsewhere (21–23). Briefly, in HUNT2, active migraine (i.e during the last 12 months) was diagnosed according to a modified version of the first version of the International Classification of Headache Disorders (ICHD-I) (24), and according to the criteria in the International Classification of Headache Disorders, second edition in HUNT3 (25).
Those who fulfilled the diagnostic criteria of active migraine in HUNT2 and/or HUNT3 were classified as having migraine, whereas remaining persons with headache were classified as having “non-migrainous headache” in the analyses of those who participated in both surveys. In the analyses of HUNT3 the merged group of “any headache” consisted of individuals with migraine, tension-type headache (TTH), or unclassified headache (21,23).
The validity of the HUNT2 questionnaire-based diagnoses has been reported previously (22): For headache, the sensitivity was 85% and specificity 83% (kappa value 0.57); for migraine, the sensitivity was 69% and specificity 89% (kappa value 0.59); and for non-migraineurs, the sensitivity was 61%, and specificity 81% (kappa 0.43). The validity of the HUNT3 questionnaire-based diagnoses has also been reported previously (23); for any headache, the sensitivity was 88 %, and specificity 86 % (kappa value at 0.70, 95 % CI 0.61–0.79); for migraine the sensitivity was 51 %, and specificity 95 % (kappa value 0.50, 95 % CI 0.32–0.68), and for TTH ≥1 days/month the sensitivity was 96% and specificity was 69% (kappa value 0.44, 95% CI 0.30–0.58).
Classification of diabetes
At HUNT2 and HUNT3, participants were asked about any history of diabetes. Participants with a positive answer to the question “Do you have or have you had diabetes?” in Q1 (self-reported DM) reported their age at diagnosis. The self-reported diagnosis of DM has been shown to have good validity (26). In the analyses of those who participated in both surveys, individuals defined as being “without DM” confirmed this status by answering “no” to the screening question both in HUNT2 and HUNT3. In the analyses in HUNT3, those “without DM” answered “no” to the screening question in HUNT3.
Participants declaring DM were invited to a follow-up investigation in the fasting state. This had a response rate of 74% in HUNT2 and 53% in HUNT3 (24). At follow-up, participants were interviewed by nurses to confirm their age at diagnosis and details on the time-point of beginning different types of diabetes treatment (16). They provided a fasting blood sample for measurements of glucose, glycosylated hemoglobin (HbA1c), serum C-peptide levels, and antibodies against glutamic acid decarboxylase (GADA) (27). In those not attending this follow-up investigation, GADA was analyzed in serum from a non-fasting state. Thus, GADA was measured in all with self-reported DM.
In the present study, the classification of DM subtypes was based on information about age of onset, diabetes medication and measurements of C-peptide and GADA in line with the definition used in previous studies (e.g. 27–31). For those not attending the follow-up, the classification of DM subtypes was based on GADA and time for the start of insulin treatment. Antibody levels were expressed as an antibody index relative to a standard serum. An index of ≥ 0.08 was considered positive. The cutoff value was used to achieve the highest possible specificity (100%) with an acceptable corresponding sensitivity (64%) (27). Patients starting insulin treatment within one year of DM diagnosis were classified as having classical type 1 DM if, in addition, they were GADA positive or had fasting C-peptide levels <150 pmol/l (27). Patients were classified as having latent autoimmune diabetes in adults (LADA) if they were GADA positive and had not been treated with insulin within one year of diagnosis. Type 2 DM was defined by being GADA negative and if currently using insulin, having had no such treatment within the first year of diagnosis (27,30). The unclassified group consisted of patients with incomplete information on insulin treatment and results of C-peptide and GADA (29).
Study participants
In HUNT2, 65,237 persons (70%) participated out of 93,898 invited, and 51,856 (56%) of them indicated whether they suffered from headache or not. In HUNT3, 93,860 persons were invited, of whom 50,807 (54%) participated, and 39,690 of whom (42%) answered the headache questions. Among the 65,237 persons who participated in HUNT2, 8,545 had died and 4,357 had moved out of the county during the period before HUNT3.
The present study was partly based on 37,071 persons who participated in both HUNT2 and HUNT3. Among them, 26,121 participants (30–97 years of age, median 58.3 years) were included in the present study because they had answered questions regarding DM and headache in both HUNT2 and HUNT3 (Figure 1). We also evaluated the relationship between DM and headache among the 39,584 participants who answered questions regarding DM and headache in HUNT3 (20–97 years of age, median 54.1 years).
The flow of participants in HUNT2 and HUNT3.
