Abstract
Introduction
The involvement of the serotonergic system of the brainstem raphe in the pathogenesis of migraine is discussed. Here we studied brainstem alterations in migraineurs using transcranial sonography and examined their relation to clinical features and self-medication.
Methods
We investigated 51 migraineurs (11 men, 40 women, mean age 29.7 ± 11.9 years) and 32 healthy individuals without history of headache or depressive disorder (eight men, 24 women, mean age 34.4 ± 13.0 years). Transcranial sonography was performed in an investigator-blinded fashion. Midbrain raphe echogenicity was quantified using digitized analysis. Migraine characteristics and the use of analgesics were evaluated by applying validated questionnaires. Eight migraineurs underwent neurophysiologic evaluation of contingent stimulus-related cortical potentials.
Results
Echo-reduced midbrain raphe was detected in 27 (53%) migraineurs, but only six (19%) control subjects (odds ratio = 4.87, p = 0.002). Lower raphe echogenicity correlated with both higher amplitude of terminal contingent negative variation (Spearman test, r = 0.76, p = 0.028) and higher use of analgesic drugs (r = −0.45, p = 0.011), but not with use of triptans or with migraine frequency or severity (all p > 0.2). Compared to migraineurs without aura, migraineurs with aura had enlarged third ventricles (t-test, p = 0.014), while the lateral ventricle widths did not differ (p = 0.62).
Conclusions
Midbrain raphe alteration is frequent in migraineurs and relates to self-medication behavior. This alteration may reflect the dysfunction of serotonergic raphe nuclei.
Introduction
Migraine is the most frequent neurological disorders in the adult population worldwide, affecting up to 12% of the general population (1). The current concepts of this disorder focus on a dysfunction of the descending pain-inhibitory system of the brainstem, leading to hyperexcitability in nociceptive areas (2). In particular, the serotonergic system emerging from the brainstem raphe nucleus has been implicated in migraine pathophysiology, and it has been suggested that a low serotonin state facilitates activation of the trigeminovascular nociceptive pathway, as induced by cortical spreading depression (CSD) (1). Abnormal cortical information processing of sensory stimuli has been proposed as a predisposing factor for migraine via CSD (3,4). In neuroimaging studies a number of morphologic, functional and metabolic alterations of brainstem structures can be linked to migraine, some of them showing a correlation to the duration of the disorder or its severity (5,6). In addition, iron accumulation in the basal ganglia was reported in episodic migraineurs, especially those with higher attack frequency and longer disease duration (7). However, it remains unclear whether these alterations of deep brain structures are caused by the disorder or cause it, and how the different aspects of the pathophysiology influence each other.
Transcranial sonography (TCS) has proved to be reliable in detecting alterations of the midbrain raphe (MBR), substantia nigra and basal ganglia in distinct neurodegenerative disorders, and can sensitively visualize trace metal accumulation (8). For example, TCS studies have consistently demonstrated increased echogenicity (‘hyperechogenicity’) of the substantia nigra in Parkinson’s disease, and of the lenticular nucleus in idiopathic dystonia, but this has not been validated against another imaging measure (8). In depressive disorders TCS has revealed reduced echogenicity (‘hypoechogenicity’) of the MBR, which has been linked to the rarefaction of crossing fiber tracts at the raphe in post-mortem studies, and to alteration of the central serotonergic system (8–10). The discovery of MBR hypoechogenicity in depressive states has been replicated by numerous independent studies (11). Here, we tested the hypothesis that hypoechogenic alteration of the MBR is frequent in migraineurs. In addition, we wanted to assess whether changes of deep brain structures relate to clinical and neurophysiologic characteristics of migraine, and to the use of analgesic medication.
