Abstract
If treatment-emergent central nervous system (CNS) symptoms following triptan therapy represent direct pharmacological effects of the drug, they should occur independent of response to active drug. However, if they represent unmasking of neurological symptoms of the migraine attack after pain is relieved, they should be more common in responders both to active drug and to placebo. To explore this issue, we evaluated the relationship between the CNS adverse events and treatment response following triptan or placebo treatment. We used pooled data from seven double-blind, placebo-controlled trials involving eletriptan 20 mg (E20, n = 402), eletriptan 40 mg (E40, n = 1870), eletriptan 80 mg (E80, n = 1393), sumatriptan 100 mg (S100, n = 275) and placebo (Pbo, n = 1024). Somnolence was more prevalent among 2 h headache responders than non-responders for all treatments, including E80 (8.8% vs. 5.0%; P < 0.05), E40 (6.4% vs. 5.0%; NS), E20 (4.0% vs. 2.0%; NS), S100 (4.7% vs. 3.2%; NS) and Pbo (7.6% vs. 3.0%; P < 0.05). Similarly, the incidence of asthenia was higher among patients who responded to treatment compared with those who did not respond to E80 (15.2% vs. 7.8%; P < 0.05), E40 (6.5% vs. 3.6%; P < 0.05), E20 (6.5% vs. 1.0%; P < 0.05), S100 (10.1% vs. 4.7%; NS) and Pbo (4.4% vs. 2.7%; NS). The generally higher rates of somnolence and asthenia in patients who respond to treatment suggests that these treatment-emergent neurological symptoms may represent the unmasking of CNS symptoms associated with the natural resolution of a migraine attack, rather than simply representing drug-related side-effects. The rate of somnolence in placebo responders is comparable to that in responders to E40 and E80, indicating that somnolence is related, at least in some important part, to headache relief and not treatment.
Keywords
Introduction
Clinical trials with serotonin 5-HT1B/1D receptor agonists, triptans (1), offer an important opportunity to gain insights into the pathophysiology of migraine. The clinical trials involving triptans were conducted in similar populations of patients defined by standard diagnostic criteria (2) and with considerable methodological homogeneity (3). Large datasets, such as those from the triptan clinical trial programmes, offer the statistical power to explore interesting and important questions ordinarily limited by smaller datasets. We have employed the eletriptan clinical trial database to explore issues around adverse events reported with triptans. The starting hypothesis was that some so-called adverse events may represent the unmasking of disease-related symptoms when headache is relieved and not a direct effect of drug.
Thus far, the triptan clinical trial datasets have largely been used to study efficacy issues. Meta-analyses of triptan studies, either from specific programmes, such as that for sumatriptan across formulations (4), or within formulations (5) or across programmes have been completed. The broad meta-analyses (6, 7) demonstrate the relative similarity of these clinical trials and thus their relative suitability for such examinations (8). Perhaps because patients (9, 10) and doctors (11) alike value efficacy so clearly over side-effects, the study of adverse events has lagged for triptans. Except for cardiovascular adverse events (12, 13), other triptan side-effects have received little attention.
Previously we have examined the relationship between efficacy and nausea (14). Such a relationship does exist in the sense that patients who respond to a triptan have relief of nausea, but slight variations in the pharmacology of the triptans, particularly their activity at the 5-HT1A receptor (15), have made this analysis complex. Somnolence and asthenia are well reported side-effects of triptans, but it is also true from clinical practice that many migraineurs value sleep to assist in terminating attacks—indeed, in children this is a very common behaviour (16). We hypothesized that if the side-effect represented a direct pharmacological effect of the drug it should be equally common in responders and non-responders. In the scenario of unmasking on-going brain events by relieving pain, we predicted that responders should have higher rates of central nervous system (CNS) adverse events than non-responders. If adverse event rates are equivalent for active drug and placebo in responders, that would suggest the entire effect was due to unmasking. Typical publications of clinical trials present data as response by treatment group. Treatment response is more likely with active drug, so that the relationships may be obscured. Accordingly, we employed the eletriptan database to explore the relationship of these two CNS side-effects, commonly associated with triptan therapy to treatment response. The work was reported in preliminary form at the Annual Meeting of the American Headache Society 11–13 June 2004 (17).
