Abstract
Aims
We conducted a cross-sectional study to re-examine the clinical profile of patients with a clinical diagnosis of classical trigeminal neuralgia (CTN).
Methods
Inclusion criteria consisted of the International Headache Society’s published classification of CTN. For the specific purposes of the study, features such as autonomic signs, persistent background pain, attack durations of >2 minutes and reports of pain-related awakening were included. The demographic and clinical phenotype of each patient were carefully recorded for analysis.
Results
The study cohort consisted of 81 patients and based on reported attack duration these were divided into short (≤ 2 minutes, n = 61) and long (> 2 minutes, n = 20) groups for further analysis. The group with short attack duration neatly fit most of the criteria for CTN while the long attack group presents a more challenging diagnosis. There were no significant differences in pain severity, quality and location between the short and long attack groups. The frequency of persistent background pain was significantly higher in the long (70%) compared to the short attack group (29.5%, p = 0.001). There were significantly more reports of pain-related awakenings in the long (55%) than in the short attack groups (29.5%, p = 0.04). There were no significant differences in the frequency of autonomic signs between the short (21.3%) and long attack groups (40%, p = 0.1). In the short attack group, the presence of autonomic signs was significantly associated with longer disease duration, increased pain-related awakenings, and a reduced prognosis.
Conclusion
There are clear diagnostic criteria for CTN but often patients present with features, such as long pain attacks, that challenge such accepted criteria. In our cohort the clinical phenotype of trigeminal, neuralgiform pain with or without autonomic signs and background pain was observed across both short and long attack groups and the clinical implications of this are discussed.
Introduction
Classical trigeminal neuralgia (CTN) is a paroxysmal, short-lasting, unilateral, facial pain of excruciating intensity. The International Headache Society (IHS) classifies CTN based on clear clinical features (1). These include recurring, unilateral, trigeminal pain that is paroxysmal and lasts seconds to 2 minutes (min). Intensity is severe and pain quality is electric-like, shooting, stabbing or sharp. Pain paroxysms are typically precipitated by innocuous stimuli to the affected side of the face (also known as triggering), but may also be spontaneous (1). The pathophysiology of most CTN cases is considered to be secondary to compression of the trigeminal root at or near the dorsal root entry zone by an aberrant blood vessel (2,3). This concept receives wide support from imaging with correlated surgical findings (4–6), and ultrastructural analysis of neuronal tissue (7,8). These confirm neurovascular compression in many patients with resultant clear histological damage to neurons and their myelin sheaths.
Diagnostic criteria for CTN (modified with permission from the IHS 2013).

The table shows the essential criteria for diagnosis of CTN and the scoring that we allotted each criterion for the study. In addition the IHS also stipulate that CTN may occur with constant background pain. aStabbing (including sharp) and electric (including shooting) were collapsed into one parameter.
CTN: classical trigeminal neuralgia; IHS: International Headache Society; ICHD-3: International Classification of Headache Disorders, third edition; VAS: visual analogue scale.
The presence of autonomic signs in CTN creates a challenging diagnosis vis-à-vis short-lasting unilateral neuralgiform headache attacks with conjunctival injection and tearing (SUNCT) and with short-lasting unilateral neuralgiform headache attacks with autonomic signs (SUNA). Although SUNCT and SUNA are characterised by intense autonomic activation, longer attack duration and the usual absence of a refractory period, there is often considerable phenotypic overlap with CTN (1). There are no data on whether SUNCT and SUNA are discrete entities or variants of the same disorder. However, at this stage the IHS has grouped these together under short-lasting unilateral neuralgiform headache attacks (SUNHA).
The aim of the study was to re-examine clinical features in a cohort of CTN patients.
In particular we were interested in examining specific features of CTN such as the presence of a persistent background pain in addition to the typical paroxysms, autonomic signs, waking from sleep and reported attack duration.
Materials and methods
All patients were interviewed and examined at the Orofacial Pain Clinic, Faculty of Dentistry, Hadassah-The Hebrew University, Jerusalem, Israel. Data collection and characterisation was prospective and the analysis performed at the end of the study. The patients were observed between the years 2005 and 2013.
Inclusion criteria and pain diagnosis
Inclusion criteria consisted of all patients with trigeminal neuralgiform pain with a tentative clinical diagnosis of CTN according to the then current classification published by the IHS (16) and updated in 2013 (1). For the aims of this study we allowed inclusion of patients with atypical symptomatology such as persistent background pain in addition to the typical paroxysms, autonomic signs, waking from sleep and reported attack duration beyond 2 min. Exclusion criteria consisted of any patient that on further history, examination or imaging was found to have underlying pathology, infection or previous trauma explaining the pain. All diagnoses were confirmed in the clinic and then re-examined following data tabulation and summary, by both senior authors (RB, YS).
Data collection
The institutional review board approved the study and informed consent was obtained from all participating patients. Eighty-six patients were included and interviewed. The resultant data, including a standard pain history, were recorded on a standard intake form.
At baseline, patients were asked to rate pain duration, quality and pain intensity of a typical paroxysm. Patients were asked to assess their pain duration based on that of their predominant attacks. Pain intensity was rated employing a verbal pain scale (VPS) where 0 is no pain and 10 the worst imaginable pain. In the pain diary patients were asked to rate each attack. The IHS criteria do not specify a minimum pain intensity score for CTN and employ the definition of ‘severe’; based on the literature we define severe as a score of >6 (17). Pain quality was assessed by asking the patients to choose one or more of the following descriptive terms: electrical (includes shooting), stabbing (includes sharp), throbbing, pressure, burning or any combination of the five terms. These terms are in routine use in our clinic to provide rapid assessment of pain quality (9,18). To test for combinations of quality descriptors, each term was coded so that when summed each combination retained a unique identifier.
