Abstract
Background
Individual differences in pain perception to a standardized nociceptive input are a well-known phenomenon within pain research. Brain structures known to play a crucial role in pain modulatory processes are the rostral/subgenual anterior cingulate cortex (sACC) as well as the periaqueductal gray (PAG), which belong to the endogenous antinociceptive system. However, the exact mechanisms possibly leading to this high level of variance in pain perception are still a matter of debate.
Methods
Pain perception within the trigemino-vascular system was investigated in 37 healthy volunteers using functional magnetic resonance imaging.
Results
Behavioral results show high levels of variance being inversely correlated to mean pain ratings as well as to an increase in BOLD signal intensity within the sACC. In addition, higher sACC activation was coupled with activation in the PAG the lower the level of intra-individual variance.
Conclusion
This study gives first indications that coupled BOLD response within brain structures of the antinociceptive system seems to rather not code pain intensity within the trigemino-nociceptive system but the stability of volunteers’ pain ratings. Intrinsic mechanisms may modulate the pain perception in the trigemino-vascular system, which is highly involved in headache disorders.
Introduction
One outstanding attribute of human somatosensory perception is that individuals tremendously differ in their conception of and sensitivity to all sensory modalities. This robust phenomenon is especially well known in pain research, showing robust painful responses to constant nociceptive stimulation in some participants while other individuals experience the same stimulation as less or even non-painful (1–3). But recent studies reported not only interindividual but also high intra-individual differences in nociceptive and somatosensory perception (4–7). It is widely recognized that a vast diversity of factors may influence pain perception and perception per se (1), such as spontaneous neuronal fluctuations (8), attention (9,10) expectation of pain (11,12), sleep habits (13) or stress (14). Concerning the trigeminal system, it could be shown that especially emotional control induced by the presentation of pleasant, unpleasant and neutral pictures could modulate trigeminal nociception (measured by the blink reflex) as well as trigeminal pain (measured by pain ratings) as such (15). It was concluded that emotional control as well as diffuse noxious inhibitory control would be good tools to study the possibility of supraspinal pain processing modulation of headache or facial pain (15). Imaging studies investigating pain perception demonstrate a crucial role of the rostral/subgenual anterior cingulate cortex (sACC/rACC), especially in pain perception processes like habituation and placebo (16–18). Another study underlined the influence of external factors on pain perception by showing decreased pain ratings associated with distraction (19). Distraction during painful stimulation was furthermore related to increased activity in the frontal cingulate cortex (pACC), the periaquaeductal gray (PAG) and thalamic regions (19). Thus, especially this well-known connectivity between sACC/rACC and PAG seem to play a very important role in central pain perception processes, i.e. pain modulation. From a clinical point of view the underlying causes of individual differences in pain perception are of major interest, as it has been suggested that altered pain sensitivity and reduced pain-inhibitory capacity might be a risk factor for the establishment of clinical pain like post-operative pain or possibly even chronic pain syndromes (20). Regarding the trigemino-vascular system, intra- and interindividual differences in pain perception are of significant importance to improve our understanding of the underlying pathophysiology of migraine as it could be shown that migraineurs differ in their pain thresholds compared to healthy controls interictally (21). Moreover, pain thresholds of migraineurs seem to be even lowered shortly before an attack compared to interictally (22).
Using a previously described standardized design for trigemino-nociceptive stimulation in functional magnetic resonance imaging (fMRI) (23), we observed prominent within-subject variability in pain perception (high level of variance) within a considerable subset of our volunteers, even though the trigeminal nociceptive stimulation was kept at a constant level throughout the entire paradigm. Other participants, however, experienced the same trigemino-nociceptive stimulation as very stable. Focusing on this phenomenon and referring to early studies, we hypothesize that sACC and PAG might possibly be involved in the variability of pain intensity perception as such.
Materials and methods
Participants
Thirty-seven healthy volunteers (18 female) with a mean age of 26.5 (range: 20 to 37 years, SD 3.69) were recruited locally. None of them had a history of neurological or psychiatric disease and particularly no history of pain syndromes. None of the participants had been under pain medication while participating in the study. Written informed consent was obtained from all participants and the study was conducted according to the Declaration of Helsinki and approved by the Ethics Committee in Hamburg, Germany. All volunteers were remunerated after participation.
