Abstract
Background
Migraine, especially chronic migraine (CM), causes substantial disability; however, health care utilization has not been well characterized among patients receiving different migraine prophylactic treatments.
Methods
Using a large, US-based, health care claims database, headache-related health care utilization was evaluated among adults with CM treated with onabotulinumtoxinA or oral migraine prophylactic medications (OMPMs). Headache-related health care utilization was assessed at six, nine, and 12 months pre- and post-treatment. The primary endpoint was the difference between pre- and post-index headache-related health care utilization. A logistic regression model was created to test the difference between onabotulinumtoxinA and OMPM-treated groups for headache-related emergency department (ED) visits and hospitalizations.
Results
Baseline characteristics were comparable between groups. The proportion of patients with ED visits or hospitalizations for a headache-related event decreased after starting onabotulinumtoxinA, but increased after starting an OMPM, for all three cohorts. Regression analyses showed that the odds of having a headache-related ED visit were 21%, 20%, and 19% lower and hospitalization were 47%, 48%, and 56% lower for the onabotulinumtoxinA group compared to the OMPM group for the six-month, nine-month, and 12-month post-index periods, respectively.
Conclusions
When compared with similar patients who initiated treatment with OMPM, onabotulinumtoxinA was associated with a significantly lower likelihood of headache-related ED visits and hospitalizations.
Keywords
Introduction
Migraine is the leading cause of years lived with disability among neurological disorders worldwide (1–3). Individuals with chronic migraine (CM) experience greater headache impact (4) and headache-related disability (5,6), decreased health-related quality of life (HRQoL) (4,6) and reduced productivity (6,7) when compared with individuals who experience episodic migraine (EM). Additionally, patients with CM versus EM are also more likely to have other chronic comorbid conditions, including obesity, vascular disease, and psychiatric disorders (e.g. depression, anxiety) (4,5). Although EM is the more prevalent of the two types, CM occurs in an estimated 1.4% to 2.2% of the global population (8) and nearly 1% of the United States (US) population (9).
As a result of headache-related effects and higher comorbidity occurrence, individuals affected by CM make more visits to health care providers (4,7), particularly headache specialists and neurologists, compared to those with EM (6,7). In addition, the direct and indirect costs of CM have been reported to be four times higher than those associated with EM (10). Overall, the economic burden of CM is considerable and is driven primarily by increased health care resource use (11).
Prophylactic therapies for CM have a potential to influence health care resource utilization and care-associated costs consequent to their impact on headache frequency, migraine symptoms (e.g. pain intensity), and benefits to HRQoL. Although several oral migraine prophylactic medications (OMPMs; e.g. antiepileptic drugs and beta blockers) have been studied for prophylaxis of EM (12), onabotulinumtoxinA is the only therapy with an approved indication (13) and international guideline recommendations (14–16) specifically for prophylaxis of headache in patients with CM. OnabotulinumtoxinA has proven efficacious in significantly reducing headache days per month, minimizing pain, severity, and symptoms caused by migraine (17–20), reducing headache-related disability (17–21), and improving HRQoL (17–21) in patients with CM. The efficacy and safety of onabotulinumtoxinA have been shown in long-term studies extending up to 56 weeks (22). Although not approved specifically for CM, OMPMs for EM have been used as prophylactic treatment for patients with high-frequency migraine (23,24) and may also be effective in CM (24–26).
Little is known about the health care resource utilization in patients with CM who are being treated with onabotulinumtoxinA or OMPMs. A small single-center retrospective study found that in the six months following initiation of onabotulinumtoxinA, in patients with treatment-refractory CM, there was a significant reduction in migraine-related hospitalizations and visits to the emergency department (ED) and urgent care centers (27). While there have been some studies exploring the impact of OMPMs on health resource utilization in patients with EM, to date there have been no such studies published among CM patients being treated with OMPMs, nor any comparative effectiveness studies between these two types of treatments. Given the burden CM imposes on the health care system, large-scale studies are needed to validate the results from the Rothrock et al. (27) study and demonstrate the real-world effects of onabotulinumtoxinA or OMPMs in the general commercially insured CM population. To address this need, using data from a large, US-based health care claims database, headache-related health care resource utilization was evaluated among patients with CM before and after treatment with onabotulinumtoxinA or OMPMs.
