Abstract
Background
Altered cerebrovascular tone is implicated in reversible cerebral vasoconstriction syndrome (RCVS). We evaluated vasomotor reactivity using bedside transcranial Doppler in RCVS patients.
Methods
In this retrospective case-control study, middle cerebral artery (MCA) blood flow velocities were compared at rest and in response to breath-hold in RCVS (n = 8), Migraineurs (n = 10), and non-headache Controls (n = 10). Hyperventilation response was measured in RCVS.
Results
In RCVS, Breath Holding Index (BHI) was severely reduced in seven of eight patients and 14/16 MCAs; seven of 16 MCAs showed exhausted (BHI < 0.1) or inverted (BHI < 0) vasomotor reactivity. Mean BHI in RCVS (0.23 ± 0.5) was significantly lower than Migraine (1.52 ± 0.57) and Controls (1.51 ± 0.32), p < 0.001. Triphasic velocity responses were seen in all groups. The maximum Vmean decline during the middle negative phase was −15.5 ± 9.2% in RCVS, −15.4 ± 7% in Migraine, and −10.3 ± 5% in Controls (p = 0.04). In the late positive phase, average Vmean increase was 6.2 ± 14% in RCVS, which was significantly lower (p < 0.001) than Migraine (30.5 ± 11%) and Controls (30.2 ± 6%). With hyperventilation, RCVS patients showed 23% decrease in Vmean.
Conclusion
Cerebral arterial tone is abnormal in RCVS, with proximal vasoconstriction and abnormally reduced capacity for vasodilation. Further studies are needed to determine the utility of BHI to diagnose RCVS before angiographic reversibility is established, and to estimate prognosis.
Keywords
Introduction
Reversible cerebral vasoconstriction syndrome (RCVS) comprises a group of conditions characterized by reversible segmental constriction and dilation of cerebral arteries (1–3). Most patients have recurrent thunderclap headaches and approximately one-third develop ischemic or hemorrhagic strokes. RCVS is a self-limited condition with angiographic normalization within three months and benign outcome in 90–95% of patients. Historically, the diagnosis has been challenging, especially upon presentation, with many patients misdiagnosed as primary angiitis of the central nervous system (PACNS).
The pathophysiology of the transient but prolonged arterial narrowing is not completely understood. Altered cerebral vascular tone, e.g. from endothelial dysfunction (4) or ultrastructural abnormalities (5), is implicated as a key underlying mechanism based on the dynamic nature of angiographic abnormalities (6), the temporal relationship of symptom onset to vasoconstrictive drug exposure (2,7), the absence of arterial inflammation on pathology (8), the presence of hemorrhages that are attributed to dynamic arterial caliber changes (reperfusion injury) (9), and the clinical-imaging overlap with posterior reversible encephalopathy syndrome (PRES) (10), in which endothelial dysfunction and altered cerebral vascular tone are considered important (11).
Cerebral vasomotor reactivity (CVR) can be assessed using bedside transcranial Doppler ultrasound (TCD) by measuring changes in blood flow velocity in response to changing arterial carbon dioxide levels (12,13). When cerebral blood flow is reduced, as in RCVS, the distal arterioles dilate at rest to compensate for the reduced blood flow, resulting in reduced capacity for further vasodilation in response to stimuli such as increases in carbon dioxide level. This results in reduced CVR. We hypothesized that CVR would be abnormal in RCVS, reflecting altered cerebral vascular tone.
Methods
Cases
From a total of 158 RCVS patients in our institutional review board (IRB)-approved institutional database (1998–2015), we retrospectively identified and included all eight patients who underwent bedside TCD with CVR testing for clinical indications (e.g. to assess stroke risk). All patients proved to have the classic features of RCVS without evidence of cerebral inflammation and documented angiographic reversal within three months, thus confirming the diagnosis of RCVS (1).
Control groups
For comparison, we used our previous TCD-CVR dataset of Migraine and non-headache Controls (14). The diagnosis of migraine was based on the International Headache Society criteria (15). Non-headache Controls included healthy individuals without prior headaches, known cerebrovascular or neurologic disease, and no history of drug abuse.