Methods
Participants
Of the 51 migraineurs (11 men, 40 women, mean age 29.7 ± 11.9 years) who participated in this cross-sectional study, 42 were recruited between April 2014 and February 2015 through an announcement sent via email to all students at the University of Rostock. The remaining nine were patients at the Department of Medical Psychology and Sociology, being trained in cognitive-behavioral pain management. The diagnosis of migraine, according to the International Classification of Headache Disorders (ICHD) diagnostic criteria, had to be confirmed by an expert experienced in the diagnosis and treatment of headache disorders (12). The exclusion criteria were (i) a history of neurological disorder other than the primary headache, (ii) a history of severe head trauma, and (iii) a history of depressive disorder involving medical consultation or treatment. For comparison, 32 healthy control subjects (students and hospital staff members) were included in this study (eight men, 24 women, mean age 34.4 ± 13.0 years), all without a history of episodic or chronic headache, or psychiatric disorders. For each participant the clinical and TCS assessments took place on the same day. The study was approved and registered by the ethics committee at Rostock University (identifier: A-2014-0167), and written informed consent was obtained from each study participant.
Clinical investigations
A validated questionnaire that assessed the pain frequency (recurrent versus permanent), aura symptoms, triggering factors and headache symptoms (13) was used to assign the migraine and tension-type headache diagnoses and record the features of the individual disorder, as well as current medication or self-medication. The data on the history and symptoms of headache were assessed by experts experienced in the diagnosis and treatment of headache disorders. To evaluate allodynia, the migraineurs were asked to rate their agreement with the phrase ‘Combing my hair hurts’ on a scale from 1 to 5. The migraine impact was assessed using the Migraine Disability Assessment Score (14). This scores the days with headache in the past three months, and the severity of the pain, on a visual analogue scale from 1 to 10. All the migraineurs completed the Beck Depression Inventory (BDI) (15).
Transcranial sonography
TCS was performed through the preauricular acoustic bone window using a diagnostic 2.5 MHz phased-array ultrasound system (Acuson Antares, Siemens, Erlangen, Germany) (8,10). The system settings were applied as follows: penetration depth 16 cm, dynamic range 50 dB, post-processing preset G. Migraineurs and control subjects were assessed on TCS in random order by an examiner with 15 years’ experience in TCS (UW), who was blinded to the diagnostic and clinical data. The echogenicity of the MBR was rated visually on a semiquantitative scale ranging from grade 1 to grade 3. Typically, the MBR presents as a clearly visible median line that is highly echogenic compared to the adjacent tissue (grade 3). It can also produce a less intense echo than usual, or the line can be interrupted (grade 2). Sometimes the raphe structures are not identifiable at all despite the clear visibility of red nuclei and basal cisterns (grade 1) (10). Grades 2 and 1 are considered to be abnormal (hypoechogenic). Further brain structures were studied in an exploratory fashion, directed according to the standard TCS protocol applied in neurodegenerative disorders (8). For assessment of the substantia nigra, the echogenic size was calculated automatically according to manually encircled boundaries in an axial scan plane (8). The greater value of the two (bilateral) echogenic sizes was used for further analysis. For the ultrasound system used here, substantia nigra echogenic sizes of <0.24 cm2 are considered normal, and sizes >0.24 cm2 (upper 25% percentile in a normal population) are considered hyperechogenic. The echogenicity of the red nuclei, lenticular nuclei and heads of caudate were rated as described earlier (8). The widths of the third ventricle and the frontal horns were measured at a standardized axial scanning plane. In addition, an off-line digitized analysis of the anonymized TCS images was performed by a second reader (AR) in order to obtain investigator-independent echo intensity values for MBR, using previously validated MATLAB-based software (16).