Methods
The current analysis was performed on data pooled from seven double-blind, placebo-controlled, parallel-group, multicentre studies evaluating the efficacy and tolerability of eletriptan in the acute treatment of migraine headaches (Table 1). The analysis reports first attack data for patients who had both pre- and post-treatment assessment for the following study treatments: eletriptan 20 mg (E20, n = 434), eletriptan 40 mg (E40, n = 1942), eletriptan 80 mg (E80, n = 1464), sumatriptan 100 mg (S100, n = 296) and placebo (Pbo, n = 1070).
Demographic and clinical characteristics of study patients: combined controlled trial data

The combined studies had similar designs, as summarized in previous reports (18–22). Briefly, adult patients were enrolled if they met the International Headache Society criteria for migraine with or without aura (2) and reported a monthly frequency of one to six attacks. Key exclusion criteria consisted of coronary artery disease, heart failure, uncontrolled hypertension or abnormal electrocardiogram (ECG); hypersensitivity or known contraindication to treatment with eletriptan or sumatriptan; misuse or abuse of alcohol or other substances, including analgesics or ergotamine; and women who were pregnant or breast-feeding. The study protocols were approved by Institutional Review Boards (Ethics Committees) at each site and written informed consent was obtained prior to study entry.
Study procedures
All studies were conducted in compliance with Good Clinical Practice guidelines. Similar data collection procedures were used in all studies. Patients were instructed to take study medication when they experienced a typical migraine attack of moderate or severe intensity that was not spontaneously improving. Patients took study treatment when the aura phase had ended and within 6 h of the onset of headache pain. Treatment with study medication was not permitted if the patient had used an analgesic or antiemetic during the previous 6 h, or had taken another triptan or ergotamine-containing or ergot-type medication (e.g. dihydroergotamine) during the previous 24 h. For all studies, the occurrence, severity and duration of adverse events were concurrently recorded by patients in diaries. Diary recording for adverse events continued for up to 7 days postdosing.
Choice of studies and CNS adverse events
Three additional studies were excluded from the analysis because they ascertained the presence of adverse events by means of retrospective assessment at the post-treatment visit instead of by means of concurrent diary assessment. The retrospective ascertainment method and the resulting heterogeneity precluded simple pooling. Dizziness was excluded because we were not able to distinguish orthostatic effects and lightheadedness from vertigo.
Statistical analyses
Data were pooled from seven double-blind placebo-controlled studies (18–22). The analysis was based on data from a single treated migraine attack per patient. In studies where patients treated more than one attack, only data from the first attack were used to avoid complexity of baseline clinical characteristics that might affect CNS adverse events.
Statistical modelling and testing was performed using Proc Logistic in the Statistical System Software (SAS Inc., Cary, NC, USA)). The response variables were CNS adverse events such as somnolence, asthenia and any of the adverse events (asthenia or somnolence combined).
Three step logistic regression models were used with terms included for baseline headache severity, gender, age, baseline nausea, baseline photophobia/phonophobia, aura and 2 h headache response. The model was then reduced to include only those terms that had a P-value of 0.1. The model was further reduced to include only the terms that had a P-value of <0.05. The affect of each term on CNS adverse events was also assessed using adjusted odd ratio (OR) (Table 2).
