Abstract
Aims
We conducted a cohort study to examine demographic and clinical features associated with the pharmacotherapeutic outcome in classical trigeminal neuralgia (CTN) patients.
Methods
Patients with a clinical profile indicating a diagnosis of CTN, as per the International Headache Society’s published classification, were enrolled prospectively. Demographic and pain-related characteristics were carefully collected. For the purposes of the study, patients with features such as autonomic signs and longer attack duration were included. All patients were then initiated on a standardised and accepted stepped pharmacotherapeutic protocol for the management of CTN. Initial pain scores and prospectively collected pain scores from pain diaries were used to assess the treatment outcome, with a ≥50% reduction considered significant.
Results
A total of 86 patients were seen, of whom five had an underlying disorder that could account for the pain. The study cohort therefore consisted of 81 patients, and based on attack duration these were divided into short (≤2 minutes, n = 61) and long (>2 minutes, n = 20) groups, for further analysis. The features of these patients and a discussion on the differential diagnosis have been presented in part 1 of this report. Employing an accepted stepped pharmacotherapeutic protocol for the management of CTN, significant improvement was more frequent in the short (74%) than in the long attack group (50%, p = 0.05). In the short attack group there were statistically significant associations between a poor treatment response and longer disease duration, the presence of autonomic signs and atypical pain descriptors for pain quality (p < 0.05).
Conclusion
This report supports previous findings that prolonged disease duration and autonomic signs are negative prognostic indicators. The present study now adds long attack duration as a further negative prognostic sign.
Introduction
Classical trigeminal neuralgia (CTN) is considered a progressive disorder with a poor long-term prognosis (1). In some series, up to 90% of CTN cases may report increased attack frequency and severity, with a time-dependent, increasing failure rate to pharmacologic and invasive therapies (2–4). However, a recent study has shown a more robust and prolonged response of CTN patients to treatment with carbamazepine (CBZ) and oxcarbazepine (5).
The standard for pharmacotherapy in CTN remains CBZ (6). Oxcarbazepine, a related drug, is quite efficient with fewer cognitive side effects than CBZ. Synergistic combinations with CBZ include baclofen (7) and lamotrigine (8). Alternatives include gabapentin, pregabalin, topiramate and some older anticonvulsants.
There are indications that specific clinical features may be associated with a higher rate of treatment failures. These features include concomitant background facial pain and autonomic signs (9–13). Background pain occurs relatively commonly (13,14) and was recently recognised by the International Headache Society (IHS) as part of the clinical CTN phenotype (15). Currently the IHS therefore classifies CTN as ‘CTN, purely paroxysmal’ or ‘CTN with concomitant persistent facial pain’ (11,15). Autonomic signs are not typical of neuropathic type pain. However, tearing has been reported in ophthalmic, maxillary and mandibular CTN (16–18). There is also evidence of autonomic activity in CTN such as facial flushing (vasodilation), increased salivation and swelling (17,19). A further feature recently reported is waking from sleep. Although CTN was considered a daytime phenomenon, reports of nocturnal CTN (14,20) have clearly established that attacks may wake from sleep.
In the first part of this report we examined the clinical phenotype of patients with trigeminal neuralgic pain presenting to the clinic. We analysed clinical features and in particular attack durations lasting longer than the stipulated 2 minutes (min). The aim of the present study was to examine features associated with the outcome in response to an accepted pharmacotherapeutic protocol in this group of patients.
Materials and methods
All patients were interviewed and examined at the Orofacial Pain Clinic, Faculty of Dentistry, Hadassah-The Hebrew University, Jerusalem, Israel.
Inclusion criteria and pain diagnosis
Inclusion criteria consisted of all patients with trigeminal neuralgiform pain with a tentative clinical diagnosis of CTN according to the classification published by the IHS (21) and updated in 2013 (15). For the aims of this study we allowed inclusion of patients with symptomatology such as persistent background pain in addition to the typical paroxysms, autonomic signs, waking from sleep and reported attack duration beyond 2 min. Exclusion criteria consisted of any patient that on further history, examination or imaging was found to have underlying pathology, infection or previous trauma explaining the pain. Initial patient intake was performed by a resident, followed by review with one of the senior authors (RB, YS). All diagnoses were subsequently reviewed in the clinic and then re-examined following data tabulation and summary, by both senior authors (RB, YS).