Potential confounders
A large number of health-related data were assessed, including factors associated with headache and migraine (32) as well as DM (28,30,31). The following variables available at HUNT3 were examined as potential confounders: age (continuous variable) (28,32); sex (32), years of education in HUNT2 (≤9, 10–12 and ≥13 years) (32); employment status in HUNT3, body mass index (BMI) (continuous variable) (28,32); smoking status (three categories; current daily smoking, previously daily smoking and never daily smoking) (32,30); frequency of physical activity (five categories; never, less than weekly, once a week, 2–3 times per week, daily) (28,32), alcohol consumption (five categories) (30); anxiety and depression as measured by the total Hospital Anxiety and Depression Scale (HADS) (continuous variable) (32,31); and systolic blood pressure (continuous variable) (32).
Ethics
This study was approved by the Regional Committee for Ethics in Medical Research, and the HUNT study was in addition approved by the Norwegian Data Inspectorate.
Statistical analysis
In the multivariate analyses, using logistic regression, we estimated the prevalence odds ratio (OR) with 95% confidence interval (CI) for the association between types of headache and types of DM. In analyses of participants in both surveys, individuals without DM in HUNT2 and HUNT3 were used as reference. In the analyses of HUNT3, persons who did not report DM in HUNT3 were used as reference. Initially, we made separate analyses for classical type 1 DM and LADA. Because LADA can be classified as type 1 DM, we also performed additional analyses for the merged group of individuals with classical type 1 DM or LADA.
We initially adjusted for age and sex, and subsequently for predefined confounding factors. To determine which factor contributed most to the effect on the adjustment, analyses were carried out with adjustment for each separate factor in addition to age and sex. In addition, multiple factors were also tested together. We excluded factors from the final model if they did not change the OR at all or just changed it marginally (≤ 0.01) when evaluating each factor separately or by including several factors grouped together. Potential interaction between two variables was evaluated by including the product of the variable in the logistic regression analyses, and the interaction was tested using Wald χ2 statistics. In the final analyses of participants in both surveys, we adjusted for age, gender, years of education, and smoking as potential confounders. Correspondingly, in the final analyses of HUNT3, we adjusted for age, gender, BMI, and smoking as potential confounders. Other factors such as alcohol consumption, total HADS score, previous myocardial infarction, and mean systolic blood pressure were also evaluated, but were excluded from the final analyses because they changed the OR by less than 0.01. Participants with incomplete data for one or several variables were included (as a separate missing category) in all analyses to reduce the impact of response bias. Based on the findings in one of our previous studies (7), we stratified analyses by age (< or ≥ 50 years), and by duration of DM (< or ≥ 2 years).
Data analyses were performed with the IBM Statistical Package for the Social Sciences, version 22 (SPSS, Chicago, Illinois, USA)
Results
Prevalence of DM among participants in HUNT2 and HUNT3
Participants in HUNT2 and HUNT3. Background data measured at HUNT3 on persons without and with known diabetes mellitus (DM) (classical type 1, type 2, LADA, and unclassified).
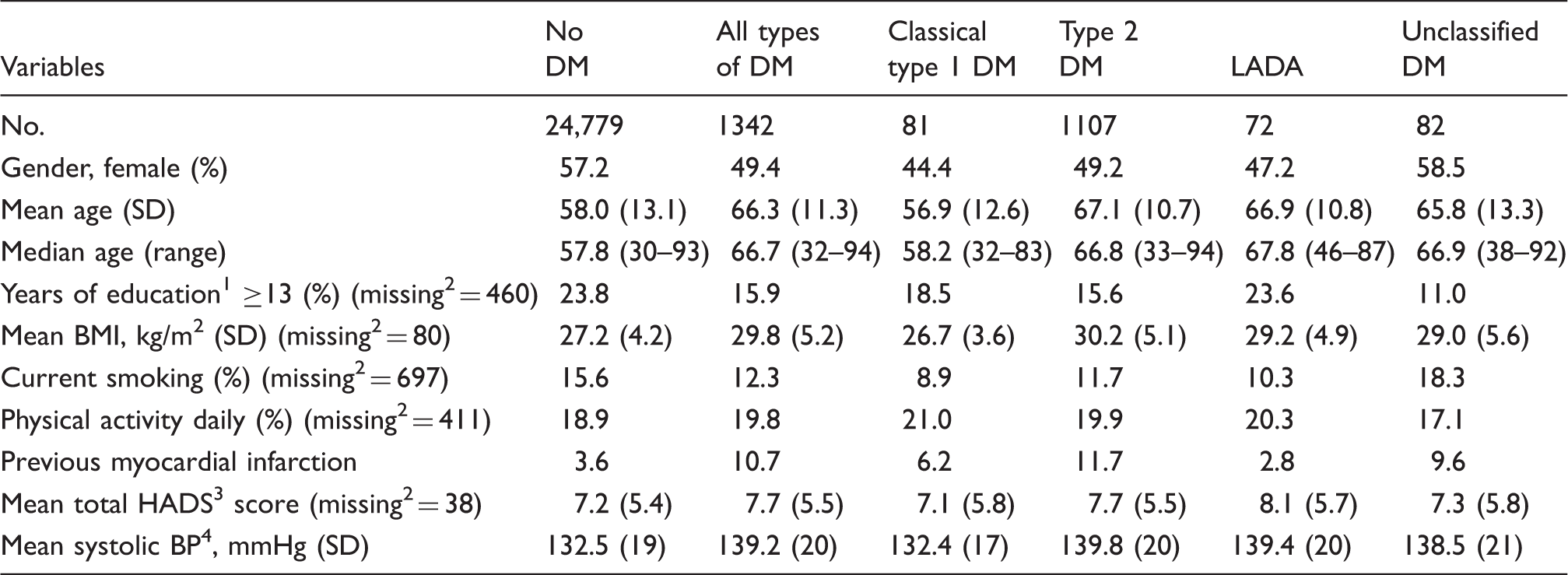
Years of education reported in HUNT2.