Patients were asked specifically about autonomic signs, i.e. tearing and redness of the ipsilateral eye, nasal congestion/rhinorrhoea, eyelid oedema or regional swelling, increased sweating, flushing, fullness in the ear and ptosis and/or miosis during pain attacks. The presence of continuous background pain, in addition to the typical paroxysms, was noted. For follow-up, pain diaries were employed so that accurate data on VPS and treatment were available. Location of pain was mapped out onto a diagram of the head and neck.
Patients were also asked via a standardised question whether the pain specifically wakes them from sleep. Answers to this question were carefully interpreted so as to ensure that the patient was reporting awakening specifically related to pain. In this manner we excluded, for example, random awakenings (to drink water, or for micturition) where the patient reported that pain was coincidentally present but had not been the reason for awakening (9).
Clinical examination
All patients underwent a thorough extra- and intra-oral examination including a cranial nerve examination. So as to ensure the correct interpretation of ‘triggered’, we assigned this only to patients in whom we could clinically identify trigger areas and induce a refractory period. The masticatory apparatus (temporomandibular joints and masticatory muscles) and neck muscles were examined for sensitivity to palpation as described previously and based on accepted protocols (18,19). Tenderness to palpation was graded and the individual scores summated to give the total tenderness score (muscle tenderness score) for each patient (20,21).
Intra-orally the examination aims to rule out gross dental, periodontal and mucosal pathology.
Imaging
All patients arrived with no previous imaging of the head and were referred by us for imaging of the brain and brainstem to exclude intracranial pathology. All patients that arrived with no dental radiographs were referred for these to exclude clinically occult pathology. These included full mouth periapicals and/or panoramic radiographs.
Treatment
Treatment was initiated as per recommended protocols (22). In brief, patients are initiated on carbamazepine (CBZ) or oxcarbazepine (OXC). If there is a partial response, baclofen is added and outcome reassessed. Patients with a rash or other allergic-type reaction were transferred to baclofen monotherapy. If there is a lack of response to CBZ or OXC, patients are offered gabapentin or pregabalin. Treatment response was assessed through pain diaries and patient interview. Significant relief was defined as an improvement of 50% or more in pain scores.
The analysis of treatment outcomes is the subject of the second part of this study.
Statistics
Data were analysed with SPSS (version 21 for Mac, IBM Corporation) with two-tailed α for significance set at 0.05. Associations between nominal variables were analysed with a Pearson’s Chi square (χ2), and between continuous variables with a regression analysis. The differences between continuous variables in dichotomous groupings were analysed with a t-test (T). Results are expressed as the mean and standard deviation.
Results
Patients
