Abstract
Background and objective
Somatosensory temporal discrimination was recently reported as prolonged during migraine attacks, which is consistent with disrupted sensorial perception in migraine. However, knowledge about central sensory processing in tension-type headache is still lacking. This prospective, controlled study aimed to investigate somatosensory temporal discrimination thresholds in tension-type headache.
Methods
The study included 10 tension-type headache patients, 10 migraine patients and 10 healthy volunteers without headache. Somatosensory temporal discrimination thresholds were evaluated during the headache attacks of tension-type headache and migraine patients.
Results
Somatosensory temporal discrimination thresholds of tension-type headache patients (39.0 ± 5.5 ms for the right hand and 40.6 ± 4.6 ms for the left hand) were significantly lower than those of episodic migraine patients (137.1 ± 35.8 ms for the right hand and 118.4 ± 34.3 ms for the left hand, p < 0.0001 and p < 0.0001 respectively), and comparable to those of healthy volunteers (38.6 ± 5.3 ms for the right hand and 38.3 ± 7.2 ms for the left hand, p = 0.79 and p = 0.45 respectively).
Conclusion
Central sensory processing, as tested by somatosensory temporal discrimination, was remarkably disrupted during the headache attacks in migraineurs, whereas it remained intact in the tension-type headache patients.
Keywords
Introduction
Somatosensory temporal discrimination (STD) evaluates the temporal threshold to distinguish two separate stimuli applied to the same region (1,2). The STD threshold (STDT) gives information about the sensory stimulus processing of the brain in differentiating the accurate entry of each external stimulus (3). The integrity of both peripheral and central sensory pathways is essential for the cortical processing of STD.
Migraine headache attacks are accompanied by characteristic symptoms of sensorial dysfunction. Tactile, visual, auditory and olfactory senses are all altered during the migraine attack and generate cutaneous allodynia, photophobia, phonophobia and osmophobia (4–8). These symptoms are independent from trigeminal nerve input (8) and reflect an altered cerebral cortical processing of sensorial information. Consistent with the latter notion, we recently demonstrated that STDT values were significantly prolonged during the headache attack and returned to normal limits interictally in migraine patients (10).
On the other hand, in tension-type headache (TTH), headache features do not match migraine criteria and sensory disturbances such as photophobia and allodynia are not prominent (4). TTH is still one of the most misdiagnosed disorders in patients with probable migraine, chronic migraine and medication overuse headache. Because of these diagnostic difficulties, TTH prevalence varies between 5.1–78% (11,12). In this study, we aim to investigate whether the disrupted sensory perception processes are shared phenomena of both migraine and TTH, or if STDT elevation is specific to migraine, by evaluating STD thresholds during the headache attacks of both migraine and TTH disorders.
Methods
Participants
Episodic tension-type headache patients and episodic migraine patients were consecutively recruited from the Gazi University Faculty of Medicine, Department of Neurology, headache outpatient clinic. Inclusion criteria for episodic tension-type headache patients were: 1) age between 18 and 65 years; 2) at least eight years of formal education; 3) a definite diagnosis of frequent episodic tension-type headache according to the International Classification of Headache Disorders 3rd edition – beta version (ICHD-3 beta) (4); 4) no prior history of probable or definite migraine attack; 5) 1–14 attacks per month; 6) no use of any preventive medication or any other regular medication; 7) agreement to be evaluated during the first six hours of a TTH attack without taking any analgesics until the end of the STD test. Inclusion criteria for the episodic migraine without aura patients were: 1) age between 18 and 65 years; 2) at least eight years of formal education; 3) diagnosis of definite migraine (episodic migraine without aura) according to ICHD-3 beta (4); 4) 1–14 attacks per month; 5) no use of any preventive medication or any other regular medication 6) agreement to be evaluated during the first six hours of a lateralised migraine attack without taking any abortive medication until the end of the STD test. Inclusion criteria for healthy volunteers were: 1) age between 18 and 65 years; 2) at least eight years of formal education; 3) no history of headache 4) no history of any neurological or chronic disease.