Missing values are given as number of individuals when not zero.
HADS = Hospital Anxiety and Depression Scale.
BP = blood pressure.
Prevalence of headache
13,209 (50.6%) out of our 26,121 participants suffered from headache in HUNT2 and/or HUNT3. Among these, 5,209 had migraine (19.9%) (30–97 years of age, median 53.7 years). The remaining group of 8,000 (30.6%) (30–97 years of age, median 56.2 years) accordingly had non-migrainous headache.
The association between headache and DM
Participants in HUNT2 and HUNT3. Prevalence odds ratio (OR) with 95% CI of types of headache related to types of diabetes mellitus (DM).

Adjusted for age and sex.
Adjusted for age, sex, years of education and smoking.
Prevalence of DM among participants in HUNT3
Participants in HUNT3. Background data measured at HUNT3 on persons without and with known diabetes mellitus (DM) (classical type 1, type 2, LADA, and unclassified).

Missing values are given as number of individuals when not zero.
HADS = Hospital Anxiety and Depression Scale.
BP = blood pressure.
Prevalence of headache in HUNT3
A total of 18,727 (47.3%) out of 39,584 HUNT3 participants had reported headache, of whom 7,327 had migraine (18.5%, 20–91 years of age, median 50.2 years), 5,420 had TTH (13.7%, 20–97 years of age, median 47.8 years), and 5,980 had unclassified headache (15.1%, 20–96 years of age, median 56.2 years).
The association between headache and DM in HUNT3
Participants in HUNT3. Prevalence odds ratio (OR) with 95% CI of types of headache related to types of diabetes mellitus (DM).

Adjusted for age and sex.
Adjusted for age, sex, body mass index and smoking.
Discussion
In this cross-sectional population-based study utilizing data from two health surveys, type 1 DM was inversely associated with any headache and migraine.
Comparison with other studies
The present results are in accordance with our study based solely on data from HUNT2 (10). However, in contrast to previous results (9,10), of which one study was performed in the HUNT2 study population (10), we could not confirm any clear association between migraine and type 2 DM. In a prescription-based study from Norway, migraine was less likely (sex and age-adjusted OR = 0.76, 95% CI 0.71–0.81) among persons using oral diabetes medication alone (9). The results were only evident among those aged 50 years or more (9), as were the majority of participants in our study. However, in spite of the fact that the statistical power was much better for type 2 DM than type 1 DM, we could not confirm the results from the prescription-based study regarding type 2 DM.
In accordance with our result, no association between migraine and type 2 DM was found in prospective analyses from the large-scale Women’s Health Study (n = 38,620 women) performed in the USA (18) and a case-control study (n = 297) from Iran (17).
The present study replicated an inverse relationship between migraine and type 1 DM previously reported in a cross-sectional study based solely on data from HUNT2 (10). In contrast to our previous study (10), the current reference group in analyses of participants in both surveys consisted of persons who had been confirmed not to have DM in both surveys. Similarly, headache-free individuals had confirmed being without headache complaints in both surveys. Thus, the possibility of misclassification was minimized, i.e. erroneous inclusion of DM patients among those without DM, and headache sufferers among those without headache. In this cohort of participants in two different surveys, we considered investigating the migraine incidence in those who had either type 1 DM or type 2 DM. However, this was not done owing to the low statistical power for classical type 1 DM. Instead, we made supplementary analyses based on participants in HUNT3.