Methods
Database
Claims data from Truven MarketScan® Databases were used. MarketScan Databases are longitudinal, patient-level, integrated sources of medical and pharmacy claims that include more than 200 million unique patients since 1995, with more than 66 million unique patients per year. Data are collected from health care plans, employers, and state Medicaid agencies, and include claims from hospitalizations, office visits, and prescription claims from all retail, mail order, and specialty pharmacies (28). Patients are enrolled in a wide range of health insurance plans (i.e. health maintenance organization (HMO), preferred provider organization (PPO), point-of-service), and the data are representative of the US commercially insured population across age, sex, and geography. The datasets are compliant with the Health Insurance Portability and Accountability Act (HIPPA) (29). All patient data are de-identified within the database; therefore it was determined that this study does not require institutional review board approval.
Observation period
The observation period was from January 1, 2008 to September 30, 2013. Headache-related health care utilization was assessed over three time periods from six, nine, and 12 months pre-treatment to six, nine, and 12 months post-treatment following initiation of either onabotulinumtoxinA or OMPMs after diagnosis of CM (Figure 1).
Study design. OMPM: oral migraine prophylactic medication.
Cohort selection criteria
Eligible patients had a diagnosis of CM (International Classification of Diseases, ninth revision, Clinical Modification (ICD-9-CM) code 346.7x (30)) and were ≥18 years of age at diagnosis. CM diagnosis had to occur within the observation period. Patients must have had a six-month washout period before CM diagnosis (i.e. no inpatient or outpatient claims for CM) to ensure that only newly diagnosed CM patients were included. In addition, patients were required to have ≥1 claim for onabotulinumtoxinA or an OMPM that was dated after the CM diagnosis date. The index date was defined as the date of first onabotulinumtoxinA injection (code J0585 with either a Current Procedural Terminology (CPT) code 64615 or ICD-9 code 346.7x on an outpatient claim) or first pharmacy claim for an OMPM that occurred after the date of CM diagnosis. Patients were excluded if they had received onabotulinumtoxinA for any indication during either the pre-index (six, nine, or 12 months prior to the index date) or washout period (six months prior to CM diagnosis).
Patients were required to have continuous medical and pharmacy coverage during the washout period and for ≥6 months, ≥9 months, and ≥12 months before and after the index date for each of the six-month, nine-month, and 12-month cohorts, respectively. During the post-index period, patients were also required to be persistent with therapy. For the onabotulinumtoxinA group, persistence was defined as ≤120 days between injections and the date of discontinuation was determined as 90 days after the last injection, because onabotulinumtoxinA has a recommended retreatment schedule of 12 weeks, per the Food and Drug Administration (FDA)-approved label (13). For the OMPM group, persistence was defined as a ≤30-day gap between two consecutive claims for the same OMPM. Date of discontinuation was determined based on the end of days’ supply of the last fill. The OMPMs included in the analysis were anticonvulsants (topiramate, divalproex/valproate, carbamazepine, gabapentin), antidepressants (amitriptyline, nortriptyline, fluoxetine, citalopram, sertraline), and antihypertensives (metoprolol, propranolol, atenolol, nadolol, bisoprolol, lisinopril, candesartan, nicardipine, nifedipine, verapamil, clonidine).
Baseline characteristics
The number of office visits to a health care provider during the pre-index period and the type of provider (neurologist versus nonspecialist) were analyzed at baseline. In addition, the use of acute migraine medications during the pre-index periods was assessed by recording the number days’ supply dispensed for each patient by drug class. Comorbidities, identified by the first or second ICD-9 code listed on inpatient and outpatient claims during the pre-index period, were reported for a set of specific disease states that were identified in previous studies (31,32) to be relevant to the EM and CM populations as well as by a commonly used aggregate score, the Charlson Comorbidity Index (33).