Exclusion criteria for episodic tension-type headache patients were concurrence of different headache types, ≥15 headache attacks per month, medication overuse, a history of alcohol/drug abuse, or any chronic disease requiring daily treatment. Exclusion criteria for migraine without aura headache patients were coexistence of different headache types, ≥15 headache attacks per month, medication overuse, migraine aura, bilateral migraine attack, history of alcohol/drug abuse, or any chronic disease requiring daily treatment in the medical history.
The patients and the healthy volunteers were recruited prospectively between January 2016 and April 2016, and included in the study by headache specialists (HB, OC) after a detailed clinical evaluation and neurological examination.
Study design
STDTs were measured at the dorsum of the hands bilaterally (dermatome of C7) once for all three groups. STDT measurements were made once during the headache attack period in the episodic tension-type headache and episodic migraine without aura groups.
Disease duration, pain intensity (scored before testing with the 10-point visual analogue scale (VAS)), clinical features during the headache attack such as nausea, photophobia, phonophobia, osmophobia, and pressure pain thresholds (PPTs) measured by algometer were also recorded before the measurement of STDTs. PPT is the minimum amount of pressure that produces pain. PPTs, to identify tender points if present, were assessed over both temporal muscles and greater occipital nerves (approximately 2 cm lateral and 2 cm inferior to the external occipital protuberance) with the pressure algometer (Commander Algometer, JTECH Medical), which consisted of a 1 cm2 hard rubber tip. The probe of the algometer was compressed against the evaluated area slowly to allow the subject time to react when pain was felt. The instrument was calibrated before each measuring session. Brush allodynia was not detected in the hands before STD evaluation.
STD procedure
STD measurements were performed as previously reported (10). Electrodes were placed on the dorsum of the hand after cleaning the skin with alcohol. Ag-AgCl skin electrodes (10 mm diameter surface) were used and the anode was placed 1 cm distal to the cathode. The tactile stimulation was done with a constant current stimulator (Medtronic, Keypoint). The duration of the square wave electrical pulse was 0.2 ms. The intensity of the stimulation was increased, starting from 1 mA with steps of 0.2 mA to determine the minimal intensity of stimulation. The measurement of minimal intensity of stimulation was repeated three times and the minimum intensity that was felt consistently was recorded as the minimal stimulation threshold. The maximum stimulus intensity was preset at 10 mA. Paired stimuli were delivered at intervals of 10 seconds with a starting interstimulus interval (ISI) of 5 ms, and ISI was increased in 5 ms steps. The ascending STDT (aSTDT) was the first of three consecutive ISI where the subjects discriminated two temporally discrete stimuli. Then ISI was decreased with steps of 5 ms until the patient recognised paired stimuli as one stimulus three times consecutively. The first of three consecutive ISIs was recorded as descending STDT (dSTDT). This process was repeated with a stimulus intensity of 1.5 times minimal sensory threshold. The arithmetic mean of the aSTDT and dSTDT at minimal sensory threshold and the aSTDT and dSTDT at 1.5 times minimal sensory threshold was calculated and recorded as the STDT value for that hand. A single stimulus was given between each three-paired stimuli to test the subject’s attention. STDT values were recorded from right and left hands in all groups. Also STDT values were recorded, according to headache lateralisation, as ipsilateral hand and contralateral hand in migraine patients. STDT values were measured by a blinded neurologist (DV). No adverse reactions were observed during the study period.
Approvals and patient consents
The study protocol and procedures conformed to the standards set by the Declaration of Helsinki and the local ethics committee approved the study (E-16-850). Each patient and healthy volunteer was included in the study with his or her approval, and written informed consent was obtained from the participants.
Statistical analysis
Data analysis was carried out in the SPSS 15.0 for Windows software. Descriptive statistics were indicated as mean ± standard deviation, while categorical variables were reported as frequency and percentage values (%). Categorical variables were analysed by Pearson’s chi-square test. Normalisation of data was investigated by Kolmogorov-Smirnov test. STDT and PPT data between all three groups were analysed by one-way ANOVA, and STDT and PPT data between two independent groups were analysed by independent t test. The paired t test was used to compare contralateral and ipsilateral STDT and PPT values in migraine patients. Pearson’s correlation analysis was used to evaluate a possible correlation between the STDT value and the clinical features of migraine patients including age, VAS score, PPTs and the presence of nausea, photophobia, phonophobia and osmophobia. Sample sizes were based on past experience (10), and no prior statistical power analysis was done. P < 0.05 was considered statistically significant.