Of the 86 patients clinically diagnosed with TN, two had meningiomas and three were diagnosed while suffering from multiple sclerosis. These lesions were considered to be causal by our consulting neurosurgeon and neurologist. These five patients were significantly younger than the CTN patients (48.7 ± 7.7 years (y) versus 61.6 ± 14.3 y, T: t = 2.6, degree of freedom (df) = 85, p = 0.05). Although not significant, the five patients were also remarkable in that four had no identifiable trigger point while this was absent in 38% of CTN patients (χ2: χ = 3.2, df = 1, p = 0.07). Additionally none of the secondary neuralgia patients had autonomic signs (CTN = 26%. χ2: χ = 1.7, df = 1, p = 0.19). They were otherwise unremarkable in their demographic or pain characteristics. Based on the history, clinical and radiological examinations, none of the patients had dental or other oral pathology that could explain the pain. The total study cohort therefore consisted of 81 patients, 43 females (age 60.3 ± 17.5) and 38 males (age 63.1 ± 9.5 y). The mean pain onset age was 61.6 ± 14.3 y and the mean disease duration (i.e. interval from onset till patients arrived at the clinic) was 25.6 ± 38.5 months.
Seven patients arrived to our clinic after having been prescribed CBZ (five on 200 mg and two on 100 mg), with no effect on their pain. All other patients were with no medication and diagnosed in our clinic.
Study population’s demographic and pain-related features.
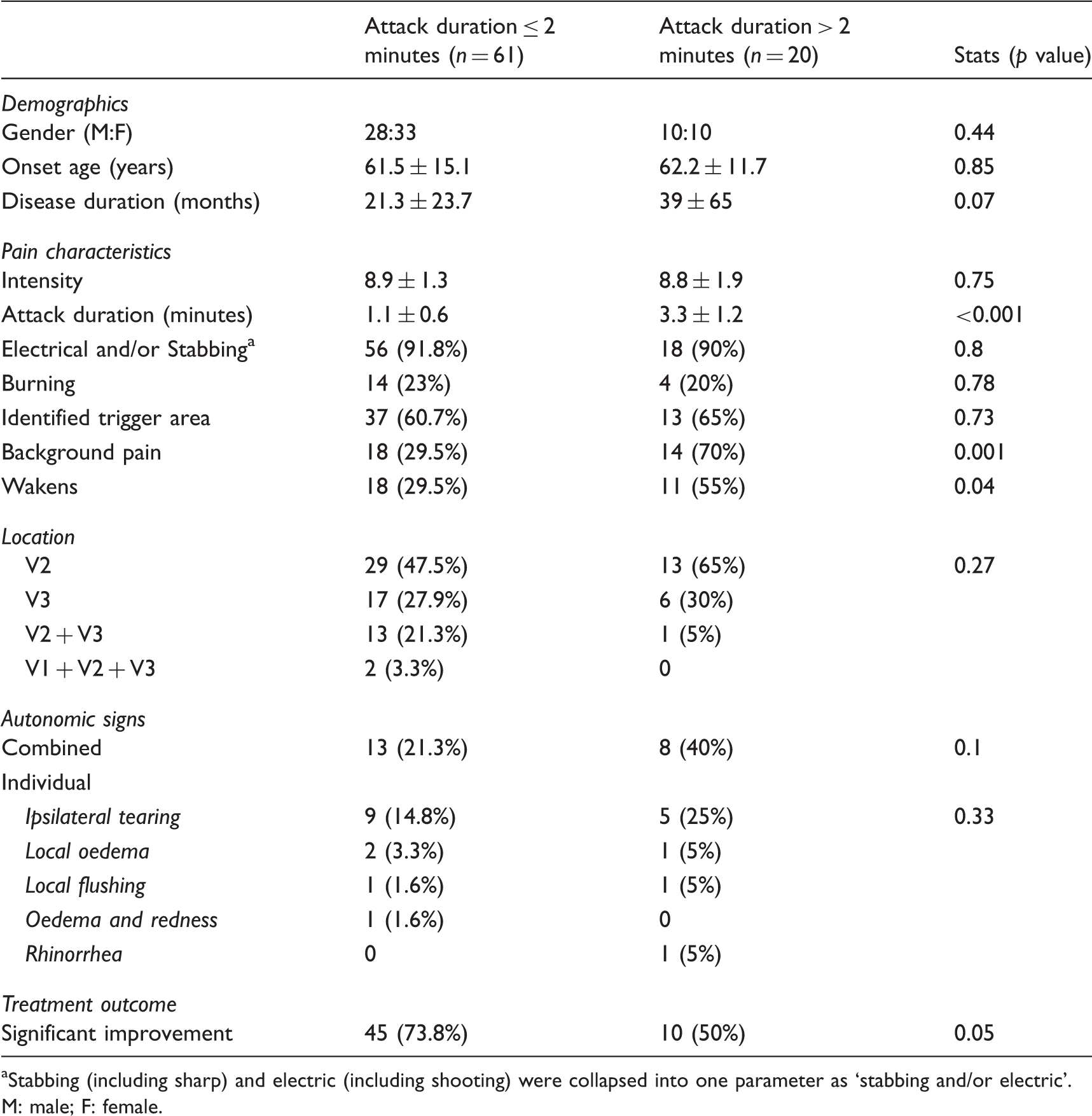
Stabbing (including sharp) and electric (including shooting) were collapsed into one parameter as ‘stabbing and/or electric’.
M: male; F: female.
Demographics and pain-related features
Gender
There was no significant difference in gender ratios between the attack duration subgroups (Table 2). In both short and long groups there was no significant effect of gender on any of the demographic or pain-related parameters collected.
Onset age and disease duration
Onset ages in the short (61.5 ± 15.1 y) and long (62.2 ± 11.7 y) duration subgroups were not significantly different (T: t = −0.48, df = 79, p = 0.85) (Table 2).
Although disease duration in the short (21.3 ± 23.7 months) was less than that observed in the long (39 ± 65 months) group, this was not statistically significant (T: t = −1.8, df = 79, p = 0.07).
Location
In both groups pain was invariably unilateral. Most patients, regardless of subgroup, reported pain solely in the mandibular dermatome followed by solely in the maxillary and then by pain in both maxillary and mandibular dermatomes jointly (see Table 2). Only two patients from the short attack group reported involvement of all ipsilateral trigeminal dermatomes.
This distribution of location was significantly different from the statistically predicted pattern (one sample χ test; short p < 0.0001, long p = 0.004). The distribution of location was not significantly different between attack duration subgroups (χ2: χ = 3.89, df = 3, p = 0.27).
Duration and frequency
Demographic and pain-related features associated with short (≤ 2 minutes) attack duration.
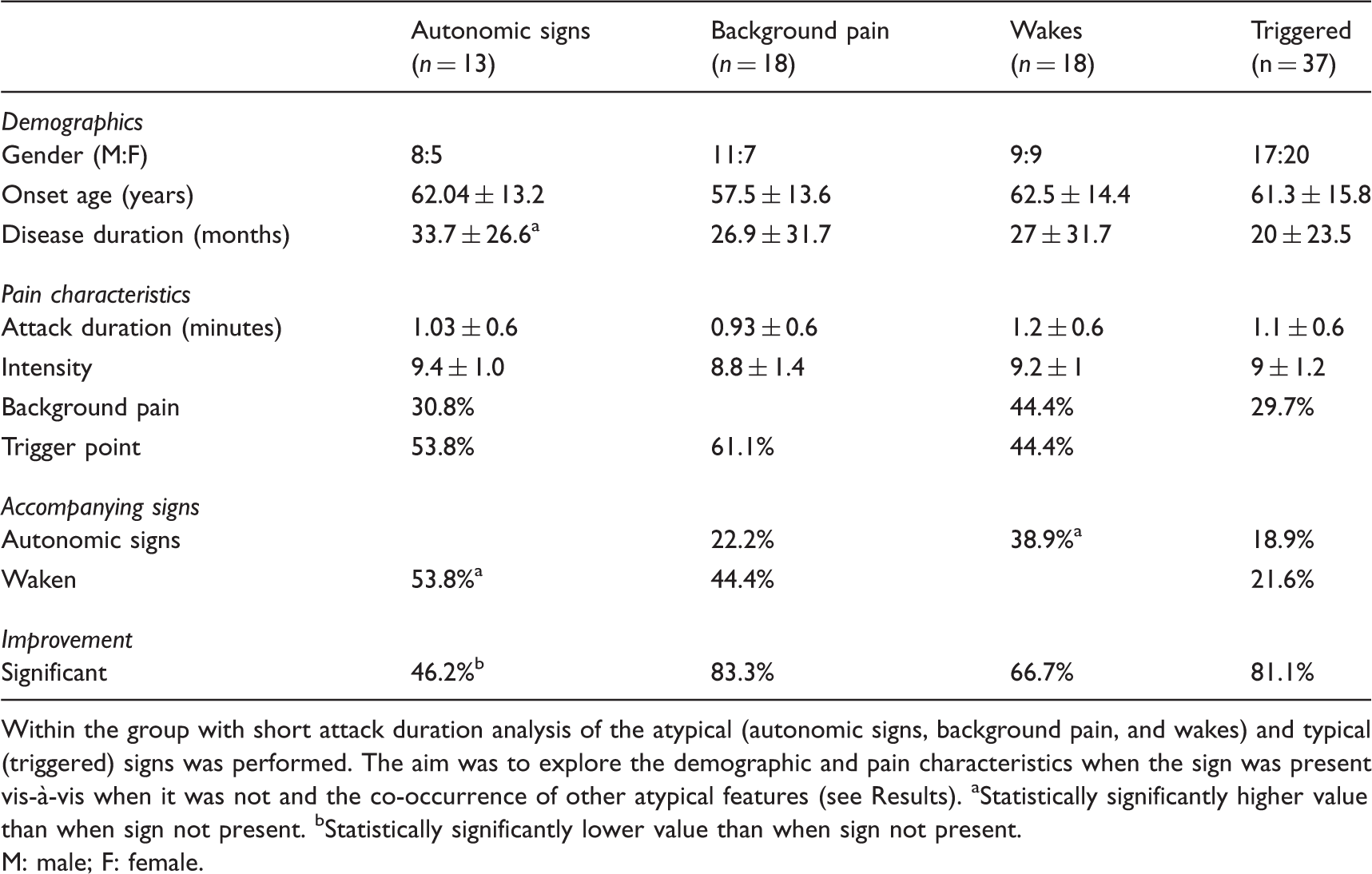
Within the group with short attack duration analysis of the atypical (autonomic signs, background pain, and wakes) and typical (triggered) signs was performed. The aim was to explore the demographic and pain characteristics when the sign was present vis-à-vis when it was not and the co-occurrence of other atypical features (see Results). aStatistically significantly higher value than when sign not present. bStatistically significantly lower value than when sign not present.
