Abstract
Background
Apart from the underlying cardiovascular (CV) risk associated with migraine, both triptans and ergotamines can induce vasoconstriction and potentially increase the risk of serious ischemic events. Because of the low frequency of such events in eligible patients, randomized controlled trials are not exhaustive to assess the drug-related CV risk. Observational studies are, therefore, an essential source of information to clarify this matter of concern.
Aim
The aim of this study was to systematically review the available published observational studies investigating the risk of serious CV events in triptan or ergotamine users, as compared to unexposed migraineur controls.
Methods
We systematically searched MEDLINE and EMBASE electronic databases for cohort or case-control studies up to December 1, 2013. Studies retrieved from CDSR, DARE and HTA databases of the Cochrane Library were used for snowballing. Studies investigating the risk of any CV outcome in patients with a migraine diagnosis and exposed to triptans or ergotamines were considered for inclusion. Selection of studies, data extraction, and risk of bias assessment were conducted independently by two reviewers. Pooled odds ratios (ORs) with 95% confidence interval (95% CI) were computed using a random-effects model for studies and outcomes judged eligible for quantitative data synthesis.
Results
From a total of 3370 citations retrieved, after duplicate removal and screening, only four studies met the inclusion criteria (three nested case-control analyses and one retrospective cohort study). These studies investigated the risk of different CV outcomes associated with either the recency or the intensity of exposure to the studied drugs. As for the intensity of use, the pooled OR of serious ischemic events was 2.28 (95% CI 1.18–4.41; I2 = 0%) for ergotamine use (two studies), whereas for triptans (three studies) it was 0.86 (95% CI 0.52–1.43; I2 = 24.5%). Recent use of ergotamines was not significantly associated with any CV outcome (only one available study). Two studies investigated the risk of stroke related to recent triptan use: the first study reported an OR of 0.90 (0.64–1.26), and the second one suggested an increased risk of 2.51 (1.10–5.71). In this case, because of the high degree of heterogeneity, results were not pooled.
Conclusions
To date, few comparative observational studies have investigated the CV safety of migraine-specific drugs in clinical practice. Evidence gathered here suggests that intense consumption of ergotamines may be associated with an increased risk of serious ischemic complications. As for triptans, available studies do not suggest strong CV safety issues, although no firm conclusions can be drawn. In particular, evidence on stroke risk is conflicting. However, if an increase of the absolute stroke risk in recently exposed patients does actually exist, it must be small. Overall, residual uncontrolled confounding factors reduce the confidence in the risk estimates collected from the included studies. Further investigations are needed to better define the risk for rare but serious CV events related to triptan and ergotamine use for treatment of migraine.
Introduction
Migraine is a chronic and debilitating neurological disorder affecting more than 10% of the worldwide adult population (1).
Different pharmacological approaches are currently available to treat acute migraine attacks. These are usually classified into two main categories: non-specific therapies with analgesic effect, such as nonsteroidal anti-inflammatory drugs (NSAIDs), paracetamol, barbiturate-containing analgesics and opiates; and migraine-specific drugs, including triptans and ergotamines (1,2)
Drug choice in clinical practice is generally based on headache features (e.g. onset rapidity, severity, early vomiting) as well as patients’ preferences and characteristics (e.g. inter-individual variability of response/tolerability, comorbidities, risk factors, co-medications) (2–4).
As for migraine-specific drugs, underlying cardiovascular (CV) diseases or risk factors represent the major limitation for their use in practice. In fact, both triptans and ergotamines can induce, through heterogeneous mechanisms, vasoconstriction that may potentially increase the risk of serious ischemic events (2,4–6).
Ergotamine and dihydroergotamine were the first specific anti-migraine agents introduced in therapy, respectively, in 1926 and 1945 (7). Their efficacy in the treatment of migraine is thought to be related to the affinity with 5HT1b/d receptors, through which these drugs act on central serotoninergic neurons, inhibit neurogenic inflammation and cause cerebral vasoconstriction (8). However, both ergotamine and dihydroergotamine also activate other 5HT receptors subtypes and α-adrenoceptors that, in turn, are mostly responsible for side effects (7–9), such as rare but potentially fatal vasospastic reactions (e.g. ergotism, gangrene) (5).
In the early 1990s, sumatriptan, the first selective agonist of the 5HT1b/d receptors, was specifically developed and marketed for the acute treatment of migraine, with the promise of a better safety profile than ergotamines. In the last two decades, a total of six other sumatriptan analogs, commonly called “triptans,” have been licensed worldwide (1). During these years, a large number of randomized clinical trials have demonstrated the efficacy of triptans, and no major CV issues have been reported from such studies (3,10,11).