Study design/Stimuli
Preceding fMRI acquisition participants completed two self-report questionnaires: (a) Beck Depression Inventory II (BDI-II) and (b) the German form of the Patient Health Questionnaire (PHQ-D). During scanning all volunteers participated in an fMRI paradigm (Figure 1) based on an established experiment by Stankewitz et al. (23). In more detail, volunteers were exposed to 15 standardized trigeminal nociceptive stimuli (gaseous ammonia) and several control stimuli through an air-proof Teflon tube which was connected to the olfactometer outside of the scanning room.
Temporal profile of the experimental paradigm. Every trial of the functional magnetic resonance imaging (fMRI) paradigm starts with a reaction task, in which participants are instructed to press a button immediately after the fixation cross on the screen changes color from white to red. Following a jittered time delay of 4–6 seconds, participants underwent the stimulation part, receiving either trigemino-nociceptive stimulation or one out of the three control stimuli (rose odor, visual stimulation or air puff). After each stimulus (painful as well as all control stimuli), participants were asked to rate the intensity and pleasantness of the stimulation on a visual analog scale (VAS). Finally, an intertrial interval (ITI) of 4–6 seconds followed prior to the next trial.
Control conditions were either an olfactory (rose odor), an odorless (air puffs) (both shown 15 times for one second each), or a visual stimulus (flickering checkerboard, shown 16 times for 4 seconds each). Prior to every stimulus presentation, participants underwent a reaction task that consisted of a white cross that changed colors after a jittered time delay of 4–6 seconds. As soon as the cross changed its color to red, volunteers had to press a button. Another jittered time delay of 4–6 seconds later participants were exposed to one of the above described stimulations. The latter were presented in a pseudo-randomized order to avoid trigemino-nociceptive stimulation being followed by the very same condition. After stimulation volunteers used a button box system to rate the intensity and pleasantness of all types of stimuli on a visual analog scale (VAS). The intensity scaling ranged from 0 (no pain/no sensation) to 100 (highest pain conceivable/highest intensity conceivable), whereas the pleasantness scale ranged from –50 (very pleasant) to 50 (very unpleasant).
Image acquisition
Functional imaging was performed on a 3 T Scanner (Siemens-Trio) using a 32-channel head coil. Depending on reaction time and time to rate the stimulus intensity/pleasantness on the VAS, 869–1202 volumes (mean 955 volumes) were acquired with 40 axial slices (slice thickness 2 mm, gap 1 mm, 2 × 2 mm in-plane resolution) using a gradient echo planar imaging sequence (Repetition time: 2.62s, echo time: 30 ms; flip angle 80 degrees, field of view: 220 × 220 mm, 110 × 110 matrix). Additionally, one high-resolution T1-weighted structural image (voxel size 1 × 1 × 1 mm) was acquired using a magnetization-prepared rapid gradient echo (MPRAGE) sequence.
Analysis of behavioral data
Behavioral data analysis was performed using MATLAB Version 8.0 (R2012b, Mathworks Inc, Natick, MA, USA). The main interest of this study lay in investigating the effect of trigemino-nociceptive stimulation in relation to the level of overall variance between the subsequent pain ratings. Therefore, we calculated the arithmetic mean and variance of all 15 pain ratings within the fMRI session for every volunteer. The same statistical analysis was conducted for the olfactory condition (rose odor) for further comparisons. Next, correlation analyses between a) the statistical variance and the arithmetic mean pain intensity scores, b) mean reaction time (RT) and variance, and c) mean RT and mean intensity rating of pain were performed for participants. In addition to the analysis of pain intensity ratings, arithmetic mean and statistical variance of all individual pain pleasantness ratings related to trigemino-nociceptive stimulation were calculated for every participant and investigated for correlation. Furthermore, a correlation analysis investigating mean intensity scores and the statistical variance between rose odor ratings was performed.
