Abstract
Background
The clinical criteria for cluster headache (CH) are included in Chapter 3 of the International Classification of Headache Disorders, 3rd beta edition (ICHD-III). CH may sometimes be secondary to other pathologies.
Case reports
We report two patients in whom the clinical features of CH initially fulfilled the ICHD-III criteria, but who later presented some radical modifications in headache natural history as a result of a secondary pathology. The first case of CH was secondary to a pontine cavernous angioma and the second to a cerebral venous thrombosis.
Conclusion
We highlight the importance of clinical modifications of CH that could suggest clinical investigations should be performed or repeated to exclude a secondary pathology in a previously diagnosed cluster headache. Some of the pathological mechanisms of CH and brain lesions are discussed.
Keywords
Introduction
Cluster headache (CH) is a rare type of headache and usually occurs between the third and fifth decades of life in men who smoke and drink alcohol; there is peak incidence for both sexes between 20 and 29 years of age (1). The clinical criteria of CH are included in the International Classification of Headache Disorders 3rd beta edition (ICHD-III) (2). CH may also be a manifestation of other pathologies but, at the beginning, symptoms may be indistinguishable from the primary form of headache. A review of the literature identified 158 published cases of cluster-like headache (CLH) since 2008 (3). Many of these papers reported only a temporal correlation with the underlying disease and the symptoms did not satisfy all the criteria, whereas others reported radical changes in the frequency, intensity of pain, or in the responsiveness to drugs. We present the cases of two patients with CH. One case was secondary to a brainstem cavernous angioma and case was secondary to a cerebral venous thrombosis (CVT). We discuss the possible relation with multiple sclerosis (MS). Written informed consent was obtained from both patients.
Case reports
Case 1
A 71-year-old man with an unremarkable familial and personal history developed severe pain in the left periorbital region at the age of 51 years. The pain was constantly associated with lacrimation and conjunctival injection, which lasted 15–20 minutes, and preferentially recurred at 2 am with a frequency of one attack every other day for two months every six months. A diagnosis of episodic CH was made and he started treatment with carbolithium in association with desimipramine with an initial benefit. He was evaluated at our headache centre at the age of 65 years, when a brain magnetic resonance imaging (MRI) scan showed a lesion in the left pontine area, compatible with a cavernous angioma. This was not considered to be related to the episodic CH and no surgery was carried out. The headaches improved on treatment with corticosteroids and methysergide. After several pain-free months, the patient discontinued the methysergide and the pain returned with the same features. Sumatriptan and high-flow oxygen inhalation (7 l/min) were not completely effective, so treatment with verapamil and prednisolone was successfully started. After three years he abruptly developed a chronic CH that was not responsive to verapamil, sodium valproate or gabapentin. A new brain MRI scan showed a slight enlargement of the cavernous angioma (Figure 1(a)) and brain surgery was performed (Figure 1(b)). Complete resolution of the pain was present at a six-year follow-up examination.
Upper panel: pre-surgery brain MRI scan. (a), (b) Sagittal T1W, (c) axial T2W and (d) T1W images. The images show a small lesion in left lateral pontine region, localized near, and superomedial to, the trigeminal nerve root entry zone. Lower panel: post-surgery brain MRI scan. (a), (b) Sagittal T1W, (c) axial T2W and (d) T1W showing the removal of a small (biopsy-proved) cavernous angioma.
Case 2
A 29-year-old man with a silent medical history presented with a first episode of episodic CH characterized by a right-sided pulsating pain localized in the fronto-orbital and nasal regions, which lasted for two hours and recurred twice a day at the same time (around 2 am and 2 pm) for two weeks; the pain was accompanied by ipsilateral conjuntival injection, lacrimation and rhinorrhoea. Three months later he reported a second period of CH, initially with the same features, but, after a week, the pain became more intense, pressing and localized all over his head and was associated with a transient, fluctuating loss of visual. After a month of persistent attacks, he presented to our emergency department with fever, signs of meningeal irritation and intracranial hypertension. During his recovery, brain MRI was performed and showed non-specific white matter lesions (Figure 2(a)) and a left external jugular vein thrombosis climbing up to the distal portion of the sigmoid sinus (Figure 2(b)). His blood results were normal, although a cerebral spinal fluid examination showed evidence of IgG oligoclonal bands. Treatment with warfarin and acetazolamide was successfully started and one year later he was still pain-free.