CVR testing
Our standard institutional protocol for TCD-CVR testing is as follows. Blood flow velocities are measured with 2-MHz monitoring transducers and a head-frame used to fix probes over temples throughout the procedure. Simultaneous recording of cerebral blood flow velocities (peak systolic velocity (PSV), mean velocity (Vmean) and end-diastolic velocities (EDV)) and pulsatility indices (PI) in the right and left middle cerebral arteries (MCA) is performed using standard techniques (16). Of note, the depth of insonation is set at 45–60 mm through transtemporal sonic bone windows. After probe fixation, a rest period of four minutes is allowed before measuring baseline mean velocities (Vbaseline) over a span of one minute, with normal respirations throughout. Then, participants are requested to initiate breath holding (apnea) for 30 seconds, or as long as possible (tapnea), taking care to avoid the Valsalva maneuver. The highest mean velocity measured within 4 seconds following termination of breath hold is defined as Vapnea (12). Breath Holding Index (BHI) is calculated as [(Vapnea − Vbaseline)/Vbaseline.tapnea %] (13). After a five-minute interval of normal respiration, Vbaseline is again recorded, after which the individual is requested to initiate hyperventilation, i.e. deep breaths at approximately 25 per minute for 30 seconds. The minimum mean velocity during hyperventilation is defined as VHPV and the time to this point is registered. Percentage change after hyperventilation (%ΔVHPV) is calculated as [(Vbaseline − VHPV)/Vbaseline)] × 100. Apnea and hyperventilation procedures are typically repeated after two-minute rest periods, and the mean of the two values noted for analysis.
Statistical analysis
SPSS v.21 was used for analysis. Wilcoxon signed-rank test, paired t-tests, and analysis of variance (ANOVA) were used as appropriate. A value of p < 0.05 (two sided) was considered significant. Data are shown as means, standard deviations, and percentages as appropriate.
Results
Clinical and imaging characteristics of RCVS patients.

ACA: anterior cerebral artery; CaCB: calcium-channel blockers; CEA: carotid endarterectomy; CTA: computerized tomography angiography; CVST: cerebral venous sinus thrombosis; DSA: digital subtraction angiography; F: female; ICH: intracerebral hemorrhage; IV: intravenous; M: male; MCA: middle cerebral artery; MRA: magnetic resonance angiography; Misc: miscellaneous (thyroid and parathyroidectomy); mRS: modified Rankin Score; ND: not done; PCA: posterior cerebral artery; PRES: posterior reversible encephalopathy syndrome; RCVS: reversible cerebral vasoconstriction syndrome; SAH: subarachnoid hemorrhage; TCH: thunderclap headache.
All RCVS patients presented with “thunderclap” headaches and had segmental cerebral artery narrowing on angiographic studies. Two patients (25%) developed infarcts located in arterial “watershed” regions, one had no parenchymal abnormality, and brain magnetic resonance imaging (MRI) findings in the remaining patients are shown in Table 1. Two cases have been published in detail (Case 4 and Case 6) (17,18) and all patients were included in a published collaborative study (2).
Ultrasound findings in RCVS patients.

PSV: peak systolic velocity; EDV: end diastolic velocity; MCA: middle cerebral artery; Vmean: mean blood flow velocity; PI: Pulsatility Index; BHI: Breath Holding Index; HPV: hyperventilation; %ΔVmean apnea: percentage changes from baseline to apnea termination; %ΔVHPV: percentage change of Vmean from baseline to minimum during hyperventilation. All velocities are cm/s.