Image processing and statistical analysis of fMRI data
Preprocessing and statistical analyses of fMRI data were performed using Statistical Parametric Mapping Software SPM8 (Wellcome Department for Imaging Neuroscience, London, UK). The first five scans were discarded. Preprocessing included slice time correction, realignment to the first volume as well as spatial normalization to the Montreal Neurological Institute (MNI) stereotactical space. Spatial smoothing was performed using an isotropic 8 mm full-width at half-maximum (FWHM) three-dimensional Gaussian kernel. To identify blood-oxygen-level dependent (BOLD) signal changes after painful stimulation, the first-level model included the following regressors: ammonia (trigemino-nociceptive stimulation), rose odor, air puffs, visual stimulation and button presses. Each of the five regressors was convolved with the hemodynamic response function. No participant needed to be excluded for excessive, abrupt or task-related motion. Movement parameters were included as regressors of no interest. Additionally, a parametric modulator of the intensity rating from every participant regarding trigemino-nociceptive stimulation was added to the model. In more detail, the first-level general linear model predicting BOLD responses to ammonia was not only constructed from onsets of ammonia stimulation (convolved with the hemodynamic response function) but contained a second regressor predicting the amplitude of every single BOLD response following ammonia stimulation (with subjective pain intensity ratings determining these amplitudes). The second level again included individual variance as a covariate for each participant to separate effects of parametric modulation from effects of individual variance. Linear contrast images of the parameter estimates were estimated for every participant testing for ammonia > air puffs. To specify the effect of nociceptive stimulation, additional contrasts testing for differences between ammonia, rose odor and air puffs (ammonia > (rose odor + air puffs)) were estimated for every individual.
Statistical tests performed at the group level
Contrast images of ammonia > air puffs as well as ammonia > (rose odor + air puffs) were inserted into two separate second-level models to analyze group effects. We further included three covariates into the model. The first covariate coded for the interindividual variability level and was operationalized as the statistical variance of every subject over the 15 pain ratings in the individual session. Second, we included the mean rating of nociceptive stimulation of every individual into the second level analysis, modeling the general effect of mean pain intensity. Finally, gender was implemented as a covariate of no interest to control for gender differences. The following analyses were conducted:
The Effect of statistical variance between pain intensity ratings (covariate): tested for a correlation between contrast estimates of the nociceptive condition (ammonia > air puffs) and the individual level of variance between the subsequent pain ratings while controlling for an effect of mean pain intensity and gender differences (covariates of no interest). Concerning previous studies, which showed a role of the sACC/rACC and the PAG in pain modulation (17–19,24), we hypothesized to find a correlation between the level of variance over individual pain ratings and these brain structures. Thus, we used small volume correction (SVC) based on family-wise error correction (FWE p < 0.05) with a sphere of 10 mm radius centered on (x = ± 6, y = 30, z = −9) for the sACC and a sphere of 8 mm radius centered on (x = ±0, y = −32, z = −10) for the PAG. Coordinates for sACC and PAG were chosen based on previous studies (17,18,24). Correlation analysis between the nociceptive condition (ammonia > air puffs) and the individual mean pain rating of the volunteers while controlling for the individual variance between pain ratings of the volunteers as well as for gender type. To ensure that the activation found in (1) is specific for pain, an additional analysis tested for a correlation between the contrast images of (ammonia> (rose odor + air puffs)) and the individual level of variance between subsequent pain intensity ratings while controlling for gender and mean pain intensity ratings. A psychophysiological interaction analysis (PPI, Friston et al., 1997) (25) assessed whether activation in the sACC (as region of interest obtained from analysis (1) interacted with activity patterns in other brain structures related to pain processing (for more information, see supplemental S1). The individual level of variance was again implemented as covariate of interest on the second level. Additionally, gender was implemented as a covariate of no interest to control for gender differences as found in previous studies (24). Knowing from earlier studies that there is a strong interaction between the rACC/sACC (as well as the perigenual ACC) and the PAG in studies modulating pain (18,19,25), we also hypothesized to find a coupling between these brain structures in correlation with the intra-individual level of variance over pain intensity ratings. Therefore, we also conducted SVC for the PAG in this PPI analysis. This was performed using FWE (p < 0.05) with a sphere of 8 mm radius centered on PAG coordinates of a previous study (0, −32, −10), which are suggested to be the geometric center of this structure (18). To relate changes in brain activation patterns to the psychophysical response of trigemino-nociceptive stimulation, a parametric modulation of ammonia with subjective pain intensity ratings as a parameter was conducted while controlling for an effect of the individual level of variance between the subsequent pain ratings. This allowed us to explore brain areas in which the hemodynamic response increases with pain.
