Abstract
Background
Intravenous infusion of adenylate cyclase-activating polypeptide-38 (PACAP38) provokes migraine-like attacks in 65–70% of migraine sufferers. Whether aggregation of migraine in first-degree relatives contributes to this discrepancy in PACAP38-induced response is unknown. We hypothesized that genetic enrichment plays a role in triggering of migraine and that migraine without aura patients with a high family load ( ≥ 2 first-degree relatives with migraine) would report more migraine-like attacks after intravenous infusion of human PACAP38.
Methods
In this study, we allocated 32 previously genotyped migraine without aura patients to receive intravenous infusion of 10 pmol/kg/min PACAP38 and recorded migraine-like attacks including headache characteristics and associated symptoms. Information of familial aggregation was obtained by telephone interview of first-degree relatives using a validated semi-structured questionnaire.
Results
PACAP38 infusion induced a migraine-like attack in 75% (nine out of 12) of patients with high family load compared to 70% (14 out of 20) with low family load (
Conclusion
Migraine response to PACAP38 infusion in migraine without aura patients is not associated with high family load or the risk allele of rs2274316 (
Keywords
Introduction
The role of pituitary adenylate cyclase-activating polypeptide-38 (PACAP38) in migraine pathophysiology has gained considerable interest in recent years (1). Intravenous infusion of PACAP38 induces migraine-like attacks in 60–70% of migraine patients and dilates extracerebral arteries (2,3). The role of genetic factors in the heterogeneous PACAP38 response in migraine patients is unknown.
Pharmacological migraine provocation may be a novel approach to explore the contribution of genetics to migraine susceptibility (4). The genetic basis in the most prevalent type of migraine, migraine without aura (MO), is complex with probably many loci determining disease susceptibility (5,6). Aggregation of migraine in first-degree relatives of probands with migraine implies enrichment of migraine susceptibility genes (7,8). A clinical study in healthy volunteers suggested that susceptibility of migraine-like headache to pharmacological provocation is associated with familial aggregation of migraine (9), but the International Headache Society (IHS) criteria for migraine were not used. Several susceptibility loci have been identified in migraine in recent years (10) suggesting many interesting pathways in migraine pathophysiology even though their exact function is unknown. One of the single nucleotide polymorphisms (SNPs) rs2274316 identified recently confers a small increased risk of migraine. It is localized within the
Here, we investigated whether the hypersensitivity to PACAP38 experienced by two-thirds of MO-patients could be explained by aggregation of migraine in first-degree relatives (family load). We hypothesized that MO-patients with high family load would report more migraine-like attacks after intravenous infusion of human PACAP38 than patients with low family load. We conducted a balanced and double-blinded study in 32 genotyped patients with MO and obtained the migraine history of their first-degree relatives by direct interview. In addition, we did an explorative analysis of whether carriers of the
Materials and methods
The patients were recruited from a cohort of 1010 unrelated MO patients from the Danish Headache Center, who were genotyped for the 12 migraine-associated SNPs (10) for follow-up studies (12,13). The risk allele C of rs2274316 (
The history of migraine of the patient's first-degree relatives (parents, siblings and children) was obtained via a telephone interview based on a validated semi-structured questionnaire (14,15). Migraine (MO or with aura) was diagnosed according to the 3rd edition of the International Classification of Headache Disorders beta version (ICHD-3β) (16). Patients identified with ≥ 2 first-degree relatives with migraine were defined as having a high family load, whereas patients identified with ≤1 first-degree relative with migraine were defined as having a low family load. Trained senior medical students conducted the interviews and were blinded in respect of the PACAP38 response and genotype of the proband.
More information about the study population, phenotyping, genotyping and risk allele has been fully described elsewhere (12).
Design
We conducted a double-blinded study, in which participants and research fellows performing all recordings were blinded in respect of the genotypes. All subjects received a continuous intravenous infusion of 10 pmol/kg/min human PACAP38 (Bachem AG, Bubendorf, Switzerland) over 20 min as in our previous provocation studies (2,3).
All participants gave their written informed consent to participate in the study. All women used sufficient contraception. Exclusion criteria were any other type of headache (except episodic tension-type headache ≤ 8 days per month); intake of any daily medication (except migraine preventives); serious somatic or psychiatric diseases. A full medical examination and electrocardiography (ECG) were performed on the day of the study. We told the patients that PACAP38 might induce headache in some individuals, but the timing or the characteristics of headache was not discussed.
The study was approved by the Regional Committee on Health Research Ethics of Copenhagen (H-2-2013-033) and the Danish Data Protection Agency and was conducted according to the Helsinki II declaration of 1964, as revised in 2008. The study was also registered at ClinicalTrials.gov (NCT01841827).