However, results from pre-approval clinical trials have a recognized limited power in detecting rare adverse reactions, as well as little generalizability toward “real-world” populations (12).
During post-marketing experience, different serious ischemic vascular events have been reported in association with the use of triptans (6,13). Nevertheless, on the basis of case reports only, the risk of such events cannot be estimated and, in most of cases, a possible causal relationship with triptan exposure cannot be definitively excluded (3).
In this context, post-marketing observational studies can provide important information on the CV safety profile of migraine-specific drugs. In fact, by involving large and unselected populations from a real-world setting, observational studies may represent the best source for estimating the risk of rare adverse drug reactions in clinical practice (12).
Therefore, the aim of this study was to systematically review the available published literature on observational studies (case-control and cohort) investigating the risk of severe CV events associated with triptan or ergotamine use among migraine patients.
Methods
Protocol registration
The study protocol was submitted in advance to the International Prospective Register of Systematic Reviews (PROSPERO) (14).
Characteristics of studies eligible for inclusion
Observational cohort or case-control studies investigating the risk of any CV outcome in patients exposed at least to a triptan (i.e. sumatriptan, rizatriptan, zolmitriptan, naratriptan, eletriptan, almotriptan, frovatriptan) or an ergotamine compound (i.e. ergotamine or dihydroergotamine) for the acute treatment of migraine were considered for inclusion. Since migraine itself may represent an independent risk factor for serious CV outcomes (15,16), we decided to exclude studies involving non-migraineurs as the control group. No restriction with respect to age or gender was applied.
Search strategy
Studies entered up to December 1, 2013, in PubMed, EMBASE, Cochrane Database of Systematic Reviews (CDSR), Database of Abstract of Review of Effects (DARE) and Health Technology Assessment (HTA) database of the Cochrane Library were retrieved using a combination of exploded terms and free text. As for PubMed, the terms “Serotonin Receptor Agonists,” Medical Subject Heading (MeSH), “triptan*,” “sumatriptan*,” “rizatriptan*,” “naratriptan*,” “zolmitriptan*,” “eletriptan*,” “almotriptan*,” “frovatriptan*,” “ergotamine*,” “dihydroergotamine*” AND “cardiovascular diseases” (MeSH) OR “all the cardiovascular events associated to triptan use as reported by Roberto et al. (13)” were used in combination with the search limit “humans.” A similar approach was used also for searching in EMBASE (full search strategy for all databases concerned is available in the online Supplementary file 1). CSDR, DARE and HTA databases were searched in order to identify other systematic reviews possibly useful for snowballing: In this case, in order to be as sensible as possible, the exposure of interest only was included in the search string, i.e. “Serotonin Receptor Agonists,” MeSH “triptan*,” “sumatriptan*,” “rizatriptan*,” “naratriptan*,” “zolmitriptan*,” “eletriptan*,” “almotriptan*,” “frovatriptan*,” “ergotamine*” and “dihydroergotamine*.”
Retrieved references were managed with Reference Manager 11.
Study selection
Two pairs of investigators (GR-ER and LV-EP), working in duplicates, independently examined all titles and abstracts of retrieved references. When characteristics for inclusion/exclusion were not clearly reported within the title/abstract, or the abstract was not available, studies were in any case retained for subsequent full-text screening. Retrieval of full-text studies was performed with the help of the document delivery system’s librarians of the University of Bologna. Only studies in English, Spanish, French or German were screened on full-text. Included studies as well as any other full paper that could include useful references (e.g. review, case-reports with literature review, guidelines, bulletin) were hand-searched for further studies of interest.
Data extraction and quality appraisal
Data from included studies were extracted by GR, ER, CP, VC working independently and in duplicate. A pre-established form concerning information on study design, source of data, population characteristics, outcomes of interests, matching criteria and confounding factors considered for statistical adjustment was used. EP and LV were consulted for discrepancies on extracted data. The risk of bias of included studies was assessed by using the Newcastle-Ottawa Scale (NOS) (17). In order to produce a graphical representation of the assessment results, the three NOS domains (selection, comparability and exposure or outcome) were imported in the Risk of Bias tool of the Review Manager software (version 5.2) provided by the Cochrane Collaboration (18). We further customized the Risk of Bias tool by adding two fields concerning “migraine ascertainment” and “company funding,” since these aspects are not specifically addressed within the NOS scale, but they should be taken into account when interpreting study results.
