Abstract
Introduction
Migraine headache is a neurological disorder whose attacks are associated with nausea, vomiting, photophobia and phonophobia. Treatments for migraine aim to either prevent attacks before they have started or relieve attacks (abort) after onset of symptoms and range from complementary therapies to pharmacological interventions. A number of treatment-related adverse events such as somnolence, fatigue, and chest discomfort have previously been reported in association with triptans. The comparative tolerability of available agents for the abortive treatment of migraine attacks has not yet been systematically reviewed and quantified.
Methods
We performed a systematic literature review and Bayesian network meta-analysis for comparative tolerability of treatments for migraine. The literature search targeted all randomized controlled trials evaluating oral abortive treatments for acute migraine over a range of available doses in adults. The primary outcomes of interest were any adverse event, treatment-related adverse events, and serious adverse events. Secondary outcomes were fatigue, dizziness, chest discomfort, somnolence, nausea, and vomiting.
Results
Our search yielded 141 trials covering 15 distinct treatments. Of the triptans, sumatriptan, eletriptan, rizatriptan, zolmitriptan, and the combination treatment of sumatriptan and naproxen were associated with a statistically significant increase in odds of any adverse event or a treatment-related adverse event occurring compared with placebo. Of the non-triptans, only acetaminophen was associated with a statistically significant increase in odds of an adverse event occurring when compared with placebo. Overall, triptans were not associated with increased odds of serious adverse events occurring and the same was the case for non-triptans. For the secondary outcomes, with the exception of vomiting, all triptans except for almotriptan and frovatriptan were significantly associated with increased risk for all outcomes. Almotriptan was significantly associated with an increased risk of vomiting, whereas all other triptans yielded non-significant lower odds compared with placebo. Generally, the non-triptans were not associated with decreased tolerability for the secondary outcomes.
Discussion
In summary, triptans were associated with higher odds of any adverse event or a treatment-related adverse event occurring when compared to placebo and non-triptans. Non-significant results for non-triptans indicate that these treatments are comparable with one another and placebo regarding tolerability outcomes.
Keywords
Introduction
Migraine is a chronic neurological disorder with a 1-year period prevalence of 18% of women and 5% of men in developed countries (1). Migraine attacks generally last between 4 and 72 hours. Migraine, with its associated symptoms including nausea, vomiting, photophobia, and phonophobia, has a substantial impact on quality of life and health-related resource utilization (2,3).
Treatments for migraine are taken either for prevention of attacks or to treat acute attacks. Abortive treatments include triptans, non-steroidal anti-inflammatory drugs (NSAIDs), and ergotamines (4,5). Triptans and ergotamines are generally known to provide better alleviation of migraine symptoms compared with NSAIDs (6). Furthermore, within the class of triptans, some drugs have been demonstrated to exhibit somewhat better population efficacy than others (6).
A number of treatment-related adverse events (TRAEs) such as somnolence, fatigue, and chest discomfort have previously been reported in association with triptans (7). Also, a number of adverse events (AEs) have also been reported in association with ergotamines and cyclooxygenase-2 (COX-2) inhibitors, such as medication withdrawal headache, nausea and vomiting (8,9). While these reports suggest that important efficacy–tolerability trade-offs may exist, the comparative tolerability of available agents for the treatment of migraine attacks has not yet been systematically reviewed and quantified.
Network meta-analysis (NMA) is an approach to assess comparative tolerability of interventions that have not all been assessed in head-to-head-trials (10,11). This study therefore aimed to determine the comparative tolerability of treatments for migraine headaches using NMA.
Methods
All systematic literature review and NMA methods followed the recent extension of the PRISMA guidelines for NMA (12).
Systematic review
Eligibility criteria
The PICOS (population, interventions, comparisons, outcomes, study design) presented in Supplementary material – Appendix A outline the eligibility criteria used for the selection of trials for inclusion in the NMA. The literature search targeted all randomized controlled trials evaluating oral abortive treatments for acute migraine in adults over the age of 18 that met the inclusion criteria (Supplementary material – Appendix A). This review focused solely on oral treatments due to the difference in tolerability profiles associated with subcutaneous injection. Treatments of interest included triptans, NSAIDs, and ergotamines, in any combination with or without caffeine or barbiturates.