Data collection
The institutional review board approved the study and informed consent was obtained from all 86 participating patients examined and interviewed. Data collection was prospective and the analysis performed at the end of the study. The patients were observed between the years 2005 and 2013. The resultant data, including a pain history, were recorded on a standard intake form. Since the aim was to test a stepped pharmacotherapeutic protocol, the endpoints of the study were defined as two staged: At 12 weeks’ review and if no significant pain relief at this time point, then completion of the study protocol to the point of the patient having received either gabapentin or pregabalin or refused to continue.
At baseline, patients were asked to rate pain duration, quality and pain intensity of an average paroxysm. Patients were asked to assess their pain duration based on that of their predominant attacks. Pain intensity was rated employing a verbal pain scale (VPS) where 0 is no pain and 10 the worst imaginable pain. Pain quality was assessed by asking the patients to choose one or more of the following descriptive terms: electrical (includes shooting), stabbing (includes sharp), throbbing, pressure, burning or any combination of the five terms. These terms are in routine use in our clinic to provide rapid assessment of pain quality (14,22). To test for combinations of quality descriptors, each term was numerically coded so that when summed each combination retained a unique identifier.
Patients were asked specifically about autonomic signs, i.e. tearing and redness of the ipsilateral eye, nasal congestion/rhinorrhoea, eyelid oedema or regional swelling, increased sweating, flushing, fullness in the ear and ptosis and/or miosis during pain attacks. Patients were asked whether they felt additional persistent pain in the affected area that lasted hours or all day. The presence of continuous background pain, in addition to the typical paroxysms, was recorded. For follow-up, pain diaries were employed so that accurate data on VPS and treatment were available. In the pain diary patients were asked to rate each attack. Location of pain was mapped out onto a diagram of the head and neck.
Patients were also asked via a standardised question whether the pain specifically wakes them from sleep. Answers to this question were carefully interpreted so as to ensure that the patient was reporting awakening specifically related to pain. In this manner we excluded, for example, random awakenings (to drink water, or for micturition) for which the patient reported that pain was coincidentally present but had not been the reason for awakening (14).
All patients underwent a thorough extra- and intra-oral examination including a cranial nerve examination. So as to ensure the correct interpretation of ‘triggered’, we assigned this only to patients in whom we could clinically identify trigger areas and induce a refractory period. The masticatory apparatus (temporomandibular joints and masticatory muscles) and neck muscles were examined for sensitivity to palpation as described previously and based on accepted protocols (14,22–25). Intraorally the examination aims to rule out gross dental, periodontal and mucosal pathology.
Imaging
All patients arrived with no previous imaging of the head and were referred by us for imaging of the brain and brainstem to exclude intracranial pathology. All patients that arrived with no dental radiographs were referred for these to exclude clinically occult pathology. These included full-mouth periapicals and/or panoramic radiographs.
Pharmacotherapeutic protocol
The clinic employs a stepped pharmacotherapeutic protocol based on relevant literature (26,27). We initiate therapy with CBZ, regular or controlled release formulation that has fewer side effects. Drug dosage is titrated according to therapeutic benefit versus side effects, taking into account the patient’s weight, habits (smoking), etc. If patients complain of immediate side effects, oxcarbazepine has fewer cognitive side effects and is our alternative to CBZ (3). Alternatively, when CBZ causes mild, dose-dependent side effects we reduce the dose and add baclofen (7,28). Add-on therapy with lamotrigine has been suggested (8), but due to lamotrigine’s side effects and slow titration schedule we very rarely employ this. Alternatives we employ include gabapentin (29) and pregabalin (30), and a trial of either one of these was considered the end of the study for any individual patient that had progressed to this point for medical reasons, side effects or failure of previous medications. All patients underwent baseline haematologic, enzymatic (liver function) and biochemical testing with a further set of tests at four to eight weeks’ post-therapy (31–33).
