Abstract
Background
The impact of adverse events (AEs) of antiepileptic drugs (AEDs) have an impact on compliance and dropouts. We compared tolerability of AEs of AEDs among patients with migraine, epilepsy, or both.
Methods
Overall, 335 patients (epilepsy (n = 142), migraine (n = 131), and both (n = 62)), were evaluated with the Liverpool Adverse Events Profile (LAEP) to assess the magnitude, profile and occurrence rate of the AEs of valproate, topiramate, and lamotrigine.
Results
AEs were significantly more common with topiramate treatment (71.0%) and among migraineurs (69.5%), the latter being more prone to discontinue AEDs (46.6%). The profile of AEs with topiramate and valproate differed among groups. Moreover, treatment with both topiramate and valproate was associated, for all groups, with a worse tolerability profile compared to lamotrigine.
Conclusion
Our data suggest a specific drug and disease AE profile of AEDs. Specifically, migraineurs are the most affected by AEs, even though they receive very low dosages of AEDs. This finding might be considered a clinical implication of central sensitization mechanisms. Both the profile and tolerability of AEs, highly influencing quality of life, depended on the underlying conditions, and deeply impacted on treatment dropout. Therefore, before starting, switching or stopping AED treatment, all options need to be considered.
Background
Migraine and epilepsy are chronic nervous system disorders generally manifesting with recurrent or episodic neurological dysfunction, with a return to the symptom-free baseline status between attacks (1). Both migraine and epilepsy are frequent in adults, affecting, in a lifetime, from 6–30% and from 5–15% of the general population respectively (2–4). Given that they share so many risk factors and pathophysiological mechanisms (5,6), several antiepileptic drugs (AEDs), which had already been widely used for epilepsy treatment, began to be prescribed for migraine prevention (7–9). Among them, valproic acid (VPA) and topiramate (TPM) are highly effective in migraine prophylaxis, while lamotrigine (LTG) efficacy is still debated, with a recent meta-analysis by Jackson and colleagues reporting equivalency of LTG and TPM in migraine prophylaxis (10–21). With regional variation in prevalence, TPM, LTG and VPA are to date among the most frequently used AEDs, the latter being used with caution among women of childbearing potential according to the reported teratogenic effects and decreased IQ scores in children of mothers taking VPA (8,9).
VPA, TPM and LTG have a specific profile of adverse events (AEs): nausea, hair loss, tremor and weight gain are the most frequent for VPA (13,22,23), cognitive dysfunction, paresthesia, weight loss and language disturbances for TPM (15,24), and skin rash for LTG (25). Negatively impacting quality of life, morbidity and mortality, AEs associated with AEDs are hurdles to surmount in achieving both patient compliance and disease control (26,27). It has been reported that migraineurs have a higher prevalence of AEs with TPM use, compared to patients with epilepsy (28). However, in clinical practice, differences in reported AEs seem to emerge with AEDs other than TPM. This observational study sought to assess the differences of AEs associated with TPM, VPA and LTG among patients with migraine, epilepsy, and both, by directly comparing AEDs’ AE tolerability among different disease groups. Having detailed and compared tolerability profiles of LTG, VPA and TPM should assist clinicians in making decisions on the best treatment options for their patients.
Methods
Setting
We performed an open, comparative, observational study to analyse the AE profiles of valproic acid (VPA), topiramate (TPM) and lamotrigine (LTG) (herein referred to as AEDs) of patients with migraine, epilepsy, or both. The study protocol was approved by our internal advisory board. Written informed consent was obtained from all participants in the study. All data were collected as a part of the routine care, since all patients during follow-up usually complete the Liverpool Adverse Events Profile (LAEP) questionnaire (29–31) (Supplementary Figure e-1), whose Italian version was previously developed according to a standardized protocol and tested, showing very high reliability (Chronbach’s alpha 0.846 95% CI = 0.826–0.863).