Results
Thirty-four subjects who agreed to participate in the study and met the inclusion criteria were evaluated between January 2016 and April 2016. One tension-type headache, one healthy volunteer and two migraine without aura subjects could not complete the study since they failed to report the correct answer when a single stimulus was given to test their attention. Ten episodic tension-type headache, 10 episodic migraine without aura patients and 10 healthy volunteers completed the study.
Demographic features of healthy volunteers, tension-type headache and migraine patients, clinical features and PPTs of patients during headache attacks before STD measurements.

† One-Way ANOVA, ‡ Pearson’s Chi-Square test, ¶ Independent t test, # Paired t test.
VAS: Visual analogue scale.
PPT: Pressure pain threshold.
GON: Greater occipital nerve.
There was a statistically significant difference between healthy volunteers and migraine group (p < 0.0001). bThere was a statistically significant difference between the tension-type headache and migraine group (p < 0.0001).
There was a statistically significant difference between healthy volunteers and the tension-type headache group (for the right temporal muscle p = 0.013, for the left temporal muscle p = 0.017, for the right GON p = 0.008, for the left GON p = 0.005). P< 0.05 are written in bold.
PPTs in healthy volunteers were significantly higher than in both TTH and migraine patients, and PPTs in migraine patients were also significantly lower than in TTH patients. PPTs over ipsilateral temporal muscle and GON were lower than on the contralateral side in migraine patients. PPT values of healthy volunteers, tension-type headache and migraine patients are summarised in Table 1. No statistically significant difference was found between the minimal stimulus thresholds of the three groups. There was no statistically significant difference between healthy volunteers and the TTH group according to STDTs of the right hand (38.6 ± 5.3 ms vs 39.0 ± 5.5 ms, p = 0.79) and left hand (38.3 ± 7.2 ms vs 40.6 ± 4.6 ms, p = 0.45) (Figure 1). In the migraine group, ictal STDTs were significantly higher (Figure 1) than ictal STDTs of the TTH patients and STDTs of healthy volunteers (137.1 ± 35.8 ms vs 39.0 ± 5.5 ms vs 38.6 ± 5.3 ms for the right hand, p < 0.0001 and 118.4 ± 34.3 ms vs 40.6 ± 4.6 ms vs 38.3 ± 7.2 ms for the left hand, p < 0.0001). Ictal STDTs of the hand contralateral to the attack hemisphere (155.8 ± 22.0 ms) were significantly higher than the ipsilateral hand (99.8 ± 20.7 ms) in migraine patients (p < 0.0001). STDT values of healthy volunteers, tension-type headache and migraine patients are presented in Table 2.
