Abstract
Objective
A prospective clinical imaging study has been conducted to investigate pain processing functional pathways during trigeminal heat stimulation (THS) in patients with migraine without aura experiencing ictal cutaneous allodynia (CA) (MwoA CA+).
Methods
Using whole-brain BOLD-fMRI, functional response to THS at three different intensities (41°, 51° and 53℃) was investigated interictally in 20 adult MwoA CA+ patients compared with 20 MwoA patients without ictal CA (MwoA CA−) and 20 healthy controls (HCs). Secondary analyses evaluated associations between BOLD signal change and clinical features of migraine.
Results
During moderate-noxious THS (51℃), we observed a significantly greater activation in (a) the anterior cingulate cortex in MwoA CA+ patients compared to HCs and (b) the middle frontal gyrus in MwoA CA+ patients compared to both MwoA CA− patients and HCs. Furthermore, during high-noxious THS (53℃) a significantly decreased activation in the secondary somatosensory cortices was observed in (a) MwoA CA− patients compared to both MwoA CA+ patients and HCs and (b) MwoA CA+ patients compared to HCs. CA severity was positively correlated with the secondary somatosensory cortices activation.
Conclusions
Our findings suggest that CA may be subtended by both a dysfunctional analgesic compensatory mechanism and an abnormal internal representation of pain in migraine patients.
Introduction
Approximately two-thirds of migraine patients complain of cutaneous allodynia (CA), which is defined as a pain perception evoked by ordinary non-nociceptive skin stimulation (1) in cephalic or, less frequently, extracephalic regions (2) during migraine attacks. Although the prevalence of central sensitisation (2) underlying CA appears highest in chronic migraine (3), episodic migraine patients experience CA with high frequency and severity as well (4). Converging evidence has suggested that CA may be underlied by the sensitisation of second-order trigeminovascular neurons, belonging to the trigemino-thalamo-cortical pathway (TTC), that could maintain their activity even in the absence of sensory inputs (5). In this context, a crucial role also seems to be played by an impaired descending pain modulatory system (DPMS) (6). In recent years, both stimulus-related and connectivity functional magnetic resonance imaging (fMRI) studies have provided further insights into the neural correlates of migraine CA (7–10). However, most of these studies have used a region-of-interest (ROI) approach, which may limit a comprehensive interpretation of CA-related functional changes in migraine patients.
Therefore, the aim of the present study was to explore the functional reorganisation both of the DPMS and ascending TTC pathway using whole-brain blood oxygen level-dependent (BOLD)-fMRI during trigeminal heat stimulation (THS) in migraine without aura (MwoA) CA+ patients compared to MwoA patients not experiencing CA (MwoA CA−) and healthy controls (HCs). Furthermore, we used voxel-based morphometry (VBM) (11) and diffusion tensor imaging (DTI) (12) to assess whether potential functional changes were dependent on structural or microstructural brain differences. We hypothesise that MwoA CA+ patients are characterised by a peculiar functional reorganisation of both the DPMS and ascending TTC pathway even during the interictal period (9,13,14).
Methods
Patient population
Clinical characteristics of patients with MwoA CA+, MwoA CA− and HCs.

MwoA CA+ vs MwoA CA−. bMwoA CA+ vs HCs. cMwoA CA- vs HCs.
MwoA CA+: patients with migraine without aura with cutaneous allodynia; MwoA CA−: patients with migraine without aura without cutaneous allodynia; HC: healthy controls; F: female; M: male; HRSA: Hamilton Rating Scale for Anxiety; HRSD: Hamilton Rating Scale for Depression; MIDAS: Migraine Disability Assessment Scale; HIT-6: Headache Impact Test; VAS: Visual Analogue Scale; ASC-12: Allodynia Symptom Checklist 12.
Twenty age- and sex-matched, right-handed Caucasian individuals were recruited as HCs via advertisements placed in the hospital (e.g. posters and flyers), via word-of-mouth referrals, and from a database of research volunteers maintained by the MRI Research Center of the Second University of Naples (Table 1).
All patients and HCs underwent preliminary MRI examination before entering the present study.
Standard protocol approvals, registrations, and patient consents
The experiments conformed to the principles of the Declaration of Helsinki and were approved by the ethics committee of the Second University of Naples. All participants provided informed, written consent after the experimental procedure had been explained.