Data synthesis
According to the Meta-analysis Of Observational Studies in Epidemiology (MOOSE) guidelines, we used broad inclusion criteria (19). For those studies and outcomes judged eligible for data synthesis, the most adjusted relative risks, odds ratios (ORs) or hazard ratios were used, with 95% confidence intervals (95% CIs), assuming that ORs, relative risks and hazard ratios were all comparable measures in the presence of relatively rare events (20). Statistical heterogeneity among studies was evaluated using Q statistic (with p < 0.10 considered significant), and I-squared statistic (I2) together with visual inspection of graphs were used to estimate the proportion of total variation contributed by between-study variance. Pooled ORs with 95% CIs were computed for the comparisons of interest, using both a fixed-effect model (inverse-variance method) (20) and a random-effects model (DerSimonian and Laird method) (21). In order to take into account differences between observational studies, the random-effects model was preferred for the presentation of results, since it represents a more conservative approach for the calculation of aggregated effect size. All statistical analyses were performed with Comprehensive Meta-Analysis Version 2 software.
As recently suggested by Barbui et al. (22), the GRADE pro Version 3.6 software was used to transparently grade the strength of evidence collected on the CV profile of triptans and ergotamines, and generate a summary of findings table.
Results
Literature search results
The literature search strategy retrieved 3370 citations (Figure 1). After automated duplicate removal and screening on title and abstract of the retrieved studies, a total of 189 full-text articles were further assessed for inclusion. Only four studies met the pre-specified inclusion criteria (23, 23–26). Four otherwise eligible case-control studies were excluded because of the use of non-migraineurs as controls (27–30). Reasons for exclusion of the remaining screened full papers are reported in Figure 1.
Selection process for study inclusion in the systematic review.
Characteristics of included studies
Characteristics of included studies: design, population, interventions and outcomes.
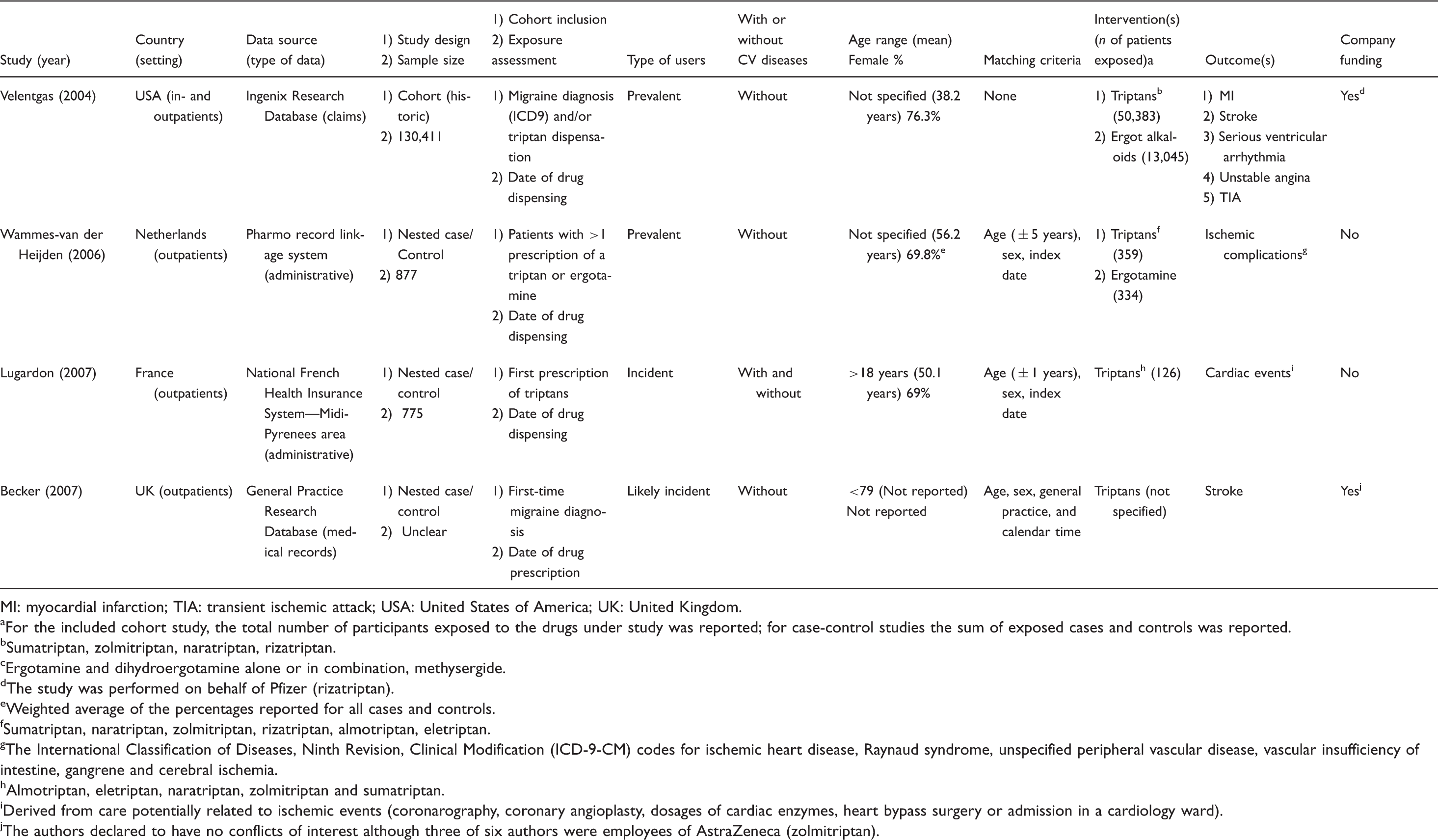
MI: myocardial infarction; TIA: transient ischemic attack; USA: United States of America; UK: United Kingdom.
