Abstract
Tolerability is an important attribute of patient satisfaction with, and consequence adherence to, migraine acute treatment. Nevertheless, the determinants of tolerability are poorly explored. Accordingly, our objectives were: (i) in subjects receiving triptans, to contrast two methods of assessing adverse events (AEs); and (ii) to explore the relationship between migraine features and treatment attributes with tolerability. We surveyed 365 migraineurs who had been using the same triptan for at least 3 months. After prospectively treating an attack, headache characteristics and treatment response were assessed using headache calendars. Subjects also completed a standardized questionnaire, first asking about any AE and then prompting patients with a list of possible AEs. We contrasted both sets of answers and conducted logistic regression to assess if headache attributes or response to therapy influenced tolerability. Using the unprompted method, AEs occurred in 11.5-36.4± of patients, depending on the triptan used. Using the prompted method, they ranged from 26.9 to 64.3±. Chest and neck tightness were spontaneously reported by 3.5± of the sample, vs. 7.4± when prompted (P< 0.05). Chest pain was not spontaneously reported and was elicited in nine patients (2.5±, P = 0.002). Feeling groggy occurred in 5.7 and 17.5± (P< 0.001). AEs were not a function of headache severity, disability, efficacy of the drug, time to meaningful relief with the drug or recurrence of pain. The report of AEs varies dramatically with the methods of assessment. However, tolerability is not influenced by the severity of the attacks or by medication efficacy.
Introduction
Several studies have shown that migraine patients consider tolerability as an important attribute for acute migraine therapy and for adherence to treatment (1–3). Nevertheless, methods for assessing tolerability have not been standardized, which complicates the comparison of this attribute across different studies (4). It has been previously shown that when assessing adverse events (AEs), the method of data collection dramatically influences the results (5); an important proportion of subjects who did not self-report AEs after using a triptan actually reported them if prompted with a list of side-effects. This has implications. First, because assessment of AEs is not standardized within different clinical trials, comparisons are problematic. Second, clinicians should be aware of these findings not to induce nocebo response when assessing the tolerability of specific migraine treatments (6).
Herein we expand our prior work (5) by first contrasting two methods of assessing AEs (where patients are first asked to report them spontaneously, and are subsequently prompted to a list of potential AEs) in migraine patients using oral forms of triptans. We then assess the possible relation between triptan's side-effects and attack severity, disability, and recurrence of headache, as well as drug response. We hypothesized that, for a particular drug, higher headache response to therapy is inversely correlated with tolerability.
Methods
Setting and inclusion criteria
This study was conducted in a Headache Centre in Italy from February 2004 to February 2006. We prospectively surveyed adult headache sufferers fulfilling the following inclusion criteria:
Diagnosis of migraine according to the Second Edition of the International Classification of Headache Disorders (7).
Had been using the same oral triptan for at least 3 months.
At least three attacks treated with the same triptan over a 3-month period.
Being followed at the headache centre for at least 1 year.
Age range from 18–65 years.
No contraindications to triptan use.
At the time of this study, naratriptan was not available in Italy.
Data collection
Subjects were requested to treat a headache attack prospectively with their usual triptan. Complete information about migraine (e.g. severity of pain, unilaterality, etc.) was captured using semi-standardized headache questionnaires. Response to therapy was assessed using headache calendars.
After treating the attack, subjects were requested to return to the clinic within 24 h. They received a definition of AEs, which, in lay terms, meant any untoward medical occurrence temporally associated with the use of medication. AEs were assessed in two different ways. First, subjects were asked if they had had any AE when using the triptan (unprompted questionnaire). Those who responded positively were then asked to list the AEs, grade their severity as mild, moderate, or severe and their duration in minutes or hours. After returning the first part of the questionnaire, subjects received a second form. The second form was answered on the same day, but subjects did not have the opportunity to check their answers given in the first questionnaire. In this questionnaire (prompted questionnaire), 49 possible AEs were listed. Most of them were known triptan side-effects; some confounders (side-effects not expected to be related to triptan use) were added.
Analysis
We used descriptive statistics and summary tables to present the demographics. Analyses were performed using
The presence of AEs (according to both methods of assessment) was modelled using headache features (frequency of headache, duration of headache, headache-related disability, response to therapy, use of preventive medication, age, race and socioeconomic status as independent variables. Continuous independent variables were evaluated for non-linearity using squared and higher order terms. Backwards stepwise maximum-likelihood estimation was used to arrive at a parsimonious model.
This study had been approved by an Investigation Review Board.
Results
Our sample consisted of 365 subjects (80% female) with a mean age of 34.6 years. Among the six triptans that are available in Italy, rizatriptan (10-mg tablet) was being used by 101 (27.6%), almotriptan (12.5-mg tablet) by 87 (23.8%), sumatriptan by 74 (20.3%), eletriptan by 49 (13.4%), zolmitriptan by 28 (7.6%) and frovatriptan by 26 (7.2%). Of sumatriptan users, 40 (10.9%) were using 50 mg, 22 (6.0%) were using 100 mg and two (0.5%) were using 25 mg.
Table 1 presents the proportion of patients, using the different triptans, reporting AEs in the unprompted and prompted methods. Using the unprompted method, side-effects ranged from happening on 11.5% of the patients (frovatriptan) to 36.4% (sumatriptan). Using the prompted method, AEs ranged from 26.9% (frovatriptan) to 64.3% (zolmitriptan). The prompted/unprompted ratio ranged from as low as 1.37 (sumatriptan) to as high as 2.33 (frovatritptan).