Treatment-emergent CNS adverse events by treatment response category: combined controlled trial data
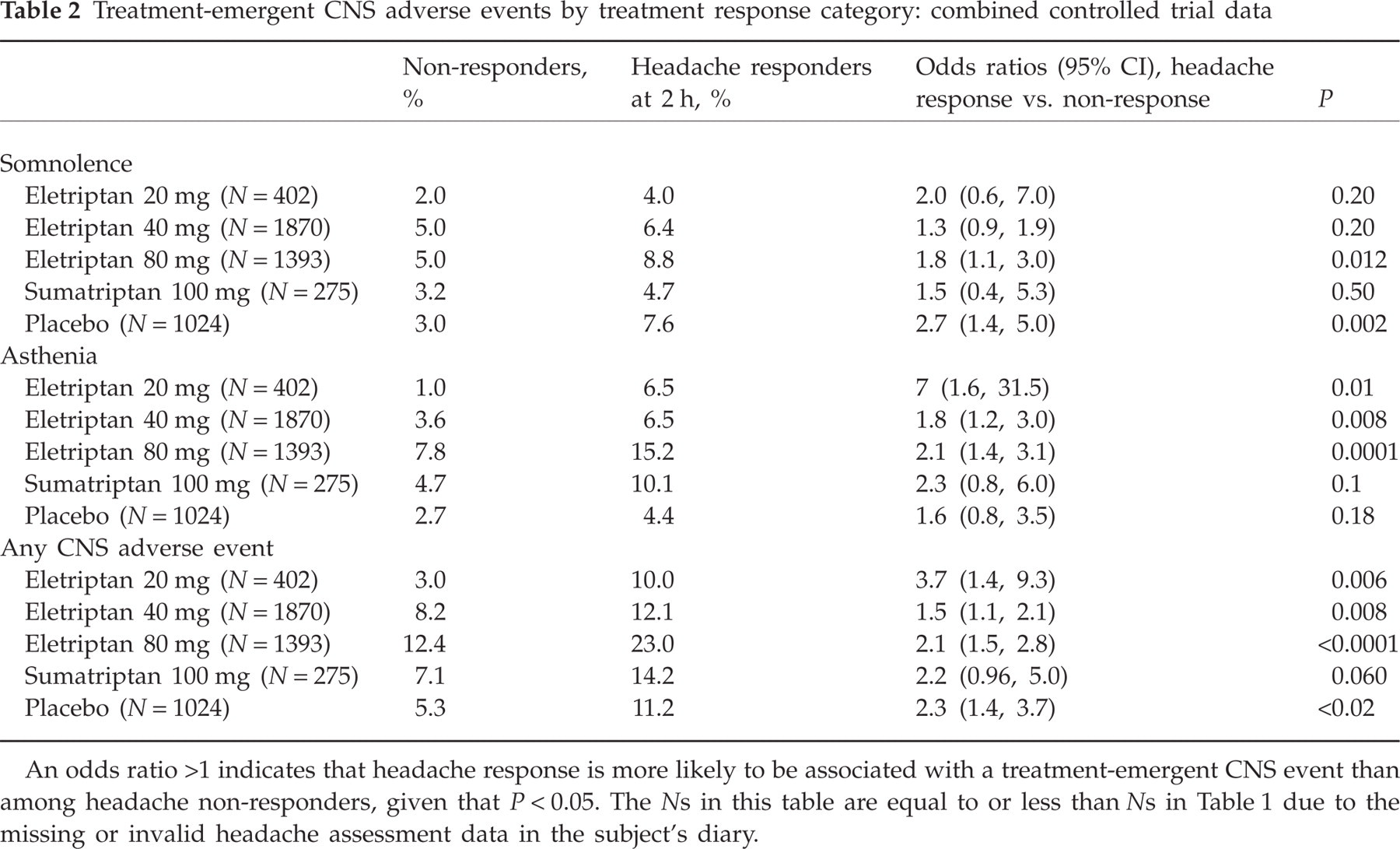
An odds ratio >1 indicates that headache response is more likely to be associated with a treatment-emergent CNS event than among headache non-responders, given that P < 0.05. The Ns in this table are equal to or less than Ns in Table 1 due to the missing or invalid headache assessment data in the subject's diary.
Finally, since treatment was also associated with both headache response and CNS adverse events, we used analysis of variance with CNS adverse event as response variable and treatment and headache response as independent variables using GENMOD procedure in the SAS (Table 3).
Incidence of treatment-emergent CNS adverse events (asthenia/somnolence) regardless of headache response status

Adjusted OR >1 indicates that active treated is more likely to be associated with a CNS adverse event when compared with Pbo given that P < 0.05. The Ns in this table are equal to or less than Ns in Table 1 due to the missing or invalid headache assessment data in the subject's diary.
Descriptive statistics rate of headache response also provided for patients with CNS adverse events and for patients without CNS adverse events (Table 4).