For the included cohort study, the total number of participants exposed to the drugs under study was reported; for case-control studies the sum of exposed cases and controls was reported.
Sumatriptan, zolmitriptan, naratriptan, rizatriptan.
Ergotamine and dihydroergotamine alone or in combination, methysergide.
The study was performed on behalf of Pfizer (rizatriptan).
Weighted average of the percentages reported for all cases and controls.
Sumatriptan, naratriptan, zolmitriptan, rizatriptan, almotriptan, eletriptan.
The International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) codes for ischemic heart disease, Raynaud syndrome, unspecified peripheral vascular disease, vascular insufficiency of intestine, gangrene and cerebral ischemia.
Almotriptan, eletriptan, naratriptan, zolmitriptan and sumatriptan.
Derived from care potentially related to ischemic events (coronarography, coronary angioplasty, dosages of cardiac enzymes, heart bypass surgery or admission in a cardiology ward).
The authors declared to have no conflicts of interest although three of six authors were employees of AstraZeneca (zolmitriptan).
Studies estimating the risk of CV events related to the intensity of migraine-specific drug use
Intensity of triptan/ergotamine use and cardiovascular outcomes as reported from the included cohort study.

IRR: incidence rate ratio; 95% CI: 95% confidence interval.
Statistically significant.
Factors considered for statistical adjustment: year of cohort entry, age, sex, current and recent use of ergot alkaloids, comorbidities (ischemic heart diseases, cerebrovascular disease, peripheral vascular disease, diabetes, hypertension and hyperlipidemic), use of oral contraceptive and/or hormone replacement therapy.
Intensity of triptan/ergotamine use and cardiovascular outcomes as reported from the included case-control studies.
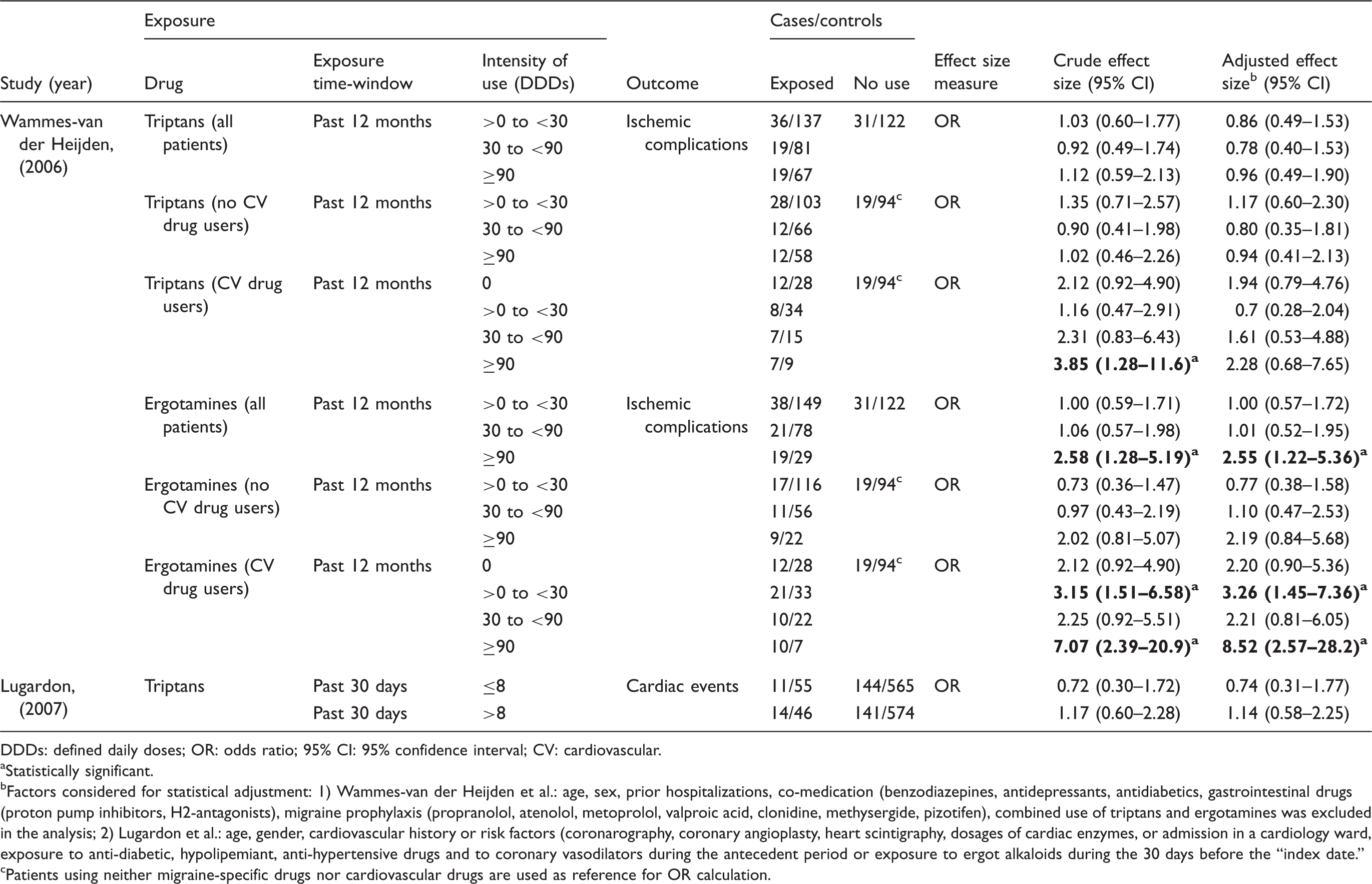
DDDs: defined daily doses; OR: odds ratio; 95% CI: 95% confidence interval; CV: cardiovascular.
Statistically significant.