STDT values obtained from both hands (C7 dermatome) in healthy volunteers, TTH and migraine groups. STDT values in TTH patients were comparable with healthy volunteers and were below 50 ms. It is notable that STDT values were elevated in the migraine group. STDT values of the contralateral hand were significantly increased compared to those of the ipsilateral hand in migraine patients. STDT values of healthy volunteers, tension-type headache and migraine patients. STDT: Somatosensory temporal discrimination threshold. One-Way ANOVA; ¶ Paired t test. There was a statistically significant difference between healthy volunteers and the migraine group (p < 0.0001). There was a statistically significant difference between the tension-type headache and migraine group (p < 0.0001).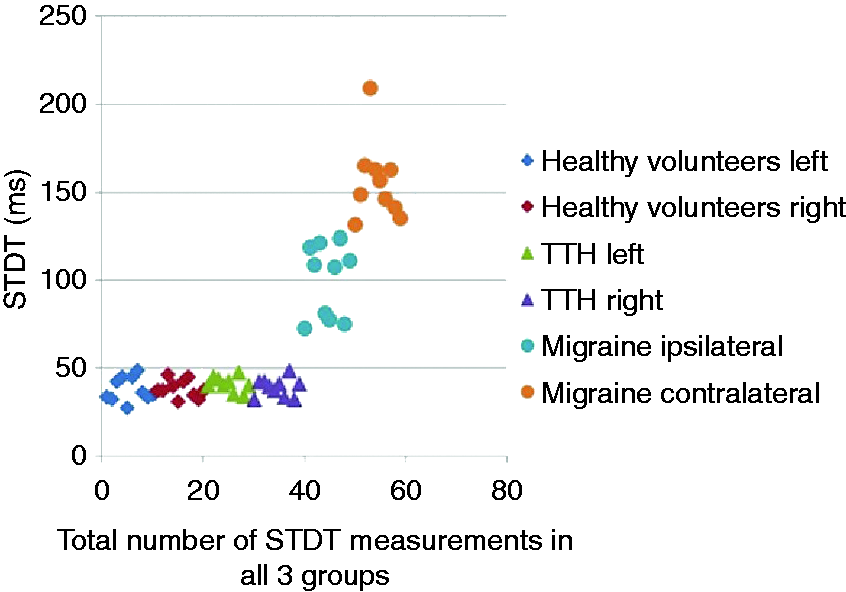

Correlation analysis
The visual analogue scale scores of the migraine patients were positively correlated with the contralateral STDT values (r = 0.85, p = 0.002). No correlation was detected between STDTs and age, gender, hand preference, disease duration, headache frequency, PPTs and clinical features such as phonophobia, photophobia, osmophobia and nausea.
Discussion
We have demonstrated for the first time that higher sensorial processing of two separate somatosensory stimuli remains intact during TTH, while it is disrupted during a migraine headache attack. The mean temporal discrimination threshold value is approximately 30 milliseconds in healthy individuals under the age of 65 (1). STDT values obtained from healthy volunteers and patients with TTH attacks were comparable, though they were significantly prolonged during migraine attacks. Elevated STDT values in migraine attacks were compatible with our previous report (10). Impaired central processing of somatosensory stimulation provides an important electrophysiological tool to differentiate migraine attacks from TTH attacks.
The diagnoses of migraine and other primary headache disorders depend solely on the history obtained from the patient. TTH is considered to be the most frequent headache worldwide, with highly variable prevalence rates in different studies. Tension-type headache is commonly confused with a mild form of migraine without aura and probable migraine. Therefore, the IHS classification committee attempted to tighten the diagnostic criteria for tension-type headache to exclude migraine patients. However, pure TTH without any prior migraine headache attacks is indeed a rare disorder, reported to have a prevalence rate of 5.1% (11).
The intact central sensory stimulus processing in TTH attacks is a novel finding. TTH is one of the least studied primary headache disorders, and pathophysiological mechanisms are not clear compared to those of migraine. Pericranial tenderness is a clinical sign of central sensitisation, and is associated with both TTH and migraine (13–15). Tenderness over temporal muscles and GON were recorded in all of our patients. While central mechanisms are involved in both headache disorders, and decreased PPT values may show sensitisation of second-order neurons at the level of the spinal dorsal horn and trigeminal nucleus, sensitisation of supraspinal neurons, and decreased antinociceptive activity from supraspinal structures, STD is disrupted only in migraine attacks. It is remarkable that STDT differentiates the central pathology of migraine from TTH.
The elevated STDTs obtained from bilateral C7 dermatomal areas highlight the interference of sensory stimuli processing with migraine pain in the central structures. The results also denote that sensorial cognitive processing of two stimuli from the contralateral hand was more significantly disrupted in a manner specific to migraine attack. Moreover, STDTs from the contralateral hand are only positively correlated with the severity of migraine pain. On the other hand, STDTs were not correlated with age, disease duration, headache frequency, hand preference, PPTs, phonophobia, photophobia, osmophobia, and nausea. A recent report also showed that cross-modal sensory perception requiring precisely-timed processing of sensorial inputs was impaired during migraine attacks (16).