Participants
Eligibility criteria included: 1) an established diagnosis of epilepsy, according to the International League Against Epilepsy (ILAE) criteria (32), or migraine, according to the International Classification of Headache Disorders (ICHD) III beta criteria (33), or both; 2) at least six weeks of AED monotherapy; 3) no previous AED treatment; 4) age ≥18 years; 5) the ability to respond to the LAEP questionnaire. Exclusion criteria included: 1) no response to treatment; 2) concomitant use of other prophylactic migraine and epilepsy drugs; 3) a history of psychiatric disorders or the use of anti-depressant drugs over the last six months; 4) previous AED treatment. All patients were recruited and followed at the Headache outpatient center and at the Epilepsy outpatient center of the Neurology Clinic. All patients, naïve for AED, were started on AEDs following standard titration schemes. There were three patient groups: E, with epilepsy (n = 131, 54% female); M, with migraine (n = 142, 82% female); ME, with both migraine and epilepsy (n = 62, 68% male). Among the patients in group ME, AED treatment was started and titrated to primarily achieve seizure control.
Interview
We interviewed patients regarding any possible AEs during their AED use over the previous four weeks. AEs were identified through the LAEP, a self-completed questionnaire consisting of 20 items (30). Patients were asked to quantify each AE on a 1–4 point scale, where 1 = never a problem; 2 = rarely a problem; 3 = sometimes a problem; and 4 = always or often a problem (30). In order to better define the item “any other problems”, we evaluated six additional AEs frequently reported during follow-up: Paresthesia, hematological abnormalities, sexual disorders, kidney stones, weight loss and language disorders. Overall scores ranged from 25 to 100. For statistical analysis, an AE was considered present when it was rated higher than two. The efficacy of AED treatment, which was comparable among all groups, was evaluated through clinical diaries, and assessed according to the International Headache Society (IHS) and the ILAE guidelines (33,34).
Analysis and statistical methods
Demographic data and clinical features of the study cohort.

A = ANOVA, K = Kruskal-Wallis test, C = Chi-square test, F = Fisher’s Exact test; GE = genetic epilepsy; M = male; F = female; SE = structural epilepsies; UE = unknown epilepsy. CM- = Chronic migraine without medication overuse; EM = Episodic Migraine; CM+ = Chronic migraine with medication overuse. LTG = lamotrigine; TPM = topiramate; VPA = valproate; ns = non significant. Data expressed as mean ± standard deviation and ranges.
Results
Characteristics of the study cohorts
Overall, 341 patients were recruited from the Headache and Epilepsy Center at the Neurology Clinic of Perugia, Italy. Six patients were excluded: five withdrew their informed consent after enrollment, one was diagnosed with generalized anxiety disorder (Supplementary Figure e-2). No significant differences regarding demographic characteristics were observed between participants and those excluded.
Among the 335 patients enrolled, 142 had epilepsy (group E), 131 had migraine (group M), and 62 had both conditions together (group ME). Regarding epilepsy diagnosis, patients with unknown epilepsy prevailed both in group E and ME, followed by structural epilepsy and genetic epilepsy. Episodic migraine was the most common migraine diagnosis both in groups M and ME, while chronic migraine with or without drug abuse was more common in group M than in group ME. Mean age was 45 in groups E and M, 41 in group ME. Gender prevalence in the three groups reflects population-based diseases prevalence. Among the investigated drugs, the most commonly prescribed were VPA for group E (47.9%), TPM for group M (68.7%) and LTG for group ME (38.7%). AED dosage differed among different groups, with patients in group E and ME receiving on average a higher dosage of each AED compared to patients in group M (Table 1). No significant differences concerning treatment efficacy were observed among groups.
Adverse events
Drug Adverse Events, drugs vs groups.
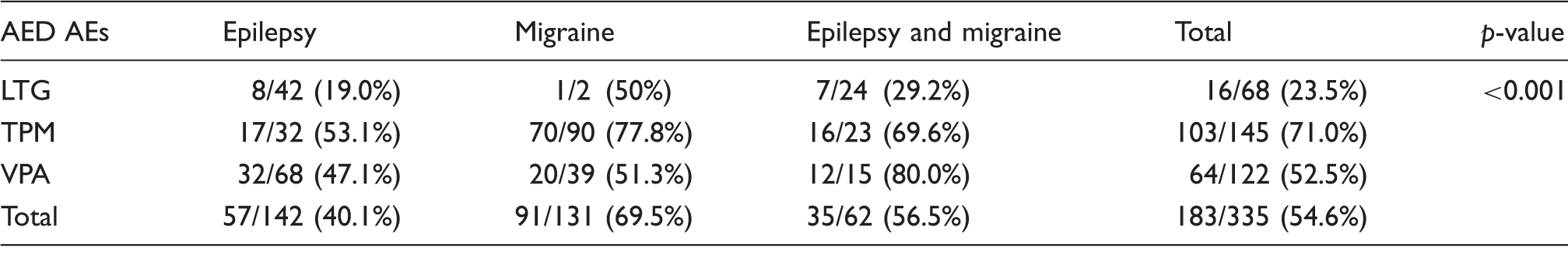
AED = antiepileptic drugs; AEs = adverse events; LTG = lamotrigine; TPM = topiramate; VPA = valproate
Multivariate logistic regressions modelling the occurrence of AEs.