Experimental protocol
All participants arrived non-fasting at the clinic between 08:00 and 11:00 hours. They had to be without any kind of headache or intake of analgesics 48 h before the study day. All fertile female patients had a pregnancy test at the beginning of each study day. Venous catheters (Venflon®) were inserted into the right and left antecubital vein for the administration of PACAP38 and drawing of blood samples (data and blood sampling protocols will be reported in a separate paper: Part II). The patients were supine in quiet surroundings and, after 15 min of rest, we measured the baseline values (at 10 min before start of infusion (T-10) and at the time of infusion start (T0)) of headache intensity and vital signs and started the infusion using an infusion pump (Braun Perfusor, Melsungen, Germany). We observed the patients for 90 min post infusion.
Headache intensity and questionnaire
Headache intensity was recorded at T-10 and then every 10 min up to 90 min after the start of infusion on a verbal rating scale (VRS) from 0 to 10: 0 is no headache; 1 represented a very mild headache (including a sensation of pressing or throbbing or otherwise altered sensation in the head not associated with pain); 5 is headache of moderate intensity; 10 is the worst headache imaginable (17). Headache localization, characteristics, associated symptoms and premonitory symptoms (unusual fatigue, yawning, thirst, craving, mood swings and neck stiffness) were also recorded. After discharge from the hospital, the patients were carefully instructed to continue recording their headache by a self-administered questionnaire every hour until 12 h after the start of infusion or until they went to bed. The questionnaire recorded headache characteristics and associated symptoms according to the IHS criteria (16) and also included questions concerning adverse events, premonitory symptoms and if the reported headache mimicked the spontaneous migraine attacks. The patients were allowed to take their usual acute migraine medication at any time.
Migraine-like attack criteria
Vital signs
Heart rate (HR) and mean arterial blood pressure (MAP) were measured at baseline (T-10 and T0) and then every 10 min until 90 min after the start of infusion using an auto-inflatable cuff (Omega 1400, Orlando, FL, USA).
Statistical analysis
Headache intensity scores, peak headache intensity and median time to onset of migraine-like attacks after PACAP38 infusion were presented as median (range). HR and MAP data were presented as mean values ±SD.
Calculation of sample size was based on the difference between two groups reporting PACAP38-induced migraine-like attacks after PACAP38 infusion (0–12 h), at 5% significance with 80% power. We assumed that PACAP38 would induce migraine-like attacks in at least 80% of patients with high family load and migraine-like attacks in less than 15% of patients with low family load. We chose 65% difference because previous pharmacological provocation studies demonstrated that about 20% of patients did not report migraine attacks after triggers (2,3,19). The hypothesis was that those patients are not sensitive to triggers because of low family load. We estimated that inclusion of 12 subjects in each group would be sufficient (http://biomath.info/power/ based on J.L. Fleiss, et al., Statistical Methods for Rates and Proportions).
The primary end-points were the difference in incidence of migraine-like attacks and the difference in area under the curve (AUC) for headache intensity scores (0–1.5 h and 1.5–12 h) between two groups. Secondary end-points were differences in incidence of any head pain, in AUC for MAP and HR during the in-hospital phase (0–1.5 h). In addition, we conducted an exploratory analysis of the difference in incidence of migraine-like attacks between patients with and without the
The incidence of migraine-like attacks, head pain, associated symptoms, premonitory symptoms and adverse events between two unrelated groups were analysed as categorical data with chi-square test except when the cell count was less than five then Fisher's exact test was applied. We calculated AUC according to the trapezium rule (20) to obtain a summary measure to analyse the differences in response between the two groups of patients. Difference in AUC for headache intensity scores were tested using the non-parametric Mann–Whitney
The
Results
Thirty-two genotyped MO-patients completed the study and family history of migraine was assessed in all patients. Twelve patients had a high family load of migraine, whereas 20 had a low family load (Table 1). In total, 101 out of 162 first-degree relatives were interviewed and migraine was diagnosed in 37 of these. In addition, we diagnosed migraine in six first-degree relatives based on information from parents or the proband. For more information, see Table S1 supplementary material.
Description of the two groups of patients with high or low family load.

Three patients in each group with high and low family load had missing values in their headache intensity scores of 1 to 5 hours, because they went to bed or tried to sleep through the attack. Data at these time points were excluded from the AUC analysis and an extrapolation was done.
Six (out of 32) patients had previously participated in a similar provocation study using calcitonin gene-related peptide (CGRP). Three of these patients reported attacks after both CGRP and PACAP38, while one patient did not report attack on both study days.
