Abstract
Objective
Specific acute treatments of migraine are 5HT1B/D receptor agonists; triptans and ergotamine, but only two-thirds of patients respond well without side effects. No migraine-prophylactic drugs are specific to migraine. Prophylactic drugs are selected by time-consuming “trial and error.” Personalized treatment is therefore much needed. The objective of this study was to test the effect of 12 single nucleotide polymorphisms (SNPs) significantly associated with migraine on migraine drug responses.
Methods
Semi-structured migraine interviews including questions on drug responses, blood samples and genotyping were performed on 1806 unrelated migraine cases recruited from the Danish Headache Center. Association analyses were carried out using logistic regression, assuming an additive model for the genetic effect. The effect on drug responses was tested for a combined genetic score and for each of the 12 SNPs. Significant findings were subsequently tested in an independent replication sample of 392 unrelated Danish migraine cases.
Results
A single risk variant, rs2651899 in PRDM16, was significantly associated with efficacy of triptans with an odds ratio (OR) of treatment success of 1.3, and a higher combined genetic score was significantly associated with efficacy of triptans with an OR of success of up to 2.6. A number of SNPs showed nominal preferential association with the efficacy of triptans and others with prophylactic drugs. Analyses of triptans and ergotamine complemented each other and gave a stronger signal when analyzed together. The associations between response to triptans and genetic load and rs2651899 were partially confirmed in the independent sample.
Conclusion
We show for the first time an association between genetic constitution and migraine drug response. This is a first step toward future individualized medicine.
Introduction
Migraine is a disabling headache disorder with a lifetime prevalence of 16% in the European population and is very costly to society (1,2). Medical treatment of migraine consists of acute treatment to ease ongoing attacks and prophylactic treatment to reduce the number of attacks. Specific acute treatment consists of the triptans, i.e. selective 5HT1B/D receptor agonists, and ergotamine, which is believed to act through the same receptors. 5HT1B/D receptor agonists are efficient in treating migraine and cluster headache but have no efficacy in other types of pain. A lack of response to triptans is, however, seen in 25%–30% of migraine patients (3). None of the widely used migraine-prophylactic drugs are specific to migraine. The different drugs were originally developed to treat other diseases and were subsequently shown to have a significant preventive effect in migraine. The most common types of prophylactic drugs are certain anti-hypertensive drugs and anti-epileptics. The mechanism of action in migraine prevention is not known. Only a sub-group of migraine patients respond positively to each drug. It is not possible to predict the effect, side effects are common, and drugs are selected by “trial and error,” satisfying neither to patient nor physician.
The prevalent types of migraine, migraine with typical aura (MTA) and migraine without aura (MO), are complex disorders caused by a combination of genetic and environmental factors and have an estimated heritability of 40%–60% (4). Twelve common single nucleotide polymorphisms (SNPs) associated with migraine susceptibility have been reported by the International Headache Genetics Consortium (IHGC) (5). Effect-sizes of these common SNPs are all quite modest, with odds ratios (ORs) ranging between 0.86 and 1.16, and they do not explain much of the observed heritability, as is the case for most common diseases. The effect-sizes are in general higher for MO than MTA, but pointing in the same direction. The mechanism of action of the risk alleles is unknown.
It is not known whether common genetic variants can explain the difference in response to migraine treatments. In order to study the effect of the 12 common SNPs associated with migraine susceptibility on migraine drug responses, we used a study population of 1806 unrelated Danish patients with MTA and/or MO, thoroughly phenotyped by a validated, semi-structured telephone interview conducted by a trained physician or a trained senior medical student. The information obtained included response to acute and prophylactic migraine drugs. The patients were genotyped for the 12 SNPs found in the meta-analysis mentioned above. We subsequently performed a replication of the significant single-SNP association in a smaller replication sample of 392 unrelated Danish migraine cases, prospectively collected and likewise thoroughly phenotyped and genotyped.
Material and methods
Study population
The study population/discovery sample, consisting of 2463 migraine cases, was recruited at the Danish Headache Center from a large migraine cohort collected in 2010–2011 and two cohorts collected in 1999–2002 and 2005–2006 (6–8). Migraine history and clinical data were obtained by a validated, extensive, semi-structured interview performed either face to face or over the telephone by a trained physician or a trained senior medical student (described in detail previously (8)). The phenotyping was performed according to the International Classification of Headache Disorders, second edition (ICHD-2) (9). Blood samples for DNA-extraction and genotyping were collected by standard venesection.
To minimize clinical heterogeneity, cases diagnosed only with probable migraine with or without aura were excluded from the analysis. Also, cases without genotypes or without interview were excluded, reducing the sample size to 2227 (1118 MTA ± MO and 1109 MO cases). After filtering for relatedness, retaining the probands and removing other family members, the effective sample size was reduced to 1806 unrelated cases, with a mean age of 44.7 (±12.4) and a male-to-female ratio of 1:4.6. Of these, 796 cases were diagnosed with MTA, with or without a co-diagnosis of MO, and 1010 cases with MO only.
A replication sample consisting of 392 unrelated cases, 124 with MTA ± MO and 268 with MO only, was collected prospectively from patients attending the Danish Headache Center between February 2014 and January 2015. The mean age was 42.9 (±10.5) and male-to-female ratio was 1:6. The same methods were used as for the study population/discovery sample.