Literature search
A comprehensive search algorithm was developed and employed within the major medical databases (MEDLINE, EMBASE, and the Cochrane Controlled Trials Register). Databases were searched from inception to week 3, 2014 (January 13–19). The search strategy is presented in Supplementary material – Appendix B. Published systematic review bibliographies were additionally searched for relevant studies. Furthermore, clinicaltrials.gov and pharmaceutical websites were searched to identify potentially eligible trials that had produced final results but were not yet published in a peer-reviewed publication format.
Data extraction
Data were extracted on each outcome of interest. Primary outcomes included any AEs, TRAEs, and serious adverse events (SAEs) (outcome definitions were left at the discretion of the RCT authors). Secondary outcomes included fatigue, dizziness, chest discomfort, somnolence, nausea, and vomiting. The secondary outcomes have all previously been reported as treatment-related outcomes, and thus were found relevant. Nausea and vomiting may also be directly linked to the effectiveness of the treatment, as they are commonly associated with migraine. Treatment characteristics (such as drug and dosage) and baseline patient characteristics (such as criteria used to diagnose migraine, history of migraine, whether concomitant prophylaxis was allowed) were also extracted.
Evidence base
A total of 929 abstracts were identified in the literature search. Among these, 188 were selected for full-text review, 55 being subsequently excluded, whereas eight unpublished trials were included, resulting in 141 trials eligible for inclusion in the NMA. A flowchart of the trial selection process is provided in Supplementary material – Appendix C, and the list of trials excluded following full-text review is provided in Supplementary material – Appendix D.
Among the 141 eligible trials included in the NMA, 59 included sumatriptan (13–62), 12 included almotriptan (20,24,53,63–71), 14 included eletriptan (27,39,47,72–82), four included frovatriptan (64,83–85), eight included naratriptan (18,32,77,86–90), 31 included rizatriptan (18,29,35,38,40,44,60,66,78,86,91–108), 17 included zolmitriptan 18,22,26,31,82,99,103,109–119), eight included acetaminophen (20,30,96,120–124), five included selective COX-2 inhibitors (9,36,125–127), one included diclofenac (128), eight included naproxen (16,25,40,51,62,129,130), one included ibuprofen (120), seven included ergotamines (61,69,74,95,131–133), 12 included combination treatment of sumatriptan and naproxen (16,51,62,129,134–137), and one included combination treatment of rizatriptan and acetaminophen (96).
Regarding the outcomes of interest, 109 trials reported on any AEs, 53 reported on TRAEs, 110 reported on SAEs, 55 reported on chest discomfort, 75 reported on somnolence, 97 reported on dizziness, 77 reported on fatigue, 68 reported on nausea and 26 reported on vomiting. A summary trial network (network of all trials included in all analyses) is presented in Figure 1. Baseline characteristics for the included trials are provided in Supplementary material – Appendix E.