Neurophysiologic investigation
Eight migraineurs (one man, seven women, mean age 40.2 ± 19.2 years) underwent assessment of contingent negative variation (CNV), a slow cortical potential recorded between two contingent stimuli (3,4). The demographic and clinical data for these subjects did not differ from the data for the other migraineurs (all p > 0.1). To analyze the CNV, the electroencephalogram was recorded over Cz with linked mastoids as a reference with an electrode resistance of approximately 6 kOhm. The electroencephalogram (bandpass: 0.03–35 Hz) was digitized with a sampling rate of 100 Hz for each channel. All the participants were seated with their eyes open in an armchair located in an electrically shielded, sound-proof room. The auditory warning (S1) and imperative stimulus (S2) were produced by a loudspeaker located behind the subject with an intensity of 75 dB. The interval between S1 and S2 was 3 s. The CNV recording consisted of 32 trials in which the subjects had to react to the imperative stimulus (GO response). To keep the subjects in a vigilant state, eight trials were randomly presented where no reaction was expected (NO-GO response). S1 for the GO response was a 1000 Hz tone which lasted 100 ms. S1 for the NO-GO response was a 200 Hz tone. S2 (f = 2500 Hz) lasted a maximum of 1500 ms and was deactivated by pressing a key. The length of each CNV recording was 6 s, with a randomized interval of 6–10 s between trials. Recordings started 1 s before S1 and ended 2 s following the onset of S2. NO-GO trials were not analyzed. In addition, we recorded a vertical electrooculogram; CNV recordings containing eye blinks or artifacts were discarded. Due to the habituation processes during rejected intervals, a protocol listed the number of rejected trials for each session. The number did not exceed a maximum of two. The periods between the recording onset and S1 were used as the baseline for each trial. The CNV session was finished after 40 artifact-free trials (32 GO, eight NO-GO trials). The 32 GO trials of each CNV session were grand averaged. The overall CNV (oCNV) was the grand-averaged mean amplitude between the onsets of S1 und S2. The initial component (iCNV) was defined as the average amplitude in a window of 200 ms around the maximal amplitude of the expectancy wave between 550–750 ms after S1 (3). The terminal CNV (tCNV) was the average amplitude during the 200 ms preceding S2. The postimperative negative variation (PINV) was the average amplitude of CNV between 500 and 2000 ms following S2. To exclude the effects of the migraine attack on the CNV amplitudes and habituation, we controlled the occurrence of an attack and omitted recordings within three days before or after an attack (4).
Statistical analyses
For the correlation of categorical data, Spearman’s non-parametric rank correlation was used. For the comparison of categorical variables, the chi-square test was used; for the comparison of non-normally distributed data, the Mann-Whitney U test was used; and for the comparison of means, the t-test for independent samples was used. Since the TCS findings were compared with five clinical domains (age, migraine duration, migraine severity, depression severity and use of analgesics), a Bonferroni correction for multiple correlations was employed by dividing a p value < 0.05 by 5; thus a value of p < 0.01 was taken here to be significant for group comparisons. The analyses were performed using SPSS 20 (IBM Corp., Armonk, New York, USA).
Results
Clinical findings
Clinical and TCS findings in migraineurs and healthy control subjects.

p: significance (significant values in bold).
chi-square test. 2t-test. 3Mann-Whitney U test.
CN ↑ denotes increased echogenicity of the caudate nucleus; DA, digitized image analysis; DD, disease duration; HDs, headache days; LN ↑, increased echogenicity of the lenticular nucleus; LV, width of the frontal horn of the lateral ventricle; MBR ↓, reduced echogenicity of the midbrain raphe; MIDAS, Migraine Disability Assessment Score; RN ↕, abnormal (reduced or increased) echogenicity of the red nucleus; SN, the substantia nigra; TTH, tension-type headache; VAS, pain severity on the visual analogue scale; and 3 V, width of third ventricle.
Transcranial sonography findings
Images of normal and hypoechogenic MBR are shown in Figure 1. The visual MBR echogenicity ratings correlated well with the echo intensity score in the digitized image analysis (Spearman test, r = 0.54, p < 0.001; see also Figure 1S in the supplementary material). Twenty-seven (53%) migraineurs showed a hypoechogenic MBR (mean visual grade 2.31 ± 0.74), but only six (19%) control subjects did (2.76 ± 0.55; odds ratio (OR) for diagnosis of migraine: 4.87, 95% CI, 1.72–13.8; χ2 test, p = 0.002; Figure 2). The TCS findings are summarized in Table 1. The migraineurs recruited at the Department of Medical Psychology and Sociology did not differ from those recruited through the email announcement with respect to the TCS findings (all p > 0.1). Of the two participants with chronic migraine, one had a normal, and the other one a hypoechogenic MBR; the other TCS findings were unremarkable.