Patients are routinely given pain diaries to record data for the time they are being treated. In this diary patients report pain severity, waking from sleep and medication compliance. Based on these pain diaries we examined therapeutic outcomes by assessing changes in pain severity and defined a significant improvement as ≥50% reduction in severity, from baseline.
Statistics
Data were analysed with SPSS (version 21 for Mac, IBM Corporation) with two-tailed α for significance set at 0.05. Associations between nominal variables were analysed with a Pearson’s Chi square (χ2), and between continuous variables with a regression analysis. The differences between continuous variables in dichotomous groupings were analysed with a Student’s t-test (T). Results are expressed as the mean and standard deviation.
Results
Total patient cohort (n = 81)
Patients
Of the 86 patients, two had meningiomas and three were diagnosed with CTN-like pain while suffering from multiple sclerosis. These five patients were excluded from further analysis.
The study cohort therefore consisted of 81 patients, 43 females and 38 males. The mean onset age was 61.6 ± 14.3 years (y) and the mean disease duration (i.e. interval from onset till patients arrived at the clinic) was 25.6 ± 38.5 months. A detailed review of the clinical features in this cohort appears in part I.
Summary of clinical phenomenology (Table 1 and for details, see part I)
Study population’s demographic and pain-related features.

M: male; F: female.
Stabbing (including sharp) and electric (including shooting) were collapsed into one parameter as ‘stabbing and/or electric’. Statistics (Stats) refer to the comparison between the short and long duration groups.
Pharmacotherapy
There were no medical issues that established an absolute contraindication to the use of CBZ. Seven patients arrived to our clinic after having been prescribed CBZ (five on 200 mg and two on 100 mg), with no effect on their pain. All other patients were with no medication and diagnosed in our clinic.
All patients were initiated on CBZ and this was continued to study completion in 54 (67%) of our patients at a mean dose of 575 ± 300 mg. In three patients CBZ was combined with baclofen (CBZ 400 ± 100 mg, 20 ± 10 mg) due to severe cognitive side effects with CBZ. Similarly, due to severe cognitive side effects, CBZ was replaced with oxcarbazepine (950 ± 500 mg) in seven patients (9%). Thus we had a total of 10 patients (12%) with severe cognitive side effects to CBZ, mostly lack of concentration and memory loss. Nearly all patients on CBZ reported some degree of dizziness (n = 60, 75%) and six (7.5%) had mild elevations in liver transaminases. There were no significant biochemical or haematologic side effects necessitating withdrawal.
A lack of response to these combinations resulted in the use of gabapentin in eight (9.9%), and pregabalin in one (1.2%) patient. Baclofen (35 ± 20 mg) as monotherapy was used in three patients who could not tolerate standard antiepileptic drugs. The rest of the patients (n = 5) had either refused therapy or had poor compliance with no data. In the 54 patients treated solely with CBZ there was no correlation between dose and disease duration, or dose and baseline VPS (Pearson’s p > 0.05).
Treatment outcomes (Table 2)
Using our standardised stepped pharmacotherapeutic protocol, 55 patients (68%) attained a ≥50% improvement in pain severity or attack frequency. Twenty-six therefore reported a non-significant response, i.e. less than 50% reduction in baseline pain severity. Patients with incomplete follow-up or who refused treatment due to side effects were included in the latter group following the principles of intention to treat.
Patients were seen at baseline then two weeks following that. Further appointments were scheduled according to response and varied from one-week to four-week visits. The follow-up period ranged from a predefined minimum of 12 weeks to a maximum of 38 weeks to complete the study protocol. Patients who improved significantly had a median of five visits (range 4–5) and those who did not, a median of six visits (range 4–8, p > 0.05).
Analysis of variables associated with significant pain relief in the total patient cohort (n = 81).

M: male; F: female.