M: male; F: female.
Demographic and pain-related features associated with long (> 2 minutes) attack duration.

Within the group with long attack duration analysis of the atypical (autonomic signs, background pain, and wakes) and typical (triggered) signs was performed. The aim was to explore the demographic and pain characteristics when the sign was present vis-à-vis when it was not and the co-occurrence of other atypical features (see Results). aStatistically significantly higher value than when sign not present. bStatistically significantly lower value than when sign not present.
M: male; F: female.
Patients reported between two and 20 attacks daily with no significant differences between the short and long groups (p > 0.05). We did not collect data on the profile of attacks, e.g. if they felt these to be ‘saw-toothed’, etc.
Severity
Pain was severe in both short (8.9 ± 1.3) and long (8.8 ± 1.9) attack duration groups, with no significant difference between them (T: t = 0.3, df = 79, p = 0.75) (Table 2).
A significant inverse correlation was noted between VPSs and attack duration in the long (R = 0.78, coefficient B = −1.2, p < 0.001), but not in the short attack group (R = 0.09, coefficient B = −0.19, p = 0.51), as shown in Figure 1(a) and (b).
Correlation between attack duration and severity. >(a) In short attack group. (b) In long attack group.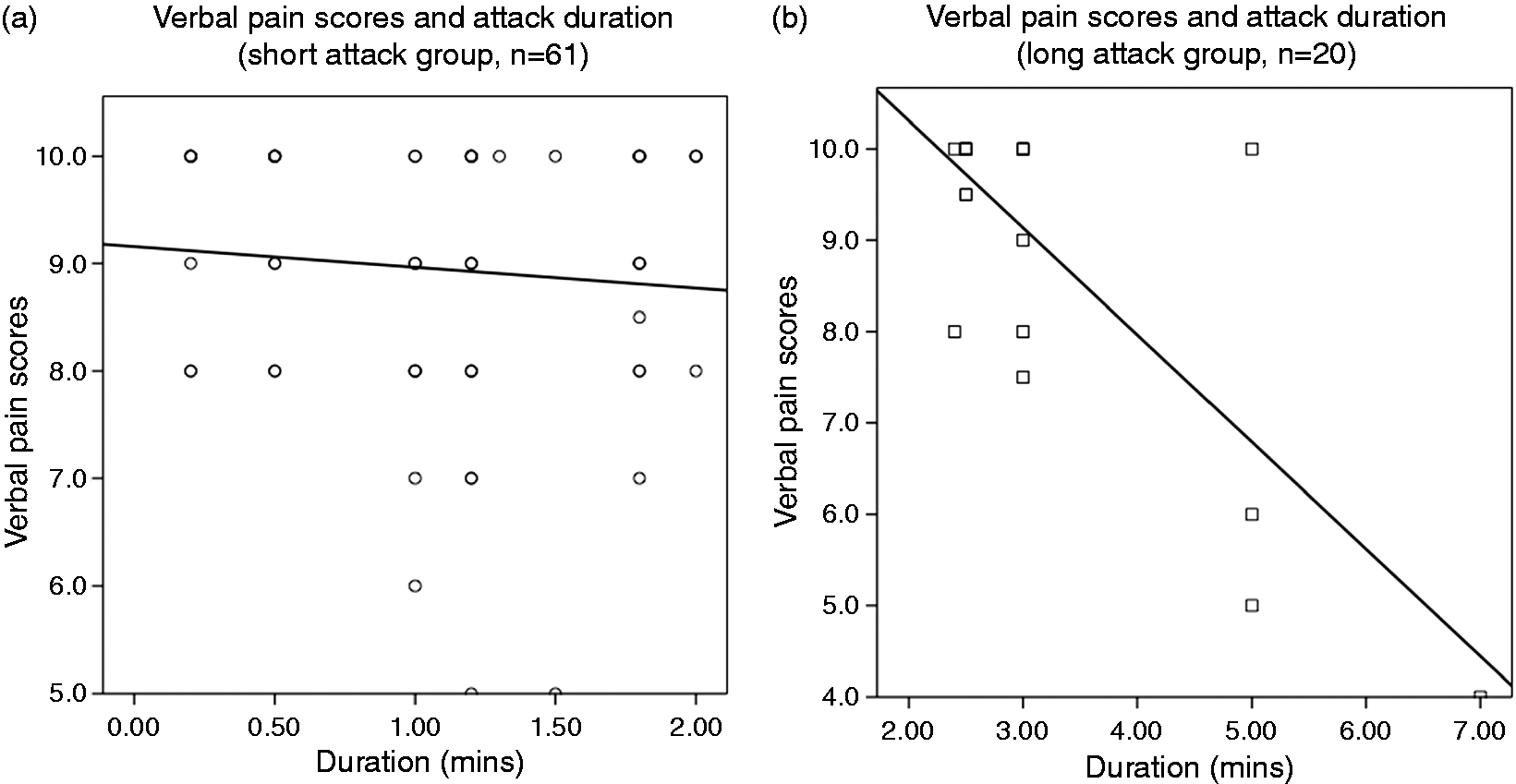
Quality
It was found that the presence of ‘electrical’ as a descriptor was significantly correlated to a description of ‘stabbing’ (χ2: χ = 14.6, df = 1, p < 0.001). Since these two qualities together form part of the definition of CTN, they were combined for further analysis into one variable (electrical and/or stabbing, see Table 1).