Results
No significant correlations between BDI II scores and the variance of perceived pain intensity or mean pain intensity ratings could be revealed. No correlation between anxiety measures (PHQ-D), somatization scores (PHQ-D) and the variance of perceived pain intensity could be found. Finally, there was no correlation between anxiety, somatization and mean pain intensity ratings.
Psychophysics
Average ratings and standard deviations of the mean (±SEM) for nociceptive sensation were 61.8 ± 2.6, anchored by 0 = no pain and 100 = worst pain imaginable. Rose odor intensity was rated 40.7 ± 2.7. Variance between pain ratings (±SEM) was 198.5 ± 36.8 on average. Variance between ratings of rose odor intensity was 161 ± 23.8 (±SEM) on average. The level of variance differed strongly between participants. Several individuals had a remarkably large inter-trial variance and others rated rather stable (example: Figure 2; for all participants, see supplemental information S2). A correlation analysis between the statistical variance between all pain intensity ratings of every volunteer within a session and the mean pain intensity scores of every participant showed an inverse correlation (r = −0.49, p = 0.0019), i.e. the lower the variance in pain intensity perception, the higher the perceived pain over all trials. No correlation between the statistical variance of intensity scores of olfactory stimulation and the arithmetic mean of these ratings could be found. In addition, no correlation between the variance of pain pleasantness ratings and the mean of participant’s pain pleasantness or intensity scores could be revealed (p > 0.05). For this reason all analyses of the imaging data focused on the effect of variance between pain intensity scores and not on variance between pain pleasantness ratings.
Exemplary pain ratings. The figure shows 15 pain ratings (visual analog scale (VAS) values) of two exemplary healthy participants after trigemino-nociceptive stimulation. While pain ratings of one volunteer showed low rating variability over time (red, variance = 2.98), the inter-stimulus variance of the other participant is high (blue, variance = 608.31).
Imaging findings
Pain intensity
Consistent with previous studies, statistically significant bilateral neuronal activation following trigeminal-nociceptive stimulation (contrast: ammonia > air puffs) was found in brain areas including the insula, putamen, thalamus, middle cingulate cortex, anterior cingulate cortex, primary and secondary somatosensory cortex (FWE corrected, p < 0.05).
Variance of pain perception/Pain stability
Complementing the negative correlation between the individual variance over 15 pain ratings and the mean pain intensity rating, we tested for the effect of the level of variance on functional neuronal activity. Regarding our hypothesis, we found an increased activation of the sACC (x = −6, y = 26, z = −16, T(33) = 4.00, p < 0.05 SVC) in pain (ammonia) vs. non-pain (air puffs) trials the lower the level of variance, i.e. the lower the within-subject variability, the higher the activity in sACC (Figure 3(a)). No significant results were found for the converse analysis (the higher the variance the more activation). Of note, sACC activation was also revealed in the same location (x = −6, y = 26, z = −16) investigating the contrast (ammonia > (rose odor + air puffs)) (T(33) = 4.09, p < 0.05 SVC). No effect of the level of within-subject variability in pain ratings was found for the PAG. An additional analysis regarding the effect of mean pain intensity rating revealed no significant effect. To exclude that the rating variation could depend on participant attention/wakefulness, we computed the time variation of the rating procedure as a covariate of no interest. This did not change the finding in the sACC. Of note, there was no correlation between the reaction task before each condition or the time variation of the rating procedure after each condition and pain ratings.