Network diagram displaying the evidence base for all trials included in the network meta-analysis.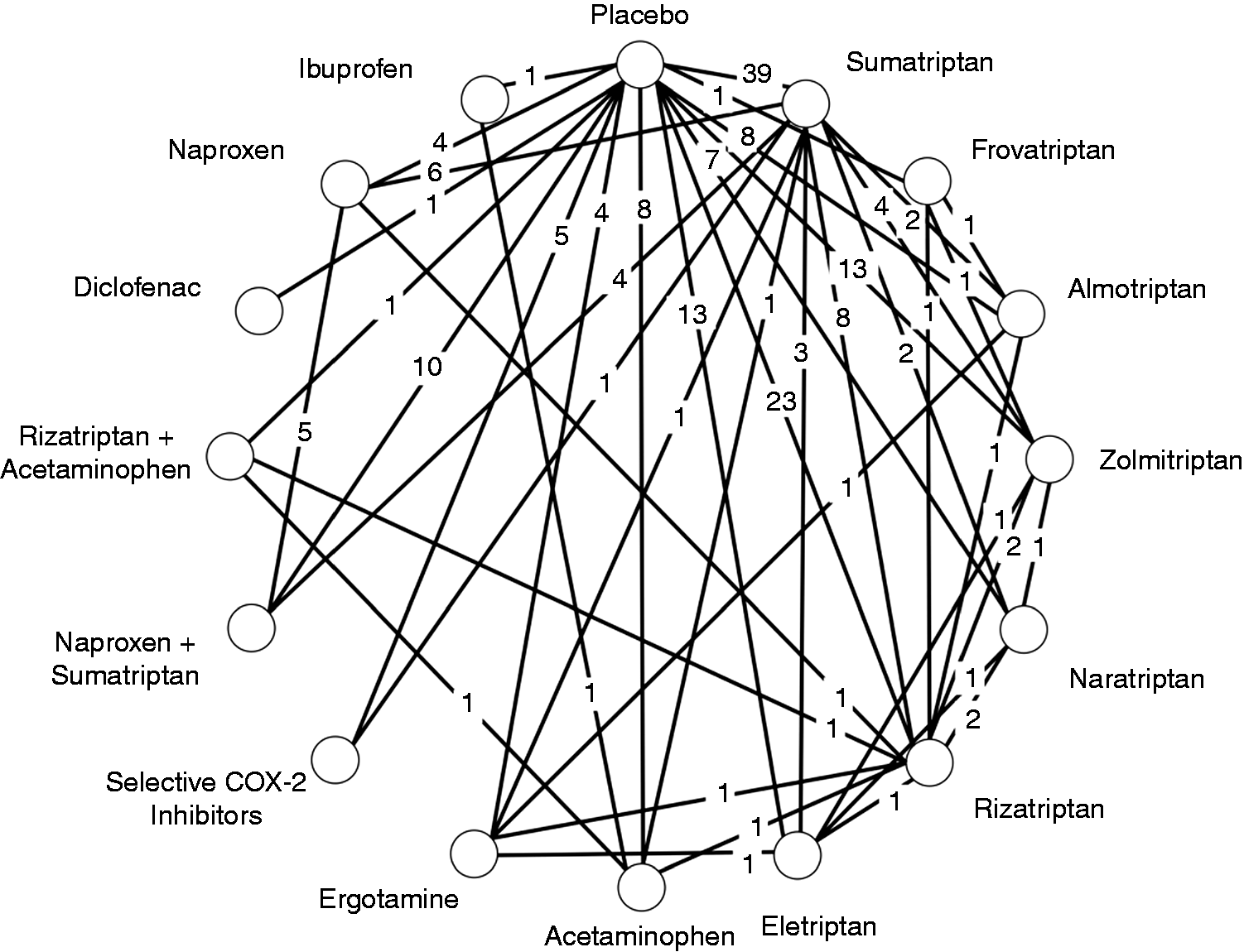
Network meta-analysis
Our analyses categorized each triptan (sumatriptan, eletriptan, frovatriptan, almotriptan, zolmitriptan, naratriptan or rizatriptan) as separate treatments. NSAIDs were classified as either non-selective COX inhibitors or selective COX-2 inhibitors (i.e., COX-2 inhibitors and other NSAIDs). Typically, COX inhibitors can be non-selective (effect both COX-1 and COX-2) or selective for COX-2. These produce different tolerability profiles, as COX-2 inhibitors selectively target the mechanism for relief of symptoms, whereas non-selective COX inhibitors can inadvertently cause AEs due to their non-selective nature, generally leading to worse tolerability profiles (138,139). Non-selective COX-2 inhibitors were further categorized as diclofenac, ibuprofen or naproxen. Finally, ergotamine, including Cafergot®, and acetaminophen were classified as single treatments.
To inform comparative tolerability between all interventions, we conducted Bayesian network meta-analyses (NMA) (140). This method provides better comparative evidence than pair-wise meta-analysis because it combines direct (i.e., head-to-head comparisons) and indirect evidence (comparisons across one or more common comparators), thus increasing the power of statistical comparisons while allowing for inferences of comparative tolerability between interventions that have not been directly compared (11,141). NMA also adds strength to comparisons that already informed head-to-head by combining evidence with all available indirect sources of evidence. We modeled log odds ratios using the conventional logistic regression NMA setup (142). Non-informative priors were assigned to all comparative treatment effect parameters (the log odds ratios in the logistic regression model) and the between-trial heterogeneity variance parameter in random effects models (142). All results for the NMA are reported as posterior medians with corresponding 95% credibility intervals (CrIs).