Distribution of quality descriptors employed by the patients with short and long attack duration (see Results).

Background pain (Tables 3 and 4)
Background pain was significantly more common in the long (n = 14, 70%) compared to the short duration subgroup (n = 18, 29.5%. χ2: χ = 10.3, df = 1, p = 0.001). We were unable to obtain reliable historical data from our patients on whether the background pain had begun with the initiation of the paroxysmal attacks or had developed later in the natural course of the pain.
A constant background pain was more common in males than in females in both attack duration subgroups, but this was not statistically significant (χ2: p > 0.05).
In both the short and long duration subgroups, pain-related awakenings were not significantly more common in patients with background pain than in those without (Tables 3 and 4).
Autonomic signs
The distribution of ipsilateral tearing during pain attacks, oedema, local flushing and combination of these is shown in Table 2. There was no statistically significant difference in the distribution of autonomic signs between the short and long attack groups (χ2: χ = 5.8, df = 5, p = 0.33). There were no significant correlations between autonomic signs and pain location in both the subgroups (χ2: p > 0.05).
For further statistical analyses, all autonomic signs were pooled into one group, and these were present in 13 (21.3%) of the short and eight (40%) of the long attack duration subgroups (χ2: χ = 2.7, df = 1, p = 0.1).
In the short attack duration subgroup, the presence of autonomic signs was significantly associated with longer disease duration (33.7 ± 26.6 versus 17.9 ± 26.22 months T: t = −2.2, df = 59, p = 0.03), a higher frequency of waking (53.8% versus 22.9%, χ2: χ = 4.7, df = 1, p = 0.03), and a lower frequency of significant improvement on standard anticonvulsant therapy (46.2% versus 81.3%, χ2: χ = 6.5, df = 1, p = 0.01). No significant associations were observed for autonomic signs in the long attack duration subgroup (see Table 4).
Waking
Pain-related awakenings were of significantly lower frequency in patients with short (n = 18, 29.5%) than in those with long attack duration (n = 11, 55%. χ2: χ = 4.3, df = 1, p = 0.04), as shown in Table 2.
In the short attack group, pain severity in patients woken from sleep was not significantly higher than in those that were not (9.2 ± 1 versus 8.8 ± 1.4, T: t = −1.1, df = 59, p = 0.28). However, patients in the long duration group woken from sleep reported both significantly more severe attacks (9.6 ± 0.8), and significantly shorter attacks (2.7 ± 0.3 min) than those not woken from sleep (severity 7.9 ± 2.4, T: t = −2.3, df = 18, p = 0.04, duration 3.9 ± 1.6 min, T: t = 2.5, df = 18, p = 0.02).
Triggering (Table 6)
An identifiable trigger was found in 60.7% (n = 37) of patients with short and 65% (n = 13) of patients with long attack duration (χ2: χ = 0.12, df = 1, p = 0.73). All of the patients with identifiable trigger areas demonstrated a refractory period. In the patients with no identifiable trigger, some reported pain triggered by movement or other innocuous stimuli (short n = 7, 11.7%; long n = 2, 10%; p > 0.05), but we were unable to replicate these latter triggers.
Comorbid muscle pain
In both duration subgroups the vast majority of patients (short n = 47/77%, long n = 14/70%) had no comorbid muscle pain. The number of pericranial and masticatory muscles involved ranged from one to eight. There was no significant difference in this distribution or in the calculated total tenderness score between the short and long attack duration subgroups (p > 0.05). In both subgroups no correlations or associations could be found between muscle tenderness and demographic or pain-related features.
Analysis of patient profiles vis-à-vis IHS criteria
Individual clinical profiles for patients with short (≤ 2 minutes) attack duration.
