Abstract
Introduction
Reduced echogenicity of the brainstem raphe nuclei (BRN) was demonstrated in major depression, possibly indicating serotonergic dysfunction. Postulating that migraine may constitute a “chronic low serotonin syndrome,” we aimed to evaluate the echogenicity of midbrain structures, including serotonergic BRN in episodic migraine.
Methods
Transcranial sonography was performed in 39 patients with episodic migraine (median age 35, interquartile range (IQR): 27–47 years; 27 women) and 35 controls (median age 31, IQR: 29–47 years; 19 women). Individuals with concomitant depression were excluded. Echogenicity of BRN, substantia nigra (SN) and third ventricle width was evaluated according to an internationally established examination protocol.
Results
Hypoechogenicity of BRN was depicted in 23.1% of migraine patients and 20% of controls, showing no significant difference. Migraine patients with hypoechogenic BRN had significantly higher attack frequency (median 3, IQR 2–5 vs. 1.5, IQR 1–2 days/month; p = 0.029) and a trend toward earlier disease manifestation. The rate of hyperechogenic SN and width of the third ventricle were similar between both groups. We did not observe any differences between migraine patients with and without aura.
Conclusion
Sonographic findings did not differ between migraine patients and controls. Hypoechogenic BRN correlated to a higher migraine attack frequency, probably indicating more severe disease activity.
Keywords
Introduction
The Global Burden of Disease Survey 2010 ranked migraine as the third most common disease worldwide and as the seventh highest among specific causes of disability worldwide (1). Despite progress in therapy in the last decades, the precise underlying pathophysiological mechanism of migraine still remains unresolved. The trigeminocervical complex serves as an afferent gate, receiving sensation from dura mater and meningeal vessels (2). The descending modulation of pain perception occurs primarily in the brainstem and diencephalic nuclei. They can facilitate, inhibit and even abolish pain sensation (3). The concept of a “migraine generator” within the brainstem was proposed by an early positron emission tomography (PET) study (4). Considering ictal and interictal changes of the serotonin metabolism, the role of brainstem raphe nuclei (BRN), as a major site of serotonergic projections within the brain, is of special interest in migraine (5). Neurons of the dorsal nucleus raphe (DNR) were activated through nociceptive stimulation of the trigeminal ganglion experimentally and in turn were able to reduce activity of trigeminal neurons (6). Reciprocal effects of cortical spreading depression (CSD) and BRN activity have also been demonstrated: CSD inhibits antinociceptive activity of raphe nuclei; degeneration of raphe nuclei in contrast facilitates CSD (6–8). No structural changes of BRN have been depicted in migraine so far (9).
Transcranial sonography (TCS) has become a valid and reliable supportive diagnostic tool in the evaluation of extrapyramidal and other neurodegenerative disorders, such as Parkinson’s and Huntington’s disease and ataxias (10–13). Depicting the echogenicity of different parenchymal regions in the brain, it provides additional diagnostic value to established brain imaging methods like computed tomography (CT) or magnetic resonance imaging (MRI). While hyperechogenicity of the substantia nigra (SN) is a typical finding in Parkinson’s disease, reduced echogenicity of the BRN was demonstrated in patients with major depression as well as in depressed patients with different neurodegenerative diseases (14–16). We hypothesized that similar to depression, changes in serotonergic metabolism in migraine may be reflected by reduced BRN echogenicity. Therefore, we investigated echogenicity of midbrain structures, including the raphe nuclei, in patients with episodic migraine compared to healthy controls.
Methods
Participants
We recruited 46 patients with episodic migraine diagnosed according to International Classification of Headache Disorders, second edition (ICHD-II) criteria (17). Clinical and demographic data have been collected based on a specially developed questionnaire and included headache frequency in the last year, family history of migraine, medications, headache features and concomitant autonomic signs. For the control group, 37 age- and gender-matched healthy volunteers were recruited from the hospital staff, the medical student body as well as from their friends and family.
In both groups we used the Beck Depression Inventory (BDI) in order to exclude those patients with concomitant depression (18). Patients with previous episodes of depression or other psychiatric or neurological diseases as well as previous therapy with antidepressants were excluded.
All individuals gave written informed consent. In accordance with the Helsinki Declaration of 1975, the study was approved by the local university ethics committee of the Ruhr University Bochum, Germany.
Four migraine patients and two controls had to be excluded because of an insufficient temporal bone window. Another three patients with migraine were excluded because of concomitant depression or a BDI score ≥ 11. Thus, 39 patients with episodic migraine and 35 volunteers were included in the study.