Mean duration of paroxysmal pain was 1.6 ± 1.2 min, and significantly shorter in patients that improved (1.4 ± 0.9 min) relative to those that did not (2.1 ± 1.6 min, T: t = 2.43, df = 79, p = 0.02). Even when attack duration was dichotomised, data analysis showed that 74% of patients with short attack duration and only 50% of patients with long attack duration reported significant improvement (χ2: χ = 3.9, df = 1, p = 0.05).
For analyses, all autonomic signs were pooled into one group (Table 2 and see part I). In patients with autonomic signs, 16% improved significantly on our pharmacotherapeutic protocol. This compares with a significant improvement of 46% in patients with no autonomic signs (χ2: χ = 8.2, df = 1, p = 0.004). The presence of ‘pressure’ as a descriptor was associated with a poor outcome (χ2: χ = 4.1, df = 1, p = 0.04). Electrical and/or stabbing quality was not associated with a significant outcome (χ2: χ = 1.0, df = 1, p = 0.31).
As stated above, the patients with a duration of ≤2 min (‘short’ group) were analysed separately from those with attack duration of >2 min (‘long’ group). The decision to analyse the separate groups is supported by the clear finding of a significantly better outcome in patients with shorter attack durations and no autonomic signs. There were also significantly higher frequencies of background pain and of pain-related awakening in patients with long attack duration, as shown in Table 2.
Short attack group results (n = 61)
Pharmacotherapy and treatment outcome
Demographics, diagnosis and treatment response in patients with short (≤2 minutes) attack duration.

All patients reported paroxysmal pain that was unilateral and dermatomal. F: female; M: male; ID: identification; IHS: International Headache Society; Dur: attack duration; BackG: continuous background pain; Aut: autonomic signs; Wake: report that pain wakes from sleep; CBZ: carbamazepine; OXC: oxcarbazepine; GABA: gabapentin; PGB: pregabalin; BAC: baclofen; CTN: classical trigeminal neuralgia; Back: with persistent background facial pain; Par: purely paroxysmal; T: tearing; O: oedema; FF: facial flushing; R: redness; Ref: refused treatment; All: allergy to carbamazepine. In the column for treatment outcome, ‘+’ signifies a significant improvement, ‘−’ no response and ‘−+’ a partial but not significant improvement with the relevant drug next to it.
Analysis of variables associated with significant pain relief in patients with short attack duration only (n = 61).

M: male; F: female.
In the patients treated solely with CBZ there was no correlation between dose and disease duration, or dose and baseline VPS (Pearson’s p > 0.05).
Demographics (Table 4)
There was no association between gender and treatment outcome (χ2: χ = 0.15, df = 1, p = 0.7), as shown in Table 2. Age of onset in patients with significant improvement (60.7 ± 15.5 years) was not significantly different from that in patients with none (63.6 ± 14.4 y, T: t = 0.66, df = 59, p = 0.55).
Pain-related parameters (Table 4)
There were no significant differences in pain severity, attack duration, presence of background pain, the frequency of pain related awakenings or the identification of a trigger in patients that improved significantly and those that did not.
Disease duration
Mean disease duration was 22.6 ± 15.1 months and patients who did not respond to treatment suffered from pain for a significantly longer time (42.1 ± 21.7 months) than patients who improved significantly (13.9 ± 19.8 months, T: t = 4.8, df = 59, p < 0.001).
Autonomic signs (see Table 1)
Autonomic signs were observed in 13 (21.3%) patients with short attack duration. The presence of autonomic signs indicated significantly less frequent improvement (13.3%) than when no autonomic signs were present (43.8%, χ2: χ = 6.5, df = 1, p = 0.01).
Quality
A reported quality of ‘pressure’ reduced the frequency of a significant outcome from 31.3% to 8.9% (χ2: χ = 4.7, df = 1, p = 0.03).
Long attack duration group (n = 20)
Pharmacotherapy and treatment outcomes
Demographics, diagnosis and treatment response in patients with long (>2 minutes) attack duration.