Transcranial sonography (TCS) images of normal and reduced echogenicity of the midbrain raphe (MBR). The midbrain is encircled for better visualization (small circles: red nuclei). (a) TCS image of normal MBR. The MBR is clearly visible thanks to its high echogenicity (grade 3; arrow). (b) TCS image of MBR with moderately reduced echogenicity (grade 2; arrow). (c) TCS image of MBR with markedly reduced echogenicity (grade 1; arrow). The MBR is not visible despite the clear visibility of the red nuclei and basal cisterns. (d) MRI plane corresponding to the TCS images shown in (a), (b) and (c). Distribution of different grades of midbrain raphe echogenicity in migraineurs and control subjects. *χ2 test, p = 0.006.

Relationship between TCS and clinical findings
Clinical data in migraineurs with normal or reduced echo signal of the midbrain raphe.
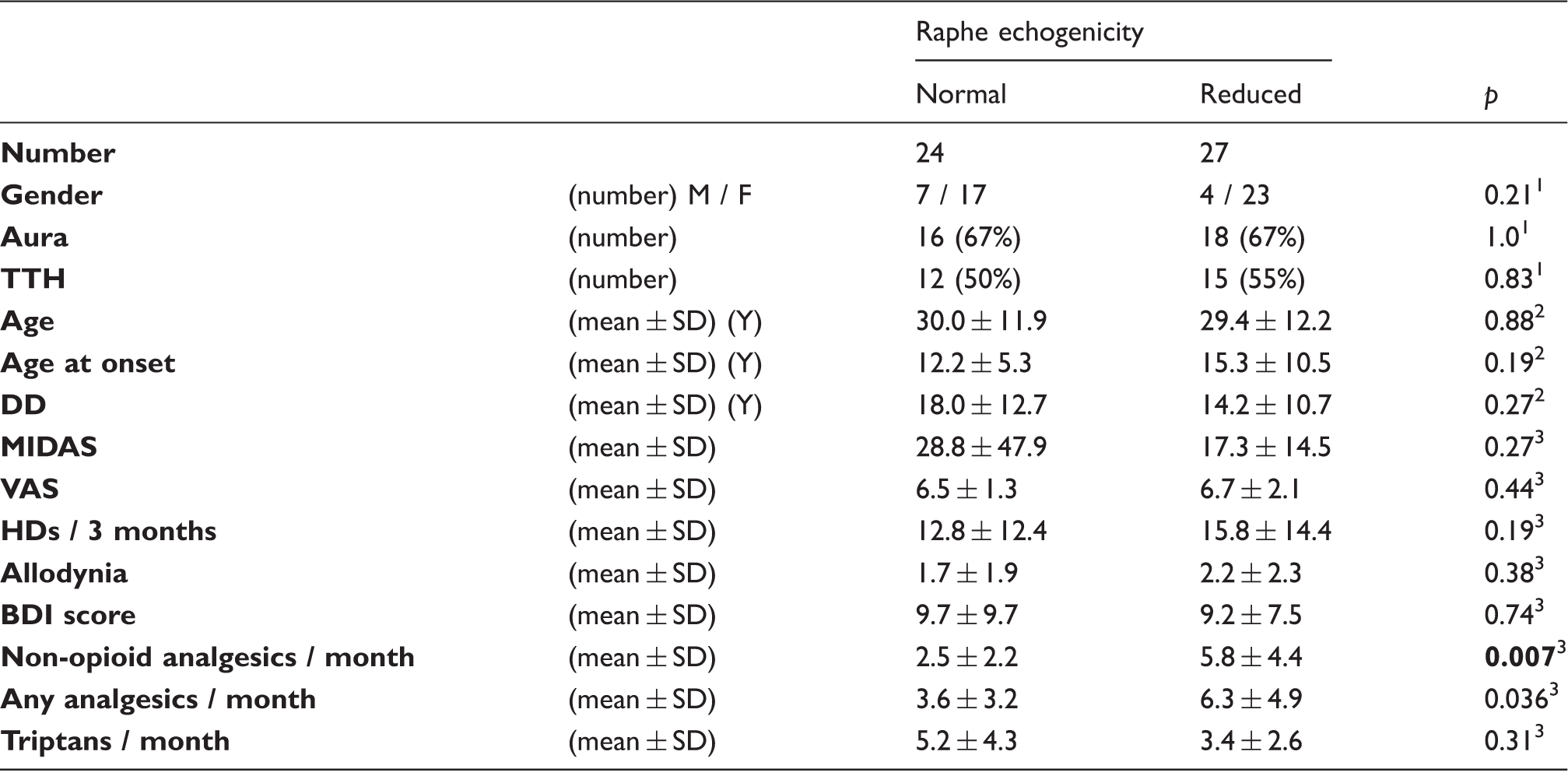
p: significance (significant values p < 0.01 in bold; for details, see text).