TCS
All patients have been investigated during the interictal period, at least 72 hours after the last migraine attack. TCS was performed by one of two experienced sonographers (IA, CK), both certified by the German Society of Ultrasound in Medicine (DEGUM). Both sonographers were blinded to the clinical data. Using a phased-array ultrasound system equipped with a 2.5 MHz-transducer (Aplio XG Ultrasound System, Toshiba Medicals, Tochigi, Japan), examination was performed through the transtemporal bone window as recommended elsewhere (19). Patients were instructed not to talk to the investigator concerning their complaints and diagnosis. A dynamic range of 45 dB and penetration of 150 mm were chosen. Image brightness and time-gain compensation were adapted as needed for each examination. The midbrain structures were visualized in the axial section, as previously recommended (19). Initially the midbrain was visualized through bilateral TCS and the side with the best visible BR structure was chosen for analysis. Echogenicity of BRN was evaluated semiquantitatively on a three-point scale, using the red nucleus or basal cisterns’ signal intensity as a reference point: 0 = brainstem raphe not visible, 1 = slight echogenic raphe, 2 = normal echogenicity (Figure 1). Grade 0 and grade 1 are classified as hypoechogenic.
Corresponding mesencephalic axial examination planes in patients with normal and hypoechogenic brainstem raphe.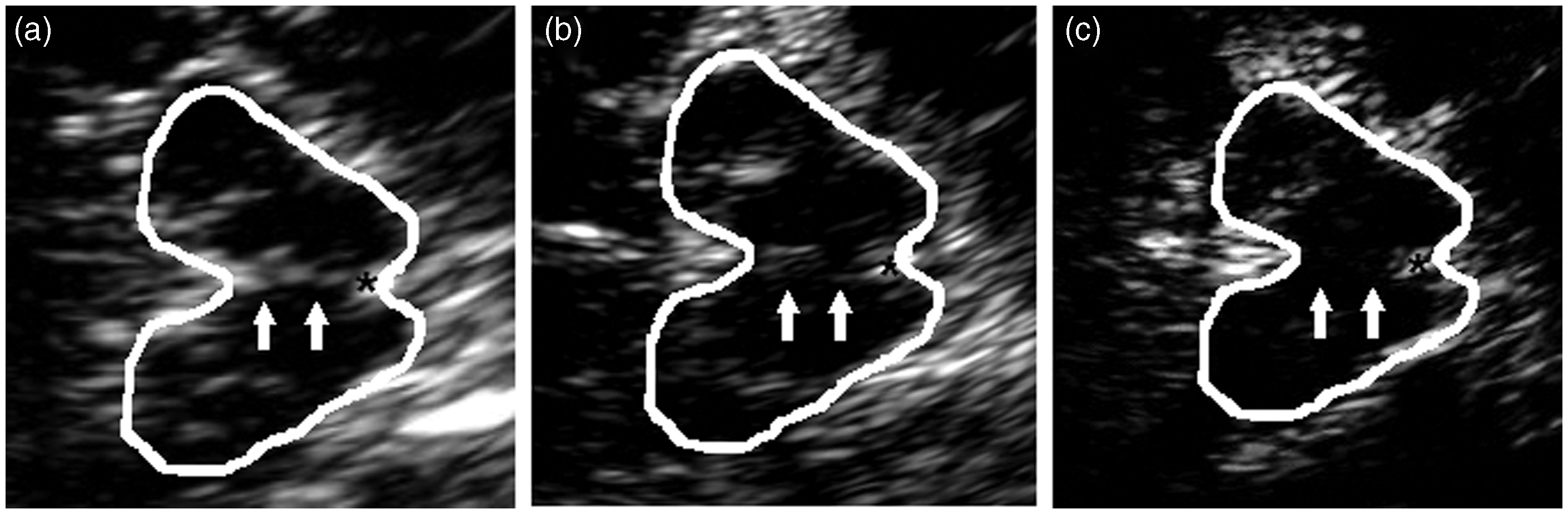
In the case of hyperechogenicity of the SN, a planimetric measurement was performed. Sizes of less than 0.20 cm2 were graded as normal, sizes between 0.20 and 0.25 cm2 as moderate and >0.25 cm2 as markedly hyperechogenic.
Minimal transverse diameter of the third ventricle was measured on a standardized diencephalic level. The sonographic findings were stored and re-evaluated “off-line” by the second investigator (IA, CK), who performed independent evaluation and classification of results. In the case of discrepant ratings a consensus was accomplished subsequently.
Statistical analysis
Descriptive statistics are given as mean ± standard deviation (SD) when normally distributed, otherwise as median (interquartile range, IQR). Inter-rater agreement was analyzed by Cohen’s kappa statistics. Statistical comparison of groups was performed by appropriate nonparametric tests (Mann-Whitney U test or Fisher’s exact test). Statistical analyses were performed using Prism 6 (GraphPad).