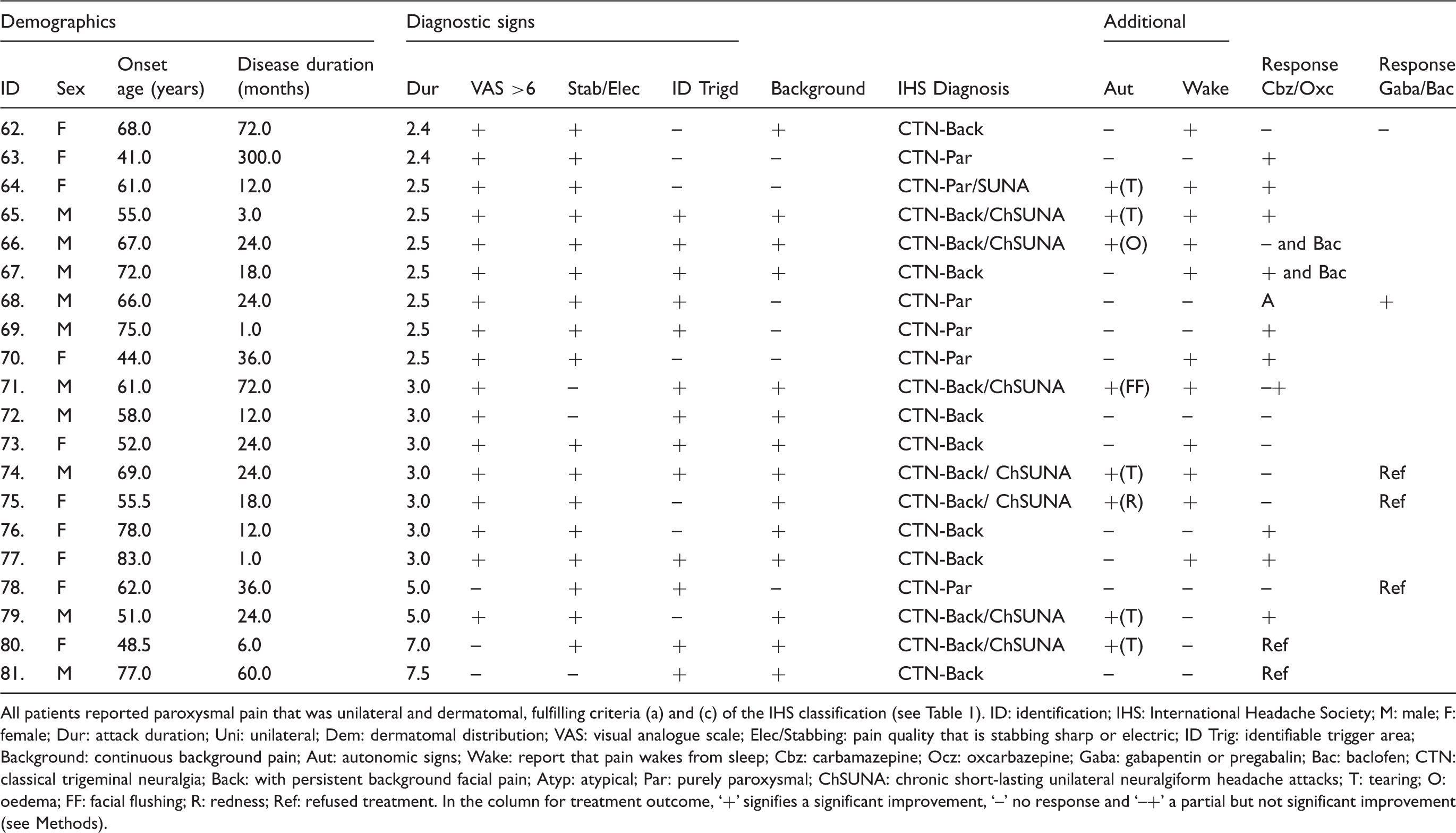
All patients reported paroxysmal pain that was unilateral and dermatomal, fulfilling criteria (a) and (c) of the IHS classification (see Table 1). ID: identification; IHS: International Headache Society; M: male; F: female; Dur: attack duration; Uni: unilateral; Dem: dermatomal distribution; VAS: visual analogue scale; Elec/Stabbing: pain quality that is stabbing sharp or electric; ID Trig: identifiable trigger area; Background: continuous background pain; Aut: autonomic signs; Wake: report that pain wakes from sleep; CBZ: carbamazepine; OCZ: oxcarbazepine; Gaba: gabapentin or pregabalin; Bac: baclofen; CTN: classical trigeminal neuralgia; Back: with persistent background facial pain; Par: purely paroxysmal; T: tearing; O: oedema; FF: facial flushing; R: redness; Ref: refused treatment; All: allergy to carbamazepine. In the column for treatment outcome, ‘+’ signifies a significant improvement, ‘–’ no response and ‘–+’ a partial but not significant improvement (see Methods).
Individual clinical profiles for patients with long (> 2 minutes) attack duration.

All patients reported paroxysmal pain that was unilateral and dermatomal, fulfilling criteria (a) and (c) of the IHS classification (see Table 1). ID: identification; IHS: International Headache Society; M: male; F: female; Dur: attack duration; Uni: unilateral; Dem: dermatomal distribution; VAS: visual analogue scale; Elec/Stabbing: pain quality that is stabbing sharp or electric; ID Trig: identifiable trigger area; Background: continuous background pain; Aut: autonomic signs; Wake: report that pain wakes from sleep; Cbz: carbamazepine; Ocz: oxcarbazepine; Gaba: gabapentin or pregabalin; Bac: baclofen; CTN: classical trigeminal neuralgia; Back: with persistent background facial pain; Atyp: atypical; Par: purely paroxysmal; ChSUNA: chronic short-lasting unilateral neuralgiform headache attacks; T: tearing; O: oedema; FF: facial flushing; R: redness; Ref: refused treatment. In the column for treatment outcome, ‘+’ signifies a significant improvement, ‘–’ no response and ‘–+’ a partial but not significant improvement (see Methods).

Distribution of number and profile of signs from International Headache Society (IHS) criteria in patients with reported attack duration of ≤ 2 min (see Table 1):

Distribution of number and profile of signs from International Headache Society (IHS) criteria in patients with reported attack duration of >2 minutes (see Table 1):
Discussion
The major finding in our patients is the high frequency of features such as background pain, autonomic signs, and waking from sleep in patients with short attack duration diagnosed with CTN. We also note a number of patients with long attack duration and re-examine their diagnosis. The analyses and discussion on treatment outcomes are covered in part II of this article.
The diagnosis of head and facial pain relies primarily on the specific combination of clinical features. In CTN the most prominent of these have been considered to be its extreme intensity, the triggering mechanism and the short attack duration. These factors together with the other signs are discussed below.
Background pain
The aetiology of the persistent pain is unclear and a faulty pain modulation system or central sensitisation have been proposed (23,24). CTN patients with persistent pain experienced no experimental induction of conditioned pain modulation, suggesting a deficient descending inhibitory system (24). Additionally they had more tender points relative both to controls and purely paroxysmal CTN patients, indicating central sensitisation extending beyond the trigeminal system (24). Central facilitation of trigeminal nociceptive processing was observed in patients with TN with concomitant chronic facial pain, indicating overactivation of central sensory transmission (25).