Migraine-like attacks and headache
Details of daily medication intake of all patients with high or low family load.

Clinical characteristics and associated symptoms of the spontaneous and provoked migraine attacks in the nine migraine patients with high family load who developed migraine-like attacks.

NS: not stated.
Headache characteristics: Localization/intensity/quality (throb: throbbing; pres: pressing)/aggravation (by cough during in-hospital phase and by movement during out-hospital phase).
Associated symptoms: Nausea/photophobia/phonophobia.
Migraine-like attacks are defined according to criteria, described in Methods.
PACAP38 infusion induced a migraine-like attack in 75% (9 out of 12) of patients with high family load compared to 70% (14 out of 20) with low family load ( Family load of the participating migraine patients (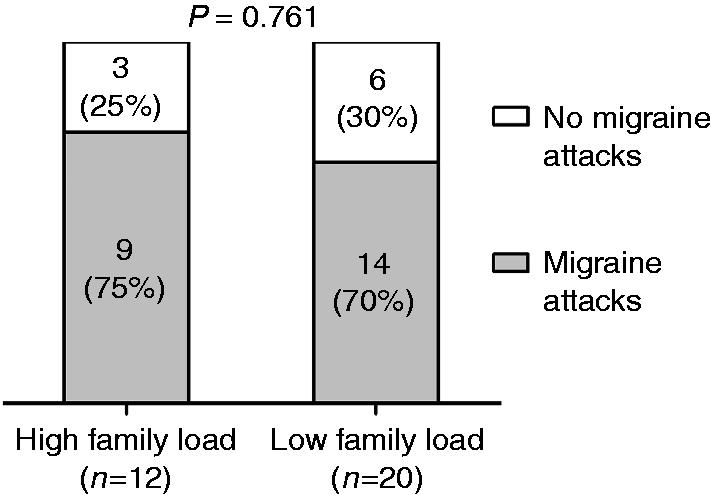
Median duration of migraine attacks for high load patients was 1 h (range 0.16–6 h) while median duration for low load also was 2 h (range 1–8 h). Seven out of 12 (58%) patients with high load took rescue medication compared to 15 out of 20 (75%) with low load (
Headache characteristics and migraine-associated symptoms induced by PACAP38 showed no difference between the two groups (
Clinical characteristics and associated symptoms of the spontaneous and provoked migraine attacks in the 14 migraine patients with low family load who developed migraine-like attacks.
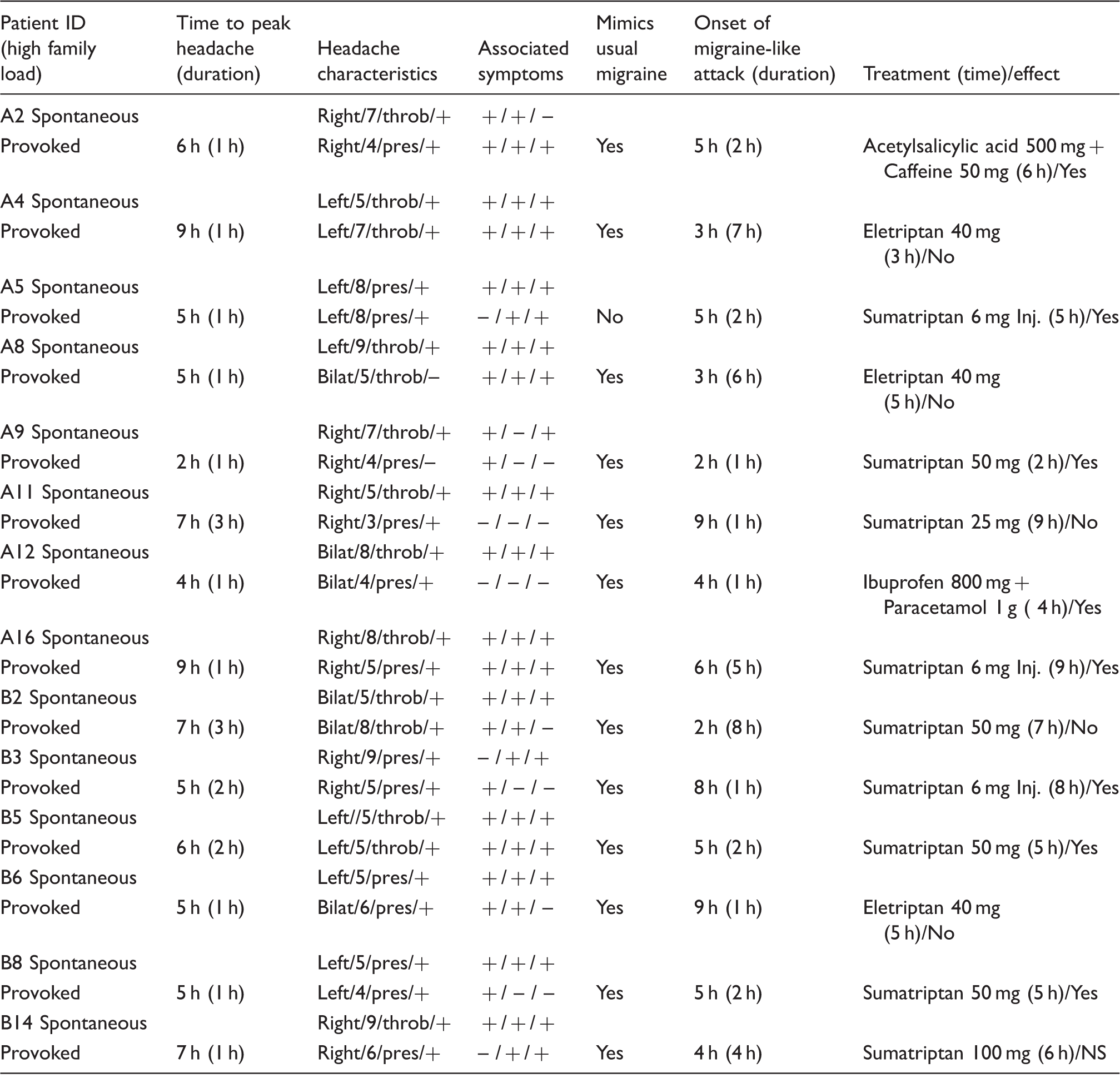
NS: not stated.
Headache characteristics: Localization/intensity/quality (throb: throbbing; pres: pressing)/aggravation (by cough during in-hospital phase and by movement during out-hospital phase).
Associated symptoms: Nausea/photophobia/phonophobia.
Migraine–like attacks are defined according to criteria, described in Methods.
There was no difference between the two groups in the incidence of any head pain or in the AUC for headache intensity over the 12 h observation period (Figure 2).
Median (thick red line) and individual (thin lines) headache intensity on a 0–10 VRS for 12 MO patients with high family load and 20 patients with low family load. Median peak headache score after PACAP38 was 4 (range 1–7) in patients with high family load and also 4 (range 1–8) in patients with low family load. There was no difference in the AUC between high and low family load (AUC0-12 h: 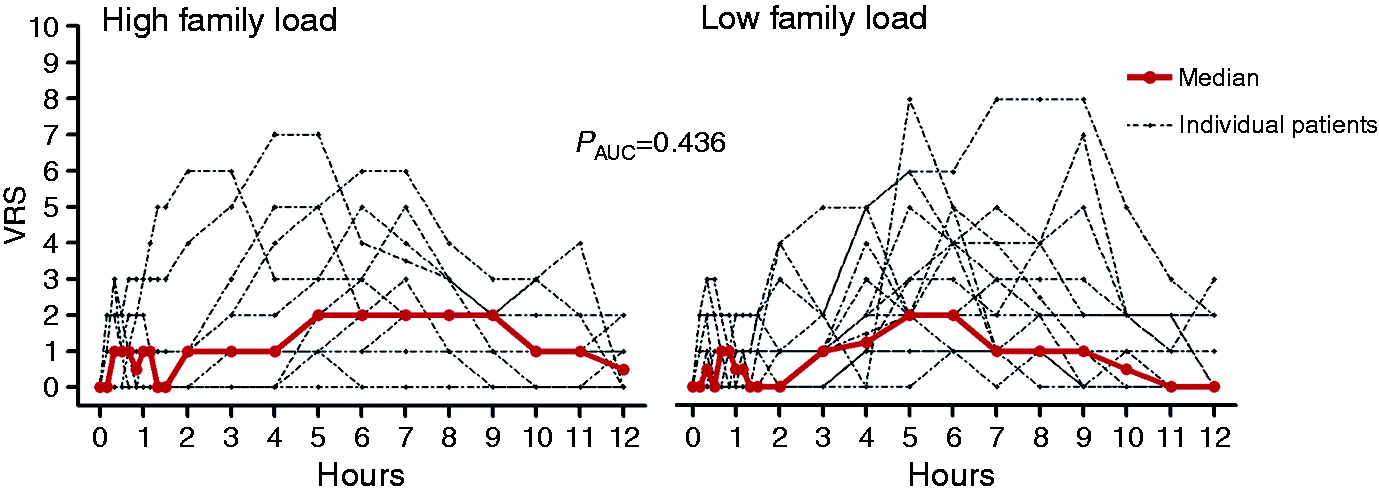
Explorative analysis of the rs2274316 risk allele (MEF2D)
Sixteen patients carried double risk alleles of rs2274316 (
HR and MAP