The study was approved by the Danish Ethical Standards Committee and the Danish Data Protection Agency, protocol number: H-2-2010-122. Written informed consent was obtained from all participants.
Phenotyping and data on medicine response
The validated, semi-structured interview was based on the diagnostic criteria of ICHD-2. In addition to the necessary clinical data for migraine diagnoses, the interview contained information on age of onset, relevant comorbidity, specific provoking factors and use and effect of medication.
Questions about drugs are shown in Table 1
Questions about migraine drug responses in our semi-structured interview.
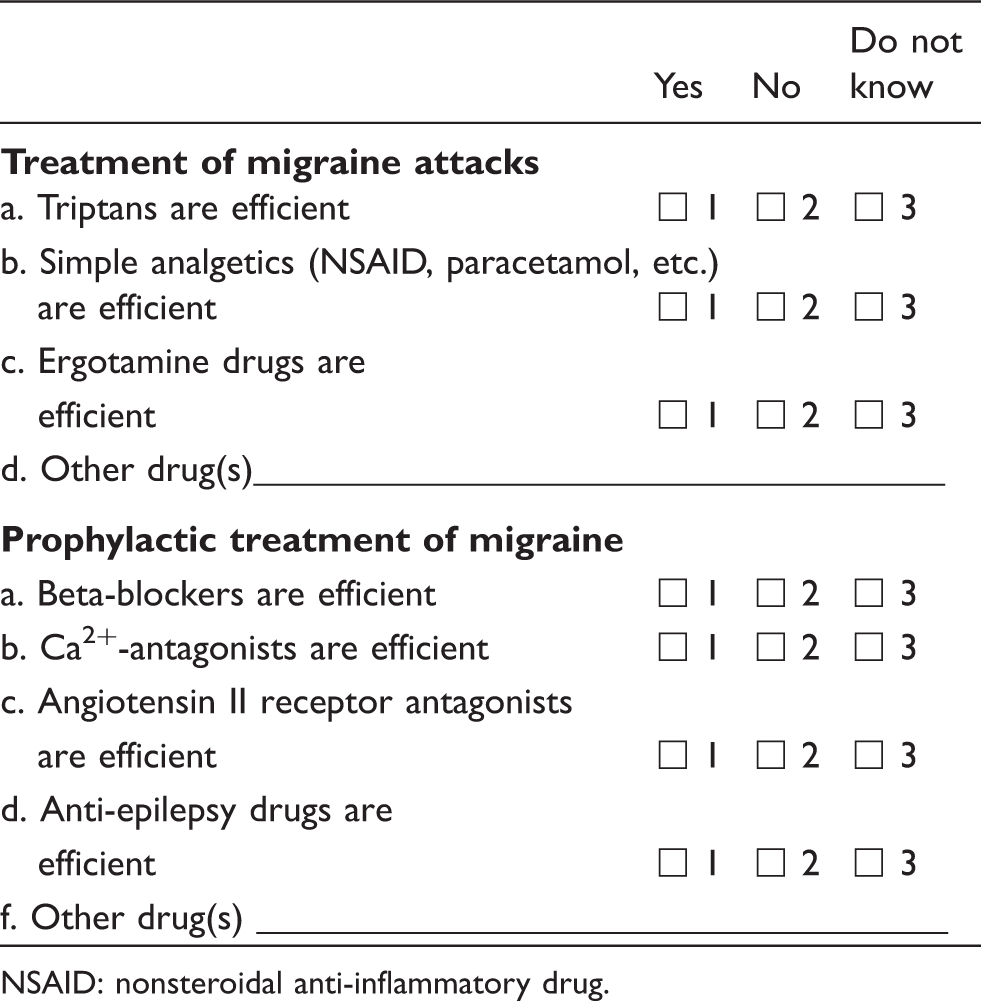
NSAID: nonsteroidal anti-inflammatory drug.
SNP selection and risk alleles
SNPs exceeding the threshold for genome-wide significant association with migraine (p < 5 × 10–8) in the migraine meta-analysis by Anttila et al. (IHCG) were chosen for genotyping (5). The 12 markers included were: rs10915437 (near AJAP1); rs11172113/LRP1; rs12134493 (near TSPAN2); rs13208321—surrogate marker for rs11759769 (FHL5); rs2274316(MEF2D); rs2651899 (PRDM16); rs4379368(c7orf10); rs6478241(ASTN2); rs6790925 (near TGFBR2); rs7577262—surrogate marker for rs6741751 (TRPM8); rs9349379 (PHACTR1); rs10504861 (near MMP16). All samples were typed using Centaurus assays at deCODE genetics, Reykjavik, Iceland. Genotyping success rate was greater than 95%. A replication analysis of the findings in the meta-analysis by Anttila et al. and an association analysis of severe migraine phenotypes in the study population/discovery sample are reported elsewhere (10).
Per convention, the OR of the minor allele was reported for all SNPs identified in the meta-analysis. Thus, an OR > 1 assigns the enhanced risk of migraine to the minor allele, while an OR < 1 assigns the enhanced risk of migraine to the major allele. In this study we focused on the risk alleles. When the OR reported in the meta-analysis was below 1, the allele coding was reversed. Hence, the following risk alleles were determined for the reported SNPs: rs10915437-A-allele; rs11172113-T-allele; rs12134493-A-allele; rs11759769-A-allele; rs2274316-C-allele; rs2651899-C-allele; rs4379368-T-allele; rs6478241-A-allele; rs6790925-T-allele; rs6741751-G-allele; rs9349379-A-allele; rs10504861-G-allele. We calculated a multi-locus cumulative genetic risk score for each individual by adding up the number of risk alleles for each of the 12 SNPs. We subsequently defined two groups representing high genetic load and low genetic load, based on the normal distribution of the risk alleles and its lower and upper quartiles.