Previous work has shown that analyses are required to be adjusted for dose as it treatment dosage acts as an effect modifier (6). We have used a similar method in our analyses. Doses were categorized as “common”, “low”, and “high” according to previous NMA on comparative efficacy of triptans (6). The common dose was used as the reference dose. See Supplementary material – Appendix F for a full listing of low, common, and high doses for all treatments of interest. The effect-modification by administering a low dose as well as the effect-modification of administering a high dose were modeled using indicator variables and assuming similar effect-modification across all treatments in the network. Note, for non-triptan interventions all doses were modeled as common dose since they fell within similar ranges. Non-informative priors (normal distributions with variance 10,000) were assigned to the two regression parameters. A uniform distribution ranging from 0 to 2 was used as the prior for the random effects heterogeneity parameter.
Multiple models were fit for each outcome. The deviance information criterion (DIC) is a measure of model fit that penalizes for model complexity (143). A smaller DIC indicates a better fit. Model selection using the DIC calculated according to Dias et al. (142) was used to assess the fit of fixed and random effects models, and to determine whether the condition of consistency required for NMA was met. In this analysis, random effects models were consistently favored, and are reported. We used an independent means model to assess consistency, rather than using
All analyses were conducted using WinBUGS version 1.4.3 (Medical Research Council Biostatistics Unit, Cambridge) and R version 3.0.1 (http://www.r-project.org/). Model convergence was assessed by running 50,000 burn-in iterations of three chains and inspecting the trace plots and Brook–Gelman–Rubin plots. No thinning was applied.
Results
Analysis of primary outcomes
Network meta-analysis results of primary outcomes.

Note: OR, odds ratio; CrI, credibility interval; —, no evidence in the network. COX-2: cyclooxygenase-2; DIC: deviance information criterion. Statistically significant values are highlighted in bold. Serious adverse event outcomes do not include trials with zero counts.

Forest plots depicting results from the network meta-analysis (NMA) for the outcome any adverse event. Comparative effects are presented as odds ratios with 95% credible intervals. Odds ratios smaller than 1.00 favor the treatments, odds ratios larger than 1.00 favor placebo.
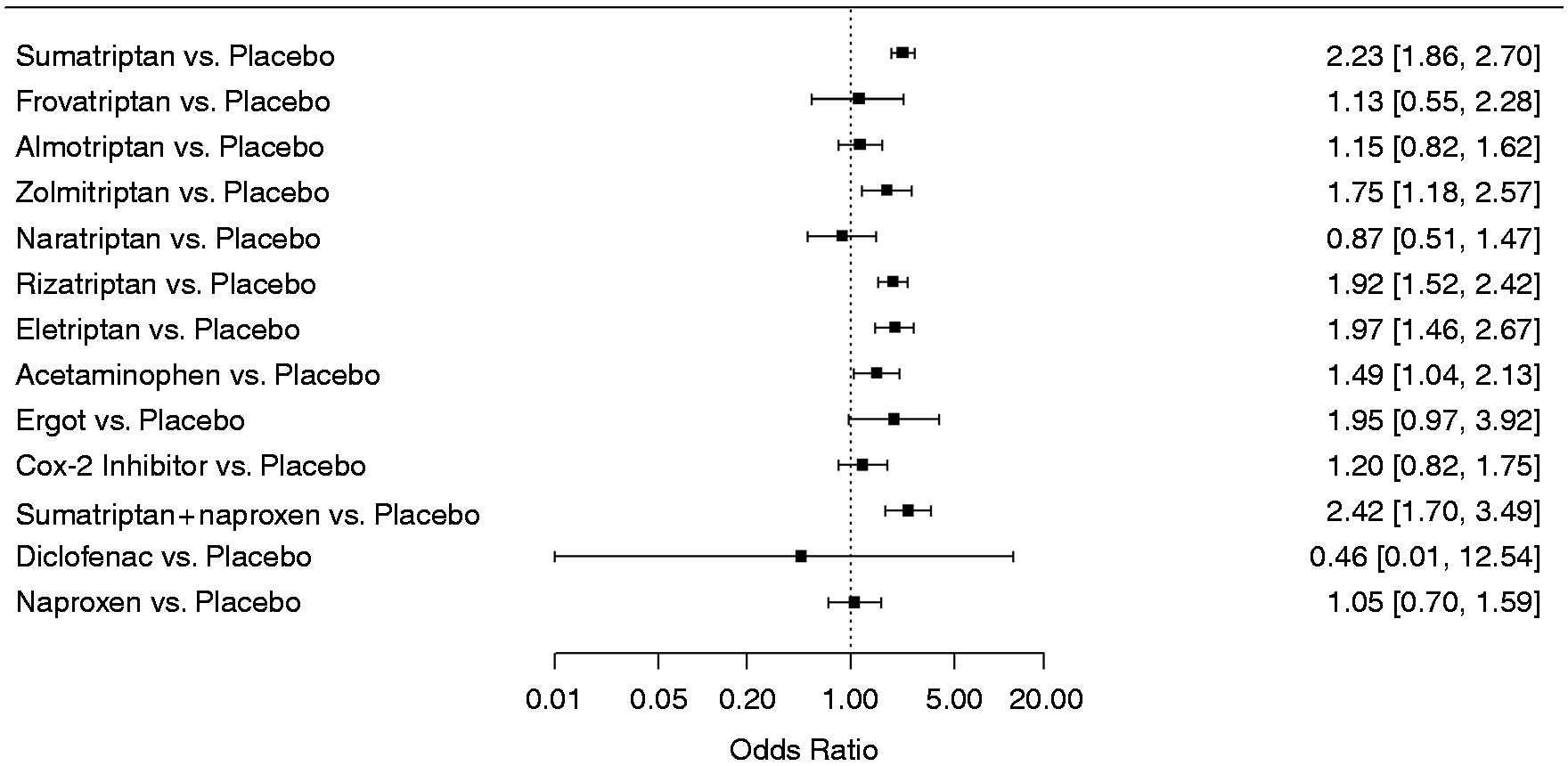
Forest plots depicting results from the network meta-analysis (NMA) for the outcome treatment-related adverse events. Comparative effects are presented as odds ratios with 95% credible intervals. Odds ratios smaller than 1.00 favor the treatments, odds ratios larger than 1.00 favor placebo.
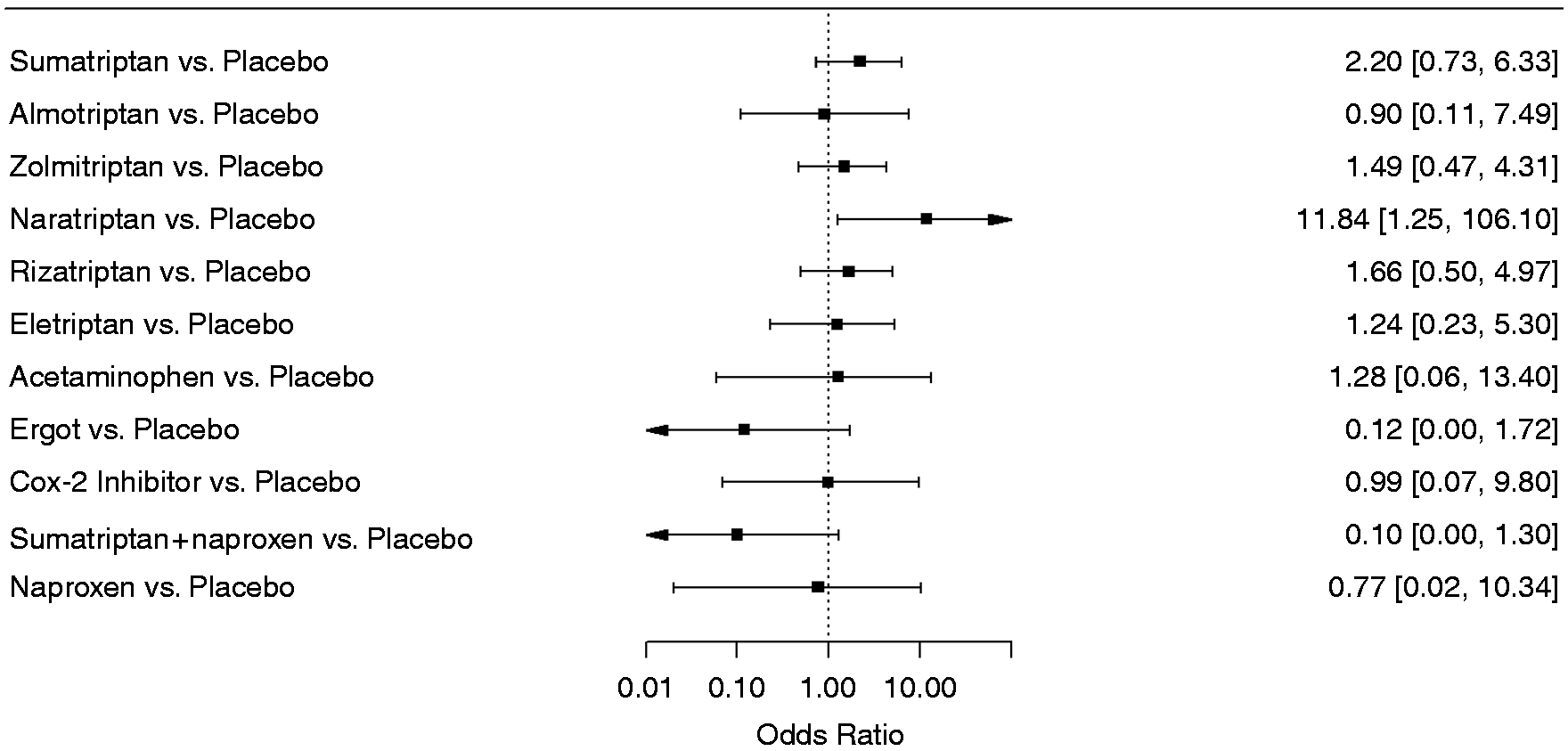
Forest plots depicting results from the network meta-analysis (NMA) for the outcome serious adverse events. Comparative effects are presented as odds ratios with 95% credible intervals. Odds ratios smaller than 1.00 favor the treatments, odds ratios larger than 1.00 favor placebo.