Effect of presence vs. absence of CNS event on headache response and pain free at 2 h: combined controlled trial data
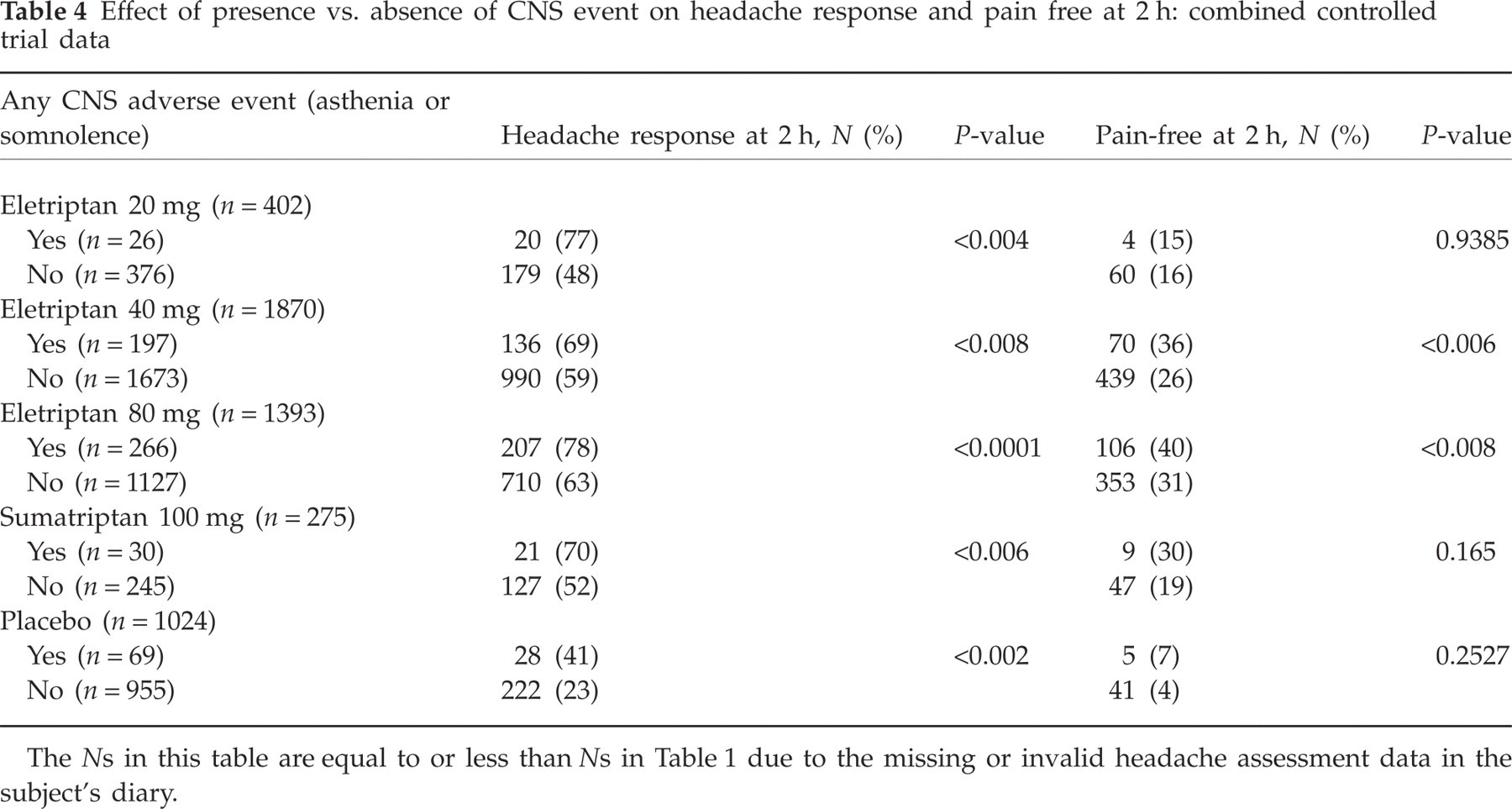
The Ns in this table are equal to or less than Ns in Table 1 due to the missing or invalid headache assessment data in the subject's diary.