Outcome measures
The study endpoint was the difference between pre- and post-index use of headache-related health care resource utilization among patients with a CM diagnosis who had received treatment with onabotulinumtoxinA or OMPMs. Resource utilization was identified as “headache related” if the ED or hospitalization claim contained a diagnosis code for headache or migraine (ICD-9 codes: migraine, 346.x; tension headache, 307.81; premenstrual tension syndromes, 625.4; headache, 784.0). Hospitalization was based on the primary diagnosis at discharge whereas outpatient claims for ED visits were based on first diagnosis code listed on the event claim. Resource utilization was assessed using patient-level and event-level data.
Statistical analysis
Descriptive statistics were generated for demographics, frequency of pre-index outpatient office visits, comorbid conditions (33,34), pre-index use of OMPMs, and days between CM diagnosis and first claim for the intervention. Categorical variables were presented as counts and proportions and continuous variables were presented as means with standard deviations. Pre- and post-index period comparisons for the study endpoint (ED visits and hospitalizations) and number of patients with ≥1 acute migraine treatment, stratified by drug class and individual drug type, were analyzed using McNemar’s Chi-square test.
For between-group comparisons, a logistic regression model was used, with treatment (onabotulinumtoxinA versus OMPMs) as the independent variable and outcome (≥1 versus 0 post-treatment headache-related ED visit or hospitalization) as the dependent variable. Aside from general demographic variables such as age, gender, and region, a number of covariates were identified as potential confounders. Specifically, insurance type, the number of headache-related office visits during the pre-index period, and whether the patient has seen a neurologist during those visits, may impact the quality and extent of health care a patient receives. Other covariates included in the model are linked to indicators of disease severity or variables that may affect the probability of having future ED visits or hospitalization. Such variables include the number of OMPMs tried during the pre-index period, the number of headache-related ED visits or hospitalizations (based on the outcome being assessed in the regression) during the pre-index period, and baseline triptan and opioid use (defined as the sum of days’ supply dispensed during the pre-index period). The adjusted model created using these variables was used to calculate odds ratios (ORs) for the occurrence of an event (i.e. ED visit or hospitalization) in the onabotulinumtoxinA versus OMPMs cohorts.
A sensitivity analysis was also completed to compare onabotulinumtoxinA to only those OMPMs that have been identified by the American Academy of Neurology 2012 guidelines as having Level A evidence (labeled as Level A OMPMs Cohort) for use in migraine prophylaxis. Medications identified as Level A are metoprolol, propranolol, topiramate, and divalproex/valproate (12). All analyses were conducted using SAS software version 12 (SAS Institute Inc., Cary, NC). Two independent programmers provided quality control of the programming codes used in the analysis.
Results
Patient disposition and demographics
Between January 2008 and September 2013, the Truven databases contained claims data for more than 262 million commercially-insured individuals. During this period, 131,448 people had an ICD-9-CM diagnosis code for CM, of whom 123,010 were aged ≥18 years (Figure 2). Ultimately, 3840 patients who received onabotulinumtoxinA and 12,170 who received OMPMs were persistent with therapy and were included in the analysis of headache-related resource use during the six-month cohort. For the nine- and 12-month cohorts, the onabotulinumtoxinA group consisted of 1831 and 936 patients, respectively, and the OMPM group consisted of 6989 and 4057 patients, respectively.
Patient selection process. BTX: onabotulinumtoxinA; CM: chronic migraine; OMPM: oral migraine prophylactic medication.
Baseline demographic and clinical characteristics by analytic cohort.

Patients having ≥ 1 claim for a neurologist visit during the pre-index period up to and including the index date.
Having ≥1 fill of OMPM before receiving the intervention of interest.
Analyzed during the pre-index period; days’ supply was summed for each patient; values are the mean days’ supply for the entire sample.
BTX: onabotulinumtoxinA; CM: chronic migraine; HMO: health maintenance organization; NSAIDs: nonsteroidal anti-inflammatory drugs; OMPMs: oral migraine prophylactic medications; PPO: preferred provider organization; SD: standard deviation.
Baseline comorbid conditions by analytic cohort.

BTX: onabotulinumtoxinA; CM: chronic migraine; OMPMs: oral migraine prophylactic medications.