We found no difference in HR and MAP between patients with high and low family load (HR AUC0-2 h,
Discussion
The major outcome of the present study is that PACAP38 infusion did not induce more migraine-like attacks among MO-patients with high family load (75%) than in patients with low family load (70%). We also found that the
Familial predisposition is a risk factor for a majority of common chronic diseases (diabetes, cardiovascular disease, asthma and several cancers) and greater increase in risk is associated with an increasing number of affected first-degree relatives (21–25). Our definition of high family load was partly based on studies of other diseases showing that two first-degree relatives significantly increased the risk of disorder (22,24). Therefore, we defined genetic enrichment in patients when at least two first-degree relatives suffer from migraine, which affects roughly 15% of the population (26,27). Hence, having one first-degree relative with migraine is likely to occur by chance. The strengths of the present study include blinding of participants and investigators in respect of family load and genotype, a well-characterized patient group and the use of direct telephone interview based on a validated semi-structured questionnaire to diagnose first-degree relatives (15,28) according to the latest IHS criteria (16). Direct interview with each relative is required to obtain accurate information on migraine in families, because proband report is not sufficiently sensitive (29,30).
We acknowledge that we could not get in touch with all first-degree relatives by phone. Accordingly, migraine diagnosis in six (out of 43) relatives was based on report from the proband or parents. Furthermore, we did not account for the number of siblings in our calculation of high and low family load. Another factor that might influence our results is a higher number of patients with high family load on preventive migraine medication compared to patients with low family load (75% vs. 30%). It is possible that preventive medication reduced the incidence of migraine-like attacks. One placebo-controlled study found that valproate reduced the number of migraine attacks induced by nitroglycerin (17% vs. 50%) (31). However, none of the participants in the present study used valproate as preventive medication. In addition, the incidence of migraine-like attack among the 15 patients who took preventive medication was 67%, which is similar to the overall migraine response after PACAP38 (60–70%).