All patients reported paroxysmal pain that was unilateral and dermatomal. Dur: attack duration; BackG: continuous background pain; Aut: autonomic signs; Wake: report that pain wakes from sleep; CBZ: carbamazepine; OXC: oxcarbazepine; GABA: gabapentin; PGB: pregabalin, BAC= baclofen, CTN= classical trigeminal neuralgia, Back = with persistent background facial pain; Atyp: atypical. Par: purely paroxysmal; ChSUNA: chronic short-lasting unilateral neuralgiform headache attacks; T: tearing; O: oedema; FF: facial flushing; R: redness; Ref: refused treatment. In the column for treatment outcome, ‘+’ signifies a significant improvement, ‘−’ no response and ‘−+’ a partial but not significant improvement with the relevant drug next to it.
Analysis of variables associated with significant pain relief in patients with long attack duration only (n = 20).

M: male; F: female.
CBZ was employed throughout the study in 13 (65%) patients at a mean dose of 650 ± 250 mg. Seven of these patients (35% of total group) attained a significant improvement. Two patients received CBZ with baclofen (CBZ 600 mg, 20 mg) due to side effects with CBZ and one attained significant improvement. Oxcarbazepine (900 ± 550 mg) was successfully employed in one patient (5%). A lack of response to these combinations resulted in the unsuccessful use of gabapentin in one (5%, 1600 mg). Baclofen (15 mg) as monotherapy was successfully used in one patient who could not tolerate standard antiepileptic drugs. Two of the patients (10%) refused therapy and three refused to progress to further alternatives after trying CBZ. There were no differences in CBZ drug dosages employed between the patients in the short and the long attack groups (T, p > 0.05).
Demographics (Table 6)
There was no association between gender, age of onset and treatment outcome (χ2: p > 0.05), as shown in Table 3.
Pain-related parameters (Table 6)
There were no significant differences in pain severity, pain quality, attack duration, disease duration, the presence of autonomic signs, frequency of pain-related awakenings or the identification of a trigger in patients that improved significantly and those that did not.
Background pain (Table 1)
A constant background pain was reported by 70% (n = 14) of patients with long attack duration. Patients with background pain reported a lower frequency of significant pain relief (50%) than those with no background pain (90%, χ2: χ = 3.8, df = 1, p = 0.05).
Discussion
The major finding in our total patient cohort is that treatment outcome is negatively impacted by a long attack duration, the presence of autonomic signs, prolonged disease duration and patient use of ‘pressure’ as a descriptor of pain quality, rarely associated with neuralgiform pain such as CTN (see part I and Cohen et al. (34)). Although the IHS criterion for CTN duration has a clear maximum of 2 min, attack duration in CTN has not been thoroughly validated and we rely on patient reports. This has been standard practice in publications studying CTN attack duration, as indeed also for almost all other headache duration, but these patient reports may be unreliable.
The patients in the long attack duration group were more resistant to standard anticonvulsants as recommended for CTN. The reasons for this are unclear but may involve disease progression as described below.
The vast majority (∼64%) of our patients with short attack duration responded significantly to CBZ or oxcarbazepine. This is in line with most of the published figures in the literature (35). However, a recent study reports that 98% of their CTN patients significantly responded to CBZ and 94% to oxcarbazepine (5), but no definition of a significant response was included.
Autonomic signs
When analysing the duration subgroups, presence of autonomic signs was still associated with a poor outcome in the short but not in the long attack group. This is an unclear and confusing finding. The presence of autonomic signs should be a negative prognostic factor irrespective of attack duration. Possibly this may be partly due to the near double frequency of autonomic signs observed in the long (40%) relative to the short (21%) attack group. Certainly the small sample size would reduce the power of our analyses. Autonomic signs in CTN have been previously reported, at a similar frequency to that observed in our short attack patient cohort and have been reported as a negative prognostic factor in surgery for CTN (17). Alternatively, as discussed in part 1, the patients with long attack duration share similarities with SUNA. It is unclear whether SUNA responds to CBZ at a similar level as CTN.