A triphasic velocity response to breath-hold was seen in all three groups, as described previously (14): a short initial positive phase in which velocities increase above baseline, a middle negative phase during breath-hold, and a late positive phase starting before the cessation of breath-hold and continuing for a period after. The maximum Vmean decline during the middle negative phase was −15.5 ± 9.2% in RCVS, −15.4 ± 7% in Migraine, and −10.3 ± 5% in Controls (p = 0.04) (Figure 1(a)). In the late positive phase, the average Vmean increase was 6.2 ± 14% in RCVS, which was significantly reduced (p < 0.001) as compared to Migraine (30.5 ± 11%) and Controls (30.2 ± 6%) (Figure 1(b)). Further, when one participant (Case 6) with near-normal CVR was excluded, the Vmean increase in RCVS was only 2.5%. Vessel-based analysis of RCVS (n = 16 arteries) from baseline to the end of the breath holding period showed only a mild increase in PSV (123 ± 45 cm/s to 126 ± 42 cm/s, p = 0.365), Vmean (80 ± 30 cm/s to 83 ± 28 cm/s, p = 0.155) and EDV (59 ± 24 cm/s to 62 ± 23 cm/s, p = 0.191) and a mild decrease in PI (0.80 ± 0.18 to 0.77 ± 0.2, p = 0.277).
CVR indices in Control, Migraine, and RCVS groups. (a) Maximum Vmean decline during the middle negative phase. (b) Maximum Vmean increase after termination of breath-hold. (c) Breath Holding Index. *p < 0.05. CVR: cerebral vasomotor reactivity; RCVS: reversible cerebral vasoconstriction syndrome; Vmean: mean velocity.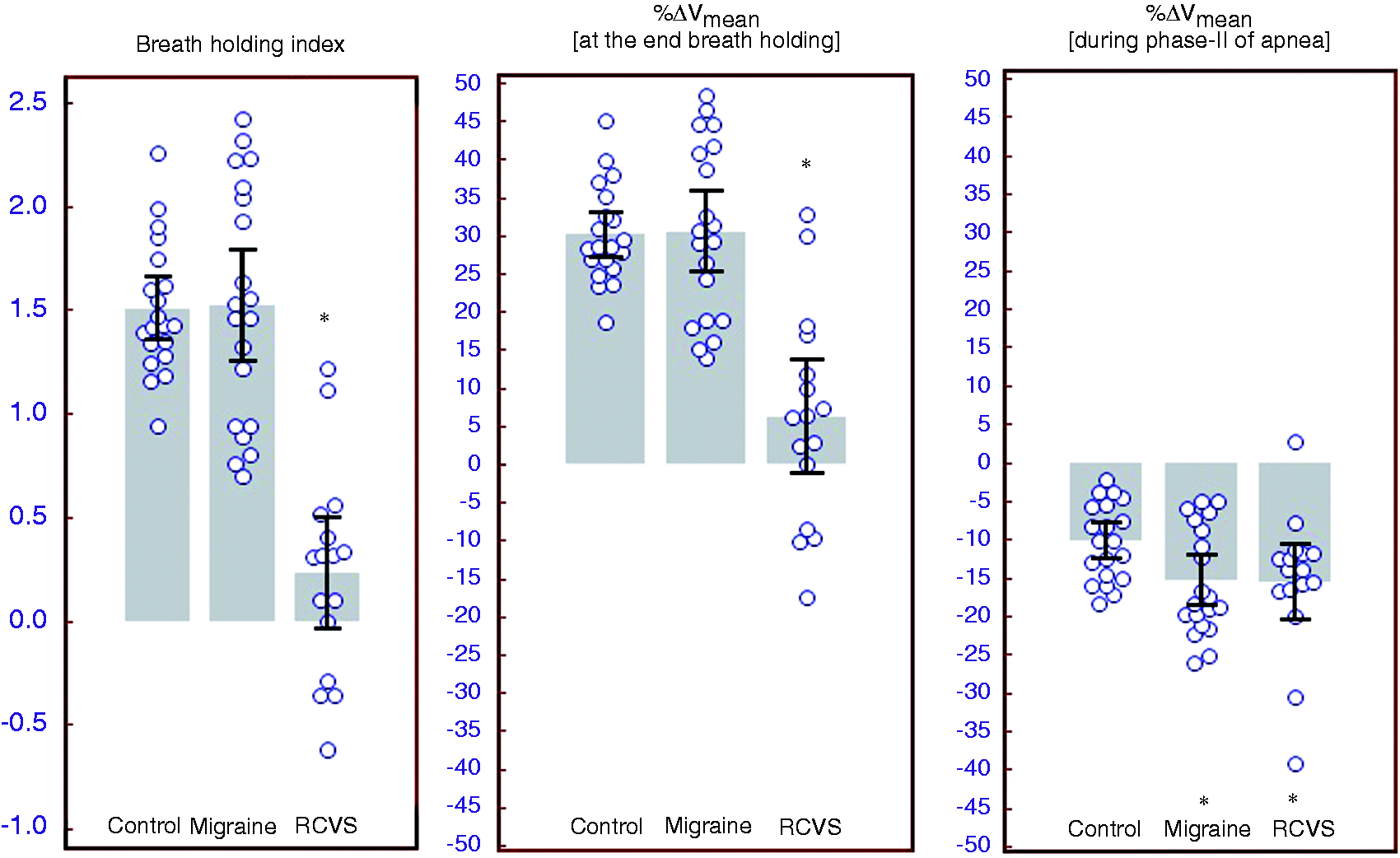
BHI in Migraine and Controls (Figure 1(c)) was within the expected normal range of 0.69–1.65 (20,21). However, seven RCVS patients (88%) had BHI values below 0.69, suggesting reduced CVR. Four RCVS patients (63%; Cases 1, 2, 3, 5, and 8) had either exhausted (BHI < 0.1) or inverted (BHI < 0) CVR. Only one RCVS patient (Case 6 (18)) had normal BHI. In contrast to the others, this patient developed RCVS of the ipsilateral MCA after left carotid endarterectomy, and did not have bilateral angiographic abnormalities (18). Among the four RCVS patients who had a history of migraine, three showed severely reduced (exhausted or reversed) BHI, and among the four without migraine, one showed severely reduced BHI (p = 0.5).