Results
A total of 5206 patients in the combined sample received study treatment for a single migraine headache and had at least one pre- and one postdose assessment for the first attack. Patients in each treatment group had similar demographic and clinical characteristics (Table 1). Consistent with the epidemiology of migraine, patients in the current treatment sample were predominantly women in the 30–50 years age range.
Treatment-emergent somnolence and asthenia
The treatment-emergent incidence of somnolence, regardless of causal attribution, was found to be numerically higher across all study treatment groups in the subgroup of patients who achieved a headache response at 2 h (Fig. 1). The difference achieved significance for eletriptan 80 mg and placebo. A similarly consistent numerical increase in the headache responder group was found for asthenia (Fig. 2), with the difference achieving significance at all three doses of eletriptan. When the two index CNS adverse events were combined (Fig. 3), the incidence was significantly higher in the headache responder group on all study treatments except sumatriptan.
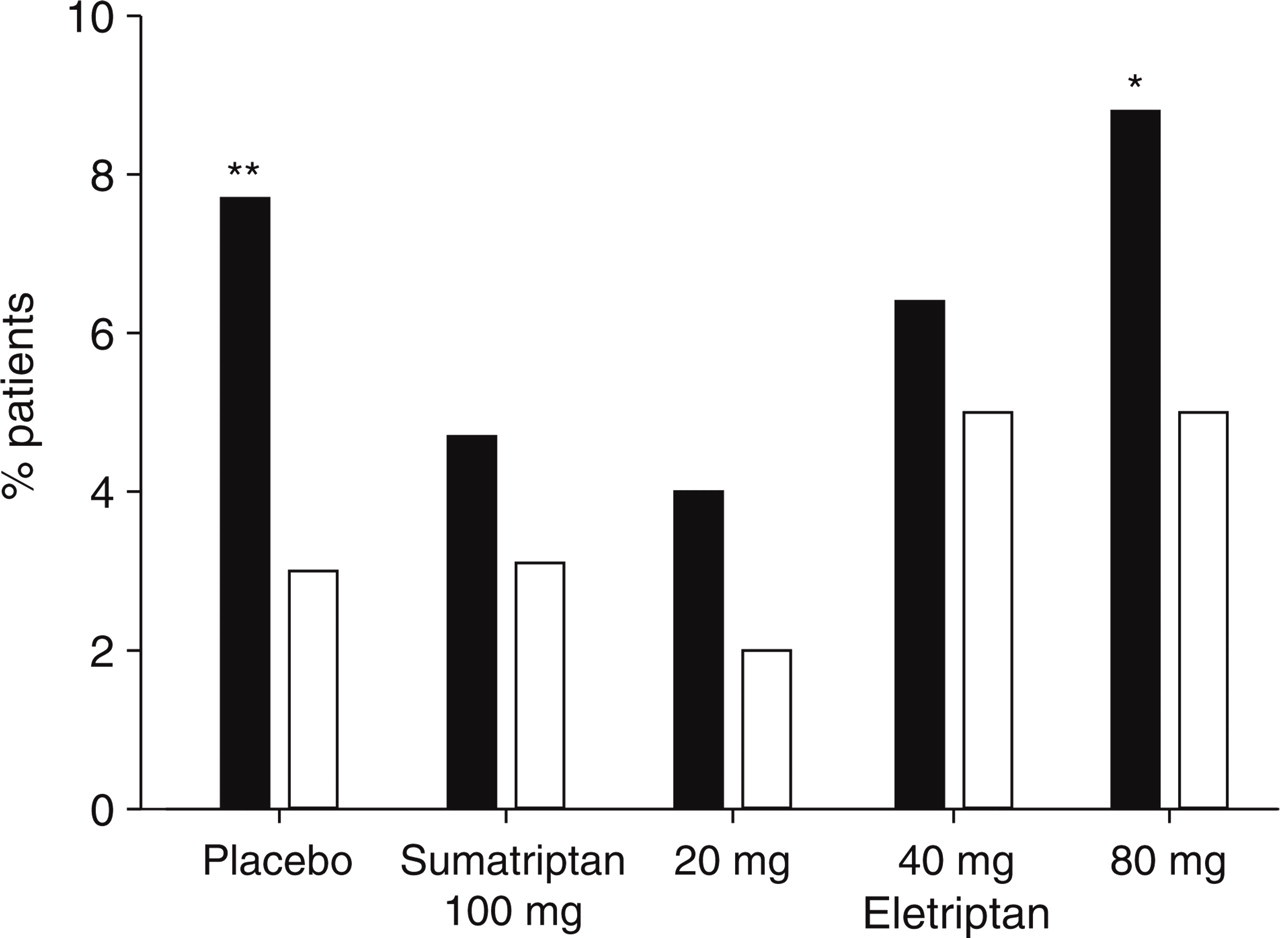
Somnolence after dosing (ordinate) with either placebo, sumatriptan 100 mg or eletriptan 20 mg, 40 mg or 80 mg in randomized controlled trials. There is a significantly increased incidence of somnolence in the placebo and eletriptan 80 mg groups when compared with responders (▪) and non-responders (□) (∗P < 0.05; ∗∗P < 0.01).