chi-square test. 2t-test. 3Mann-Whitney U test.
DD denotes disease duration; HDs, headache days; BDI, Beck Depression Inventory; MIDAS, Migraine Disability Assessment Score; TTH, tension-type headache; and VAS, migraine pain severity on the visual analogue scale.
Relationship between TCS findings and self-medication
The migraineurs with hypoechogenic MBR had more days per month with use of non-opioid analgesics compared to those with normal echogenic MBR (Mann-Whitney U test, p = 0.007). A lower grade of MBR echogenicity correlated with both an increasing number of users (Spearman test, r = −0.32, p = 0.024; Figure 3) and the frequency of the intake of non-opioid analgesics (r = -0.45, p = 0.011). MBR echogenicity was unrelated to the number of triptan users (p = 0.96) and the frequency of triptan intake (p = 0.39). With the two chronic migraineurs excluded, all the results remained on the same level of significance.
Percentage of patients taking analgesics (over-the-counter (OTC) analgesics and non-steroidal anti-inflammatory drugs (NSAIDs)) and triptans for acute pain relief according to ratings of the midbrain raphe echogenicity. Sums of more than 100% per group may be possible due to individuals taking both medications. Lower raphe echogenicity correlated with a higher number of users of analgesics (Spearman test, r = −0.32, p = 0.024) and a higher frequency of intake of analgesics (r = −0.45, p = 0.011; not shown). Raphe echogenicity was unrelated to the number of triptan users (p = 0.96) or the frequency of triptan intake (p = 0.39; not shown).
Relationship between TCS and neurophysiologic findings
The digitized measures of MBR echogenicity correlated with the amplitude of tCNV (r = 0.76, p = 0.028; see also Figure 2S in the supplementary material), but not iCNV, oCNV, or PINV (all p > 0.3). The other TCS findings were unrelated to CNV measures.
Discussion
The data obtained in this study show that a hypoechogenic MBR is a frequent TCS finding in migraineurs with or without aura compared to healthy controls. Lower MBR echogenicity in migraineurs is associated with higher use of analgesics, but not with use of triptans or with migraine frequency or severity. Compared to migraineurs without aura, those with aura have larger third-ventricle widths. Other TCS findings do not differ between diagnostic groups or subgroups. The potential limitations of this study include varying treatments, with some migraineurs taking multiple drugs, some staying on one drug, some receiving psychological therapy, and others being treatment naïve. The study heavily relied on interview data. To minimize the potential bias caused by the latter we applied only validated interviews. In addition, the recruitment of the majority of the migraineurs from a student population rather than from a head pain clinic may have caused a bias. In particular, the higher prevalence of aura in our cohort (67%) compared to larger populations of migraineurs (about 30%) could have contributed to the effects seen (17).