LTG = lamotrigine; TPM = topiramate; VPA = valproate
Drug and group specific AEs spectrum
Multivariate analysis of AE occurrences revealed qualitative and quantitative differences in reported AEs depending on disease and AED treatment (Figure 1). For this analysis, we evaluated AEs with LAEP items ≥2 for all subgroups. Patients in group E reported more tiredness than those in group M, where the latter group reported more unsteadiness, hair loss, upset stomach and paresthesia. Group ME had more unsteadiness, nervousness/agitation, weight gain and paresthesia compared to group E. Group ME had the highest prevalence of hand tremor. No significant intra-group differences were noted depending on specific epilepsy or migraine diagnosis.
Multivariate logistic regressions of adverse events (AEs) among groups (Gr).
Among AEDs, TPM and VPA resulted in having a worse AE profile than LTG. In particular, regardless of the group, patients on TPM had significantly more unsteadiness, anger, nervousness, sleepiness, and paresthesia than those on LTG. Like TPM, VPA had a worse AE profile than LTG, especially regarding nervousness, shaky hands, and weight gain. Though both TPM and VPA significantly manifested worse LAEP scores when compared to LTG, they had their own AE spectrums. Specifically, TPM caused upset stomach, shaky hands and weight gain less frequently than VPA, while it significantly exceeded VPA in causing depression, paresthesia, weight loss and language disturbances. When comparing AEs with LAEP scores over two among AEDs used in each group, significant differences in AE profiles were observed (Figure 2). That is, for group M, upset stomach and weight gain were significantly more frequent with VPA treatment, while paresthesias and language disorders were both more frequently reported with TPM treatment. Likewise, paresthesias and language disturbances were also significantly more prevalent in group ME. In the same group, significant differences were found between VPA and LTG groups for nervousness, hand tremor and weight gain, all of which were highly prevailing in the former group.
Odds ratio (OR) of Liverpool Adverse Events Profile (LAEP) scores higher than 2 among groups depending on treatment.
Dropouts
Dropouts from AED treatment.

LTG = lamotrigine; TPM = topiramate; VPA = valproate
Discussion
AEs associated with AEDs negatively impact quality of life, disability morbidity and mortality (27). This study used a patient-completed questionnaire to define the profile, frequency and magnitude of AEs associated with TPM, LTG and VPA among patients suffering from migraine, epilepsy, or both. The findings evidenced quantitative and qualitative differences for AEs associated with disease and AEDs among the three patient groups. Specifically, a higher prevalence of AEs due to AEDs was found for migraineurs, even though these patients were treated with the lowest dosage of AEDs. The entire cohort received AEDs following international guidelines for migraine and epilepsy treatment (32,33), and was negative for previous psychiatric conditions (35). Thus, since the higher prevalence of AEs in group M emerged despite the lowest dosages of AEDs, our results suggest that migraineurs were the most susceptible to AEs. This finding might be a clinical implication of central sensitization mechanisms, or the result of an abnormal network plasticity in the migraine-affected brain (36). In particular, given that migraine involves various cortico-subcortical circuits implicated in the transmission and emotional processing of pain, it could represent a substrate triggering AEs, as already suggested for language disturbances due to TPM (24). Moreover, since language areas are also known to influence the emotional elaboration of pain during migraine attacks, a remapping of this cerebral network in a “more-vulnerable” state might account for the higher prevalence of language disturbances and other AEs among migraineurs (36–38).