Disease duration
Similarly disease duration was still a significant predictor of outcome in the short attack but not in the long attack group. In the short attack group, mean disease duration in patients with treatment failure (39 months) was twice that in patients with significant relief (19 months). CTN has been recognised as a progressive disease and some series report increased attack frequency and severity over time (2,3). However, in other reports there seems to be little change in response to treatment over time, but a change in symptomatology such as longer attack duration and increased reports of persistent pain (5). As a short-term study we could not accurately evaluate these changes prospectively but we found no association between reported disease duration and pain severity in either group. Therefore our study is inconclusive on this point and possibly treatment failures remain in the system longer.
Similarly, initial response to CBZ for CTN is reported around 70%, which is similar to the rate achieved in our patients using standard anticonvulsive pharmacotherapy (particularly CBZ). This is a particularly encouraging result considering our total cohort was made up of a number of heterogeneous cases.
Disease duration and the central nervous system (CNS)
A reduction in grey matter volume has been documented in a cohort of CTN patients (36), similar to that in other nerve injury models. Interestingly, the reduction of grey matter in some areas correlated with longer disease duration suggesting deleterious effects of disease duration. In a study examining the effects of triggering pain in CTN on CNS structures (37), evidence was found for pathological hyperexcitability of the trigeminal nociceptive system. The pain neuromatrix showed significant activation during nonpainful stimulation of the trigger zone, suggesting a state of persistent sensitisation of the trigeminal nociceptive system in CTN (37). These findings support the involvement of central mechanisms in CTN and the progressive, time-dependent nature of these changes.
Background pain
There are indications that concomitant background facial pain is a clinical predictor of poorer treatment response, both pharmacologic and surgical (9–13). Microvascular decompression (12,38) and rhizotomy (39) are less successful in CTN cases with background pain. In contrast, however, other centres report no differences in surgical or gamma knife outcomes (40,41). The reasons for different centres reporting such discrepancies are unclear but suggest that this group of CTN patients may not be homogeneous, and this may be related to different inclusion criteria. For example, we found a significant effect of background pain on treatment outcome in the long but not the short attack group. Moreover, treatment outcome in patients with background pain deteriorates with disease duration and possibly centres with negative outcomes had patients with longer disease duration.
Treatment outcome
Depending on how our patient cohort was examined, different variables appeared to significantly impact treatment outcome. Based on the literature and our findings, we hypothesised a model that may underlie treatment response in patients with CTN (Figure 1). While we recognise that correlation is not causation, our data and the literature lend support for this model. Disease duration appears as a strongly associated factor and we propose this as the distal variable. Disease duration in CTN may drive changes in CNS structure and reported levels of therapeutic outcomes. Possibly CNS changes may also underlie the appearance of autonomic signs, background pain and increased attack duration. An interesting and alternative explanation is that there may be a time-dependent continuum between CTN and SUNA (42). SUNA is associated with autonomic signs, longer attack duration, and possibly an increased frequency of background pain.