Stimuli
THS was performed using the contact heat-evoked potential stimulator (CHEPS) (Medoc Ltd, Ramat Yishai, Israel) as in our previous study (14). The CHEPS has a probe with (at one end) a thermode area of 572.5 mm2 and a heating thermo-foil (Minco Products Inc, Minneapolis, MN, USA), covered with a 25-lm layer of thermo conductive plastic (Kapton®, thermal conductivity at 23℃ of 0.1–0.35 W/m/K), characterised by a rapid rising time at high temperature (up to 70℃/s) developed to study pain activation related to the thermal and nociceptive pathways. Previous fMRI studies (17) mainly stimulated the first branch of the trigeminal nerve (V1) alone or along with the second branch of the trigeminal nerve (V2). Herein, we chose a selective stimulation within the receptive field of the maxillary division of the trigeminal nerve (V2) to avoid interferences due to preferential V1 first-order trigeminovascular neurons sensitisation in the course of episodic MwoA attacks (5,18). Indeed, sensitisation of the first-order neurons provides the neural basis for the migraine headache that is localised in the oculo-fronto-temporal region in the majority of patients (19). Patients were tested on the side of the head more frequently affected during migraine attacks whereas the HCs were matched with regard to the side of the face tested in migraine patients. A low-innocuous stimulus at 41℃ (14) and two painful heat stimuli at 51° and 53℃ (to provide a moderate-noxious and a high-noxious stimulus) were used (9,14). To minimise the effects of both phenomena of habituation and expectation, all experimental stimuli were given in random modality.
fMRI
Experimental protocol
Migraine patients and HCs performed three consecutive fMRI sessions for each of the three different thermal stimuli (41°, 51° and 53℃) according to event-related experimental designs as previously reported (14). In each fMRI session, by means of the fMRI-compatible thermode, 28 short thermal stimuli (600 ms) were applied in each of three fMRI sessions at three different intensities (41°, 51° and 53℃) with a jittered inter-stimulus interval (ISI) of 14 ± 1 s (total session duration 7 min 45 s). Prior to the fMRI experiment, outside the scanner, migraine patients and HCs were fully informed about the characteristics of the applied THS. After each fMRI session, there was a delay of about 30 s during which participants, inside the scanner, had to verbally rate the intensity perception of the experimental stimulus by means of a numerical rating scale (NRS) ranging from 0 (‘no pain’) to 10 (‘worst pain imaginable’), which were then logged in by the experimenter (20).
Imaging parameters
MRI was performed on a 3-T scanner (HDxt, GE Healthcare, USA) equipped with an eight-channel parallel head coil. Each fMRI scan consisted of 300 volumes of a repeated gradient-echo echo planar imaging (EPI) sequence (repetition time (TR) = 1500 ms, echo time (TE) = 30 ms, number of axial slices = 29, matrix = 64 9 64, field of view (FOV) = 256 mm, thickness = 4 mm, interslice gap = 0 mm). Three-dimensional T1-weighted images (FSPGR BRAVO sequence, voxel size = 0.9 × 0.9 × 1.2 mm3) were acquired in the same session in order to have high-resolution spatial references for registration and normalisation of both functional images and atrophy measures.
Whole-brain DTI was performed using a gradient recalled echo (GRE) EPI sequence (TR = 10,000 ms, TE = 88 ms, FOV = 320 mm, isotropic resolution = 2.5 mm, b value = 1000 s/mm2, 32 isotropically distributed gradients, frequency encoding right-left (RL)). A T2-fluid-attenuated inversion recovery (T2-FLAIR) sequence (RT = 9.000 ms, ET = 1.200 ms, IT = 2.500, axial slices = 44, matrix = 224,448, FOV = 240 mm, thickness = 3 mm, interslice gap = 0 mm) was also acquired in all participants.
fMRI imaging pre-processing and statistical analysis
fMRI data pre-processing and statistical analyses are reported in the Supplementary material.
Structural MRI and white matter (WM) hyperintensities load
Structural (VBM and DTI) data pre-processing and statistical analyses are reported in the supplemental data. WM hyperintensities load was calculated both in migraine patients and HCs (see Supplementary material).
Results
Behavioural data (pain ratings)
Pain intensity ratings were not significantly different between MwoA CA+ patients, MwoA CA− patients and HCs at any level of experimental stimuli (Figure 1).