About 30% of our short attack duration patients reported a persistent background pain in addition to paroxysmal pain, making this a reasonably common clinical sign. These findings are in agreement with the literature describing between 35% and 49% of patients reporting concomitant background pain (9,10). Background pain may be described as dull, throbbing and burning (3), and in our cohort a number of patients with background pain described pain as burning or throbbing, but this was not statistically significant. Background pain was significantly more common in the long attack duration subgroup and this possible relationship is discussed below.
Irrespective of subgroup, patients with background pain were more frequently male and of similar onset age, contrasting with other reports that CTN patients with background pain are younger and more often females than in purely paroxysmal CTN (10). Nevertheless, the data on features suggest that patients with background pain are a distinct subgroup of TN.
Autonomic signs
Why autonomic signs appear in CTN is unclear. Painful experimental stimuli in areas innervated by trigeminal nerve divisions 1 and 2 will cause ipsilateral lacrimation and nasal stuffiness or rhinorrhoea (26). These effects are largely considered secondary to initiation of a parasympathetic reflex via trigeminal nerve activation; the trigeminoparasympathetic reflex (TPR). Interestingly, in our cohort there were no patients with pain in the first trigeminal division. Our interpretation is that patients tend to choose their medical specialist based on pain location. Thus cases with tooth/jaw pain (second and third divisions) tend to choose dental practitioners whilst the presence of eye pain may guide them to neurologists and ophthalmologists. Rarity of first-division pain has been a consistent finding in our reports of CTN patients.
Appearance of autonomic signs in some headaches is variable and has been linked to pain severity (27,28). If so, then when CTN is particularly severe trigeminal-autonomic activation may occur.
Although our patients with autonomic signs had slightly more intense pain, this was not statistically significant. Possibly there may be autonomic dysfunction in CTN and this is suggested in a case reported of cardiac arrest in association with CTN (29).
In the trigeminal autonomic cephalalgias (TACs) there is evidence of hypothalamic dysfunction that is purported to disinhibit the TPR and underlie associated sleep disorders (30). To our knowledge, however, there have been no reports of hypothalamic dysfunction in CTN. On the other hand, hypothalamic stimulation has been successful in a group of TN patients with multiple sclerosis (31), particularly for pain in the ophthalmic branch. However, it is unclear whether this is acting via hypothalamic modulation of the trigeminal nociceptive system or acting directly on the hypothalamus. It is also unclear how much we should derive from findings in TN associated with multiple sclerosis, and apply these to mechanisms of CTN.
Waking
Pain-related awakenings are usually associated with pain intensity (9,10) or shared pathophysiology with the pain syndrome (e.g. cluster headache). We observed pain-related awakening in a significantly higher proportion of the long attack duration group. In the short attack duration group there was no difference in pain intensity between patients woken and those not. This may not be surprising due to the fact that the two groups, those that woke and those that did not, had severe pain (VAS 9.2 versus 8.8). Alternatively, pain severity may not be a delineating factor.
Pain-related awakenings were reported by 29.5% of our short attack duration patients. This is similar to the frequencies reported recently for CTN (9,32). In contrast to our previous report (9), this phenomenon was not related to pain intensity but to the presence of autonomic signs.
Others have suggested that pain-related awakenings are related to triggering mechanisms. We did not note any difference in frequency of an identifiable trigger area in both patient subgroups woken from sleep. Atypical phenomena may not always be related to pain characteristics such as severity and triggering but may reflect other mechanisms such as hypothalamic dysfunction. Hypothalamic dysfunction, as suggested by functional imaging (33), may explain the reports of pain-related awakenings, and autonomic signs in SUNA patients. Our group of patients with long attack duration may all be SUNA. This would have predicted that the pain-related awakenings would have been significantly associated with autonomic signs and background pain. Yet, in this group, pain-related awakenings were significantly associated with more severe pain and shorter attack duration.
Attack duration
According to IHS criteria CTN duration has a cutoff at 2 min (1). Individual attacks are typically characterised by a rapid onset and peak, and then subside lasting overall from 10 seconds up to 2 min (34). It is important to note that attack duration in CTN has not been thoroughly validated and we continue to rely on patient reports. We noted 20 patients with attacks reported as longer than 2 min. Interestingly in the group with long attack duration the intensity was inversely related to the duration of the attack. The significance of this finding is unclear but does differentiate between the two duration subgroups: Pain severity in the short attack duration subgroup was unchanged by its duration.
Is it possible that CTN may occur with attacks of >2 min? CTN patients may report a dull aching pain that continues for a considerable amount of time after the paroxysm (35). Possibly our patients with attacks > 2 min are reporting both of these types of pain: the paroxysm and the ‘after-pain’ as one. Additionally some CTN patients report multiple overlapping attacks that may feel as one longer attack. This would, however, be an unlikely occurrence in the presence of a refractory period where the first attack would inhibit subsequent attacks.
Constant background pain was present in most of the patients with long attack duration. Possibly the paroxysms may have an ‘after-pain’ that continues to increase in duration till it becomes a constant background pain. This would additionally require that disease duration be longer in patients with long attack durations and background pain. However, although longer disease duration occurred in patients from the long attack duration subgroup, this was not statistically significant (p = 0.07). Furthermore, the presence of background pain was not associated with longer disease duration in both subgroups.
Nevertheless, a recent study reports that 2% of their patients developed increased duration of paroxysms and constant pain after 4.5–7 years during a study of the natural history of CTN (36). In a previous study, we found a significant correlation between attack duration and disease duration, with some CTN patients reporting attacks of 2–3 min (37).
Therefore, based on the literature and the data presented, there seems to be an association between long disease duration, presence of background pain, pain-related awakenings and long attack duration. A large prospective study on the natural history of CTN is needed to confirm this. Again, when considering duration it is important to stress that this is assessed via interview and patient recall and is established empirically. This has the potential for error but is still in widespread use.