Among the investigated AEDs, TPM was the drug with the highest prevalence of AEs, especially among migraineurs, in line with previous reports (28,39,40). In particular, a focused meta-analysis on TPM use by Luykx and colleagues reported that migraineurs experience different AEs compared to patients with epilepsy, and are more likely to drop out of TPM because of such AEs (28). Moreover, migraine has been associated with a higher risk of specific AEs such as TPM-induced paresthesia (39). The influence that diseases can have on AEs associated with TPM has also been confirmed by a systematic review of placebo multiple-indications controlled trials on TPM (40). However, to date, no study has addressed AEs associated with AEDs other than TPM among patients with migraine or epilepsy, and no study has ever examined the tolerability of AEDs among patients with both migraine and epilepsy. Our results come from the first-ever study to directly compare patients with migraine, epilepsy or both. These results strongly suggest that the disease for which AEDs are prescribed does in fact influence AEs, not only for TPM, but also for VPA and LTG. Thus, clinicians need to consider these conditions to tailor AED treatment to their patients.
Besides migraineurs being more sensitive to AEs, they were also more likely to discontinue AED treatment due to AEs, in line with previous reports (28). At the same time, M and ME groups tended to discontinue TPM treatment (more than with VPA or LTG), even though the AED most frequently causing AEs was VPM. These results might be influenced by AE profile. In particular, migraineurs reported vastly different profiles of AEs between VPA and TPM, with paresthesias and language disturbances more often associated with the latter. The consistent negative impact of these AEs on quality of life most likely explains why many patients decided to discontinue TPM treatment. Conversely, the overlapping profile of AEs associated with VPA and TPM in the ME group might explain their similar dropout rates. Thus, the AE profile of each AED seems to vary depending on what ails patients, and therein directly influences dropout rates. Being so, clinicians should routinely monitor AEs and consider switching treatment depending on the risk of dropout and the impact of AEs on a patient’s quality of life. Accurate tailoring of AEDs to the patient remains pivotal to obtaining compliance, low dropouts and, most of all, disease control.
When AEs were measured through LAEP, TPM, VPA and LTG unveiled diverse AEs profiles. Specifically, the prevalence and magnitudes of AEs depended on the prescribed drugs as well as on the underlying disease for which they were used. The findings of this study indicate that migraineurs are more likely to both experience AEs and drop out from treatment. Since AEs of AED negatively influence quality of life, compliance and dropout rate (43), our finding that the underlying disease influences the AE profile leads us to recommend consideration of all options before starting, switching or withdrawing AED treatment.
Strengths and shortcomings of the study
The present study is the first to compare the tolerability of different AEDs in a consistent cohort of patients with epilepsy, migraine or both. The appropriate and strict inclusion criteria allowed the comparison of diverse AEDs in different settings, with a reproducible protocol assessing AEs through a widely used questionnaire. Prevalence, magnitude and profile of AEs associated with AEDs were collected together with clinical and demographic data. Direct comparison between different cohorts as well as different treatments allowed a focused analysis on differences in tolerability and their impact on dropouts. Sample selection bias is unavoidable when selecting a specific cohort among the general population or among the large cohort of patients referred to third level centres dedicated to migraine and epilepsy. Thus, the high comorbidity between migraine and epilepsy is the result of a selective inclusion, not representing population-based comorbidity of such disorders. Limitations to the study might also be identified in the possible under-reporting of AEs, especially among patients with epilepsy, which might be concerned about seizure occurrence in case of a treatment switch. A physician dedicated to informing patients and programmed strict follow-up has been implemented to reduce the above-mentioned issue (41,42). Further shortcomings, such as over-reporting due to anxiety or depression disorders (35,42), have been limited using strict inclusion criteria, excluding patients with past or present psychiatric comorbidity, and continuing monitoring during the study to assess any variation in their mood. Nonetheless, the influence of anxiety disorders on adverse event reporting should be considered when translating the results of our study into clinical practice, since patients with such disorders might emphasize or over-report AEs.
Clinical implications
Adverse events of antiepileptic drugs are the main hurdles to surmount in achieving patient compliance and disease control both in epilepsy and migraine. The profile, magnitude and prevalence of adverse events of valproic acid, topiramate and lamotrigine vary depending on the disease, with migraineurs showing a very high susceptibility. Topiramate seems to carry a higher risk of adverse events among migraineurs compared to lamotrigine and valproate. Lamotrigine was associated with low prevalence and magnitude of adverse events in patients with epilepsy, migraine or both. Clinicians should therefore consider all options when prescribing antiepileptic drugs for migraine prophylaxis and epilepsy.
Footnotes
Acknowledgements
The authors would like to thank all the patients and their parents/guardians.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