Study endpoints
ED visits
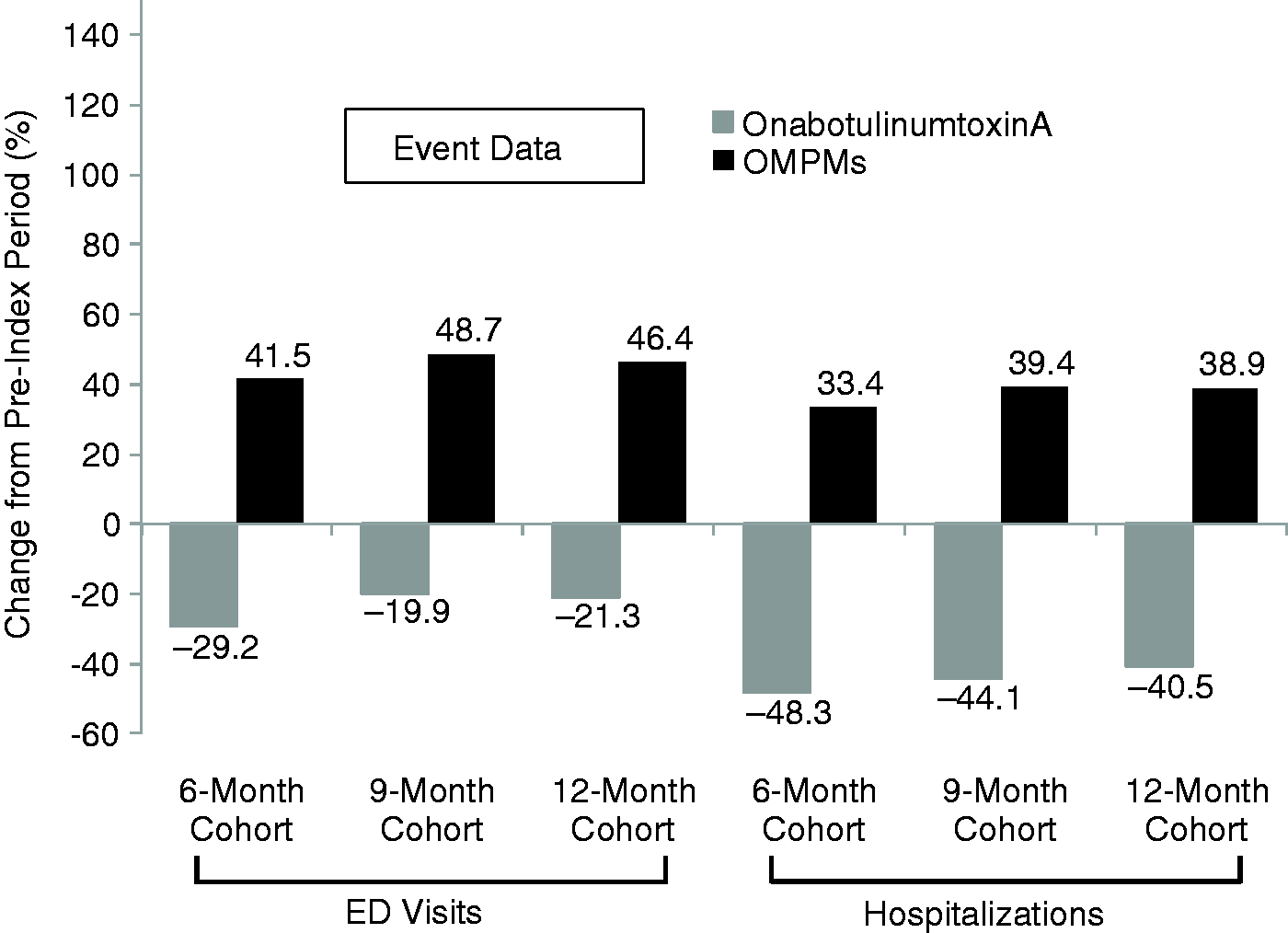
Compared with the pre-index period, fewer patients in the onabotulinumtoxinA group had ED visits during the post-index period. In contrast, the proportion of patients in the OMPM group with ED visits increased during the post- versus pre-index period. In the onabotulinumtoxinA group, 542 (14.1%) patients had a headache-related ED visit during the six-month pre-index period and 369 (9.6%) patients had a headache-related ED visit during the six-month post-index period, corresponding to an absolute change of −4.5% (95% confidence interval (CI), −3.4 to −5.6 (p < 0.001)) and a relative decrease of −31.9% (4.5%/14.1%). In the OMPM group, 892 patients had a headache-related ED visit during the six-month pre-index period, and 1064 had a headache-related ED visit during the six-month post-index period, corresponding to an absolute change of +1.4% (95% CI, 0.8–2.0 (p < 0.001)) and a relative increase of +19.0%. Similar changes for the pre-index versus post-index proportions of patients with headache-related ED visits occurred in the nine-month and the 12-month cohorts and remained statistically significant for all time points. Relative change results are presented both for patient- and event-level data in Figures 3 and 4, respectively.