Statistical analysis
The aim was primarily to test the cumulative effect of the 12 risk alleles, subsequently the effect of the individual SNPs, on the response to each specific type of medication, i.e. the two types of acute medication (triptans and ergotamine) and four types of prophylactic medication (beta-blockers, calcium antagonists, angiotensin II receptor antagonists and anti-epileptics). The number of treated patients, and thus the power of the test, was considerably higher for acute treatment compared to prophylactic treatment. Therefore we chose effect on response to acute treatment as our primary test, and the less-powered prophylactic treatments as secondary tests. As ergotamine is believed to act through the same receptors as the triptans, we first tested the combined response to specific, acute treatment: positive effect of either triptans or ergotamine or both, versus no effect of any of the two, thereafter testing them separately. As the effect of prophylactic drugs is unpredictable, we hypothesized that they act through different pathways or different levels of the same pathway, leading to prevention of migraine in some patients, depending on their genetic constitution of migraine. The known pharmacological/physiological targets of the different anti-hypertensive drugs and anti-epileptics are quite different. Also, being a non-responder to one prophylactic drug does not predict the tendency of being a non-responder to other prophylactic drugs. Therefore, we primarily wanted to test the effect of the prophylactic drugs separately. However, as the different prophylactic drugs do have one thing in common—they prevent migraine—we cannot rule out that they might have an effect on a yet unknown common pathway. Therefore, we decided also to test the response to all four drugs together, i.e. testing patients who have had an effect from one or more of the prophylactic drugs versus patients without an effect from any of the prophylactic drugs. Furthermore, as anti-hypertensive drugs constitute a logic class of drugs that, despite different ways of action, are known to have some effect in common, i.e. lowering blood pressure, we also decided to test all three anti-hypertensive drugs together. See Figure 1 for the testing strategy.
Testing strategy. Groups of drugs that were assessed in the analyses. The effect on treatment response of both genetic load and the 12 individual risk alleles were tested.
Using logistic regression, assuming an additive model for the genetic effect, we investigated the association of the cumulative genetic score, based on the total risk allele count in each individual, and the single locus association with the binary variable on medication response: “Effect” or “No effect.” Using an additive model means that we were treating the number of risk alleles, i.e. zero, one or two for each individual, as a continuous variable, assuming the same difference in effect on the response to medication when going from zero to one risk allele as compared with going from one to two risk alleles. The additive model was chosen because it was the model used to identify the risk variants in the meta-analysis.
Analyses were performed for all migraine cases, i.e. both MTA ± MO and MO cases together, and for MTA ± MO cases and “MO only” cases alone. All analyses were controlled for gender.
Results were presented with an OR, 95% confidence interval (CI) and a p value. In the primary test, the effect on response to acute treatment, p = 0.05 was considered statistically significant when testing the cumulative effect of the 12 risk alleles, genetic load. When testing the effect of the 12 risk alleles individually, a p value corrected for multiple testing, p = 0.0042, was considered statistically significant (p = 0.05/12 = 0.0042). For replication, the significant single-SNP association in the discovery sample was tested in the replication sample and p = 0.05 considered statistically significant. Statistical analysis was performed using statistical software SAS version 9.3 (2002–2008 by SAS Institute Inc, Cary, NC, USA) in Microsoft Windows 7.0.
Results
The 12 SNPs associated with migraine susceptibility, the risk alleles and proximity genes in the study population/discover sample.