Factors considered for statistical adjustment: 1) Wammes-van der Heijden et al.: age, sex, prior hospitalizations, co-medication (benzodiazepines, antidepressants, antidiabetics, gastrointestinal drugs (proton pump inhibitors, H2-antagonists), migraine prophylaxis (propranolol, atenolol, metoprolol, valproic acid, clonidine, methysergide, pizotifen), combined use of triptans and ergotamines was excluded in the analysis; 2) Lugardon et al.: age, gender, cardiovascular history or risk factors (coronarography, coronary angioplasty, heart scintigraphy, dosages of cardiac enzymes, or admission in a cardiology ward, exposure to anti-diabetic, hypolipemiant, anti-hypertensive drugs and to coronary vasodilators during the antecedent period or exposure to ergot alkaloids during the 30 days before the “index date.”
Patients using neither migraine-specific drugs nor cardiovascular drugs are used as reference for OR calculation.
Studies estimating the risk of CV events related to the recency of migraine-specific drug use
Recency of triptan/ergotamine use and cardiovascular outcomes as reported from the included cohort studies.

MI: myocardial infarction; TIA: transient ischemic attack; IRR: incidence rate ratio; 95% CI: 95% confidence interval.
Factors considered for statistical adjustment: 1) Triptans: year of cohort entry, age, sex, current and recent use of ergot alkaloids, comorbidities (ischemic heart diseases, cerebrovascular disease, peripheral vascular disease, diabetes, hypertension and hyperlipidemic), use of oral contraceptive and/or hormone replacement therapy; 2) Ergot alkaloids: year of cohort entry, age, sex, current and recent use of triptan drugs, comorbidities (ischemic heart diseases, cerebrovascular disease, peripheral vascular disease, diabetes, hypertension and hyperlipidemic), use of oral contraceptive and/or hormone replacement therapy.
Recency of triptan/ergotamine use and cardiovascular outcomes as reported from the included case-control studies.

OR: odds ratio; 95% CI: 95% confidence interval.
Statistically significant.