The frequency of MBR hypoechogenicity in our control group (19%) corresponds to the mean frequency of 15% seen in healthy subjects assessed in a recent meta-analysis (11). This rate clearly deviates from that in our migraine group (53%), which, however is lower than the rate in patients with depressive disorders (∼67%) (11). Our findings are in contrast to the previously reported equal prevalence of MBR hypoechogenicity in healthy subjects and migraineurs (18). This recent TCS study included a smaller sample of patients and applied only visual and not digitized image analysis. Still, this discrepancy may be due to the rigorous exclusion of migraineurs with a score of ≥11 on the BDI in the earlier study, while we only excluded subjects with a history of depressive disorder that involved medical consultation or treatment. At the mesencephalic level, the brainstem raphe contains the superior central nucleus and the dorsal raphe nucleus, which is responsible for the major part of the serotonergic innervation of the prefrontal cortex (19). Neuroimaging studies have revealed changes in this system in migraine patients, including altered 5-HT1A receptor availability in the pontine raphe nucleus (20), and functional and structural alterations in the dorsal rostral brainstem in the region of the locus coeruleus, MBR and periaqueductal grey matter (5,6).
Reduced MBR echogenicity was reported earlier as a typical feature of depressive disorders (8–11). As we excluded patients with a history of medically treated depression in our study, the true prevalence of reduced MBR echogenicity may be even higher in an unselected sample of migraineurs. What causes the change in MBR echogenicity on a histological level is currently a matter of debate. Preliminary post-mortem findings in individuals with depressive disorders have linked this alteration to a shift in tissue cell density and the disruption of fibre tracts passing the pontomesencephalic midline (9). It has been discussed that the reduced echo signal of the raphe reflects an alteration of the raphe nuclei rather than the fibre tracts in migraineurs (18).
As a correlate of the abnormal cortical information processing of sensory stimuli in migraineurs, often an altered CNV amplitude is observed, which seems to be an endophenotypic marker for migraine (3,4). While the iCNV is thought to indicate the level of expectancy mediated by the noradrenergic system, the tCNV rather reflects motor preparation and mobilization, and has been discussed as being related to the activity of the dopaminergic system (3,4,21). The differences in iCNV between migraineurs and controls are pronounced and highly significant (4). However, the functional meaning of tCNV is less clear. We found a correlation between lower MBR echogenicity and higher tCNV amplitude. A number of studies have demonstrated that the mesial wall motor areas (supplementary/cingulate motor areas) as well as the primary motor and sensory cortex participate in generating the tCNV (22). Serotonergic neurons within the rostral raphe complex of the mesencephalon and rostral pons (caudal linear, dorsal raphe and median raphe nuclei) project primarily to the forebrain (19). The findings of the present study link morphologic alteration of the MBR and tCNV, which might therefore reflect disturbance of the serotonergic rather than the dopaminergic system. However, one earlier TCS study on healthy subjects that compared MBR echogenicity and the loudness dependence of auditory evoked potentials, thought to be an indirect indicator of central serotonergic activity, failed to show a correlation between these two measures (23).
In another study of depressed patients, reduced MBR echogenicity was related to higher responsivity to serotonin reuptake inhibitors (10), also pointing towards an alteration of the serotonergic system. Interestingly, a lesion of the dorsal raphe nucleus induced supersensitivity to serotonin in isolated cerebral arteries in cats (24). Genetic studies on migraine showed an association of polymorphisms in the serotonin transporter (5-HTT) with migraine (25,26). It needs to be elucidated whether the subgroup of migraineurs with reduced MBR echogenicity might be characterized by genetic polymorphisms related to altered serotonin metabolism.
Another finding of the present study is that migraine patients with a hypoechogenic MBR take more non-opioid analgesics (paracetamol and NSAIDs) than patients with normal MBR echogenicity. It is generally accepted that these analgesics exert, in addition to cyclooxygenase inhibition, an effect on the central modulation and integration of noxious stimuli, with the exact mechanisms still remaining under discussion. The analgesic effect of NSAIDs is accomplished by the activation of descending, pain-inhibitory serotonergic pathways (27). Pharmacological blocking of serotonin receptors results in extensive or total loss of analgesic efficacy. Using a novel MRI technique that measures regional cerebral blood flow, not only could it be shown that the rostral ventromedial medulla, including the raphe nuclei, is involved in the perception of pain, but it could also be identified as a main site of analgesic interaction with ibuprofen (28).