Behavioural response to innocuous and noxious THS in MwoA CA−, MwoA CA+ and HCs. THS: trigeminal heat stimulation; MwoA: migraine without aura; CA: cutaneous allodynia; HC: healthy controls.
Imaging data (fMRI, VBM and DTI)
During THS at three different intensities, functional changes were identified in the three groups in several regions known to be involved in pain processing (14) (p = 0.05 cluster level corrected).
During the low-innocuous THS (41℃), no differences in BOLD-response were observed between MwoA CA+ patients, HCs and MwoA CA− patients.
During the moderate-noxious THS (51℃), when comparing patient groups among them and with HCs, we observed (a) a significantly greater activation in the perigenual part of the left anterior cingulate cortex (ACC) (Figure 2(a)) in MwoA CA+ patients compared to HCs and (b) a significantly greater activation in the left middle frontal gyrus (MFG) in MwoA CA+ patients compared both to HCs and MwoA CA− patients (Figure 2(b)).
Significant different activation in group comparisons between patients with MwoA CA−, MwoA CA+ and HCs. MwoA: migraine without aura; CA: cutaneous allodynia; HC: healthy controls.
Furthermore, during high-noxious THS (53℃) a significantly decreased activation in the bilateral secondary somatosensory cortex (SSC) was observed both in MwoA CA+ and MwoA CA− patients compared to HCs. In the same region, a significant decreased activation was observed in MwoA CA− patients compared to MwoA CA+ patients (Figure 3). Talairach coordinates of significant cluster differences between groups are reported in Table 2.
Significant different activation in group comparisons between patients with MwoA CA−, MwoA CA+ and HCs. MwoA: migraine without aura; CA: cutaneous allodynia; HC: healthy controls. Significant cluster differences between patients with MwoA CA+, MwoA CA− and HCs (p < 0.05 cluster level corrected). MwoA CA+: patients with migraine without aura with cutaneous allodynia; MwoA CA−: patients with migraine without aura without cutaneous allodynia; HCs: healthy controls; BA: Brodmann area; L: left; R: right.

Mean values of percentage BOLD signal changes extracted from the perigenual part of ACC, MFG and SSC clusters are reported in Figures 2 and 3.
There were no local regional grey matter (GM) differences in both the whole-brain and ROI-based analyses between MwoA CA+ patients, HCs and MwoA CA− patients, neither at a statistical threshold corrected for multiple comparisons (p < 0.05 familywise error rate (FWE)) nor at an uncorrected threshold (p < 0.001; cluster size:100).
Tract-based spatial statistics showed no significant differences among MwoA CA+ patients, HCs and MwoA CA− patients for fractional anisotropy, mean diffusivity, radial diffusivity and axial diffusivity maps in whole-brain and ROI-based analyses (p < 0.05 cluster level corrected). Furthermore, the three patient groups were similar in terms of WM hyperintensities load (mean ± SE MwoA CA+: 28.12 ml ± 9.81; MwoA CA−: 12.19 ml ± 2.94; HCs: 10.61 ml ± 4.48; MwoA CA+ vs MwoA CA− p = 0.12; HC vs MwoA CA+ p = 0.11; HC vs MwoA CA− p = 0.76).
Correlation analyses
There were no statistically significant correlations between any clinical parameters of migraine severity and BOLD signal changes during moderate-noxious THS (51℃). During high-noxious THS (53℃), a significant negative correlation was found between BOLD signal change in SSC and mean pain intensity in the course of migraine attacks (assessed using the VAS) both in MwoA CA+ patients (p = 0.01; r = −0.57) and MwoA CA− patients (p = 0.05; r = −0.43) (i.e. the lower the activation, the greater the mean pain intensity during migraine attacks). Furthermore, a significant positive correlation was found between BOLD signal change in SSC and CA severity (p = 0.02; r = 0.52) in MwoA CA+ patients (i.e., the higher the activation, the greater the CA severity) (see Figure 4). Interestingly, there were no statistically significant correlations between perceived pain intensity and BOLD-fMRI signal changes at any level of experimental stimuli in MwoA CA+ patients and MwoA CA− patients.