Patients with “recently diagnosis migraine” represented a sub-group of subjects with “first-time diagnosis of migraine.”
Age, sex, smoking status, BMI, comorbities (diabetes mellitus, hypertension, hyperlipidemia), number of GP consultations, NSAIDs.
Age, sex, smoking status, body mass index (BMI), comorbidities (diabetes mellitus, hypertension, hyperlipidemia), number of general practitioner (GP) consultations, nonsteroidal anti-inflammatory drugs (NSAIDs), health resource utilization (0, 1, 2–4, 5+ contacts with health professionals because of migraine prior to the index date).
Risk of bias
Overall, most of the items considered for the risk of bias assessment were judged unclear (Figure 2). In particular, exposure was assessed on the basis of the date of the drug prescription (one study) or dispensation (three studies), assuming that patients had actually used the entire amount of prescribed/dispensed of medicines. Migraine ascertainment, as criteria for inclusion in the study population, was entirely based on diagnosis only in the study by Becker et al. (26). The latter also was the only one of the four included studies in which a case validation was performed. All studies attempted to control for potential confounding factors, although, in all cases, one or more important variables were missed (e.g. smoking status, oral contraceptive use). The study from Velentgas et al. (23) reported to have conflict of interests. Becker et al. (26) did not disclose any competing interests; however, three out of six authors were AstraZeneca employees.
Risk of bias graph.
Among studies assessing the risk for CV outcomes associated with the intensity of migraine-specific drug use, the study from Wammes-van der Heijden (24) showed the highest methodological quality. As for studies concerning CV risk related to the recency of drug exposure, the study by Becker et al. (26) had the lowest risk of bias.
The specific results obtained from the application of the NOS are reported in the online Supplementary file 2.
Data synthesis
Risk of CV events and intensity of migraine-specific drug use
Studies investigating the risk of CV outcomes on the basis of the intensity of drug use were pooled considering the highest exposure category reported in the relevant analysis (Figure 3). As for the study by Velentgas et al. (23), data on intensity analysis were obtained from the “current use” group.
Random-effects meta-analysis of cardiovascular outcome risk estimates related to the intensity of ergotamines or triptans use. Squares indicate study-specific point estimates; square size is proportional to the estimate precision; horizontal lines indicate the 95% CI; diamonds indicate the overall effect size for each exposure group (i.e. ergotamines or triptans). OR: odds ratio; 95% CI: 95% confidence interval.
The pooled-risk estimate for ergotamines resulted from two eligible studies that generated a significant OR value of 2.28 (95% CI 1.18–4.41; I2 = 0%) in absence of statistical heterogeneity.
Conversely, the pooled-risk estimate calculated for triptans (three eligible studies) was OR = 0.86 (95% CI 0.52–1.43; I2 = 24.5%), not statistically significant. Although the overall estimate showed a low I2 value, the forest plot inspection highlighted certain heterogeneity of the effect size reported from the three studies concerned.
Risk of CV events and recency of migraine-specific drug use
Only two studies investigating the risk of stroke in association with the recency of exposure to triptans were potentially comparable and eligible for the quantitative pooling of results (Figure 4). However, considering the high heterogeneity of the estimates respectively reported by the only two studies concerned (i.e. Velentgas (23) and Becker (26); test for heterogeneity: p = 0.02, I2 = 80.4%), we did not calculate the overall size effect size for this comparison.
Stroke risk estimates related to the recency of ergotamine or triptan use.
Only one study (i.e. Velentgas et al. (23)) evaluated the risk of stroke associated with ergotamine exposure (OR = 1.49; 95% CI 0.93–2.40).
Grade Application
On the basis of the GRADE methodology application, the overall quality of evidence was rated as “very low” both for ergotamines and triptans in studies investigating the CV risk related to either the intensity or the recency of use, meaning that estimates of effect were very uncertain for all the outcomes considered (online Supplementary file 3).
Discussion
Summary of findings
The present systematic review of the published literature identified only four observational studies (three case-control (24–26) and one cohort design (23)) in which the risk of serious CV outcomes associated with ergotamine or triptans was investigated among migraine patients.
In general, the retrieved studies addressed two distinct research questions: the risk of CV outcomes associated with either the intensity or the recency of drug use.
As for studies focusing on the intensity of drug use, the pooled estimate from the two papers concerning ergotamines (23,24) suggested, in the absence of statistical heterogeneity, that the intense use of these anti-migraine agents is associated with a more than two-fold increased risk of serious ischemic events. Additionally, among individuals with CV co-medications, the study by Wammes-van der Heijden (24) reported an eight-fold increased risk. Conversely, in the case of triptans, both the three individual studies (23–25) and the resulting overall effect size estimate did not highlight an increased occurrence of CV events among intense users. However, considering the small number of studies available together with the heterogeneity in terms of design, type of triptan users, outcomes and direction of the effect sizes reported, no firm conclusions can be drawn.