In the RCVS cohort, hyperventilation resulted in a significant decrease in blood flow velocities (PSV 115 ± 45 cm/s to 98 ± 36 cm/s, p = 0.001; Vmean 73 ± 25 cm/s to 56 ± 18 cm/s, p = 0.001; EDV 52 ± 17 cm/s to 35 ± 9 cm/s, p = 0.001) and a significant increase in PI (0.85 ± 0.22 to 1.10 ± 0.25, p = 0.002). The nadir occurred at 11.0 ± 1.8 seconds after the initiation of hyperventilation, and then the Vmean “plateaued” until the termination of hyperventilation at 34 ± 13 seconds. The percentage decrease in Vmean during hyperventilation, %ΔVHPV, was 23 ± 8% (range, 12.5–29.8%), which is less than the percentage decrease (38–50%) reported in normal individuals (21). The time to nadir was also shorter than expected since the average time is ∼20 seconds in normal individuals (21).
Although TCD-CVR testing was performed to assess stroke risk, only one RCVS patient (Case 8) developed new ischemic lesions consistent with RCVS progression on day 3 after the study.
Discussion
TCD evaluation of CVR using response to apnea and hyperventilation is a standardized, reproducible technique to study cerebral hemodynamics (13). It is considered safe, making it suitable to investigate stroke risk in vulnerable populations such as severe carotid artery or MCA stenosis. Multiple studies have shown that the BHI in normal individuals exceeds 1.0, with values ≤0.69 considered abnormal (12,20,21). As expected, BHI was normal in our Migraine and Control cohorts. In this first study of BHI in RCVS, we observed that 88% of RCVS patients have reduced BHI, with 50% showing severe reductions, suggesting a significantly impaired vasodilatory reserve at least during the acute phase. Importantly, BHI was reduced despite the heterogeneity of brain lesions (cSAH, infarcts, hemorrhages). Further, in normal individuals, velocities decrease by up to 50%, typically over 20 seconds, in response to hyperventilation (21). However we observed only a mild to moderate decrease occurring over a shorter time period, suggesting a relatively preserved vasoconstrictive capacity. Since blood flow velocity changes are primarily driven by responses in the distal vascular bed, our results provide insights into cerebral hemodynamics in RCVS, where the more proximal arteries are known to have prolonged segmental vasoconstriction, but knowledge about distal arteries changes is limited.