Relation between SSC cerebral activity during high-noxious THS and clinical parameters in patients with MwoA CA+. SSC: secondary somatosensory cortex; THS: trigeminal heat stimulation; MwoA: migraine without aura; CA: cutaneous allodynia.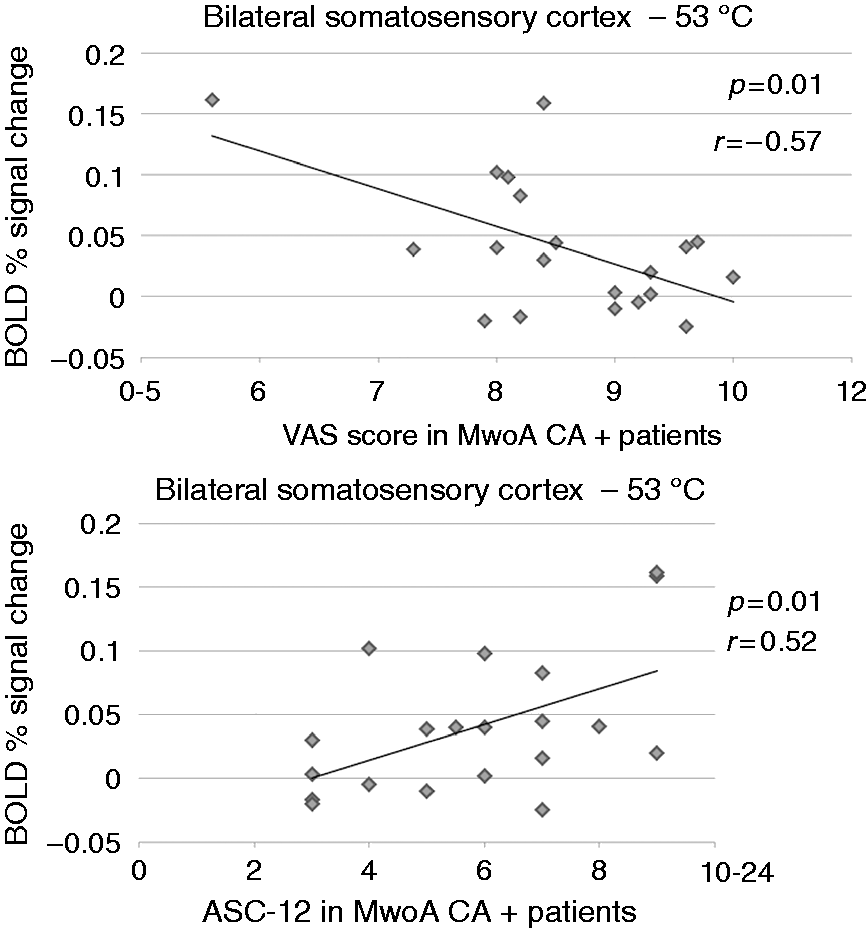
Discussion
In the present study, using a whole-brain BOLD-fMRI approach, we have demonstrated that MwoA CA+ patients exhibited a peculiar functional reorganisation of the pain-processing network. In detail, an increased activation of the prefrontal cortex (PFC), centred in the MFG, during a moderate-noxious stimulus and a reduced activation of bilateral SSC during a high-noxious stimulus were observed in MwoA CA+ patients when compared to HCs. However, during the latter stimulation, the SSC exhibited a greater activity in MwoA CA+ than in MwoA CA− patients. In this context, allodynia severity was positively correlated with SSC functional response. Interestingly, pain intensity ratings were not significantly different between the three groups at any level of THS.