Remarkably, the distribution of triptan users was equal across the groups with different ratings of MBR echogenicity. Triptans which act as 5-HT1B/1D agonists are suggested to achieve their analgesic effect via disruption of the trigemino-cortical connectivity, thus explaining their analgesic effect being specific to headache pain (29). In the human brain, 5-HT1B receptors are mainly expressed in the basal ganglia and in the frontal cortex, where they convey presynaptic inhibitory effects (30). 5-HT1D receptors are expressed to a lesser extent, e.g. as auto receptors in the dorsal raphe nucleus. Our data do not show an association between the use of 5-HT1B/1D agonists and echogenicity of the basal ganglia or the MBR. According to our findings, the effect of triptans appears to be less dependent on the state of the serotonergic MBR nuclei, which supports the idea of different targets for triptans versus classical analgesics. The missing relation between MBR echogenicity and triptan use is also supported by our finding that MBR echogenicity is unrelated to the frequency or severity of migraine pain. Taken together, our findings suggest that MBR echogenicity is associated with a change in the reaction to the headache rather than the intensity of the headache.
The TCS findings for other deep brain structures were unremarkable in the migraineurs. Abnormal hyperechogenicity of the substantia nigra, which is present in about 9% of the healthy population, indicates subclinical malfunction of the nigrostriatal dopaminergic system and has been identified as a risk factor for Parkinson’s disease (8,31). The results of a recent cohort study suggest a common vulnerability to, or common consequences of, migraine and late-life Parkinsonism (32). However, we did not detect an increased frequency of substantia nigra hyperechogenicity in migraineurs, which separates migraineurs from other risk groups for subsequent Parkinson’s disease (8). In our cohort, third-ventricle widths were larger in migraineurs experiencing aura symptoms compared to those without aura, but neither group differed from healthy subjects. A similar group difference has been reported recently which however failed statistical significance (18). Given the limited size of our study and the earlier study cohorts, this finding remains to be validated. It was proposed earlier that a cyst of the third ventricle causes migraine with aura (33). Volumetric changes of pain-related cortical areas as well as thalamic nuclei in migraine patients have been demonstrated, however, irrespective of association with aura symptoms (34,35). One voxel-based morphometry MRI study that compared migraine patients with aura to those without aura detected an increased density of the periaqueductal gray matter and of the dorsolateral pons in migraineurs experiencing aura (34). Further study is warranted to assess the changes of structures near the third ventricle that may be specific for migraine with aura.
Conclusions
Reduced MBR echogenicity is frequent in migraineurs with or without aura compared to healthy controls. This finding is likely to reflect an alteration of the serotonergic raphe nuclei and relates to higher use of non-opioid analgesics (NSAIDs and paracetamol), but not to use of triptans. MBR echogenicity is, however, unrelated to migraine severity. Compared to migraineurs without aura, those with aura have larger third-ventricle widths. The sonographic findings for the basal ganglia and substantia nigra are unremarkable in migraineurs.
Clinical implications
Midbrain raphe alteration is frequent in migraineurs and relates to self-medication behavior but not to frequency or severity of migraine. Our findings support the idea that TCS might be useful to detect migraineurs with chronically altered serotonergic system. Further studies will clarify the individual course of raphe echogenicity and its diagnostic value for the identification of individuals at risk of developing chronic headache in association with medication overuse.
Footnotes
Acknowledgement
The authors wish to thank Ms Elaine Reape for careful reading and critical discussion on the manuscript.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: GH, PK, BM and AR report no competing interests. TPJ has served on advisory boards for Pharmallergan and received honoraria from Autonomic Technologies, Inc., Hormosan, MSD, Pfizer and Pharmallergan. UW received speaker honoraria and travel grants from Abbvie, Bayer Vital, Ipsen Pharma, Merz Pharmaceuticals and Pfizer.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