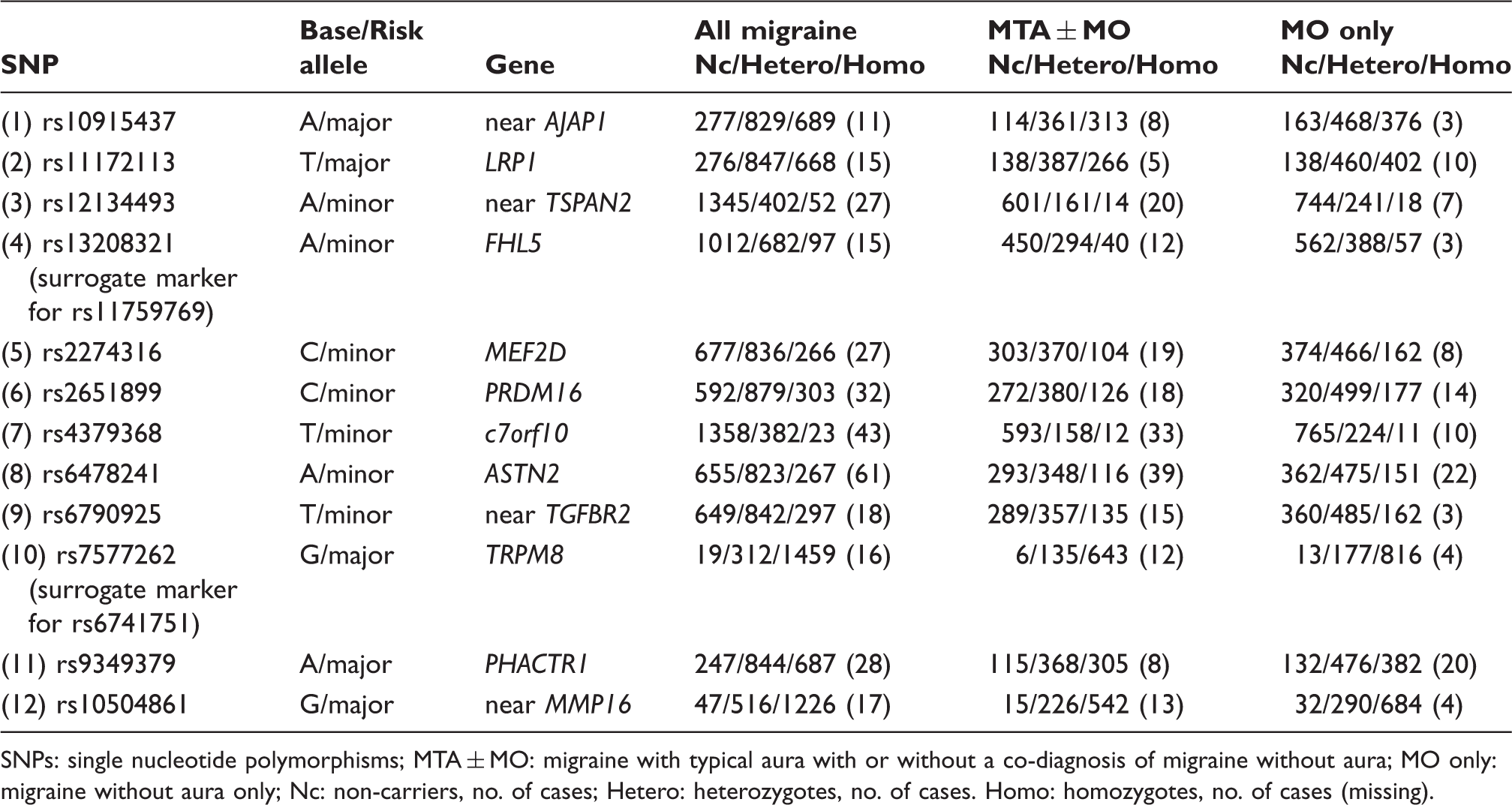
SNPs: single nucleotide polymorphisms; MTA ± MO: migraine with typical aura with or without a co-diagnosis of migraine without aura; MO only: migraine without aura only; Nc: non-carriers, no. of cases; Hetero: heterozygotes, no. of cases. Homo: homozygotes, no. of cases (missing).
Acute treatment
Response to acute treatment in the study population
The effect of migraine drug responses associated with genetic load based on the total risk allele count in each individual, using an additive model for the genetic effect.

Genetic load was first assessed as a continuous variable and then dichotomized into two groups representing high and low genetic load (defined by the upper and lower quartile). Statistically significant results are in bold (p < 0.05). SNP: single nucleotide polymorphism; MTA ± MO: migraine with typical aura with or without a co-diagnosis of migraine without aura; MO only: migraine without aura only; Acute: specific acute migraine treatment (effect of triptans and ergotamine analyzed together); OR: odds ratio; CI: confidence interval.
The effect of specific acute migraine treatment associated with genetic load based on the total risk allele count in each individual, excluding the SNP rs2651899 from the analysis.

Genetic load was first assessed as a continuous variable and then dichotomized into two groups representing high and low genetic load. Significance level p=0.05. Border significant result in italic. MTA±MO: Migraine with typical aura with or without a co-diagnosis of migraine without aura. MO only: Migraine without aura only. Acute: specific acute migraine treatment (effect of triptans and ergotamine analyzed together).
The effect of 5HT1B/D receptor agonists associated with carrying each of the 12 risk variants using an additive model.

Statistically significant results in bold (p < 0.0042), and nominal associations in italic (p < 0.05). SNP: single nucleotide polymorphism; MTA ± MO: migraine with typical aura with or without a co-diagnosis of migraine without aura; MO only: migraine without aura only; OR: odds ratio; CI: confidence interval. Reference = 0 risk alleles for each SNP. SNP: single nucleotide polymorphism; MTA ± MO: Migraine with typical aura with or without a co-diagnosis of migraine without aura; MO only: migraine without aura only.
The effect of triptans associated with carrying each of the 12 risk variants using an additive model.

Reference = 0 risk alleles for each SNP. Statistically significant results in bold (p<0.0042), and nominal associations in italic (p<0.05). MTA ± MO: Migraine with typical aura with or without a co-diagnosis of migraine without aura. MO only: Migraine without aura only.
The effect of ergotamine associated with carrying each of the 12 risk variants using an additive model.

Reference = 0 risk alleles for each SNP. Statistically significant results in bold (p < 0.0042), and nominal associations in italic (p < 0.05). MTA ± MO: Migraine with typical aura with or without a co-diagnosis of migraine without aura. MO only: Migraine without aura only; OR: odds ratio; CI: confidence interval.
The effect of any type of prophylactic drug versus effect of no prophylactic drugs associated with carrying each of the 12 risk variants using an additive model.