Proportion of patients reporting adverse events in the unprompted and prompted methods

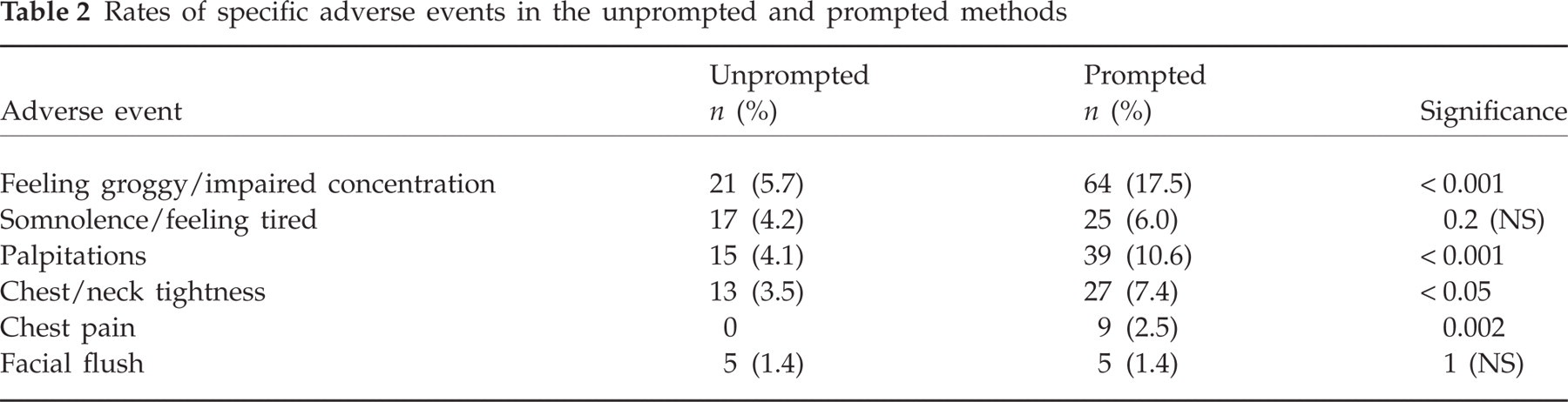
Several specific AEs were reported significantly more commonly after being prompted (Table 2). Chest and neck tightness were spontaneously reported by 3.5% of the sample vs. 7.4% when prompted (P < 0.05). Chest pain was not spontaneously reported, and was elicited in nine patients (2.5%, P = 0.002). Feeling groggy or unable to concentrate was reported by 5.7% and elicited in 17.5% of the sample (P < 0.001).
Rates of specific adverse events in the unprompted and prompted methods
Logistic regression was used to assess if selected headaches or treatment attributes predicted AEs in the unprompted and prompted methods. The proportion of side-effects, using both methods of assessment, as a function of headache severity, attack-related impairment and efficacy of the drug in treating the headache is presented in Table 3. In the logistic models, reporting AEs was not a function of headache severity, disability or efficacy of the drug. Similarly, it were not a function of having aura, unilateral pain, throbbing pain or associated symptoms.
Adverse events as a function of headache and efficacy of medication

We also assessed if time to meaningful relief with the drug and recurrence of pain were associated with reporting AEs (Table 3). These two attributes were also not predictive of tolerability in the logistic models.
Discussion
Triptans are highly effective migraine drugs (4). They are the class of choice for the acute treatment of migraine with attack-related impairment, according to several National Guidelines (8–10). Nevertheless, despite their well-established efficacy, endorsement by the accepted guidelines, and the demonstrable need to employ effective intervention to reduce migraine-associated disability, triptans are not widely used in the population (11). Although the relatively restricted use of triptans may be attributed to several factors, research suggests that prescribers’ concerns about safety and tolerability figure prominently in limiting their use (12). Furthermore, an important proportion of migraineurs that receive a triptan switch to a second triptan or to a medication from a different class (13). It has been demonstrated that a variety of treatment attributes are important in determining the reasons for switching a triptan. Assessing these attributes is increasingly important to understand patients’ attitudes and preferences regarding various treatment characteristics (14).
A survey of a population-based sample of triptan-naive migraineurs has indicated that, although complete efficacy is the most important desirable attribute of treatment in a drug, good tolerability is the second one (12). When physicians are surveyed, the results are similar, and tolerability ranks as the second most important attribute of a migraine drug, significantly more important than consistency [30%, 95% confidence interval (CI) 27, 34, vs. 21%, 95% CI 19, 24] (11).
When assessing AEs, the method of data collection may dramatically influence the results. In a clinic-based study, of those subjects who did not self-report AEs after using a triptan, most reported positively if presented with a list of side-effects (13). We have confirmed this finding in our current study. Accordingly, caution is strongly advised when contrasting the tolerability of different drugs across studies.
We had also hypothesized that tolerability would be a function of efficacy. Individuals with good response to therapy would be less likely to report side-effects. Analogously, individuals with recurrence of pain, delayed meaningful response and important attack-related disability would be more likely to report AEs. We conducted logistic regression to test our hypotheses and did not confirm them. In our data, tolerability appears to be a domain not influenced by the severity of the attack or the response to therapy.
Our data should be seen with caution. First, we assessed patients using triptans only. Therefore, our data cannot be extrapolated for other classes of medication. Second, our study was done in a single headache centre. Third, both methods of assessment of AEs (prompted and unprompted) were conducted consecutively. Also, it has been demonstrated previously that AEs are reported more frequently when a new treatment is initiated (15). Here, our patients have been on the same treatment for at least 3 months (4). Finally, we are underpowered to conduct dose- and formulation-related analyses. Strengths of our study include the sample size and the fact that headache information was prospectively collected using headache calendars.
The report of AEs varies dramatically with the methods of assessment. However, tolerability is not influenced by the severity of attacks or by medication efficacy attributes.