Relative change in proportion of patients with HRU events after initiating onabotulinumtoxinA or OMPMs. Absolute change shown in parentheses above each bar. *p = <0.001; †p = 0.003; ‡p = 0.020; ED: emergency department; HRU: headache-related resource utilization; OMPM: oral migraine prophylactic medication. Relative change in HRU events after initiating onabotulinumtoxinA or OMPMs. ED: emergency department; HRU: headache-related resource utilization; OMPM: oral migraine prophylactic medication.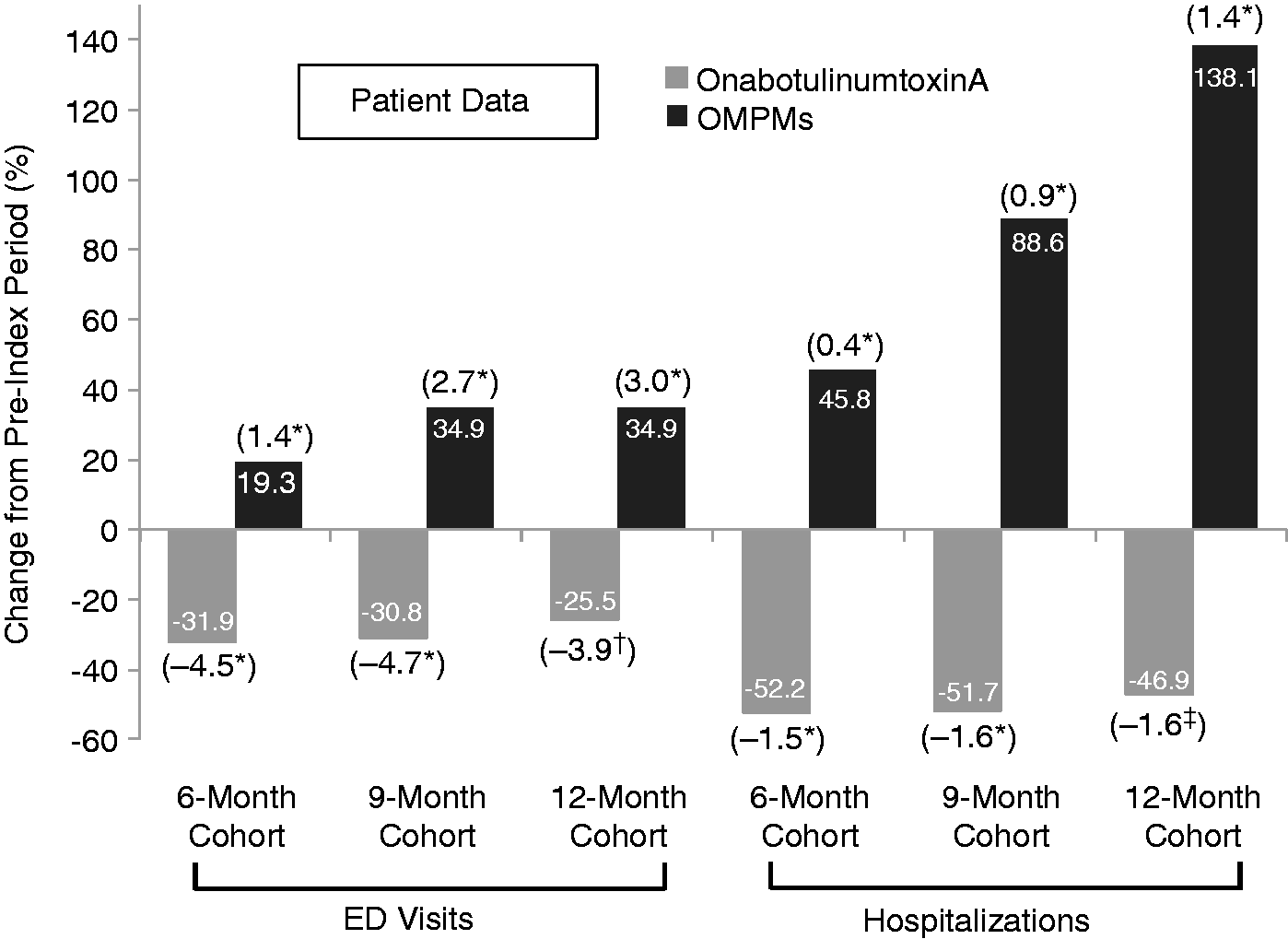
Regression analyses: odds ratios for emergency department visits and hospitalizations post-index for onabotulinumtoxinA versus OMPMs and for onabotulinumtoxinA versus level A OMPMs (15).