Reported symptoms in patients with high and low family load 0–12 h after administration of PACAP38.

Data are shown as % (
The rs2274316 conferring risk of migraine is localized intronically within the
The findings of the present study are in line with our recent provocation study using CGRP in 40 MO patients, which showed no association between family load and migraine response induced by CGRP (40). This study also reported that a high number of the currently known risk-conferring SNPs of MO did not contribute to the susceptibility of CGRP-induced migraine attacks. Interestingly, both CGRP and PACAP38 are endogenous neuropeptides that activate adenylate cyclase and increase production of signalling molecule cyclic adenosine monophosphate (cAMP) (41,42). Collectively, these data suggest no association between familial predisposition and hypersensitivity to the cAMP-signaling pathway in migraine patients. It would be interesting to investigate the contribution of familial predisposition to other signalling pathways that are implicated in migraine, e.g. the pathway of cyclic guanosine monophosphate (cGMP)-signalling (43). Another approach would be to investigate the susceptibility of induced migraine-like attacks in large families with many affected individuals.
In conclusion, we used a novel approach to investigate the functional consequences of migraine genetics by the provocation model of PACAP38 (4). We were unable to show an effect of family load or the risk allele of rs2274316 (
Article highlights
Family history of migraine cannot explain the hypersensitivity of MO patients to PACAP38 infusion. The risk allele of rs2274316 (
Footnotes
Acknowledgement
The authors thank all participating patients.
Declaration of conflicting interest
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Jes Olesen has received grants and/or research support from, has been a consultant and/or scientific adviser for, and has been in the speakers' bureau of Allergan Inc, AstraZeneca Pharmaceuticals LP, Boehringer Ingelheim, Eli Lilly, GlaxoSmithKline, Janssen Pharmaceutical Products, Lundbeck, Merck, Amgen, Alder and Pfizer. Messoud Ashina is a consultant and/or scientific adviser/speaker for the ATI, Allergan, Amgen, Alder and Eli Lilly. Song Guo has received travel grant from the ATI.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: grants from Novo Nordisk Foundation (NNF11OC1014333), Independent Research-Medical Sciences (FSS) (DFF-1331-00210A), Lundbeck Foundation (R155-2014-171) and the FP7-EUROHEADPAIN (602633).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