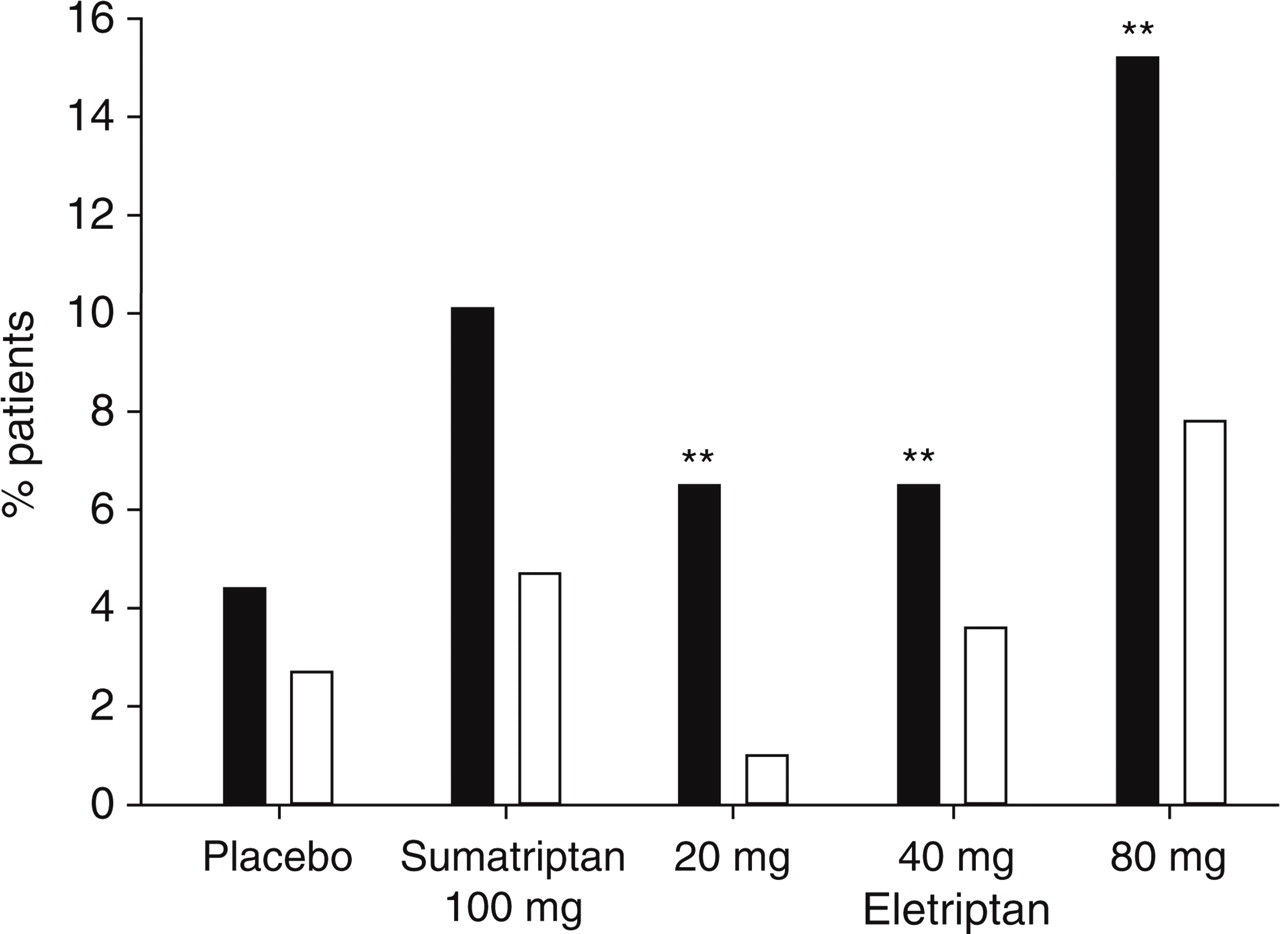
Asthenia after dosing (ordinate) with either placebo, sumatriptan 100 mg or eletriptan 20 mg, 40 mg or 80 mg in randomized controlled trials. There is a significantly increased incidence of asthenia in each eletriptan group when compared with responders (▪) and non-responders (□) (∗P < 0.05; ∗∗P < 0.01).