OMPM: oral migraine prophylactic medication; CI: confidence interval.
Hospitalizations
The proportion of patients who were hospitalized for a headache-related event was also lower after starting onabotulinumtoxinA, but higher after starting an OMPM for all three cohorts (Figure 3). In the onabotulinumtoxinA group, 113 patients were hospitalized for headache during the pre-index period, and 54 were hospitalized during the post-index period, corresponding to an absolute change of −1.5% (95% CI, −0.9 to −2.1, p < 0.001) and a relative decrease of −52.2%. For the OMPM group, 120 patients had a headache-related hospitalization during the pre-index period, and 175 were hospitalized during the post-index period, corresponding to an absolute change of +0.4% (95% CI, 0.2 to 0.7, p < 0.001) and a relative increase of 46.0%. This pattern of fewer patients hospitalized in the onabotulinumtoxinA group and more hospitalized in the OMPM group persisted in the nine-month and 12-month cohorts and remained statistically significant for all time points. Relative changes in event-level headache-related hospitalizations are shown in Figure 4. Although the event-level data (Figure 4) were generally similar to the patient data (Figure 3) for onabotulinumtoxinA, in the OMPM group, there were differences between the patient-level and event-level data for hospitalizations.
As with ED visits, adjusted regression analyses showed significant reductions in the odds of having a headache-related hospitalization for patients who received onabotulinumtoxinA versus those who received an OMPM. Following a diagnosis of CM, the odds of being hospitalized for a headache-related event six, nine, or 12 months after initiating prophylactic therapy were 47% (OR 0.53, 95% CI, 0.36 to 0.78), 48% (OR 0.52, 95% CI, 0.32 to 0.85), and 56% (OR 0.44, 95% CI 0.24 to 0.80), respectively, lower for onabotulinumtoxinA than OMPMs. When the regression analyses were further narrowed to evaluate only Level A OMPMs, the odds of a headache-related hospitalization for onabotulinumtoxinA versus Level A OMPMs were 45% (OR 0.55, 95% CI, 0.35 to 0.85), 45% (OR 0.55, 95% CI, 0.31 to 0.98), and 57% (OR 0.43, 95% CI, 0.21 to 0.86) lower for the six-, nine-, and 12-month cohorts, respectively (Table 3).
Discussion
In this analysis of claims data from a large insurance database, we compared the use of headache-related health care resources, including ED visits and hospitalizations, among patients with CM six, nine, and 12 months before and six, nine, and 12 months after treatment with onabotulinumtoxinA or OMPMs. The treatment groups were comparable in terms of demographic and clinical characteristics, and the entire sample was found to be largely representative of the general commercially insured CM population in the US based on demographics (9,35), geographic locations, and insurance types.