Concerning the risk associated with the recency of use of ergotamines or triptans, different CV outcomes (i.e. myocardial infarction, stroke, serious ventricular arrhythmia, unstable angina and transient ischemic attack) were individually investigated in the cohort study by Velentgas et al. (23). This study did not report any significant associations, either with ergotamines or with triptans. Becker et al. (26), instead, provided evidence on the risk of stroke only among migraineurs recently exposed to triptans (ergotamines were not investigated) and found a more than two-fold increased risk for triptan users compared to unexposed migraineurs. Although in the latter study the exposure to triptans was consistently associated with an increased risk of stroke across the different analyses performed, the authors preferred to be cautious with the interpretation of such results: when they additionally calculated the risk of stroke associated with recent triptan exposure among “recently diagnosed migraineurs,” they found an OR of 1.56 with large and non-significant confidence intervals (95% CI 0.26–9.53) in an over-selected sub-population (Table 3(b)). On the basis of the two above-mentioned studies, the only comparison eligible for quantitative data synthesis concerned triptans and stroke. However, we did not pool the risk estimates provided by Velentgas (23) and Becker (26) since, in this case, we considered the reporting of an overall risk estimates potentially misleading rather than useful. In fact, the effect size estimates from the two studies (23,26) had opposite directions around the unit. Such heterogeneity was possibly related to specific characteristics of the two studies (see Table 1), such as the type of triptan users concerned (prevalent vs new users), the length of the exposure time-windows considered (six months vs two months, respectively) as well as the confounding factors included in the statistical adjustment.
Overall, despite the fact that both triptans and ergotamines are largely used and have been prescribed worldwide for quite a long time (1,7,9), evidence from clinical practice experience concerning the risk estimate for rare but serious CV events are still scarce. The quality of the evidence gathered here, as rated using the GRADE approach, was “very low,” meaning that “we are very uncertain about the estimates.” Notably, in each of the four included studies at least one important confounding factor was not considered for the statistical adjustment, reducing the confidence in the relevant risk estimates reported. Finally, since triptans and ergotamine are contraindicated in patients with pre-existing CV risks, they are unlikely to be prescribed in these individuals. Therefore, due to possible residual confounding by indication or prescription bias in observational studies, the CV risk may remain underestimated.
Potential clinical/public health implications
To date, few studies have investigated the risk of rare but serious CV adverse reactions associated with triptans and ergotamines in real-world clinical practice. Notably, ergotamine and sumatriptan, although representing the oldest molecules of the respective drug class, remain the most preferred migraine-specific drugs worldwide (1).
As for ergotamines, according to the well-known effect on the peripheral vascular system (7–9), available data suggest that the intense consumption may be associated with an increased risk of serious ischemic complications, particularly in patients with pre-existing CV disorders. These findings should be taken in particular account in those countries where ergotamine can be freely purchased as an over-the-counter drug, including Italy (31). In fact, ergotamines are known to cause medication-overuse headache (7,8), a disorder that can lead patients to intensify drug consumption for the relief of increasingly frequent migraine attacks (32). Therefore, physicians and pharmacists should check the baseline CV risk of patients and inform them about the risk of ischemic complications associated with frequent use of ergotamines, discouraging self-medication.
Although studies on triptan safety do not suggest strong CV issues, firm conclusions cannot be drawn because of the very low quality of evidence. As for the specific risk of stroke, results from the two available studies are conflicting (23,26). However, considering that patients treated with triptans are generally at low baseline CV risk compared to other migraineurs (33), if an increase of the relative stroke risk does actually exist, the absolute risk increase must be small. In fact, only sporadically have triptans been reported in association with stroke cases (13,34,35) and no strong safety signals of such have been risen from the large number of clinical trials performed so far (3,11). Nevertheless, in light of the competing benefits expected from the treatment with these drugs (i.e. migraine relief), the possibility of an increased stroke risk, even if remote, should be taken into account when a choice must be made in clinical practice.
Strengths and limitations
The first strength of this review is represented by the sensitivity of the literature retrieval used, both as regards the database search strategy, the full-text screening and the hand-searching (see paragraphs 2.2 and 2.4 and 2.5). Such an approach allowed us to also identify important data reported in the study by Becker et al. (26), which, on the basis of the present systematic review, represents the only available evidence suggesting the possibility of an increased risk of stroke associated with recent triptan use. To the best of our knowledge, this study was never cited before in the context of the CV safety of triptans (36), not even in a recently performed pharmaco-epidemiologic systematic review on triptans (37).