(a) Brain MRI FLAIR sequences showing the alteration of white matter in the proximity of the corpus callosum and in the left periventricular region. (b) Angiography MRI sequences showing, in the venous phase, thrombosis of the distal portion of the left sigmoid sinus and the left external jugular (highlighted by the white arrow) and probable thrombosis of the proximal transverse ipsilateral sinus.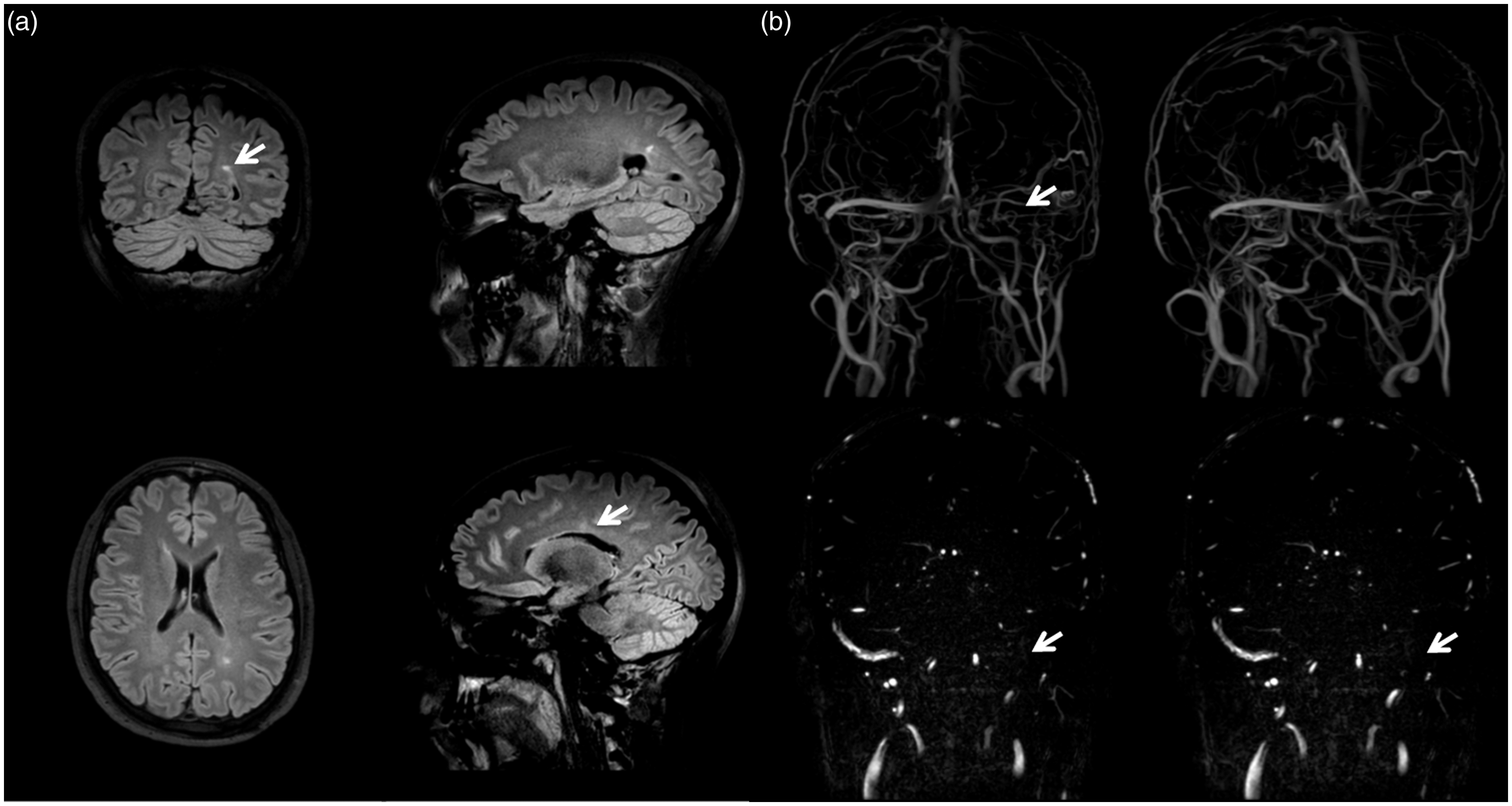
Discussion
The pathophysiology of CH is complex and still unclear; there is evidence to suggest the involvement of the trigemino-vascular system and central brain areas, but evidence on the link between them is lacking. The peripheral mechanism could explain CH related to facial pathologies in the trigeminal innervation areas. Lesions involving the ipsilateral trigeminal root support the role of the trigemino-vascular system in the pathogenesis of pain; its stimulation seems to be the key to the ignition of a process that successively involves the hypothalamus as the synchronizing structure of pain development. However, this peripheral pathway is not enough to explain the pain of CH. Matharu and Goadsby (4) reported a patient in whom sectioning of the trigeminal roots did not solve the pain; surgery performed on the trigeminal nerve is not associated with long-lasting benefits (5). The activation of central brain structures, in particular the hypothalamus, is confirmed by CH secondary to hypothalamic-hypophyseal pathologies, by positron emission tomography studies and by the efficacy of hypothalamic neuromodulator agents. All the available data suggest an integrated hypothesis that connects the two pathways, as noted by Van Vliet et al. (6) and by some ‘experimental clinical models’ represented by CLH (3).