Results
Main clinical and demographic characteristics of the groups
Main clinical and demographic characteristics of the migraine patients with and without migraine and healthy controls.

All migraine vs control group. bMigraine without aura vs. migraine with aura.
F: female; M: male; IQR: interquartile range; BDI: Beck Depression Inventory.
Baseline characteristics of excluded migraine patients were as follows: gender (female:male 6:1); age 39 (IQR 33–47) years; disease duration 17 (IQR 12–24) years; BDI score 5 (IQR 2–13); attack frequency 2.0 (1.0–4.0). As expected, there was a trend toward a higher BDI score in excluded patients, comparing to those included; however, none of these parameters reached a level of significance.
TCS findings
Transcranial sonography findings of patients with migraine and healthy controls.

All migraine vs control group. bMigraine without aura vs. migraine with aura.
Comparison of clinical parameters in patients with normal and hypoechogenic BRN
Comparison between migraine patients with normal and pathological echogenicity of brainstem raphe nuclei.
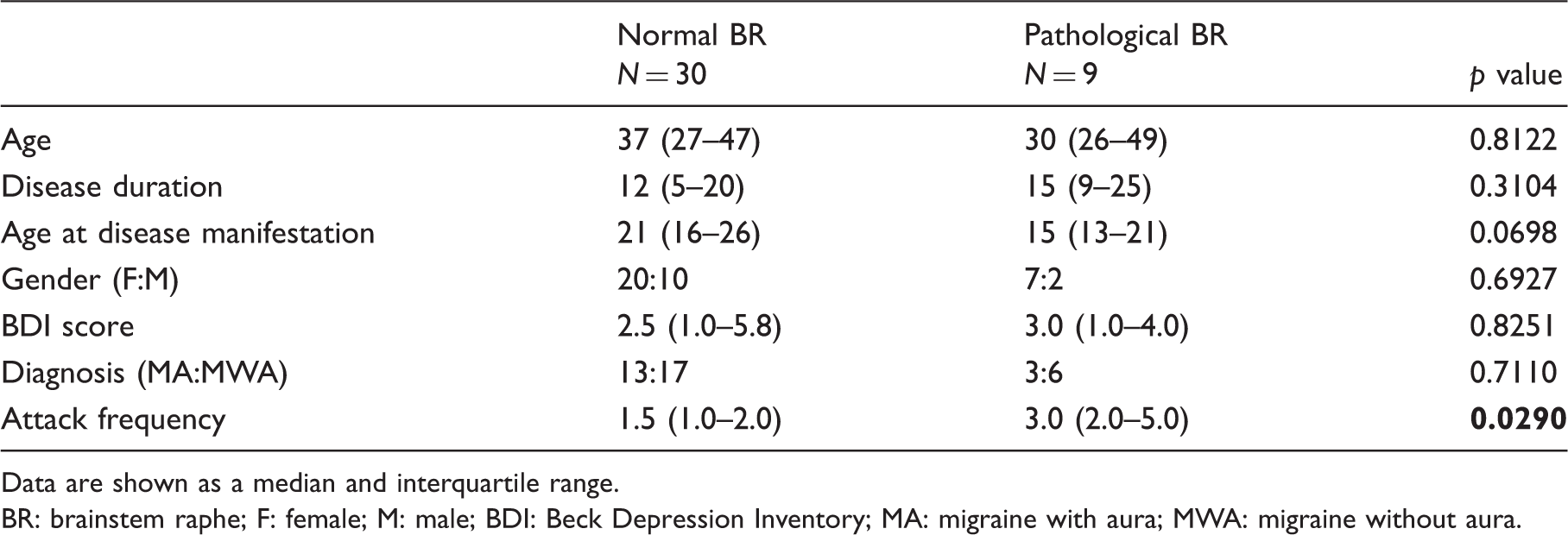
Data are shown as a median and interquartile range.
BR: brainstem raphe; F: female; M: male; BDI: Beck Depression Inventory; MA: migraine with aura; MWA: migraine without aura.
Discussion
BRN hypoechogenicity was associated with increased migraine attack frequency in episodic migraine and may deserve further investigation toward the development of a useful tool for identifying patients with increased disease activity. Moreover, patients with hypoechogenic BRN showed a trend toward earlier disease manifestation, pointing at some predisposing serotonergic deficiency that may lead to or favor the onset of migraine in these patients.