Our finding suggests that STD prolongation in the migraine attack is unlikely to be a nonspecific companion of the headache perception and headache intensity; instead, it seems specific to the migraine attack, as the mean VAS values were not significantly different between migraine and TTH groups. Consistent with our previous study findings (10), STDT values were significantly higher in migraine patients even during mild migraine headache attacks. STDT values of TTH and migraine patients having the same VAS score were clearly distinct and no overlap was observed between STDT values of the two groups (Figure 1). Furthermore, we also detected significant prolongation of STD during bilateral migraine attacks (17). Since there is a clear difference in STDT values between the TTH and migraine groups, with TDTs elevated by four times in the contralateral hand in the migraine group, STD may be employed in differentiating migraine from TTH. STDT values obtained from the hand contralateral to the attack hemisphere were significantly higher than that of the hand ipsilateral to the attack hemisphere in migraine patients. The most significant disruption of sensorial information from the hand contralateral to the attack hemisphere during migraine pain confirms our previous findings, which revealed notable affection of STD in the contralateral hand and ipsilateral face (10). Such differential involvement of sensorial processing in migraine attacks needs further consideration. These findings imply that according to the attack hemisphere of a migraine headache, the spinal lemniscus transmitting information from the contralateral hand and the trigeminal lemniscus transmitting information from the ipsilateral face are preferentially affected. The only close proximity of those two pathways is in the mid-pons region ipsilateral to the attack hemisphere, before the trigeminal lemniscus cross over. It is important to note that the area matching where the trigeminal lemniscus travels near the spinal lemniscus in the pons region has already been implicated in migraine attacks by neuroimaging techniques (18).
Even though STD requires higher cognitive processing, a circuit of prefrontal cortical areas, thalamus and cerebellum connected via the pons plays a critical role in temporal processing of somatosensory stimuli (19). In addition, the involvement of the cortical centres for pain perception, such as the insula and anterior cingulate cortex, basal ganglia, mid-brain and the brain stem, are revealed during migraine pain (19–23). Understanding the pathways and structures contributing to STD is important not only to uncover the migraine-specific pathophysiology but also to distinguish the features of migraine from other primary headaches. Recently, a central route from the cerebral cortex to the ipsilateral trigeminal nucleus caudalis was also implicated, in addition to the well-known peripheral route, in experimental animal studies (24–26). The dysfunction of the cortex activating the ipsilateral trigeminal pain nucleus through the subcortical and brain stem structures was proposed to be the predominant route only in fully awake subjects (24). By re-interpreting the above results, greater STD prolongation preferentially in the hand contralateral to and the face ipsilateral to the attack hemisphere in migraine patients might indicate transient dysfunction of a central route from the somatosensory cortex down to the mid-pons region.
The strengths of the study are 1) that it confirmed the original findings of STD prolongation during migraine headache attacks without aura; 2) that it showed the specificity of STD disruption to migraine attacks compared to TTH; 3) it included patients with pure TTH attacks without any prior definite or probable migraine headaches; 4) there was no interference of preventives or abortive medications during the STD test.
Its limitations are 1) that it was conducted in a small of number of patients, due to the clear difference between the two primary headache groups; 2) to reduce the time spent on the test and increase the cooperation of the patient with headache, STDTs were not measured in the face, and 3) the length of time of STD prolongation from the initiation of an attack is unclear.
In conclusion, thresholds for central processing in discrimination of somaesthetic stimuli were significantly higher in migraine attacks, whereas STDTs were normal in TTH attacks. Despite the presence of pericranial tenderness in all patients, reflecting central sensitisation, STDTs remained intact in TTH but disrupted in migraine. The study provides evidence that STD may have a diagnostic value in differentiating migraine without aura from TTH for the first time.
Key findings
Somatosensory temporal discrimination (STD) remain intact in tension-type headache (TTH) whereas it is remarkably disrupted in migraine attack, even though decreased pressure pain thresholds are present and central mechanisms are involved in both headache disorders. Impairment of STD during the migraine attack does not seem to be a nonspecific companion of the headache perception. STD thresholds are clearly distinct between the migraine and TTH attacks. STD prolongation is specific to the migraine attacks.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