Overview of links between main imaging results of the study. Figure 3(a) shows imaging results related to pain perception and specifically the effect of the negative covariate (level of variance) on ammonia>air puffs. The lower the rating variability of the participants was, the higher the resulting activation in the subgenual anterior cingulate cortex (sACC) (x = −6, y = 26, z = −16). Activation is shown at threshold of p < 0.001 (uncorrected) and is overlaid onto the average structural image of all participants. Cutout: Threshold of sACC activity is also set to p < 0.001 (uncorrected) for visualization purposes. Activation was plotted onto a single case structural image. Figure 3(b) shows the result of the psychophysiological interaction (PPI) analysis. The PPI analysis contrasted ammonia (pain) > air puffs (no pain) modeling regional coupling between the seed region (sACC) and other brain regions. As hypothesized, a stimulus-dependent increased coupling between the sACC and the periaquaeductal gray (PAG) (x = −2, y = −36, z = −16) was found. This interaction was inversely correlated with lower rating variance of the participants. Bold signal is shown at threshold of p < 0.001 (uncorrected), cluster >100 voxel for visualization purposes. Color scale refers to T values of individual voxels.
PPI
Results of the psychophysiological interaction (PPI) analysis using the exploratory threshold of p < 0.001 (uncorrected) once an extended voxel threshold of 100 voxels is reached.

Individual pain perception
A parametric modulation of pain could show that the higher the pain perception of the volunteers, the higher the activation in several pain processing structures including the bilateral thalamus, bilateral putamen, bilateral primary somatosensory cortex, middle cingulate cortex, anterior cingulate cortex, paracingulate gyrus, brainstem and cerebellum. Notably, the parametric modulation did not reveal any activation in the sACC and PAG. A combined figure of the activation related to the effect of the negative covariate (variance), the PPI and the parametric modulation is shown in Figure 4.
Activation pattern after painful stimulation in comparison to its parametric modulation. Figure 4 demonstrates descriptive differences in activation between the effect of the negative covariate and the parametric modulation. Pain-evoked activation corresponding negatively to the height of the within-subject variability (in red) was found in sACC (x = −6, y = 26, z = −16). This activation was coupled to activity in the PAG (x = −2, y = −36, z = −16, green) the lower the rating variance of the participants. The parametric modulation (in yellow) shows activation corresponding to the perceived nociceptive stimulation i.e. in the bilateral thalamus, bilateral putamen, anterior and middle cingulate cortex, paracingulate gyrus, cerebellum and brainstem. Activation maps were plotted at a threshold of p < 0.001 (uncorrected). All three activation patterns were overlaid onto the average structural image of all participants. Color scale refers to T values of individual voxels.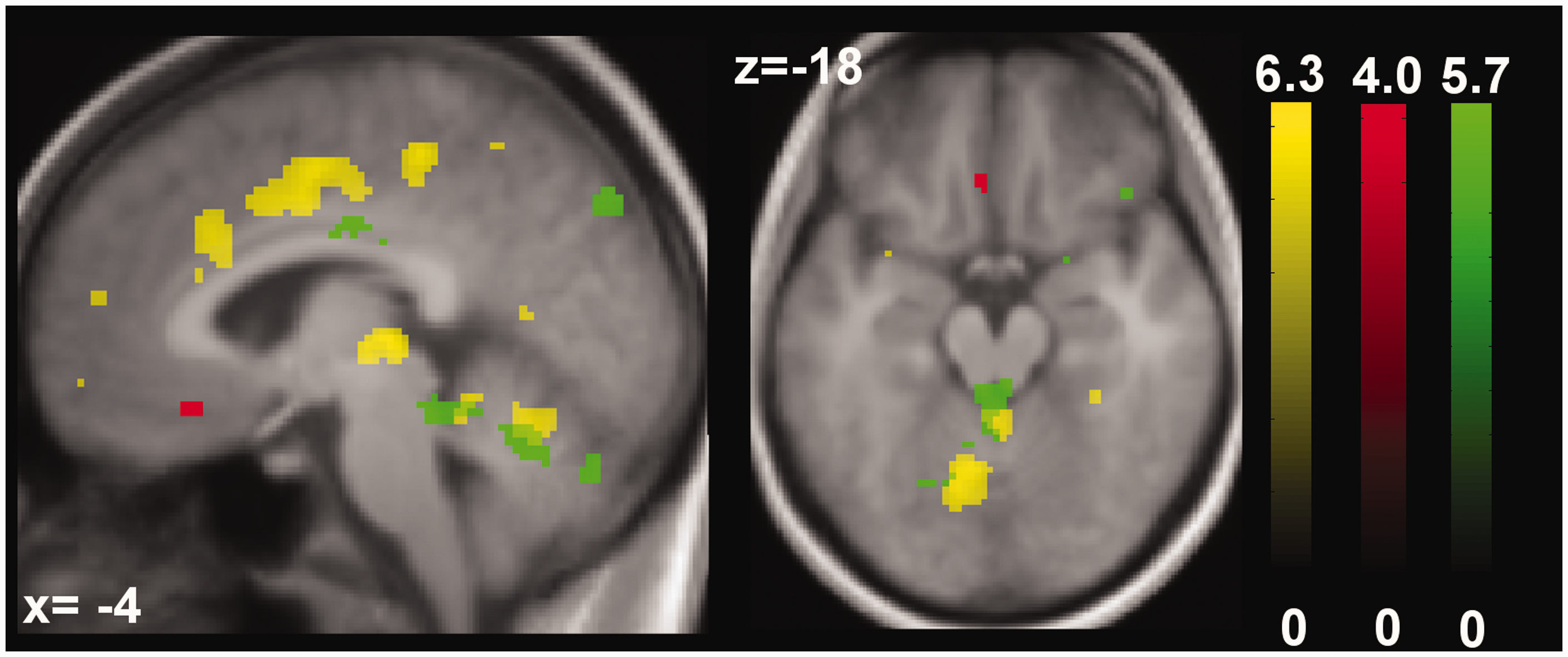
Discussion