Results from the meta-regression reported that dose of triptans had a significant effect on any AEs and TRAEs. Any AEs odds ratios would be altered by a factor of 0.76 (95% CrI 0.65–0.91) for low dose treatment and 1.28 (95% CrI 1.19–1.49) for high dose treatment. Similarly, for TRAEs, odds ratios would be altered by a factor of 0.73 (95% CrI 0.58–0.92) for low dose treatment and 1.45 (95% CrI 1.19–1.74) for high dose treatment. Results from the meta-regression indicated that dose did not play a statistically significant role for SAEs; however, the evidence base was limited.
Analysis of secondary outcomes
Network meta-analysis results of secondary outcomes.

Note: OR, odds ratio; CrI, credibility interval; —, no evidence in the network. COX-2: cyclooxygenase-2; DIC: deviance information criterion. Statistically significant values are highlighted in bold.
Overall, triptans were also associated with increased odds of dizziness, with the exception of frovatriptan and almotriptan, both of which were associated with decreased odds. Other treatments were generally associated with an increase in odds of dizziness compared with placebo, with the exception of naproxen (OR=0.88, 95% CrI 0.51–1.52) and ibuprofen (OR=0.45, 95% CrI 0.10–1.61). Compared with placebo, triptans were generally associated with increased odds that chest discomfort would be experienced, with eletriptan being associated with the highest odds ratio (vs. placebo) compared with all other treatments (OR=4.67 95% CrI 2.44–8.78). However, no significant difference was observed for almotriptan, frovatriptan, and naratriptan. Compared with placebo, the combination treatment of rizatriptan and acetaminophen was associated with the lowest odds ratio of chest discomfort (0.08, 95% CrI 0.01–3.08); however, the evidence base was limited. The evidence base for non-triptans was also limited, thus resulting in wide 95% CrIs.