Reference = 0 risk alleles for each SNP. Nominal associations in italics (p < 0.05). No p values would withstand correction for multiple testing. SNP: single nucleotide polymorphism; MTA ± MO: migraine with typical aura with or without a co-diagnosis of migraine without aura; MO only: migraine without aura only; OR: odds ratio; CI: confidence interval.
The effect of any type of anti-hypertensive drug versus effect of no anti-hypertensive drugs associated with carrying each of the 12 risk variants using an additive model.

Reference = 0 risk alleles for each SNP. Nominal associations in italics (p < 0.05). No p values would withstand correction for multiple testing. SNP: single nucleotide polymorphism; MTA ± MO: migraine with typical aura with or without a co-diagnosis of migraine without aura; MO only: migraine without aura only; OR: odds ratio; CI: confidence interval.
The effect of beta-blockers associated with carrying each of the 12 risk variants using an additive model.

Reference = 0 risk alleles for each SNP. Nominal associations in italics (p < 0.05). No p values would withstand correction for multiple testing. SNP: single nucleotide polymorphism; MTA ± MO: migraine with typical aura with or without a co-diagnosis of migraine without aura; MO only: migraine without aura only; OR: odds ratio; CI: confidence interval.
The effect of calcium antagonists associated with carrying each of the 12 risk variants using an additive model.

Reference = 0 risk alleles for each SNP. Nominal associations in italics (p < 0.05). No p values would withstand correction for multiple testing. SNP: single nucleotide polymorphism; MTA ± MO: migraine with typical aura with or without a co-diagnosis of migraine without aura; MO only: migraine without aura only; OR: odds ratio; CI: confidence interval.
The effect of angiotensin II receptor antagonists associated with carrying each of the 12 risk variants using an additive model.

Reference = 0 risk alleles for each SNP. Nominal associations in italics (p < 0.05). No p values would withstand correction for multiple testing. SNP: single nucleotide polymorphism; MTA ± MO: migraine with typical aura with or without a co-diagnosis of migraine without aura; MO only: migraine without aura only; OR: odds ratio; CI: confidence interval.
The effect of anti-epileptics associated with carrying each of the 12 risk variants using an additive model.

Reference = 0 risk alleles for each SNP. Nominal associations in italics (p < 0.05). No p values would withstand correction for multiple testing. SNP: single nucleotide polymorphism; MTA ± MO: migraine with typical aura with or without a co-diagnosis of migraine without aura; MO only: migraine without aura only; OR: odds ratio; CI: confidence interval.
The reported responses to triptans and ergotamine in the study population/discovery sample.

All mig: all migraine; MO only: migraine without aura only; MTAMO: migraine with typical aura with a co-diagnosis of migraine without aura; MTA only: migraine with typical aura only.
Genetic load
The analysis of the multi-locus cumulative genetic score treated as a continuous variable showed a statistically significant increase in the effect of 5HT1B/1D receptor agonists (i.e. triptans and ergotamine) and of triptans alone with increased genetic score for “all migraine” (p = 0.0043 and OR = 1.1 for response to 5HT1B/1D receptor agonists, and p = 0.013 and OR = 1.1 for response to triptans). Analysis of MTA ± MO and “MO only” separately showed that the effect was all accounted for by the MTA ± MO group (p = 0.0057 and OR = 1.1).
Likewise, the analysis comparing the effect of medication in the groups with low genetic load versus high genetic load showed a statistically significant increase in the effect of 5HT1B/1D receptor agonists and triptans alone in cases with high genetic load for “all migraine” (p = 0.013 and OR = 1.5, respectively, p = 0.030 and OR = 1.4). This effect was again accounted for by the MTA ± MO group (p = 0.0048 and OR = 1.9 for 5HT1B/1D receptor agonists and p = 0.013 and OR = 1.7 for triptans alone).
Tests for linearity of the cumulative genetic score showed that the linearity model was a good approximation for the association between the score and effect of acute treatment in all three groups, “all migraine,” MTA ± MO and “MO only.”
In the analysis of the effect of the individual SNPs on acute treatment (see below), the presence of rs2651899 was also associated with an increase in the effect of 5HT1B/1D receptor agonists and triptans. We therefore performed an analysis of genetic load after excluding rs2651899 to examine whether the effect of a higher genetic load was a true polygenic signal, or whether the effect of genetic load was driven by this one SNP. Excluding rs2651899, now only assessing 11 SNPs (and thereby losing power), we found the same tendency as before: an increase in the effect of 5HT1B/1D -receptor agonists and triptans with increased genetic score for “all migraine” and MTA ± MO, but the association was no longer significant (see Table 3(b)). However, for the MTA ± MO group and the effect of 5HT1B/1D receptor agonists, the association was borderline significant when assessing the cumulative genetic score as a continuous variable (p = 0.052 and OR = 1.1).
5HT1B/1D receptor agonists
A statistically significant association was found for rs2651899 in the analysis evaluating the influence of the individual risk alleles on the effect of 5HT1B/1D receptor agonists, indicating an enhanced effect of 5HT1B/1D receptor agonists in carriers of the risk allele. An OR of 1.4 with p = 0.0005, respectively, p = 0.0034 was found for “all migraine” (n = 1549) and MTA ± MO (n = 611). For rs11172113 and rs12134493 in ‘all migraine’ and MTA ± MO, a nominal association (p < 0.05) with enhanced effect, with OR ranging from 1.3 to 1.6, was found.