Combined somnolence/asthenia after dosing (ordinate) with either placebo, sumatriptan 100 mg or eletriptan 20 mg, 40 mg or 80 mg in randomized controlled trials. There is a significantly increased incidence of combined somnolence/asthenia in each eletriptan group when compared with responders (▪) and non-responders (□) (∗P < 0.05; ∗∗P < 0.01).
ORs (odd ratios for headache responders relative to non-responders) for the two index CNS adverse events are summarized in Table 2. The odds of developing somnolence achieved significance for placebo (OR 2.7) and eletriptan 80 mg (OR 1.8). The odds of headache responders developing somnolence was higher for eletriptan 20 mg (2.0), but the smaller sample size reduced the ability to detect significance at this OR. Similar trends were observed for asthenia, with higher ORs observed across all study treatments (ranging from 1.6 to 2.3). For both CNS events combined, ORs were significant for all study treatments except sumatriptan. For the three doses of eletriptan, there was a step-wise increase in the incidence of CNS adverse events (AEs) (combined), both in the non-responder subgroup and, at a higher rate, among responders. There is a suggestion of a U-shaped curve, with the lowest OR occurring at the 40-mg dose of eletriptan. The placebo treatment group provided the strongest data regarding whether emergent CNS events are treatment related or disease related. Compared with non-responders, the OR of developing a CNS AE was 2.3-fold higher in patients who achieved a response at 2 h on placebo (Table 2).
Similar trends among patients reporting any CNS AE were observed for pain free at 2 h (vs. not pain free), with higher ORs on eletriptan 40 mg, 1.5 (95% confidence interval 1.1, 2.1; P = 0.006); eletriptan 80 mg 1.5 (1.1, 1.9; P = 0.008); sumatriptan 100 mg 1.8 (0.8, 4.2; P = 0.17); and placebo 1.7 (0.7, 4.6; P = 0.23), but not eletriptan 20 mg 0.9 (0.3, 2.9; P = 0.9).
Headache response and somnolence or asthenia
An additional analysis of variance was performed using treatment and headache response as independent variables. The
Finally, we evaluated headache response and pain-free rates at 2 h for patients who experienced relative to patients who did not experience a CNS AE. As expected, headache response at 2 h was consistently higher in patients reporting treatment-emergent occurrence of a CNS event (Table 4). Similarly, higher pain-free rates were observed at 2 h in patients reporting treatment-emergent CNS AEs (Table 4). Unfortunately, data are not available on the time of onset of CNS events.
Discussion
The analysis of the data from seven large placebo-controlled trials suggests somnolence and asthenia occurring with eletriptan treatment result, at least in part, from processes that are involved in the natural resolution of the attack. This conclusion is strengthened by the fact that effective placebo treatment can result in somnolence and asthenia, and that placebo responders have higher rates of CNS symptoms than placebo non-responders. These data do not exclude that some part of the somnolence observed with eletriptan, or indeed other triptans, is a CNS side-effect, but it does suggest caution in attributing the causality of adverse events in acute migraine studies. The data raise questions as to whether other triptans, or indeed other acute therapies produce these symptoms, some of which may be seen in the premonitory phase of the attack (23). Moreover, the data raise the interesting question of what brain mechanisms may be at work in producing these symptoms as the acute attack is resolved?