Over recent decades, compelling evidence has suggested the existence of endogenous mechanisms that contrast pain inputs through a descending inhibition network (i.e. DPMS) (18,21) opposite to ascending TTC projections involved in nociceptive inputs driving to the SSC (22). Specifically, DPMS may produce inhibition or facilitation effects on nociceptive transmission by descending influences via brainstem structures anatomically connected to each other as well as to higher-order pain processing regions in the frontal lobes, thalamus, cingulate cortex, amygdala, insula, and hypothalamus (18). Among these brain regions, ACC has been considered a key structure in analgesia mechanisms as well as in the affective and emotive dimension of pain, such as self-related negative emotional states (23). The role of ACC in pain processing has been demonstrated in our previous study (14), in which we observed an increased BOLD response of this area during moderate-noxious THS, likely related to an enhanced functional antinociceptive mechanism in patients with MwoA. A different specific function in pain processing is associated to PFC, which is involved in modifying pain perception independently of nociceptive inputs changes and in sustaining internal representations of pain expectations (21). Previous imaging studies (24,25) have demonstrated a correlation between increased PFC activity during experimentally induced CA, reflecting its involvement in this specific phenomenon. Furthermore, increased PFC activity has been observed in emotional states that could modify pain perception (26,27). Mainero and colleagues (8) demonstrated a reduced functional resting-state connectivity between the PAG and several brain regions with a predominant role in pain modulation such as PFC and ACC in a patient group including patients with MwoA and migraine with aura (MwA) compared with HCs, suggesting an interictal impairment of the descending pain modulation, likely leading to loss of pain inhibition. Interestingly, this pattern of significantly reduced connectivity between PAG, PFC and ACC was also observed in patients with migraine with CA compared to patients with migraine without CA, corroborating the hypothesis that pain modulatory neurons might be involved in the development of CA. More recently, a seed-based fMRI study (10) has shown functional connectivity abnormalities between frontal regions and brainstem areas involved in the DPMS in MwoA CA+ patients. On the other hand, the SSC subserves the sensory-discriminative dimension of pain processing within the TTC pathway (21,22,27). Previous imaging studies have demonstrated an interictal SSC hypometabolism (28) in migraine patients and a reduced activity in episodic (14) and chronic migraine (29) patients during experimental nociceptive stimulations. Recently, a BOLD-fMRI study (7) has shown a reduced, interictal, SSC response to transcutaneous electrical nerve stimulation, in MwoA CA+ patients when compared with MwoA CA− patients. These findings have been interpreted as a functional response to repeated and consistent painful stimulations, which likely decreases painful inputs to the cortex by a cortical downregulation (14,20,30).
In previous fMRI studies (17) THS has been applied with an MRI-compatible contact thermode with the temperature individualised to each patient. The behavioural findings of these studies have suggested a low heat pain threshold in migraine patients during the interictal period. This could be considered inconsistent with our findings on pain intensity ratings. However, it is well known that pain intensity is different from pain threshold (31) and to the best to our knowledge, data on pain intensity ratings of noxious stimuli and relative neural correlates in MwoA CA+ patients are missing to date and could, therefore, provide further insight in understanding pathophysiological mechanisms of CA. Therefore, our study demonstrated that, during the interictal period, pain intensity ratings are not significantly different between both CA+ and CA− migraine patients and migraine patients as a group compared to HCs. Therefore, taken together, our findings may suggest that during moderate-noxious THS, the ACC activation could represent an analgesic compensatory mechanism, or alternatively a prominent affective and emotive response to pain in migraine patients regardless of CA. Moreover, we have observed an increased response in the MFG only in MwoA CA+ patients, which may represent a recruitment of additional resources to meet analgesic demands and to maintain proficiency in patients reporting CA during attacks (5). However, because of PFC involvement in emotional and cognitive aspects of pain (21), we cannot rule out the possibility that MFG activation might be related to pain-negative cognitive content and consequent inadequate pain coping in MwoA CA+ patients, as in other neuropathic conditions (30). Nevertheless, although abnormal activity of the ACC and frontal regions has been reported in patients with depression (32), patients with migraine (both with MwoA CA− and MwoA CA+) did not show significant impairments in tests exploring both depression and anxiety, compared to HCs.
Furthermore, the observed pain network functional reorganisation also involves the SSC as a complementary TTC analgesic mechanism aimed at reducing high-noxious painful input to the cortex in migraine patients (14,29). Interestingly, this SSC-mediated functional downregulation seems to be more prominent in MwoA CA− patients than MwoA CA+ patients. We believe that an imbalance between the inhibition and the facilitation of pain dynamics might contribute to dysfunctional analgesic mechanisms in migraine. This could lead to ictal CA in the course of attacks in patients with MwoA CA+. We cannot exclude that this functional analgesic reorganisation may become progressively less efficient over time, leading to the appearance of CA during the interictal period as well (6). Our hypothesis is further corroborated by correlation analyses revealing that the SSC functional activity, during high-noxious THS, is negatively associated with the mean pain intensity in the course of migraine attacks (i.e. VAS scores) both in MwoA CA− and MwoA CA+ patients. Furthermore, CA severity (i.e. ASC-12 score) was positively correlated with SSC activity in MwoA CA+ patients (i.e. the greater the SSC activation, the worse the CA).