ORs for the effect of 5HT1B/1D receptor agonists in “MO only” in association with the single-locus effect were non-significant, but the effect sizes were comparable to those of “all migraine” and MTA ± MO.
Because of the significant findings in the MTA ± MO group, both for rs2651899 and genetic load, we performed an analysis on MTAMO cases (having a diagnosis of both MTA and MO) and “MTA only” cases (having a diagnosis of MTA only) separately. A significant difference in the number of patients having an effect from 5HT1B/1D receptor agonists compared to the number having no effect between MTAMO and “MTA only” was found using Fisher’s exact test: 274 having an effect versus 87 having no effect for MTAMO, and 108 having an effect versus 157 having no effect for “MTA only,” giving an OR = 0.22 (0.15; 0.31) for “MTA only” compared with MTAMO and p < 0.0001. Testing the single-locus effect on treatment response and effect of the multi-locus cumulative genetic score in MTAMO and “MTA only” separately did not show any significant associations or any differences between the two groups (data not shown). Neither was a difference in the distribution of risk alleles between the two groups was found.
Triptans
The results of the analysis evaluating the effect of the individual risk alleles on response to triptans alone were largely comparable to those of the combined analysis (5HT1B/1D receptor agonists). For rs2651899, the ORs were comparable, but the enhanced effect in carriers a bit less significant, though still statistically significant in “all migraine.” For rs11172113 and rs12134493 the association was a bit stronger with comparable ORs, but did not reach statistical significance.
Ergotamine
Analyzing the effect of ergotamine alone, rs12134493 was the only risk allele found to be nominally associated (p < 0.05) with an enhanced effect, this in MTA ± MO. No nominal associations were found for “all migraine” or “MO only,” but the presence of rs12134493 tended to lower the effect of ergotamine in MO. However, the number of cases was relatively small (93–97 responders and 140–147 non-responders in “all migraine”).
Prophylactic treatment
Genetic load
No significant or nominal associations with genetic load were found for any of the prophylactic treatments, neither in combination nor separately. Tests for linearity showed that the linearity model was a good approximation for the association between risk score and the different types of prophylactic treatment in all three groups, “all migraine,” MTA ± MO and “MO only,” apart from anti-epileptics in “all migraine.”
All prophylaxis
Analyzing the effect of any prophylaxis compared to no effect of any prophylaxis (i.e. beta-blockers, calcium antagonists, angiotensin II receptor antagonists and/or anti-epileptics) in association with the presence of the individual risk alleles, nominal association (p < 0.05) was found for three SNPs: rs13208321 (surrogate marker for rs11759769), rs2274316 and rs10504861.
All anti-hypertensive drugs
A nominal association with higher effect of anti-hypertensive drugs was found for rs2274316 in MTA ± MO. In “MO only” an opposite, but only near-nominal, association was found for rs2274316: The effect of the drugs tended to be lower in carriers.
Beta-blockers
Rs2274316 was nominally associated with a lower effect of beta-blockers in “MO only.” Rs2651899 was nominally associated with an enhanced effect of beta-blockers in MTA ± MO.
Calcium antagonists
No risk alleles had a nominal association with the effect of calcium antagonists. The number of cases was, however, relatively small, with only 30 responders and 150–156 non-responders depending of the risk-allele questioned.
Angiotensin II receptor-antagonists
A nominal association with a lower effect was found for rs11172113 in MTA ± MO. For rs12134493, a nominal association with a higher effect of the drug was found in “all migraine” and in “MO only.” For risk-allele rs6790925, a nominal association with a higher effect was found in MTA ± MO. Finally, a nominal association with a higher effect was found for rs10504861 in “all migraine” and “MO only” (OR for MTA ± MO was similar but did not reach nominal preferential level).
Anti-epileptics
Rs2651899 was nominally associated with a lower effect in “MO only.” Rs10504861 was nominally associated with a higher effect in “all migraine.”
Gender
In all analyses performed, we controlled for gender to test if the effect of the SNPs on drug responses was dependent on gender (data not shown). A difference in drug response between genders was seen for “all migraine” in the analysis of effect of 5HT1B/1D receptor agonists with p values and ORs ranging between p = (0.032; 0.10) and OR = (0.71; 0.77) for all 12 SNPs, with females as reference. Thus, males were less likely to have an effect from 5HT1B/1D receptor agonists than females independently of the individual SNPs. For response to triptans, a similar difference between the genders was seen in “all migraine” (p = (0.044; 0.12) and OR = (0.72; 0.78) for all 12 SNPs). A difference between genders was also seen in response to “all prophylaxis” (the combined analysis) in “MO only” with p = (0.033; 0.099) and OR = (0.61; 0.68) for all 12 SNPs, suggesting that males may benefit less from prophylactic treatment in general (independently of the 12 SNPs). All other analyses showed no difference between the genders. After correction for multiple testing, none of the differences found were significant. The ORs and p values were comparable amongst the 12 SNPs in all the three mentioned cases, indicating that the difference, if any, was independent of the 12 SNPs. Thus, gender did not alter the effect of the SNPs on drug responses.