We found an inverse correlation between individual pain intensity ratings and intra-individual variability in trigeminal pain perception in healthy volunteers. The obtained negative correlation between inter-trial rating variability and sACC activity gives furthermore first indications of possible underlying mechanisms concerning pain processing within the trigeminal system. The results show, in addition, that sACC activation is strongly coupled to neural activity in the PAG, the lower the rating variability of the participants. This interaction nicely fits early studies that could show heightened coupling between these antinociceptive structures investigating pain modulatory mechanisms (16,18). However, controlling for the effect of mean pain intensity, the combined activation of sACC and PAG in the present study seems to be not further related to individual pain intensity ratings per se. This is underlined by a descriptive comparison of the effect of pain stability (variance) to the neuronal correlates of parametric modulation (Figure 4), as the latter does not reveal any sACC or PAG activation. Earlier studies already reported that across- and within session variability following heat pain stimulation differs widely. More important, an inverse correlation between individual pain ratings and levels of rating-variability was revealed, suggesting that volunteers perceiving the stimuli as less painful show a higher rating-variability. This was interpreted as probably mirroring an intrinsic characteristic of the participants (6).
We could replicate this inverse correlation between rating-variability and subsequent pain perception within the trigemino-nociceptive system. Moreover, we revealed interesting first indications that increased sACC activity is associated with lower rating variability and thus might play a relevant role in coding the variability of pain perception. As movement parameters were included as covariates of no interest in the fMRI analysis, we could already reduce the possibility that an interaction with head motion may contribute to our findings. We additionally calculated the mean of displacements (Euclidean distance) per volume as well as the variance of displacement over volumes on our movement parameters, which did not show any correlation with the variance of the obtained pain ratings (both p > 0.07, r < 0.3).
One then might argue that interindividual variance might generally lead to diminished activation. However, neither sACC nor other pain-specific brain areas displayed enhanced BOLD signal associated with the level of variance between subsequent intensity ratings of rose odor or visual stimulation. Hence, the increased activity within the sACC seems to be specific to trigeminal pain. An alternative similar analysis, now contrasting air puffs and rose odor to painful stimulation, supports this finding as it also reveals sACC activity. A very crucial question is, whether low(er) pain ratings and high variability could both be due to inattention. However, the reaction time before each trial run, which was inserted into the study design to specifically control for vigilance (23), was not different between individuals with a stable and a variant rating.
It is known from earlier studies that external modifications like distraction and expectation can be responsible for activation of the antinociceptive network and thus altered pain perception (27,28). However, in our study no external modificator was presented and as we controlled for mean pain intensity, the coupled activity of sACC and PAG reflects a pattern that seems to influence the variance but not the strength of pain perception per se (Figure 4). Quite the contrary: Pain intensity ratings seem to be regulated by brain areas related to pain transmission like the bilateral thalamus or middle cingulate cortex (Figure 4). This leads to the question why there should be a divergence in neuronal processing between variability and intensity of pain ratings.