Cluster headache and cavernous angioma
In previous reports, CLH secondary to different vascular diseases has accounted for 31.6% of reported cases, but none was related to cavernous angioma (3). We found reports of patients who developed migraine secondary to lesions in the midbrain and raphe-magnus nucleus, which stresses the importance of the brainstem in the pathogenesis of migraine (7). In our patient, the lesion mainly affected the root of the trigeminal nerve in the pontine insertion point, but an involvement of the central and parasympathetic areas has been to be considered in the pathogenesis of pain in this patient.
Cluster headache and cerebral venous thrombosis
The manifestations of CVT depend on the affected structure and the extent of venous thrombosis; it could mimic other diseases. The pain probably results from stimulation of the pain-sensitive large dural sinus and the sensory innervation of the dura mater and its vessels (the ophthalmic branch of the trigeminal nerve); their irritation could activate the autonomic pathway responsible for the pain syndromes that usually persist until the normalization of venous flow (8). Depending on the growth of the thrombosis, the onset of headache is gradual, diffuse and usually constant; there can be also sudden or explosive onset headaches (as in thunderclap headaches or subarachnoid haemorrhages) (9) or intermittently sharp headaches (as in migraine). There are few reports of CLH associated with CVT and there is no unequivocal theory to explain the symptoms. The cavernous sinus has been suggested as a pathophysiological locus. Orbital phlebograms obtained during attacks demonstrated an inflammatory vasculitic process in the ipsilateral-superior ophthalmic vein and in the cavernous sinus, which is probably due to venous stasis that obliterates the venous outflow (10). CVT in the straight sinus could involve the hypothalamus, explaining the periodicity of CH, whereas in superior sagittal sinus thrombosis an intracranial hypertension syndrome may develop.
Cluster headache and multiple sclerosis
Headache is not generally secondary to MS and the relationship is poorly understood; however, recent data show that headache is more common in patients with MS (about 50%) than in the general population (11). We found few cases of CH related to MS (12) and all patients presented demyelinating lesions in the pons or in the brainstem. The autonomic signs associated with CH could be mediated by the same central autonomic reflexes involved in the pathogenesis of MS. The demyelinating lesions might stimulate the trigemino-vascular system through the trigeminal (or cervical superior nerve) afferent fibres, triggering the process that leads to secondary CH. CH attacks as clinical overture symptoms of MS have to be considered as an exceptional condition, but a growing number of such cases have been described over the last few years (12).
Conclusion
There are few reported cases of CH related to CVT and even fewer of CH related to cavernous angioma. In these two patients, we observed a course of headache history that changed with an abrupt switch into forms that no longer fulfilled the ICHD criteria and were refractory to treatment. The cavernous angioma and CVT seemed not to be initially involved in the CH pain, but later became relevant to its pathogenesis and maintenance. Therefore we considered a causal connection between the CH and the presence of a midbrain cavernous angioma or an ongoing process of CVT because the patients remained pain-free a long time after the resolution of the underlying pathology. We could not make a clear diagnosis of MS, but the evidence of white matter demyelinating lesions on the brain MRI scan and oligoclonal bands on cerebrospinal fluid analysis requires close radiological and clinical follow-up.
Our experience allows some considerations about the mechanism of CH because there was a correlation between the lesions and CH pain in these two patients. Any drastic change in the clinical features of CH suggests that clinical investigations should be performed or repeated because the change could represent a red flag for a secondary pathology. In addition to the involvement of the hypothalamus and trigemino-vascular system, we also have to consider the brainstem (in particular the pons and midbrain) as a strategic area of functional linkage between the peripheral and central pathways of CH pain.
Clinical implications
Cluster headache could be the manifestation of a secondary neurological pathology such as cavernous angioma or cerebral venous thrombosis. Cluster-like headache helps to understand some mechanisms of cluster headache attack and can identify some strategic brain areas. Variations in headache features (i.e. duration or response to treatment) suggest that clinical investigations should be performed or repeated to exclude a secondary pathology in a previously diagnosed cluster headache.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