Replication results
In the independent replication sample, a near- significant association with higher chance of effect of triptans was found for genetic load as a continuous variable in all migraine with an OR of 1.1 (0.99; 1.3) with p = 0.081, where n = 392 (324 responders; 68 non-responders) (high genetic load vs. low genetic load: OR = 1.8 (0.86; 3.9); p = 0.11; n = 196). rs2651899 was insignificantly associated with higher chance of effect of triptans in all migraine with an OR = 1.3 (0.88; 1.9)); with p = 0.9 n = 392 (324 responders; 68 non-responders).
Discussion
This is the first study to investigate the influence of common genetic risk variants on migraine drug responses. In summary, we found that the presence of the single risk allele, rs2651899 in the PRDM16 gene, was associated with an enhanced effect from triptans in all migraine and especially in MA. This association withstood correction for multiple testing. Furthermore, this study shows that the response to triptans can be predicted based on the number of common risk alleles for migraine: The higher the number of risk alleles in an individual, the higher the chance of effect. Replication in an independent sample is often required in genetic studies. For this kind of study it is very difficult, as there are few well-studied clinic samples of sufficient magnitude and none that have asked the same questions about drug responses. However, we prospectively collected a smaller replication sample at the Danish Headache Center, and the associations between response to triptans and genetic load and rs2651899 were partially confirmed in this independent sample.
Previous literature
Knowledge about migraine pharmacogenetics is very limited. Few studies have been performed, and they are all based on the candidate gene approach (11,12). The research on acute treatment has focused on polymorphisms in seven genes involved in the neurotransmitter systems known to be affected by triptans, including 5HT1B/1D receptors/transporters and dopaminergic receptors, and in triptan metabolism, including cytochrome P450 (11). A polymorphism in a serotonin transporter (STin2 VNTR) has been associated with an increased risk of inconsistent response to triptans (13). Apart from this, there are no notable results. Identification of strongly predictive variations in candidate genes associated with triptan response is complicated by the complexity of the molecular events involved in the pharmacokinetics and pharmacodynamics of triptans (11). The pharmacokinetics and -dynamics are also affected by different drug formulations (tablets, nasal sprays, injections) and properties of the blood-brain barrier (14,15). As pointed out by reviews on migraine pharmacogenetics, a low number of patients and inconsistency in the definition of responders and non-responders are limitations in these studies (11,12). Even less research has focused on pharmacogenetics of prophylactic treatment. Only two inconclusive studies on the influence of the ACE-genotype on response to angiotensin-converting enzyme (ACE)-inhibitors/sartans and the influence of mitochondrial DNA (mtDNA) on the response to riboflavin have been performed (12). As we have no knowledge about the mechanism of action of the migraine prophylactic effect, the hypothesis-driven approach, as used in the research on triptans, is not very obvious.
Our study, focusing on common migraine risk variants, is a new approach to migraine pharmacogenetics. Genetic variants that contribute to pharmacogenetic traits often have little phenotypic consequence before administration of a drug (16,17). Therefore, they have escaped natural selection, in contrast to genetic variants with large effects on disease traits that decrease the number of offspring. Strong pharmacogenetic traits are therefore more likely to be common in the normal population. The variants involved in migraine drug responses are likely to be found among the risk variants of migraine or in the same pathways. A retrospective example of a common variant with a small effect size on the disease, but a large effect as a treatment target, is the statins in the treatment of high levels of blood low-density lipoprotein (LDL) cholesterol (18–20). HMGCR is identified as an important genome-wide significant locus for LDL cholesterol, and statins, the most commonly prescribed drug to lower blood LDL cholesterol, act by inhibiting the HMGCR enzyme.
5HT1B/1D receptor agonists
Comparing the groups representing “high genetic load” and “low genetic load,” defined by the lower and upper quartiles of the normal distribution of the multi-locus cumulative genetic risk score, we found that the chance of effect of 5HT1B/1D receptor agonists in cases with MTA ± MO was doubled in the group with high load. The effect was also significant in the “all migraine” group, but no association was found in “MO only.” The analysis of the genetic risk score, not dichotomized into groups but treated as a continuous variable, also showed significantly increased effect of 5HT1B/1D receptor agonists with increased risk score for MTA ± MO cases, here with OR = 1.1. Thus, having one more risk allele increases the chance of being responsive to 5HT1B/1D receptor agonists by 1.1. At first glance this effect looks modest. But, having n more risk variants enhances OR by OR n . Hence, having 10 more risk alleles increases the chance of being responsive by 1.110 = 2.6. The same significant effect was found in “all migraine,” but again not for “MO only.” Both tests on genetic score were also significant for triptans alone, but the association was stronger when assessing triptans and ergotamine together. Because triptans and ergotamine are believed to act through the same receptors, hence the common name 5HT1B/1D receptor agonists, we would expect to see this enhancement of the association and effect when assessing them together. Seeing this strengthens our result.
Analysis of the effect of genetic load after excluding rs2651899 to examine whether the effect of genetic load was a true polygenic effect, or whether the association was driven by rs2651899, showed the same: increased effect of 5HT1B/1D receptor agonists with increased risk score in “all migraine” and MTA ± MO cases, though the association was now just below the significance threshold. However, this analysis was less powered, including only 11 and not 12 SNPs. Thus, the association found is likely to be a true polygenic effect.