It has been widely accepted since the first triptan studies were published with subcutaneous sumatriptan that increasing doses of a triptan would produce increasing side-effects (24). Efficacy with triptans other than eletriptan comes with the price of increased somnolence and asthenia; however, in general terms, without pooling of large datasets one can make only qualitative observations. Asthenia and somnolence rates are double or more for rizatriptan compared with placebo (25–27). Moving to ergotamine, a large placebo-controlled modern standard study has suggested that it was more likely to produce asthenia and somnolence than placebo (19), even though it was compounded with caffeine as it was tested. More interesting are medicines not generally considered to have such CNS effects. In a double-blind, placebo-controlled study of diclofenac in the acute treatment of migraine, both asthenia and somnolence were numerically more evident in the diclofenac-treated compared with the placebo group (28). Diclofenac was also more effective than placebo in the treatment of acute migraine. In a double-blind, placebo-controlled study, rofecoxib was more effective in the treatment of acute migraine and produced numerically more somnolence than placebo (29). One can imagine two control experiments: if triptan-induced somnolence is prominent in migraine because it is part of the resolution of the acute attack, does it occur in other uses of the same medicine? Considering sumatriptan or zolmitriptan in the treatment of acute cluster headache either as sumatriptan subcutaneous injections (30), or nasal spray (31), or zolmitriptan as nasal spray (32) or tablet (33), no excess of somnolence or asthenia is reported. Perhaps these studies are too small to detect the change. As a positive control with a larger sample size, COX-II inhibitors do not produce more asthenia or somnolence than placebo in the treatment of arthritis (34), albeit used in continual dosing. Taken together, admitting the imperfections arising from sample size issues and the difficulties of using patient-provided as opposed to physician-elicited AE data, the broad message is that effective treatments of acute migraine produce somnolence and asthenia, be they triptan or non-triptan, while the same therapies used for other headache or pain indications do not. What might be the mechanism of this potential difference?
Migraine is a complex disorder, likely to be fundamentally a disorder of the brain (1). It is clear, since not all attacks are treated, either because it is the very first or for the myriad reasons patients do not have treatments available, that the natural history of the acute attack is to resolve. Indeed, this is the basis for the need for placebo control in acute studies. In a typical study with a design such as those we have analysed here, the 2-h rate for a reduction from moderate or severe headache to nil or mild headache is typically about 30% (6). This natural history typically involves the patient wishing to withdraw from the surroundings and, especially in children, to sleep. Brain imaging studies have consistently demonstrated activation in the dorsolateral pons in acute migraine exacerbations (35–39). One structure in this region which is attractive in terms of being a site for dysfunction in migraine is the brain's main repository of noradrenaline, the nucleus locus coeruleus (40). This same region is involved in pain modulation (41), sleep (42), attention (43), satiety with its orexinergic input from the posterior hypothalamus (44) and brain blood flow (45). Taken together, the new data from our analysis, combined with the latest data from brain functional imaging in migraine, suggest the possibility that by facilitating the termination of an attack, successful acute therapies will unmask processes, such as somnolence, that are part of the underlying physiology of the resolution of the attack.
An analysis of so-called CNS side-effects of eletriptan demonstrates that they are seen in patients with migraine treated with placebo, if they are responders, and are more common in eletriptan-treated responders than in non-responders. The traditional view of adverse events needs to carefully consider the possibility that some symptoms associated with drug treatment reflect patients reporting accentuated aspects of the natural history of their attack. This does not suggest that adverse events seen with eletriptan, or any triptan, cannot or are not caused by the medicine, but simply that such a link needs to consider how the treatment itself might interact with the natural history of the disorder itself.
Acknowledgements
This work was supported by Pfizer. The research question was conceived by the academics (P.J.G., D.W.D., R.B.L., H-C.D., P.T-H.) and the manuscript prepared independent of the sponsor by the first author except for the methodological section, where M.A. had a more substantial role. Pfizer collated the database and ran the statistical analysis (M.A.). The data, including structure of original diary cards, and the primary data output were freely available to each of the investigators.