One common factor modulating consecutive pain perception is the reliability (or uncertainty) of a following painful stimulation. The hyperalgesic effect of experiencing uncertainty as well as a certain expectation toward unpleasant events seem to be positively correlated with rACC and PAG activity (12,29). A positive correlation between greater activation and higher pain scores due to uncontrollable conditions was furthermore reported in the pregenual ACC (pACC) and PAG (30). Taken together, precariousness and expectation about a possibly upcoming negative stimulation, as well as loss of control seems to increase pain intensity ratings. Because the present study gives first indications that the coupled activation of sACC and PAG is reactivated in individuals with lower rating variability, while they remained inactive in processing the intensity of pain ratings as such, we would have to assume that these participants possibly had a more negative expectation of the incoming stimulus. Following, the higher expectation would indeed lead to higher pain ratings as a consequence or variation of “self-fulfilling prophecy.” Differences in stability of pain perception could therefore be due to the participants being more or less concerned, therefore more anxious and thus more focused on the upcoming event. Interestingly, it could be shown that individuals differ in their level of spontaneous disengagement of attention (“mind wandering”) to subsequent pain stimuli, which could be shown to be associated with stronger functional connectivity between the default mode network and the PAG (31). We note that ratings in our study might be influenced not only by mind wandering but also possibly by memory processes and in combination with fluctuations, in attention it may be difficult to discern which processes were driving the variability. Personality traits, like anxiety, which are shown to correlate with sACC and PAG activation (32–34), situational parameters such as nervousness and individual strategies in form of, for example, distraction techniques (31) are therefore promising factors to influence pain perception. That this is not only the case in heat pain paradigms but also suits that trigemino-nociceptive stimulation is supported by a study showing a significant modulatory effect of emotion control on trigeminal pain perception and nociception as such (15). One major strength of the study lies in the statistical exclusion of several different factors that could have influenced the results concerning the variability of pain perception. We could show that neither gender, pain perception (measured by mean pain intensity scores) nor vigilance showed a significant influence on the obtained BOLD signal activation in the sACC (and its coupling with the PAG) the lower the within-subject variability in trigeminal pain perception. Furthermore, we could also proof with additional statistical analyses that the found results are not due to generally diminished activation. Importantly, body and head movement of the participants could also be ruled out as a factor explaining the intra-individual variability in pain perception. Last but not least, an effect of the experimenter or calibration procedures could also be ruled out. Nevertheless, further studies are clearly needed to link our results to personality traits and/or genetic determinations. The inquiry of additional physiological measurements like skin conductance or pupil diameters could moreover help to gain a better understanding of factors influencing inter- and intra-individual differences in pain perception.
Footnotes
Conclusion
In conclusion, we could reveal a neuronal activation of sACC and a higher functional coupling of this structure to the PAG, the lower the within-subject variability in trigeminal pain perception. As this activation seems to code pain stability as such, we propose that the interindividual variability in response to trigeminal pain may be influenced by intrinsic factors. These modulatory effects on pain processing in the trigemino-vascular system are of significant interest concerning a better understanding of trigemino-nociceptive processing which is of great need for patient populations suffering from disorders of the trigeminal system (e.g. headache patients). However, further research is needed to support our findings and find possible links to personality traits or genetic differences.
Article highlights
There is an inverse correlation between individual pain intensity ratings and intra-individual variability in trigeminal pain perception.
The lower the rating variability, the higher the blood oxygen level-dependent (BOLD) response in the subgenual anterior cingulate cortex (sACC) and its functional coupling to another part of the antinociceptive system: the periaqueductal gray (PAG).
sACC and PAG activation rather code pain stability than pain intensity perception.
Intrinsic factors may modulate pain processing within the trigemino-vascular system, which is highly involved in headache disorders.
Acknowledgments
The authors thank Isabel Ellerbrock, Sophia Schneider and Eszter Schoell for helpful advice with data analysis, and Katrin Bergholz, Kathrin Wendt and Timo Krämer for assistance with data acquisition.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the German Research Foundation, DFG 1862/12-1 and the 7th Framework EU-Project EuroHeadPain (#602633) to A.M.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