BHI reductions below 0.3–0.4, especially paradoxical flow reductions during apnea or negative BHI values, are usually associated with intracranial vascular steal (12), described as the “reversed Robin Hood” effect in patients with significant internal carotid artery or MCA stenosis (22). We found significantly reduced BHI in nearly all RCVS patients, and regions of hypoperfusion were documented in all patients undergoing perfusion-MRI, suggesting a high risk for ischemic stroke. The reversed Robin Hood effect might explain the frequent arterial “watershed” location of infarcts in RCVS. Three patients (Table 1) had hemorrhagic lesions (one intracerebral hemorrhage (ICH) and convexal subarachnoid hemorrhage (cSAH)), and recent studies have shown that patients with hemorrhagic lesions have a higher risk for infarcts as compared to patients without hemorrhages (9). Two of our eight patients (25%) had infarcts on admission, including one with cSAH who developed additional infarcts three days after the TCD study. Of note, patients had developed brain lesions prior to the TCD study; however, they were still able to cooperate with the test. The procedures may not be possible in patients with substantial lesion burden. Further studies are required to determine the safety and utility of BHI to predict infarct risk in RCVS, and whether patients with severe BHI reductions require close in-hospital monitoring until the BHI starts normalizing.
Of note, the only patient with near-normal BHI (Case 6) developed ipsilateral RCVS two days after left carotid endarterectomy (18). Some patients with severe, chronic carotid artery stenosis develop cerebral hyperperfusion syndrome from impaired hemispheric autoregulation (23); it is conceivable that these chronic effects resulted in a near-normal BHI despite RCVS.
Standard diagnostic TCD without any challenge has been used successfully to follow the clinical course of RCVS where improvement in blood flow velocities has been observed within days to weeks (7,24). Of note, only two of eight patients had Vmean fulfilling criteria for “mild” vasospasm, and the rest had marginally elevated Vmean. However, all except the patient with post-carotid endarterectomy RCVS had impressive reductions in BHI. We suggest that bedside TCD-CVR testing may be useful for the diagnosis of RCVS in the acute stage (before angiographic reversibility is documented). This may be particularly useful at the time of admission, since angiographic changes may progress from distal to proximal, being less apparent on initial studies (3).
Recent studies characterizing RCVS have made it relatively easy to diagnose this condition based on admission clinical-radiological features (25). Yet, some authors have proposed invasive angiography with intra-arterial vasodilator infusion to diagnose and distinguish RCVS from mimics (26,27). Unfortunately this strategy seems unjustified since it is invasive and carries the risk of stroke and reperfusion injury (8,25). We suggest that TCD-CVR may have significant advantages as a noninvasive, low-risk study for this purpose.
In summary, our novel results show that patients with RCVS have ubiquitous abnormalities on TCD-CVR testing. The features of our patients (Table 1) are similar to larger RCVS cohorts, and showed no significant differences from the 150 patients who did not undergo TCD-CVR testing in our cohort (data not shown), suggesting the generalizability of our results. Despite the consistency in results, we acknowledge limitations such as the low number of patients, treatment heterogeneity, and a retrospective study design that used previously collected TCD-CVR data for comparison. This resulted in a different mean age across groups; however, there was significant overlap in age, and in the absence of vascular risk factors it is highly unlikely that age had a significant impact on the observed differences in TCD-CVR. Another possible limitation is that the cohorts were studied in different settings (RCVS were inpatients and controls were outpatient/research), with different operators. While we were careful to avoid recordings after an inadvertent Valsalva, it is possible that the first phase of the triphasic response was partly explained by this maneuver. However the three groups were studied using an identical protocol and only the RCVS group had a highly abnormal response. We believe this low cost bedside test with an established safety profile may have utility in the acute diagnosis and prognosis of patients with features of RCVS. Further prospective studies are warranted to confirm these preliminary observations.
Clinical implications
These results support the hypothesis that abnormal cerebral autoregulation is involved in the pathophysiology of reversible cerebral vasoconstriction syndrome (RCVS). Cerebral vasomotor reactivity testing using transcranial Doppler ultrasound may be a useful, low-cost bedside test for the acute diagnosis and prognosis of RCVS.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