For the study endpoints, the results showed a statistically significant reduction in headache-related ED visits and hospitalizations following treatment with onabotulinumtoxinA subsequent to CM diagnosis for all time cohorts using both patient- and event-level data. By contrast, CM patients who received OMPMs showed statistically significant increases in headache-related ED visits and hospitalizations (versus declining resource utilization seen in the onabotulinumtoxinA group; Figures 3 and 4). Moreover, in a regression analysis that controlled for underlying demographics and disease severity differences in the two groups, the odds of having a headache-related ED visit or hospitalization after treatment with onabotulinumtoxinA (compared with all OMPMs) was significantly reduced after six months (21% or 47%, respectively), nine months (20% or 48%, respectively), and 12 months (19% or 56%, respectively). Given the lack of evidence for efficacy with some OMPMs in the prevention of migraine, we conducted sensitivity analyses to include only those with Level A evidence (12). Reductions in hospitalizations remained significant in the six-, nine-, and 12-month cohorts and reductions in ED visits remained significant for the nine-month cohort only. The observed narrowing of ED visits among the six- and 12-month cohorts is consistent with the lack of evidence for efficacy for the excluded OMPMs.
In the current study, at baseline, 61% to 66% of patients with CM in the onabotulinumtoxinA groups were seen by a neurologist compared to only 14% in the OMPM group, possibly because neurologists are more likely to be trained in the use of onabotulinumtoxinA (i.e. injection techniques) than non-neurologists. This discrepancy is supported by some epidemiology evidence showing differential rates of diagnosis and treatment for patients with CM. The Chronic Migraine & Epidemiology Outcomes (CaMEO) Study assessed headache frequency, disability, and barriers to medical care in individuals with migraine, including CM. In CaMEO, CM diagnosis rates among patients who sought specialty care by a neurologist, headache specialist, or pain specialist were more than twice that than for patients who consulted health care professionals who were not specialists (i.e. 36.0% versus 15.8%, respectively) (36); furthermore, among all patients with CM who were diagnosed and received treatment for CM, only 4.5% received a prescribed acute or preventive treatment (37). This disparity in the rates of specialist diagnoses (versus nonspecialist) in the CaMEO Study, together with the reduction in headache-related ED visits and hospitalizations in the onabotulinumtoxinA group from the current study (who were mainly treated by neurologists), suggests treatment by a specialist may positively influence patient outcomes, and may have contributed to the reduction in ED visits and hospitalizations observed among patients treated with onabotulinumtoxinA in this study. Additionally, this finding supports the need for education about CM for nonspecialist health care professionals in order to facilitate correct diagnoses and appropriate treatment and management of this condition.
The findings of the current analyses are generally consistent with those reported by Rothrock et al. (27), who demonstrated a significant reduction in headache-related resource utilization (e.g. ED visits, hospitalizations) after onabotulinumtoxinA treatment in 230 patients with CM at a single headache specialty center (27). The percentage of patients with a headache-related ED visit or hospitalization was reduced by 41.6% and 52.9%, respectively, after treatment with onabotulinumtoxinA (27). In general, changes in headache-related health care resource utilization were comparable to the findings of Rothrock et al.; however, that patient sample (highly refractory to treatment) likely had greater CM severity. In addition to the data of Rothrock et al., there have been several analyses of OMPMs (most frequently, topiramate) on health resource utilization in the migraine population using claims databases; however, none have exclusively examined the CM population (38–41).
The results of this study provide promising findings that onabotulinumtoxinA significantly reduces both ED visits and hospitalizations, as also seen in Rothrock et al. (27). OnabotulinumtoxinA was approved in 2010 and is the only medication specifically approved for prophylaxis of headaches in adult patients with CM (13). Since approval, there had been no direct health care resource utilization analyses comparing onabotulinumtoxinA with OMPMs for migraine. The current study found that headache-related ED visits and hospitalizations were significantly reduced in the general commercially insured CM population following treatment with onabotulinumtoxinA. Additionally, the analyses also showed that both ED and hospital visits were significantly increased in the six, nine, and 12 months following the initiation of an OMPM. The current study provides the first real-world evidence that onabotulinumtoxinA reduces health care resource utilization, especially when compared with OMPMs, and even when comparing to those OMPMs that have been identified as having Level A evidence for use in migraine prophylaxis.