For somnolence, almost all treatments were associated with odds ratios larger than 1.00 when compared with placebo, but only sumatriptan, eletriptan, rizatriptan, zolmitriptan, sumatriptan+naproxen, and COX-2 inhibitors were statistically significant. Most triptans were associated with an increase in odds for nausea. Almotriptan and naratriptan, however, yielded odds ratios close to 1.00. Acetaminophen and COX-2 inhibitors also yielded odds ratios (vs. placebo) close to 1.00, whereas ibuprofen and naproxen yielded substantially lower odds of nausea compared with placebo, albeit not statistically significant.
For vomiting, almotriptan yielded an approximately 7 times higher relative odds compared with placebo, which was statistically significant. No other triptan versus placebo comparisons were statistically significant. Triptans were generally associated with decreased odds of vomiting (with the exception of almotriptan). Acetaminophen was associated with statistically significantly lower odds of vomiting compared with placebo (OR=0.43, 95% CrI 0.19–0.90). Ibuprofen yielded the lowest odds ratio, but was not statistically significant (OR=0.13, 95% CrI 0.01–1.06).
Results of the meta-regression indicated that low triptan dose treatments had a statistically significant impact on estimates regarding fatigue (OR=0.68, 95% CrI 0.57–0.89), dizziness (OR=0.71, 95% CrI 0.56–0.87), chest discomfort (OR=0.45, 95% CrI 0.27–0.75), and somnolence (OR=0.59, 95% CrI 0.45–0.76). High dose treatments had a statistically significant impact on estimates regarding fatigue (1.73, 95% CrI 1.41–2.08), dizziness (1.30, 95% CrI 1.09–1.56), chest discomfort (1.74, 95% CrI 1.26–2.45), and nausea (1.25, 95% CrI 1.01–1.56).
Discussion
This study examined the comparative tolerability of treatments for acute migraine. For most outcomes, triptans tended to report slightly higher odds of an event occurring, with the exception of SAEs and vomiting. The forest plots show that selective COX-2 inhibitors have the lowest odds for any AEs and were significantly lower than most triptans. For all other primary outcomes, there were no clinically important differences. For the secondary outcomes, eletriptan, sumatriptan, zolmitriptan, rizatriptan, and combination treatment of sumatriptan and naproxen were all associated with significantly higher risks when compared with placebo, except for the outcome vomiting. However, for the included trials, placebo risks were very low, suggesting low clinical relevance of this finding.
As previously mentioned, a NMA has already been conducted to determine the comparative efficacy of triptans (6). This study focuses on the comparative tolerability of triptans, as well as other common treatments for migraine headaches (60,144). The results indicate that overall, none of the comparative risks differed to an extent that would necessitate priority of one treatment over another based solely on tolerability outcomes.
This study has several strengths. The literature searches were broad and sensitive, ensuring that all eligible published trials have been included. The search identified 141 eligible trials, which provided a highly informed evidence base. The majority of the included trials utilized eligibility criteria following the International Headache Society (IHS) guidelines, and was very similar concerning the recorded baseline characteristics. Our study also has a number of limitations. In analyses of AEs, it is almost inevitably the case that the number of events will be low, and for this reason, play of chance impacts the comparative tolerability estimates. For this reason, those of the considered outcomes with low placebo proportions (all but any AEs) should be interpreted with caution. For SAEs in particular, most trials reported zero events in all treatment arms. Most trials include “hospitalization for any reason” as a SAE. Thus the apparently paradoxical reduction in SAEs for several triptans compared with placebo may reflect efficacy by avoiding migraine-related hospital admissions. For SAEs, a potential concern is that the definition was left to the discretion of the trial investigators, and thus, may have produced some heterogeneity in the outcome definition. However, SAEs for migraine have been defined according the to the ICH-1, and as most, if not all, studies used Good Clinical Practice Guidelines (GCP), it stands to reason that the definitions were sufficiently similar across the included trials. Whether included in the analysis or not, such data provides little information about the expected AE risks, and so, even though several trials informed the outcome, only a limited number of trials were useful for this analysis. Furthermore, the reporting of secondary outcomes was highly inconsistent across trials, with each of the secondary outcomes being reported, on average, in only about half of the included trials because most studies only report AEs that occur in enough patients. To this end, as multiple outcomes with partially overlapping data sources were analyzed with multiple models, some concerns about multiplicity (false positive and false negative findings due to multiple testing) are warranted. As such, the findings of this study should be interpreted with caution.