It is noteworthy that the observed functional differences are not related to any structural changes as suggested by our negative VBM and DTI results. Although structural and microstructural abnormalities have been previously reported, GM and WM involvement in migraine patients is still under debate (33). However, a higher sensitivity of surface-based methods in the detection of migraine-related structural changes has been demonstrated (34), and a recent study has revealed an atypical correlation between cortical thickness in the superior temporal/inferior parietal region and cutaneous pain thresholds in patients with migraine (35). In our opinion, the present study has some strengths compared to previous observations. The stimulation paradigm has been conducted by specific stimuli in trigeminal areas on the side of the head more frequently affected during migraine attacks, compared to prior investigations in which a transcutaneous electrical nerve stimulation was applied on the medial forearm (7). Furthermore, migraine patients and HCs have been investigated by means of a multiparametric (i.e. functional, structural and microstructural) MRI approach. Finally, in comparison to earlier investigations (9), we report group differences that are statistically significant over the entire brain without a priori regional hypothesis on specific pain processing areas.
Nevertheless, we are aware that our study is not exempt from some limitations as well. First, we cannot completely rule out the possibility that our data may be partially influenced by a habituation or sensitisation phenomenon associated with repetitive THS, although the absence of increased or decreased pain intensity ratings during the stimulation session may support the exclusion of these mechanisms. However, this is a limitation that may affect all task-based studies with painful stimuli due to repetitive stimulations according to the event-related design paradigms. Furthermore, it is well known that several physiological studies have shown an absence of normal habituation in the brains of patients with migraine during the interictal period in response to various stimuli. The decrease in habituation probably relates to dysfunctional inhibition or increased facilitation of sensory information and perhaps further contributes to atypical pain processing in migraine (see Schwedt et al. (17) for a systematic review). Similarly, we cannot rule out a potential influence on our data of fluctuating brain functional changes between ictal and interictal periods, as recently observed in patients with migraine (36). Second, we cannot answer the question as to whether the observed functional differences in DPMS and TTC activations are acquired through a genetic predisposition or as a result, over time, of migraine burden on the higher order pain-modulating areas. Finally, because of the small sample size of different groups, further studies should be performed to confirm and extend our observations.
In conclusion, our findings suggest that CA may be subtended both by a dysfunctional analgesic compensatory mechanism and an abnormal internal representation of pain in migraine patients.
We believe that a better understanding both of migraine and CA pathophysiology is crucial for the comprehension of mechanisms involved in central sensitisation and migraine chronification.
Article highlights
Approximately two-thirds of migraine patients complain of cutaneous allodynia (CA) during migraine attacks. Although the prevalence of CA appears highest in chronic migraine, episodic migraine patients experience CA with high frequency and severity as well. CA may be subtended by both a dysfunctional cortical analgesic compensatory mechanism and an abnormal internal representation of pain, with consequent inadequate pain coping, in migraine patients. Progress in the knowledge of CA pathophysiology is crucial for the comprehension of mechanisms involved in central sensitisation and migraine chronification.
Footnotes
Acknowledgments
Author contributions include: Dr Russo: experimental design, image data analysis, results interpretation, manuscript drafting and revision; Prof Esposito: image data analysis, statistical analysis and results interpretation; Dr Conte: clinical data analysis; Dr Fratello: image data analysis and results interpretation; Dr Caiazzo: image data analysis and results interpretation; Dr Marcuccio: literature review, experimental design, results interpretation; Dr Giordano: literature review, experimental design, results interpretation, manuscript revision; Prof Tedeschi: experimental design, results interpretation, manuscript revision; Prof Tessitore: experimental design, image data analysis, results interpretation, manuscript revision.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Professor Tedeschi has received speaker honoraria from Sanofi-Aventis, Merck Serono, Bayer Schering Pharma, Novartis, Biogen-Dompé AG; has received funding for travel from Bayer Schering Pharma, Biogen-Dompé AG, Merck Serono, Novartis and Sanofi Aventis; and serves as an associate editor of Neurological Sciences. Professor Tessitore has received speaker honoraria from Novartis, Schwarz Pharma/UCB, Lundbeck, Abbvie and Glaxo. The other authors have nothing to declare.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study has been conducted with a grant from the Italian Foundation of Headaches (Fondazione Italiana Cefalee FI.CEF.).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