It should also be highlighted that the analyses of participants in both surveys was mainly based on participants who developed DM after the previous HUNT2 study. Thus, only 22% of those with DM (330 out of 1499 patients with DM in HUNT2) included in our previous study (10) were also participants in the present study. In this way, the present results were generated on a mainly new group of patients with DM. Furthermore, in contrast to the previous cross-sectional study (10), individuals without DM and without headache confirmed this status in two surveys.
Interpretation
The inverse relationship between migraine and type 1 DM may indicate that in some way DM protects against migraine, or vice versa. In a cross-sectional design, the direction of causality cannot be determined with certainty. However, the present results may be of relevance for the ongoing debate about migraine pathophysiology (33).
One possible explanation is that macrovascular complications of classical type 1 DM causing intimal thickening and stiffness of blood vessels (34) may be protective for migraine. If true, vascular reactivity may be more involved in the migraine pathophysiology than recently suggested (33). On the other hand, microvascular alterations occurring in type 1 DM causing diabetic neuropathy (34) may also be involved, causing less pain as well as reduced cerebrovascular reactivity (35). Furthermore, we cannot rule out the possibility that insulin treatment in type 1 DM may play a role. For example, one case-control study reported altered insulin metabolism in migraine patients compared to controls, suggesting that insulin is involved in the pathogenesis of migraine (36). The present results may be explained by the highly regulated lifestyle among type 1 DM patients, but adjusting the analyses for lifestyle factors such as physical activity, smoking, and use of alcohol did not change the results. Finally, one may not exclude that there might be underlying shared genetic factors that account for the observed association between type 1 DM and migraine (37,38). It is unclear why the inverse relationship with migraine was present for type 1 DM and not type 2 DM. However, we may suggest that some unmeasured factor may be involved, because different genetic and environmental factors are involved in the etiology of type 1 and type 2 DM (15,16). To our knowledge, migraine does not have the same environmental triggers as type 1 DM, whose triggers may include infections and toxins (15).
Strengths and limitations of the study
The major strengths of this study are the population-based design and the use of validated diagnoses of headache (22,23). The classification of DM was based on interview-based information about age of onset, diabetes medication and C-peptide and GADA measurements, in line with the established definition used in many previous studies (e.g. 27–31). In contrast to the majority of other studies, we were able to perform separate analyses for types of DM. As mentioned above, combining data from two health surveys made it possible to compare migraine patients with persons who had been confirmed to be headache-free 11 years apart as controls. Similarly, it was possible to compare DM patients with persons who have been confirmed not to have DM in both surveys. In the multivariate analyses, we were able to adjust for a large number of potential confounding factors, but the possibility of residual confounding by an unrecognized factor cannot be ruled out.
Several study limitations should also be considered. Firstly, the cross-sectional design does not permit any conclusions about causality. Secondly, because few participants with classical type 1 DM had migraine (10 out of 81), incidental findings cannot be ruled out. However, we also observed a lower prevalence of migraine in the combined group of classical type 1 DM and LADA, which included 17 participants with migraine (out of 153). Furthermore, the inverse relationship between classical type 1 DM and migraine was also found in analyses based on HUNT3 participants. Thirdly, for several reasons, generalization of results to the whole population must be made with caution. The majority of included participants were middle-aged, and our results may not be valid for individuals below 30 years of age. Furthermore, only 50% of those who answered headache questions in HUNT2 were included in the present study (61% of those eligible for HUNT3), and we cannot be certain that dropouts have occurred at random. A possibility for selection bias regarding DM in HUNT3 should not be ignored, partly because people with DM were less likely to participate in HUNT3 (39), and partly because people with DM were less likely to survive from HUNT2 until participation in HUNT3 due to higher age-increased mortality in individuals with DM (40). The influence of such selection bias on our results is difficult to predict. The prevalence of headache and self-reported migraine was stable in HUNT2 and HUNT3 (21), and no consistent differences in prevalence of headache and self-reported migraine were found between participants and non-participants in HUNT3 (39), suggesting that headache did not substantially influence participation in HUNT3.
Conclusions
In this cross-sectional population-based study, an inverse relationship between type 1 DM and migraine was found. The present results are of relevance in the ongoing discussion of migraine pathophysiology.
Footnotes
Clinical implications
Type 1 diabetes mellitus was inversely associated with any headache and migraine.
No association was found between type 2 diabetes mellitus and headache.
The present results may be of relevance for the ongoing debate about migraine pathophysiology.
Acknowledgements
The Nord-Trøndelag Health Study (The HUNT study) is a collaboration between the HUNT Research Centre, Faculty of Medicine, the Norwegian University of Science and Technology (NTNU); Norwegian Institute of Public Health; and the Nord-Trøndelag County Council.