A further strength is represented by the inclusion of studies enrolling migraine patients only, since migraine is considered an independent risk factor for CV diseases (15,16). Therefore, this approach strengthens the directness of the estimates collected, both single and pooled.
None of the four included studies actually provided single risk estimates per specific agent or formulation. In particular, this limitation could be particularly relevant for ergotamine-based medicines that frequently contain other active compounds such as NSAIDs or caffeine (5,31). Notably, the study by Velentgas et al. (23) used an even broader exposure definition (i.e. ergot alkaloids) in which, other than any formulation containing ergotamine or dihydroergotamine, methysergide was also included, the latter being an active principle with slightly different pharmacological properties, mainly used for migraine prevention and with a different pattern of consumption. We recognize the possibility, though remote, that some patients exposed to “ergot alkaloids” could have been exposed to methysergide only, and not “at least to” ergotamine or dihydroergotamine, as required by the inclusion criteria.
However, this represents a minor concern given that the study by Velentgas (23) did not report any significant results for the conclusions drawn in this review and, at the same time, its exclusion wouldn’t have meaningfully affected any of the pooled risk estimates reported here.
Although we did not force the quantitative data synthesis of results from studies with very different research questions (e.g. intensity vs recency of use), we pooled risk estimates coming from studies investigating heterogeneous CV outcomes related to the intensity of migraine-specific drug use (i.e. ischemic complications, cardiac events, stroke). In the specific case of triptans, the clinical heterogeneity of pooled studies, together with the differences in design features and effect size direction, further reduce the confidence in the overall risk estimate obtained.
Furthermore, no sensitivity analyses were possible because of the small number of studies available.
Recommendations for future studies
A paucity of comparative observational studies on the CV safety of migraine-specific drugs was highlighted by the results of this review. Therefore, further studies are needed to better define the risk for rare but potentially fatal CV events related both to triptans and ergotamines. In order to reduce the influence of potentially modifying factors, future studies should focus on populations of migraine patients only, possibly selected through direct diagnosis (e.g. Becker et al. (26)). Especially for triptans, the risk in new user populations should be further investigated, since the depletion of susceptible patients may cause dilution of the risk observed in prevalent users (25).
Although when using observational data the actual date of consumption of the prescribed/dispensed migraine-specific drug cannot be accurately ascertained (23–26), choosing an adequate length of the exposure time-window can mitigate such bias. In particular, it should be preferably narrower (e.g. two months or less) for the investigation of the acute CV toxicity related to the recency of exposure, while a wider time-window has to be preferred for the risk assessment related to the intensity of use (e.g. ≥6 months) in order to more likely identify patients with a consolidated pattern of intense use.
A validated case definition would reduce the risk of misclassification bias, which can have a strong impact on study results when rare events occur.
As shown from the four studies collected here, different factors should be considered as potential confounders: age, sex, comorbities, smoking status, body mass index and co-medications (e.g. NSAIDs, oral contraceptives and hormone replacement therapy). Moreover, stratification on the basis of CV status would provide important information on the risk related to these medicines in not-recommended situations.
As suggested by Becker et al. (26), the stroke risk for triptans should be further assessed among recently diagnosed migraine patients.
Furthermore, studies performed at the active substance level would help address treatment choice within the same drug class.
Finally, a meta-analysis of the risk estimates of serious CV events from all randomized clinical trials so far performed would provide, in parallel, complementary information for the overall assessment of the CV safety of triptans and ergotamines.
Clinical implications
Pre-existing cardiovascular (CV) risks represent the major limitation to the use of triptans and ergotamines. To date, few comparative observational studies have investigated the CV safety of migraine-specific drugs in “real-world” clinical practice. As for triptans, evidence collected here does not suggest strong CV safety issues, although a firm conclusion cannot be drawn. In particular, findings on stroke risk are conflicting. However, if an increase of the absolute stroke risk in patients recently exposed to triptans does actually exist, it must be small. Because of the above considerations, clinicians should maintain attention to patients’ baseline CV risk before prescribing triptans or other “first-line” attack therapies, such as nonsteroidal anti-inflammatory drugs (NSAIDs) or paracetamol. Health care professionals should inform patients using ergotamines that intense consumption may increase the risk of serious ischemic complications, particularly in individuals with baseline CV risks.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
None declared.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