This is the first study of transcranial brain sonography in patients with migraine, so comparison of data remains difficult. Some imaging studies in humans as well as some animal studies point at the brainstem as paramount for migraine pathophysiology and serotonin as the major substance responsible for migraine symptoms and pain. While previous structural voxel-based MRI morphometry (VBM) studies revealed no specific changes in raphe nuclei, but increased density of the periaqueductal gray matter (PAG) and some cortical regions in migraine (20,21), a number of functional imaging studies, using PET and MRI, demonstrated activation of the dorsal pons during the headache phase in migraine and reduced stimulus-dependent activation of the nucleus cuneiformis (NCI) interictally (22–25). As known, PAG and NCI are densely connected to serotonergic raphe nuclei, forming an important part of the descending pain-modulatory pathway. Stereotactic interventions on PAG and dorsal nucleus raphe result in migraine attacks even in non-migraine patients (26–28). A radio ligand single-photon emission CT study revealed an increased availability of the serotonin transporter in the raphe nuclei, endorsing the biochemical theory of migraine as a “chronic low serotonin syndrome (5,29). This may be dependent on the attack interval and current state of the migraine. A PET study with a selective 5-HT(1A) antagonist tracer showed an increased binding potential in the BRN in the acute migraine attack compared to headache-free migraineurs, as well as in the left orbitofrontal cortex, precentral gyrus and temporal pole. This clearly emphasizes the role of the serotoninergic system in the migraine attack and may explain sonographic changes with increasing attack frequency (30). Experiments in rats showed that chronic degeneration of dorsal raphe serotonergic neurons modulated CSD as possible involvement in migraine pathology (8).
Regarding the association of BRN alterations and the higher attack frequency, our findings might be interpreted as a further indication of this hypothesis. However, the morphological and pathophysiological interpretation of reduced BRN echogenicity remains speculative. A correlation of this finding to a signal alteration on MRI studies has been reported previously in a major depression, suggesting that hypoechogenicity reflects a structural disruption of the mesencephalic fiber tracts of the dorsal raphe nucleus (31). Taking into account a high concentration of serotonergic neurons, its dysfunction has been supposed to underlie this phenomena in patients with depressive disorders (14,15,32,33). Interestingly, patients with hypoechogenic BRN responded better to selective serotonin reuptake inhibitor (SSRI) therapy than those with normal findings (15). Having excluded migraine patients with depressive symptoms, patients with episodic migraine showed similar prevalence of hypoechogenic BRN (23.1%) and hyperechogenic SN (7.7%) as healthy controls. This is in line with previous TCS studies demonstrating echogenicity variation of BRN in about 20% and of SN in about 10% of healthy individuals (11,14–16,33,34). As there was no association between migraine and BRN hypoechogenicity in our study, we suppose that our finding could reflect a kind of constitutional “weakness” of the pain-modulating serotonergic network, resulting in a more severe course of migraine.
Several general limitations of transcranial sonography must be taken into account. Because of an insufficient transtemporal bone window, in approximately 10% patients midbrain structures can be only partially assessed (10). Furthermore, the reliability of the findings is dependent on the quality of the ultrasonography system and on the qualification of the investigator (10). With regard to the present study, a further important limitation lies in known reduced echogenicity of BRN in patients with depressive disorders. These patients had to be excluded from our cohort. However, considering the high prevalence of comorbid depression and depressed mood in patients with migraine, this selection could cause a potentially strong bias in our study (35). An overall low attack frequency in recruited patients makes a conclusion of possible association of BRN hypoechogenicity with attack frequency speculative. However, detected differences indicate that in patients with higher attack frequency an even stronger association could exist. A number of other structures, including first of all PAG and rostral pons, have been supposed to be involved in migraine pathogenesis; however, because of technical considerations its sonographic evaluation is limited and not standardized.
Conclusion
Sonographic echogenicity of BRN and SN did not differ between patients with episodic migraine without depression and healthy controls. However, hypoechogenic BRN correlated to higher migraine attack frequency, and thus may indicate more severe disease activity. This may be related to serotoninergic dysfunction, and a correlation of the BRN echogenicity and serotonin turnover would be highly interesting to investigate in the future. Further TCS studies in frequent episodic and chronic migraine are needed in order to evaluate a possible link between abnormal echo signal of BRN and migraine chronification.
Key findings
There are no specific changes in the echogenicity of brainstem raphe nuclei and substantia nigra in episodic migraine. Patients with hypoechogenic brainstem raphe nuclei have higher migraine attack frequency and a trend toward earlier disease manifestation. Further studies, comparing episodic migraine with low and high attack frequency and chronic migraine, will clarify if abnormal echo signal of raphe nuclei is associated with headache chronification and can serve as a biomarker for a more severe course of migraine.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