Randomized clinical trials in the therapeutic area of migraine are not designed to conclusively demonstrate superiority or inferiority of treatments on tolerability outcomes. While NMA is strong in that it allows for rigorous synthesis of all randomized evidence to date, it falls short in that evidence sufficiency is never guaranteed, and some heterogeneity between trials may exist. Our NMA provide insight as to how the considered interventions are likely to compare on their tolerability profiles, but definite answers to this question should be sought in large well-designed cohort studies.
Migraine treatments are frequently being administered at different doses according to the perceived severity of the migraine and patient preferences. Our analyses cast light on the increased risk of AEs one might expect with taking a medication at a higher triptan dose, and thus can inform physician and patient choices of interventions. Our results should also be interpreted in connection with previously established estimates of comparative efficacy when making decisions about which treatment option to choose (6).
In summary, triptans and ergotamine were associated with higher odds of any AE and treatment related adverse events compared with NSAIDs. The tolerability profile of triptans versus NSAIDs and ergotamines are mixed across the analyzed individual outcomes. Overall, results between and amongst treatment classes therefore indicate comparability regarding tolerability profiles.
Article highlights
This study examined the comparative tolerability of treatments for acute migraine using a systematic review and NMA. Triptans and ergotamine were associated with higher odds of any AE and treatment related adverse events when compared with NSAIDs. The tolerability profile of triptans versus NSAIDs and ergotamines were mixed across the analyzed individual outcomes. Overall, results indicate comparability between and amongst treatment classes regarding tolerability profiles. As triptans are frequently administered at different doses according to perceived severity, this analysis cast light on the increased risk of AEs one might expect with taking medication at a higher dose and can be used to inform physician and patients about choices of interventions.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Thorlund has previously consulted to Boehringer Ingleheim, Merck, Pfizer, Novartis, Janssen, Roche, Novo Nordisk, UCB, and Gilead on systematic reviews and network meta-analysis. In addition, Drs. Thorlund and Mills have received grant funding from the Canadian Institutes of Health Research (CIHR) Drug Safety & Effectiveness Network to develop methods and educational materials on network meta-analysis. Keith Chan, Eric Druyts, Kabirraaj Toor and Ping Wu are employees of Redwood Outcomes. Drs Bhambri and Ramos are full-time employees of Pfizer Inc. Dr. Donnet has received payments from Allergan, Almirall SAS, Astellas, AstraZeneca Pharmaceuticals, Grunenthal, Lilly, MSD, Orkyn, Pfizer, Saint-Jude, Sanofi-Aventis, Teva, Zambon. Dr. Stark has received payment from Allergan for advisory boards, consulting, lectures and clinical trials; he also has received payment for lectures from Pfizer and MSD. Dr. Goadsby reports grants and personal fees from Allergan, grants and personal fees from eNeura Inc, personal fees from Autonomic Technologies Inc, grants and personal fees from Amgen Inc; personal fees from Alder Biopharmaceuticals, Pfizer Inc, Dr Reddy's Laboratories, Zosano Pharma Corporation, Colucid Pharmaceuticals, Ltd, Eli-Lilly and Company, Avanir Pharmaceuticals, WL Gore & Associates, Heptares Therapeutics, Nupathe Inc, Teva, Cipla Ltd, Ajinomoto Pharmaceuticals Co, Akita Biomedical, Wells Fargo, Ethicon, US, EMKinetics, Promius Pharma, MedicoLegal work, Up-to-Date, Journal Watch, outside the submitted work. In addition, Dr. Goadsby has a patent Magnetic stimulation for headache pending.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by Pfizer Inc. Redwood Outcomes conducted this study. Dr. Thorlund is a founding partner of Redwood Outcomes and were paid consultants for conducting this study and for the development of this manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