Significance of longer attack duration
Patients with attacks longer than 2 min may challenge the diagnosis of CTN. This may indicate that longer attack durations are part of the CTN spectrum, or may suggest that the patients with long attack duration may form part of a spectrum with trigeminal autonomic cephalalgias (TAC), particularly SUNCT and SUNA. However, although patients with long attacks had more frequent autonomic signs (40%) than patients with short attack duration (21%), this was not statistically significant (p = 0.1). There was also no difference in location between patients with prolonged attacks versus those with typical attack duration. Location in SUNCT and SUNA is considered less critical than previously thought, as pain in these may occur throughout the trigeminal system (38).
When examining individual sign and symptom profiles in patients with long attack duration, some patients demonstrated a phenotype similar to that of SUNA. Based on long attack duration, the presence of one autonomic sign and a constant interictal pain, seven of eight patients (Patients 65, 66, 71, 74, 75, 79 and 80) could be diagnosed as chronic-SUNA while Patient 64 could be an episodic SUNA (Table 7). Alternatively, we are witnessing CTN with autonomic signs and long attack durations, longer than previously recognised. Conversely, 11 patients (Patients 62, 67–70, 72, 73, 76–78 and 81) have significantly prolonged attacks but no autonomic signs. Under current classifications these could not be diagnosed as SUNA or SUNCT, supporting the above proposition that longer attack durations are indeed part of the CTN spectrum (but see discussion below and Lambru and Matharu (39)).
Moreover, within this last group of patients, eight of them (Patients 67–69, 72, 73, 77, 78 and 81) demonstrated an identifiable trigger area and refractory period (38). However, SUNCT has also been reported with a preserved refractory period (40).
The IHS defines intensity of pain in CTN as ‘severe’ (41,42). We routinely employ a pain scale and it is widely accepted that a score of >6 is severe (17). Severe pain was reported by 59 (97%) of the patients with short attack duration and by five (25%) of our patients with long attack duration. Moderate pain severity is more in line with the IHS’ definition of SUNA.
In summary, we have a number of patients with a combination of features that could be an unusual CTN or are similar to SUNA (41,43). The complexity of this differential diagnosis is recognised by the IHS’ recent classification, which recommends giving both SUNA and CTN diagnoses to such cases (1).
CTN and SUNA as a spectrum
SUNA is now classified as a TAC (1), based on the clinical phenotype of a unilateral headache involving autonomic activation (38) and results from neuroimaging studies (44). However, the clinical phenotype is distinctly similar to that of CTN, particularly since tearing has been documented in what seems to have been CTN (13,15,42). Parallel studies on SUNA have shown that it does not exclusively occur in the ophthalmic region, and over a half have maxillary and one-third mandibular pain (38). Moreover some pathophysiologic findings, such as vascular trigeminal nerve root compression (45,46), and therapeutic strategies, both pharmacologic and surgical, are often shared with those for CTN (47–49).
Recently, Lambru and Matharu (2013) suggested that when SUNA occurs in the maxillary and mandibular dermatomes, tearing and conjunctival injection may be rare or absent (39). With this in mind, it is possible that SUNA patients with little tearing or conjunctival injection are often diagnosed as CTN. This in fact raises the question whether the reported series of patients termed ‘CTN with tearing’ are indeed CTN or SUNA? Another possibility to the somewhat complex diagnostic challenge is to view CTN and SUNA as part of a spectrum (39). At one end are patients with short paroxysmal attacks with no autonomic signs, and no background pain. In the middle are patients with longer attacks, longer disease duration, increased reports of waking from sleep, background pain and autonomic signs. This intermediate stage is supported by our findings of patients with short attack duration and autonomic signs. These are characterised by longer disease duration and increased reports of pain-related awakenings. Finally at the other end of the spectrum are patients with significantly longer paroxysms of pain, constant background pain, intense autonomic activation, and a high frequency of waking.
Further and larger patient trials with functional imaging studies are needed to address this question. While the possibility of a spectrum may be important for the understanding of underlying mechanisms, clinically the delineation between TN and SUNA may be dictated by response to therapy, such as the need to utilise lamotrigine in patients that do not respond to CBZ.
Study limitations
Our sample size was relatively small and may have failed to demonstrate all the range of clinical phenotype. The diagnosis of CTN still rests on the clinical interview and examination and the exclusion of pathology by imaging and other additional tests. As such, it is reliant on patient reports. For example patients may not be consistently reliable in the report of attack duration and may tend to overestimate, but this is however a problem associated with much of the research on duration of facial pain and headache syndromes. Functional imaging would have been extremely useful in the patients with long attack duration to examine hypothalamic activation, usually detected only in TACs such as cluster headache and SUNA (44,50).
The gender ratio in the CTN group (male:female) was 1:1.2 and the female preponderance slightly lower than that observed in other studies (51). This is probably the result of a relatively small sample size and does not in our view introduce significant bias. Our inclusion of triggering may have been overly strict, including only those with clinically verifiable trigger areas.
Conclusions
It is becoming increasingly clear that CTN is a more complex disease entity than previously appreciated. Historically, CTN was not considered to present with constant background pain, or autonomic signs nor was it accepted that the pain could wake patients. However, reports of CTN with such symptomatology suggest an expanded clinical phenotype or possible subgroups. Additionally our finding of attacks lasting for more than the defined 2 min needs further investigation and corroboration. Alternatively, our data may indicate a clinical continuum with the features of SUNA suggesting that CTN and SUNA may be variants of the same disorder.
Clinical implications
The clinical phenotype of classical trigeminal neuralgia (CTN) is heterogeneous and may include background pain, autonomic signs, prolonged attack duration and awakening from sleep. There is considerable overlap in the clinical phenotype of CTN and short-lasting unilateral neuralgiform headache attacks with conjunctival injection and tearing (SUNCT)/short-lasting unilateral neuralgiform headache attacks with autonomic signs (SUNA), with many cases presenting a challenging profile, suggesting a spectrum between CTN and SUNCT/SUNA.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