Hypothetical concept on factors influencing outcomes in classical trigeminal neuralgia (CTN).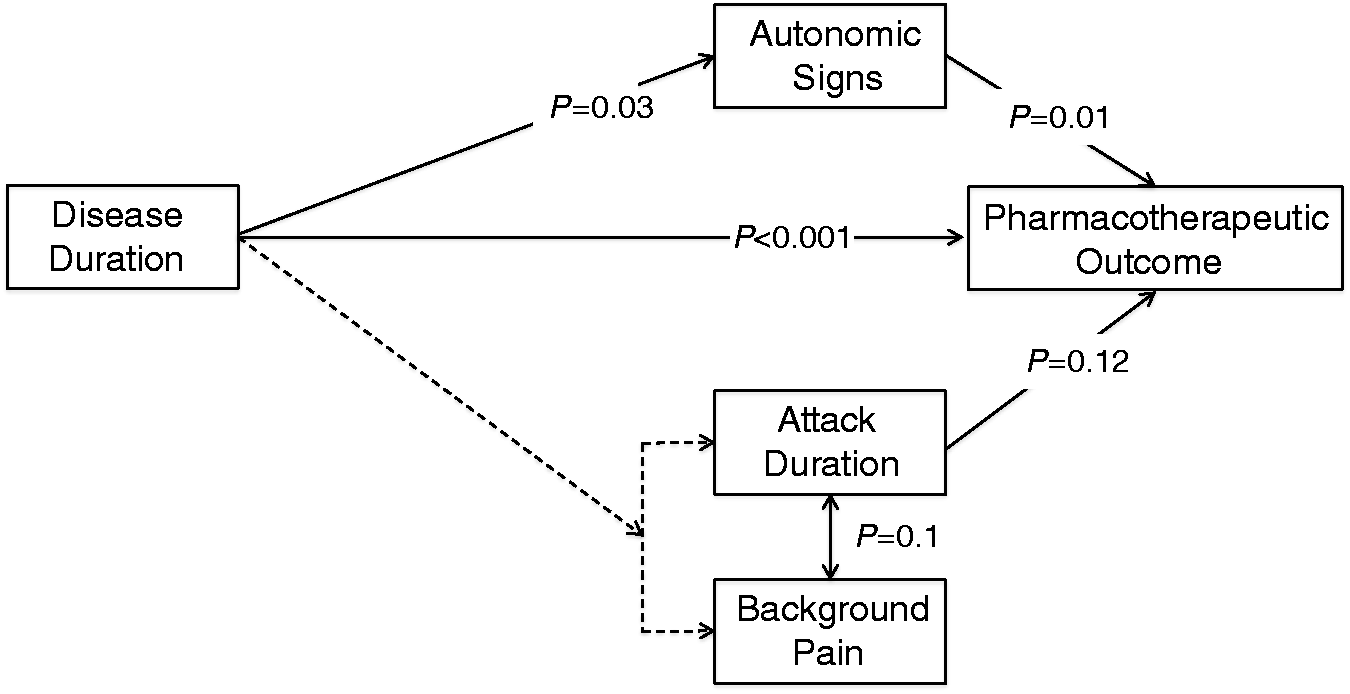
There is evidence in the literature that CTN is associated with progressive, time-dependent structural changes in the central nervous system (CNS) (33). At the same time our study and others show a clear reduction on prognosis as disease (CTN) duration is prolonged (3,4). We hypothesise that CNS changes may drive the onset of atypical signs and poor treatment outcome. Autonomic signs are a clear negative prognostic indicator in our and other studies (13). Additionally disease duration may drive the onset of background pain and the inter-related attack duration (see Discussion). Both of these may be associated with a poor prognosis and there is evidence in the literature for the association between background pain and poor outcomes (5–9). These same features: autonomic signs, background pain and attack duration may also mark the slow transition of CTN into a more atypical phenotype.
Natural history of CTN
Long-term follow-up of CTN patients reveals that there are well-defined periods of pain attacks variably followed by periods of remission that may last from weeks to years. The median active period is reported at about 49 days followed by remission of some months (36%), weeks (16%) or even days (16%). Only 6% may look forward to remissions of more than a year and about 20% may suffer from incessant attacks. The question then arises when a patient is asymptomatic on medication: Is he in remission or enjoying the benefits of therapy? There are not enough data available comparing remission patterns of CTN patients with different clinical signs. In patients with autonomic signs, resistance to treatment may also raise the pertinent question of whether these are CTN or SUNA (42).
Study limitations
Our patient cohort is relatively small and possibly some treatment effects would have been clearer in a larger sample. This is particularly true in the group with attack duration of >2 min. Some of these patients remain questionable as either atypical CTN or atypical SUNA (see part I).
Footnotes
Clinical implications
Prolonged disease duration and the presence of autonomic signs are negative prognosticators for classical trigeminal neuralgia (CTN).
Patients with CTN-like symptomatology but long attack duration are less responsive to standard anticonvulsive therapy.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for for the research, authorship, and/or publication of this article.