At first, it looks like the association applies only to MTA ± MO cases. However, the association is still significant in “all migraine,” meaning that the more abundant “MO only” cases do not dilute the signal. So, the effect is maybe not restricted to MTA ± MO. There are many more non-responders in the MTA ± MO group, and thus the difference between responders and non-responders becomes detectable and significant. The effect could be the same in “MO only,” but because of a very few non-responders not detectable. We also considered whether increased chance of effect in MTA ± MO was accounted for by MTAMO cases (MTA cases with a MO co-diagnosis, in contrast to “MTA only” cases) because they might be more similar to “MO only” cases. A significant difference in the number of patients with effect of 5HT1B/1D receptor agonists compared to the number without effect between MTAMO and “MTA only” was found, MTAMO cases being more than four times as likely to have an effect than “MTA only” cases. However, analyzing single locus effect, effect of genetic risk score and risk allele distribution in MTAMO and “MTA only” separately, no difference between the groups was found and no associations were significant in any of the groups, likely because they were too small.
Cases with low genetic load may have more rare variants as the background of their migraine, representing sub-phenotypes that are less likely to respond to 5HT1B/1D receptor agonists. This is in line with the fact that the association with migraine for some of the known risk variants is stronger for MO than MTA, and the hypothesis that MTA might be caused by more rare variants with larger effect sizes or has a higher degree of genetic heterogeneity (5).
The role in migraine of the risk allele found to influence the response to 5HT1B/1D receptor agonists, rs2651899 in PRDM16, is not known. The PRDM16 gene on chromosome 8 (8q21) encodes a transcription factor that regulates leukemogenesis, palatogenesis and brown-fat development. Furthermore, it is important for stem cell function throughout the nervous and hematopoietic systems, and deficiency leads to enhanced oxidative stress, which is possibly relevant to migraine (21). An overview of the known functions of the 12 risk variants is given in Supplementary Material Table 1.
An important limitation of our study is the self-reported effect. Ideally, patients would prospectively complete a headache diary (22). For a relatively large sample size like ours, this would be very difficult, time consuming and probably with low compliance. Regarding the self-reported effect of ergotamine, we expect a substantial proportion of patients to confuse unpleasant side effects with lack of effect. We did not include a measure of severity of migraine in this study, as we have previously analyzed the association between the 12 SNPs and traits representing severe migraine in the same study sample (discovery sample) without significant findings (10).
Prophylaxis
We found a number of genetic variants that were nominally associated (p < 0.05) with the efficacy of prophylactic drugs, but no associations would withstand correction for multiple testing. The lack of significance can be caused by a relatively small sample size, the difficulties in assessing the response to the drugs, recall bias, and the fact that the different prophylactic drugs are not receptor specific like the 5HT1B/1D receptor agonists. Thus, our findings are hypothesis generating and must be confirmed or refuted in future studies.
The real benefit of individualized treatment is in prophylaxis because selection of drugs today is exclusively by trial and error. Each drug has to be tried for three months before a decision about efficacy can be made. Since many drugs have proven efficacy, albeit of modest size, it can take years before the optimal drug is found for an individual patient. If genotyping can point to the most effective type of drug, even perhaps only a class of drugs, this would save a lot of time and effort. Furthermore, many patients drop out before all possibilities have been tried and this could be avoided. Our findings are not yet helpful toward drug selection but they do suggest that in time this will become possible.
Summary and future perspectives
In this study we take a first step toward genetically based individualized migraine drug treatment. We obtained an OR of treatment success for triptans of 1.3–2.6 depending on single SNPs or genetic load of 12 SNPs associated with migraine. This is not enough to advocate clinical use of genetic testing of patients given the ease of just trying the triptans in practice, but it indicates a possible road toward future individualized medicine (23). Unfortunately, the results for prophylactic therapies achieved only nominal association. It is in the prophylaxis of migraine that individualized treatment is likely to provide the biggest gain. Future studies should therefore focus on prophylaxis and should use not just variants significantly associated with migraine, but create a cumulative genetic risk profile from variants across the genome with predictive ability that may plateau at a p value cutoff of ∼0.1–0.2. Future genome-wide association studies (GWAS) meta-analyses focusing on pharmacological traits in migraine patients should also be performed. Notably, the omics approach, integrating genomics, transcriptomics, proteomics, metabolomics, etc., holds a great perspective (24,25).
Article highlights
Personalized treatment in migraine is much needed. We show an association between genetic constitution and triptan response. This is a first step toward future individualized medicine.
Footnotes
Funding
This work was supported by grants from the Lundbeck Foundation and a PhD scholarship from the University of Copenhagen.
Acknowledgments
We would like to thank all participating patients and the employees at the Danish Headache Center for their help during the recruitment of the patients. Also thanks to the Department of Biostatistics at the University of Copenhagen.
Declaration of conflicting interests
Jes Olesen has within the last two years received grants/research support from and/or has been a consultant/scientific advisor for, and/or has been on the speaker’s bureau of Alder Biopharma, Proreo Pharma and Amgen. Thomas Werge has been a consultant/performed lectures for H. Lundbeck A/S and received grants from the Lundbeck Foundation. Kári Stefánsson is the CEO of the company deCODE genetics in Iceland. Hreinn Stefánsson is an employee of the company deCODE genetics in Iceland. Ann-Louise Esserlind and Anne Francke Christensen have nothing to declare.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