Limitations
These findings should be interpreted with some limitations in mind. When using large claims databases, some assumptions are made. For example, analyses based on ICD-9-CM codes must assume that such codes were used appropriately and accurately describe the patients’ migraine diagnosis and comorbidities. Of note, the baseline rate of comorbid cancer in all cohorts was unexpectedly high (up to 25%), and is likely attributed to the inclusion of every possible ICD-9 code for cancer. This would include patients who had been screened for cancer, but may not have had a cancer diagnosis, and result in inflated cancer rates. Claims are also not designed to collect details for some patient demographics such as race or socioeconomic data and thus, such characteristics could not be controlled for in the regression models. Additionally, claims do not capture any direct disease severity information such as headache days. In the absence of such data every attempt was made to control for confounding effects of disease severity using proxy variables such as acute medication use, physician type seen by the patient, or the number of office visits patients had. In terms of study design, the index date determines the pre- and post-index periods, which may result in patients having some or all of their pre-analysis time collected before receiving their CM diagnosis. This was done because a large percentage of patients receive their first onabotulinumtoxinA injection within 90 days of CM diagnosis and thus, an exclusion criterion that requires inclusion of the entire pre-analysis period (six to 12 months) to be after CM diagnosis would exclude almost all patients. This potentially makes our findings conservative because health care resource utilization was captured during the time when patients were not yet diagnosed with CM.
Conclusions
In patients with CM, headache-related ED visits and hospitalizations were significantly reduced six, nine, and 12 months after initiating treatment with onabotulinumtoxinA, which is consistent with a previously published report of health care resource utilization among patients with more severe, treatment-refractory CM treated with onabotulinumtoxinA at one headache center in the US. Additionally, this study also demonstrated that, when compared with similar patients who initiated treatment with OMPMs, onabotulinumtoxinA was associated with a significantly lower likelihood of headache-related ED visits and hospitalizations.
Clinical implications
Little is known about health care resource utilization in patients with chronic migraine receiving prophylactic treatment for headaches. This research adds meaningful new findings that fill an important gap in the literature. Headache-related emergency department (ED) visits and hospitalizations were significantly reduced after prophylactic treatment with onabotulinumtoxinA and significantly increased after prophylactic treatment with oral migraine prophylactic medications (OMPMs). After controlling for potential confounders, onabotulinumtoxinA was associated with lower likelihood of headache-related ED visits and hospitalizations when compared with OMPMs. This study provides the first real-world evidence that onabotulinumtoxinA is associated with reduced health care resource utilization.
Footnotes
Acknowledgments
The authors would like to thank Jenny Chia, PhD, formerly of Allergan plc, and Guo Li, MS, of Allergan plc, for their assistance in ensuring the quality of the coding and the statistical analyses. Writing and editorial assistance was provided to the authors by Kristine W. Schuler, MS, from Complete Health care Communications Inc. (Chadds Ford, PA), Dana Franznick, PharmD, and Sally K. Laden, MS, and was funded by Allergan plc (Dublin, Ireland). All authors met the International Committee of Medical Journal Editors (ICMJE) authorship criteria. Neither honoraria nor payments were made for authorship.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Z Hepp is employed by Allergan plc. NL Rosen is on the speaker panel, has participated in advisory boards, and conducted research with Allergan plc; he has participated in an advisory board for Zogenix, has received honoraria from the American Headache Society, and has received options for consultation work with Curelator. PG Gillard is employed by Allergan plc and reports having received stock and/or stock options from Allergan. SF Varon is employed by Allergan plc and reports having received stock and/or stock options from Allergan. N Mathew receives personal compensation from Allergan for consulting. DW Dodick has within the past 12 months served on advisory boards and/or has consulted for Allergan, Amgen, Alder, Merck, ENeura, Eli Lilly & Company, Autonomic Technologies, Teva, Tonix, Novartis, and Alcobra; received royalties, funding for travel, speaking, or editorial activities from Haymarket Media Group Ltd, SAGE Publishing, Synergy, Allergan, Lippincott Williams & Wilkins, Oxford University Press, Cambridge University Press; Up-to-Date; and has received publishing royalties for Wolff's Headache, 8th edition (Oxford University Press, 2009) and Handbook of Headache (Cambridge University Press, 2010).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Allergan plc (Dublin, Ireland).
